Abstract
It has been suggested that hormonally controlled submandibular salivary gland (SSG) development and secretions may be affected by endocrine disruptor compounds. We investigated the effects of oral gestation-lactation exposure to 1 mg/kg body weight daily dose of the estrogenic soy-isoflavone genistein and/or the anti-androgenic food contaminant vinclozolin in female rats. The SSGs of female offspring were collected at postnatal day 35 to study gland morphogenesis and mRNA expression of sex-hormone receptors and endocrine growth factors as sex-dependent biomarkers. Because of high expression in neonatal SSG, mRNA expression of transforming growth factor α was also studied. Exposure to genistein, vinclozolin, or a genistein+vinclozolin mixture resulted in significantly lower numbers of striated ducts linked to an increase in their area and lower acinar proliferation (Ki-67–positive nuclei). Exposure to the mixture had the highest significant effects, which were particularly associated with repression of epidermal growth factor, nerve growth factor, and transforming growth factor α expression. In conclusion, early exposure to low doses of genistein and vinclozolin can affect glandular structure and endocrine gene mRNA expression in prepubertal SSG in female rats, and the effects are potentialized by the genistein+vinclozolin mixture. Our study provides the first evidence that SSG are targeted by both estrogenic and anti-androgenic disrupting compounds and are more sensitive to mixtures.
Keywords
Introduction
Salivary glands are exocrine and endocrine organs involved in homeostasis. Salivary gland dysfunctions can lead to oral disease, taste perception loss, and/or partial or total xerostomia, and they can occur as a consequence of drug intake, aging, and some pathology such as diabetes mellitus, hypertension, neurological disorders, and depression. Epidemiological studies have revealed evidence of interrelationships between the modification of salivary gland secretions and decrease in sex hormone production; salivary composition changes with aging, but the mouth dryness associated with menopause in healthy women could be corrected by hormonal therapy replacement (Laine and Leimola-Virtanen 1996). Sjogren’s syndrome, an autoimmune disease predominant in women that affects lachrymal and salivary gland exocrine functions, is associated with a defect in the production of androgens in the salivary glands (Porola et al. 2007). In rodents, as in humans, the submandibular salivary gland (SSG) strongly expresses androgen (AR) and, to a lesser extent, estrogen receptors (ER) and progesterone receptors (PR; Campbell et al. 1990; Ozono et al. 1991; Xu 2003).
The sexual dimorphism of salivary glands has been established, as the involvement of sex hormones in their development and function (Rins de David et al. 1990). Estrogens modulate salivary flow and electrolyte composition (Houssay and de Harfin 1973), whereas androgens induce the synthesis of proteins such as proteinases and kallikreins (Treister et al. 2005). More specifically, both estrogens and androgens regulate the synthesis of two hormonal factors that could be seen as SSG endocrine secretions, namely, nerve growth factor (NGF) and epidermal growth factor (EGF). These factors play a physiological key role, since they are released into the bloodstream (Boyer et al. 1991; Hwang et al. 1991) and are involved with ontogenetic development. Nerve growth factor acts on the sympathetic nervous system, particularly under stress conditions, whereas EGF acts primarily on mammary gland morphogenesis, uterine growth, and gametogenesis (Rougeot et al. 2000). In addition, EGF protects the oral cavity epithelium and maintains taste bud integrity (Morris-Wiman et al. 2000), whereas NGF is more involved in taste perception (Suzuki et al. 2007), behavior, and brain plasticity (Saruta et al. 2010). As a consequence, SSG sialoadenectomy results in taste loss, oral illness, and reproductive disorders (e.g., increased abortion rate and spermatogenesis impairment), which demonstrates that salivary secretions contribute to the regulation of many physiological processes, including sensory perception and immune and inflammatory reactions, but also reproductive and peripheral organ development and/or regeneration (Mathison 1995).
Taken together, these data underline the impact of sex hormones on the systemic role of salivary glands and the deleterious consequences of their dysfunction in homeostasis. Consequently, they lead us to consider salivary glands as potent endocrine disruptor targets. In this way, SSG share some similar characteristics with mammary glands in terms of morphology, sex hormone dependency, cellular mechanisms, and tumor histopathology (Actis 2005). We therefore hypothesized that female SSG morphogenesis and properties could be similarly affected by early exposure to endocrine disruptors (El Sheikh Saad et al. 2011).
We report here on the effect of prepubertal development of female rat SSG following an in utero
Materials and Methods
Chemicals
Genistein was synthesized with a purity of 99% (LCOO, Université Bordeaux 1, Talence, France). Vinclozolin was extracted from the commercial formulation Ronilan (BASF, Levallois-Perret, France) according to Bursztyka et al. (2008). The extract was dried in vacuo, and then recrystallized from methanol. Its purity, as verified by HPLC-DAD (192–400 nm) and GC-MS analysis, was higher than 96% (data not shown). In addition, the absence of the degradation products M1 and M2 was verified by LC-MS.
Animals and Experimental Procedures
All procedures involving rats were approved by the local authorities in accordance with the French Ministry of Agriculture ethical guidelines for the care and use of laboratory animals. Animals used in this study were from the same pool of animals as those studied in the recent reports of Lehraiki et al. (2011) and El Sheikh Saad et al. (2011), and our study is part of a larger study aiming at investigating the developmental effects of low doses of genistein and/or vinclozolin on several sex hormone–regulated organs. A total of sixty specific-pathogen–free (SPF) female and sixty SPF male Wistar Han rats at eight weeks of age (Harlan France SARL, Gannat, France) were acclimatized to SPF housing conditions (22°C room temperature with 55% relative humidity and a twelve-hour light/dark period) for four weeks before mating. To avoid xeno-hormone residues, they were housed in individual polypropylene cages and were allowed ad libitum access to charcoal-filtered water and a purified phytoestrogen-free diet (INRA, Jouy-en-Josas, France; Stroheker et al. 2003).
After mating, females were examined daily to determine the first gestational day (GD1) by examining the presence of spermatozoids in vaginal smears. Dams were randomly allocated to four groups corresponding to control (CTRL), genistein (GEN), vinclozolin (VIN), and genistein/vinclozolin (GEN+VIN) groups (fifteen rats per group). On parturition day, the litters were weighed, sexed, and standardized at ten offspring. From GD1 to weaning (postnatal day [PND]21), dams received the xeno-hormones orally at the 1 mg/kg BW dose, alone or in combination. Genistein and/or vinclozolin were dissolved in corn oil (Carrefour, Dijon, France) in order to give 2 mL/kg BW. Control animals received the same volume of vehicle alone (2 mL/kg BW). To prevent photolysis and oxidation, solutions were stored at 4°C in closed vials wrapped in aluminum foil.
In the present study, only female pups were studied. Thus, at weaning, ten female offspring per group (one per litter) were marked with implanted chips, and gathered in polypropylene cages (five per cage). They had ad libitum access to charcoal-filtered water and were fed a soy-free purified diet until sacrifice (PND35). Body weight and food consumption were measured twice a week.
Concomitantly, the anogenital distance (AGD)—a well-known sensitive endpoint for estrogenic and anti-androgenic chemical compounds—was measured at weaning (PND21) by considering up to three females per litter (n = 40/group). In addition, to determine the time of puberty, one female pup per litter was monitored daily for vaginal opening (VO) from PND21 to PND35.
Sacrifice and Sampling
At PND35, female pups were anesthetized with isoflurane (2.5%) followed by rib cage opening and dorsal aorta exsanguination. Submandibular salivary glands were carefully excised, then weighed. The left SSG was immediately frozen in liquid nitrogen and stored at -80°C for mRNA analysis. The right SSG was embedded in O.C.T. compound (Sakura Finetek, France), frozen (-56 to -62°C) in an isopentane histobath (Thermo/Shandon Histobath, Cergy Pontoise, France), then stored at – 80°C for histological and immunohistochemical analysis.
Quantitative Immunohistochemical Studies
Longitudinal sections of the whole SSG were taken at 5 μm on a Cryostat Leica CM3050 S (Leica, France) at -20°C cut temperature. Slides were air dried and stored at -80°C until analysis.
For histological analysis, cryosections were fixed for ten seconds in RapidFix (Thermo Shandon, France) and stained with Masson Trichrome. Slides were examined with a Nikon Eclipse 600 microscope with an attached DXM1200C camera (Nikon, version 5.03). Microscopic images were captured using a 40× objective (area = 0.0285 mm2) and analyzed using NIS-Elements Br 3.0 imaging software (Nikon, France). The number and area of striated ducts were determined as the means of thirty fields per animal (six animals per group) to determine the effect of chemical exposure on SSG development. The area of striated ducts was expressed in µm2 and the count of striated ducts was expressed as their number per unit surface area (number/mm2).
Both apoptosis and proliferation were assessed jointly by means of the Novolink Polymer Detection System (Novocastra Laboratories, UK), which detects the anti-active caspase-3 (Promega, Madison, WI, USA, 1/1,000) and the Ki-67 protein (Dako, Glostrup, Denmark, 1/1,500), respectively. In accordance with the manufacturer’s recommendations, sections were fixed in cold acetone for ten minutes, then rinsed twice for five minutes in buffered saline (Tris, pH 7.4) to be immunostained. The sites of cytoplasmic caspase-3 and nucleus Ki-67 immunoreactive antigens were detected on separated sections with DAB-H2O2, which generated a brown color. Negative controls were run in parallel by replacing the primary antibody with non-immune immunoglobulins. Sections were then counterstained with hematoxylin and mounted with EUKITT mounting medium (O. Kindler GmbH, Germany). To detect apoptotic and proliferative cells, respectively, the numbers of caspase-3 and Ki-67-positive cells were also determined as the mean of thirty fields (×40) per animal (six animals per group), as described above. The results were expressed as the number of labeled cells/mm2.
RNA Isolation and Quantitative Real-Time Polymerase Chain Reaction
The mRNA expression of SGG sex hormone receptors (ERα, AR, PR) and growth factors (EGF, NGF, TGFα) was measured by quantitative real-time polymerase chain reaction (PCR). Total RNA was extracted from submandibular glands using TRIzol Reagent (Invitrogen, France). The concentration and the integrity of total RNA were assessed with the Experion system (BioRad, France). cDNAs were synthesized using the iScript cDNA synthesis kit (BioRad) by using 50 ng total mRNA. Ribosomal protein S9 was used as an internal control. Primer sequences and qRT-PCR conditions are listed in Table 1. All oligonucleotide primers were designed using Beacon Designer 4.0 software (Bio-Rad Laboratories, Marne-La-Coquette, France). Amplification and detection of targeted and housekeeping genes were performed in duplicate for each animal (ten per group) using iQ SYBR Green Supermix (BioRad, France) on an iCycler MyiQ Single Color Real-Time Detection System (BioRad, France). The PCR program consisted of an intitial denaturation at 95°C for thirty seconds, followed by forty PCR cycles: 95°C for five seconds, then 60°C for ten seconds. Amplification of specific transcripts was confirmed by melting-curve profiles generated at the end of the PCR program between 65°C and 95°C. The relative expression of transcripts was based on the following formula (Pfaffl 2001):
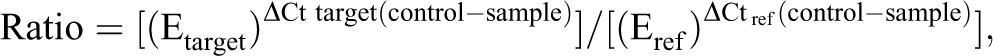
Primers Used for Real-Time PCR Analysis
Statistical Analysis
For each group, the data were expressed as mean ± SD. Statistical analyses were run using BMDP statistical software (Statistical Solutions Ltd, Cork, Ireland; Release 8.0, 2000). Comparison of the different parameters studied in the four exposure groups was made using standard one-way ANOVA. When appropriate, inter-group comparisons were performed using the Tukey test, and
Results
Effect on Food Consumption, Growth Rate, and Development
Daily exposure to 1 mg/kg BW of genistein and/or vinclozolin during the gestation/lactation period did not affect food and water intakes of dams and offspring (data not shown). These exposures also had no significant effect on the litter size, the sex ratio, or the pup BW at birth (Table 2). Nevertheless, using ANOVA, the BW at weaning (PND21) was significantly different among the four exposure groups (F = 8.98;
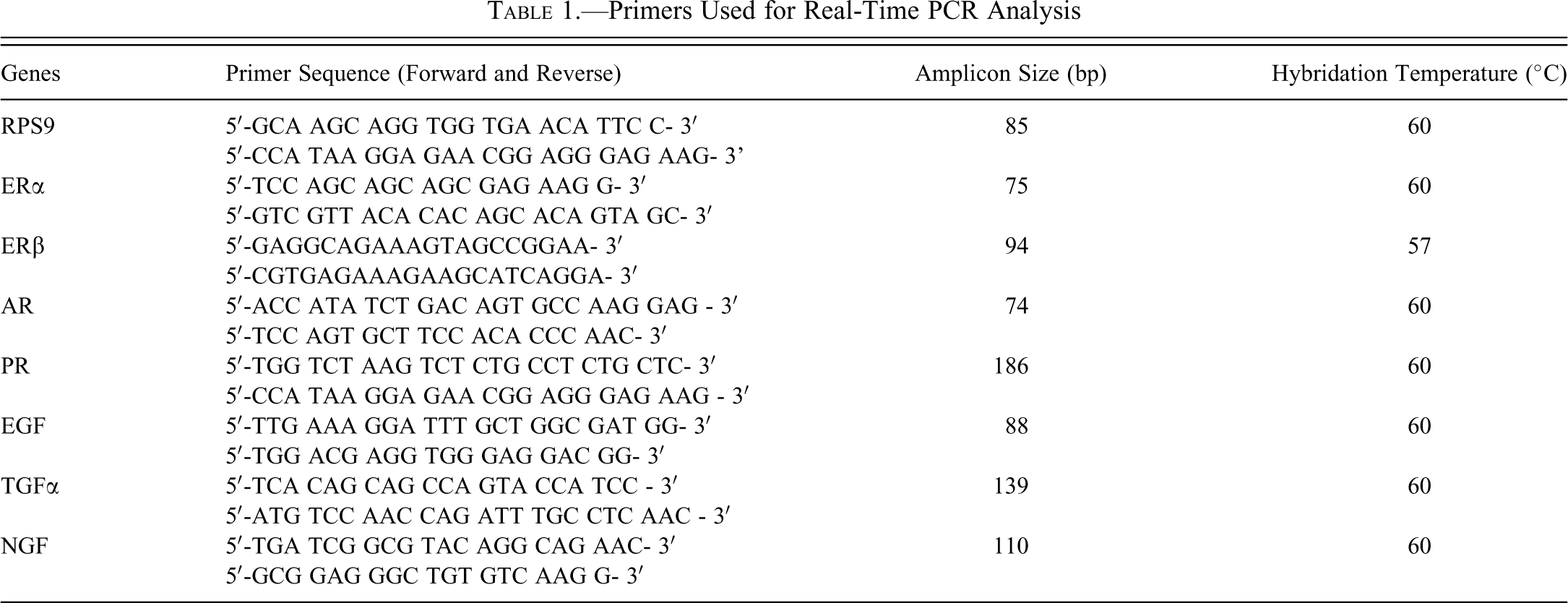
Timing of vaginal opening in female rat offspring (n = 10) after gestational and lactational exposure to genistein (GEN), vinclozolin (VIN), or the mixture (GEN+VIN) at 1 mg/kg BW oral dose. Each point on the graph represents the cumulative percentage of rats with vaginal opening. ANOVA indicated a slight difference among the four groups (F = 2.35;
Effect of Gestational and Lactational Exposure to Genistein, Vinclozolin, and Genistein+Vinclozolin (1 mg/kg body weight) on Developmental Parameters (mean ± SD) in Female Offspring
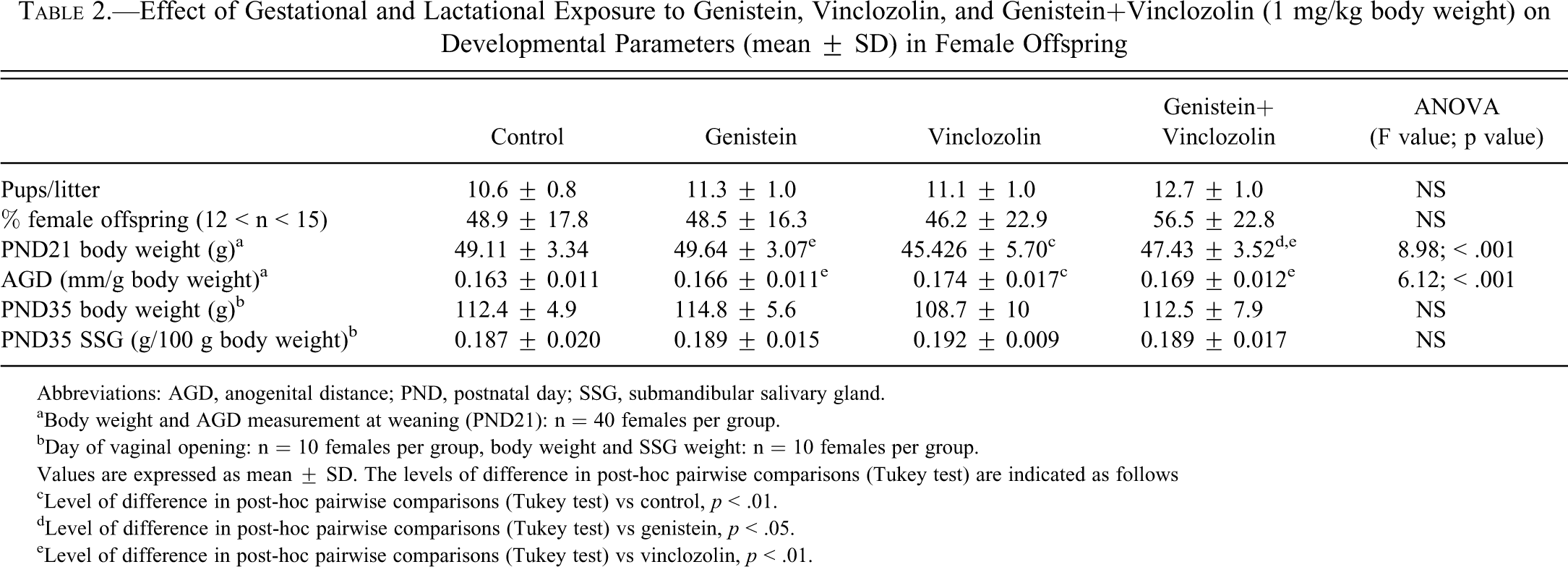
Abbreviations: AGD, anogenital distance; PND, postnatal day; SSG, submandibular salivary gland.
aBody weight and AGD measurement at weaning (PND21): n = 40 females per group.
bDay of vaginal opening: n = 10 females per group, body weight and SSG weight: n = 10 females per group.
Values are expressed as mean ± SD. The levels of difference in post-hoc pairwise comparisons (Tukey test) are indicated as follows
cLevel of difference in post-hoc pairwise comparisons (Tukey test) vs control,
dLevel of difference in post-hoc pairwise comparisons (Tukey test) vs genistein,
eLevel of difference in post-hoc pairwise comparisons (Tukey test) vs vinclozolin,
SSG Morphometry
Masson trichrome staining at PND35 indicated typical immature SSG in controls, with sero-mucous acini (pink-white), connective tissue (green), and striated ducts (red-pink) that differentiate into the granular convoluted tubules (GCT), a specific segment of adult SSG in rodents. In all rats, whether exposed to the chemicals separately (Figures 2B and 2C) or in mixture (Figure 2D), most of the striated ducts appeared wider and less frequent than in the controls (Figure 2A). This finding was confirmed by the ANOVA for both their number (F = 21.38;
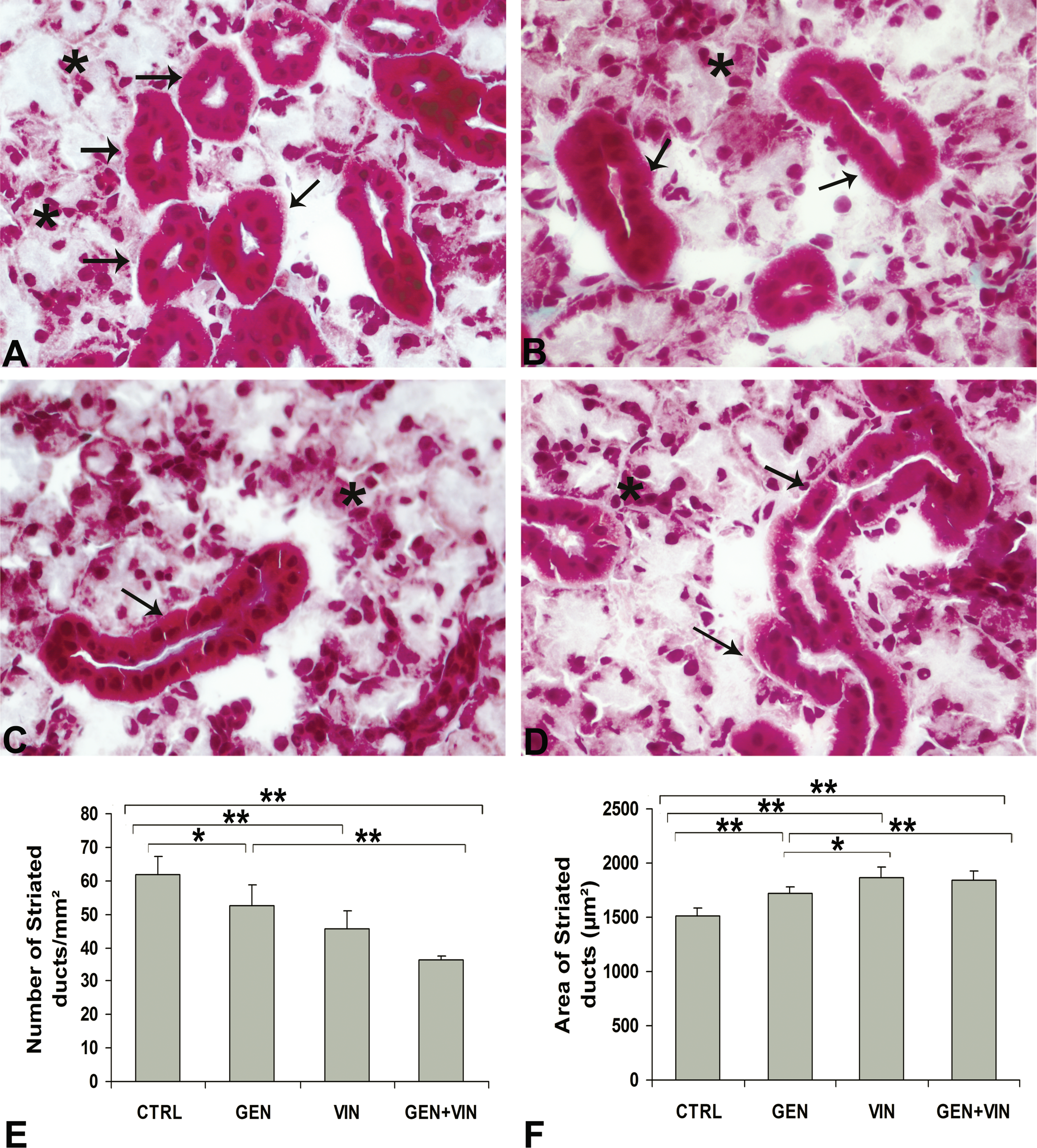
Submandibular salivary gland histology and morphometric results in female rat offspring at PND35 after gestational and lactational exposure to 1 mg/kg body weight per day of genistein (GEN), vinclozolin (VIN), and their mixture (GEN+VIN). Representative photomicrographs of Masson’s trichrome–stained sections (5-µm sections, ×400 final magnification). Sero-mucous acini are indicated by stars and striated ducts by arrows. The female CTRL (A) group SSG had normal-sized striated ducts, whereas the SSG in the female GEN (B), VIN (C), and GEN+VIN (D) groups had larger striated ducts. The number of striated ducts per mm2 (E) and the area of striated ducts expressed in µm2 (F) are represented as the mean value ± SD for each group (n = 6). Intergroup differences (Tukeys test) are indicated by the following symbols: **
Assessment of Cell Proliferation and Apoptosis
At PND35, the caspase 3 protein was not expressed in SSG of controls nor in those of any treated rats (data not shown). In contrast, Ki-67 positive cells (dark brown nuclei) were seen, especially in the acini (Figure 3A-D), and the number of positive nuclei was significantly different among exposure groups (F = 8.20;
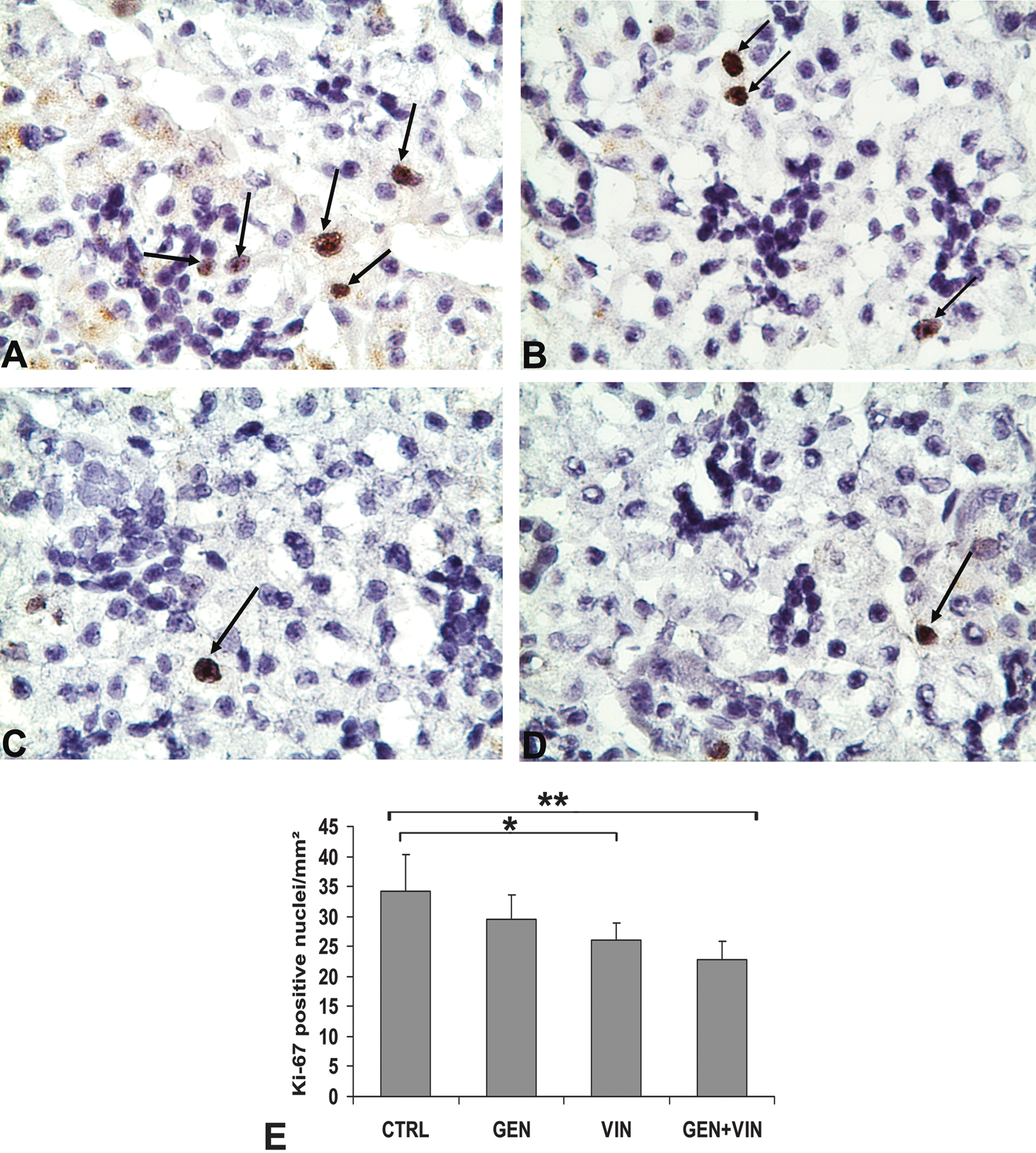
Proliferation of submandibular salivary glands in PND35 female rat offspring after gestation-lactation exposure to 1 mg/kg body weight of genistein (GEN), vinclozolin (VIN), and the mixture (GEN+VIN). Immunohistochemical photomicrographs of Ki-67 (×400) expression in submandibular salivary gland (SSG) sections. The Ki-67–positive cells have brown-stained nuclei. Note the presence of numerous Ki-67–positive nuclei (arrows) in the CTRL group (A) and the presence of rare mitotic nuclei in the GEN- (B), VIN- (C), and GEN+VIN- (D) treated groups for these sections. The number of Ki-67–positive nuclei decreases in the VIN and GEN+VIN groups (E). Values are expressed as mean ± SD per mm2 (n = 6). Different intergroup values (Tukey test) are indicated by the following symbols: **
Expression of Sex Hormone Receptors and Endocrine Growth Factors
At PND35, sex steroid receptor transcripts were differently expressed in the SSG from control group animals; AR was the most abundant sex steroid receptor transcript (Ct = 26) in comparison to ERα (Ct = 30) and PR (Ct = 29), and ERβ transcripts were not detected (Ct > 36), although the β primer used was fully efficient for detecting ERβ transcripts in a control uterine sample (Ct = 25; data not shown). The levels of sex hormone receptor transcripts were not greatly affected by the treatments (Figures 4A-4C). We found only a tendency for AR (F = 2.24,
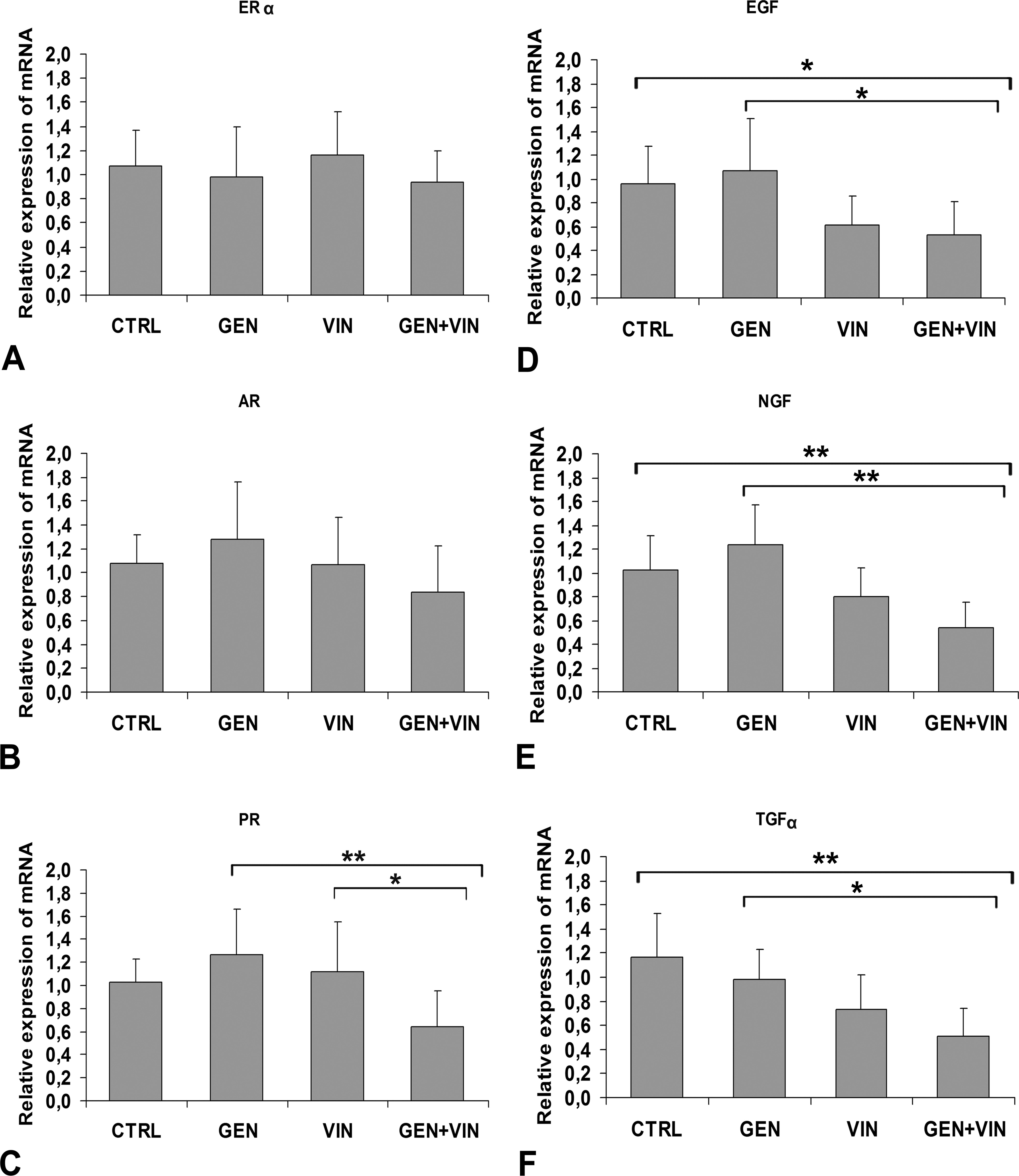
Effect of gestational and lactational exposure to 1 mg/kg body weight of genistein (GEN), vinclozolin (VIN), and the mixture (GEN +VIN) on the mRNA expression of sex hormone receptors (A, B, C) and growth factors (D, E, F) in the submandibular salivary gland of female rats at postnatal day 35 using a real-time PCR method. Values are mean ± SD (n = 10). Intergroup differences (Tukey test) are indicated by the following symbols: **
At PND35, the EGF growth factor transcripts were highly expressed in the CTRL SSG of the unexposed animals (Ct = 22), whereas TGFα and NGF mRNAs were less expressed (Ct = 33 and Ct = 29, respectively). The mean level of the EGF expression was significantly different in the four groups (F = 4.84;
Taken together, our results show that both morphogenesis of SSGs and mRNA expression of the related endocrine growth factors in the SSG of female rats were more affected by the mixture of xeno-hormones than by the compounds alone, and that the glands were more affected by vinclozolin than genistein.
Discussion
This study provides new evidence that SSG could be targeted by steroid-like xenobiotics, as suggested by several previous reports (Carvalho et al. 2011; De Rijk et al. 2003; Sakabe et al. 2000), and it gives the first evidence that relatively low doses of estrogenic and anti-androgenic dietary endocrine disruptors are capable of disrupting the development and secretions of SSG in immature female rats. Interestingly, exposure to genistein+vinclozolin strongly increased normal differentiation of striated ducts into GCT but decreased the mRNA expression of endocrine biologically active peptides (i.e., EGF and NGF), whereas sex-dependant biomarkers were weakly affected. This increase in the effect of exposure to the genistein+vinclozolin mixture compared with exposure to each compound alone is reminiscent of our previous results reported for the female mammary gland by El Sheikh Saad et al. (2011) as well as for male reproductive organs (Eustache et al. 2009), two other studies based on a similar exposure protocol. Overall, these results give supplementary data to argue that the effect of a perinatal exposure to genistein and/or vinclozolin on developmental programming of the SSG could be linked to sex hormone–dependent disorders.
Comparing AGD and the time of VO suggests that early exposure to genistein and vinclozolin did not greatly affect sex hormones at the 1 mg/kg daily dose. Nevertheless, early exposure to vinclozolin led to some female virilization, as shown by AGD at PND21, suggesting a predominantly androgenic effect, which was previously reported for the female reproductive tract (Buckley et al. 2006). This apparent discrepancy could be the result of the androgen agonist activities of the M2 vinclozolin metabolites (Molina-Molina et al. 2006; Wong et al. 1995), since a complementary study indicated that M2 metabolite, but not vinclozolin, was recovered in the plasma and milk of lactating mothers (Auger et al. 2010). In contrast, because of a specific estrogen-like response, the time of VO was not affected by vinclozolin alone, as it was by anti-androgenic compounds (Matsuura et al. 2005; Mylchreest et al. 1998), whereas estrogenicity mediated by genistein was increased by concomitant exposure to vinclozolin, provoking slight estrogenic modifications at the onset of puberty. Overall, our results showing endocrine effects on AGD and time of VO, coupled with our previous finding concerning the female mammary gland (El Sheikh Saad et al. 2011) and male reproductive organs (Eustache et al. 2009; Lehraiki et al. 2011), led us to speculate that these molecules could produce different and significant sex hormone–like effects at low doses, alone or in mixture, depending on the sex-dependent target. Consequently, because of sensitivity to genistein and vinclozolin exposure, SSG may be considered as an endocrine disruptor target for natural and synthetic xeno-hormones.
Similar to the mammary glands, salivary gland development is regulated by both estrogen and androgen, and maturity is reached at puberty (Gresik 1980). Sexual dimorphism of SSG, resulting in larger granular convoluted tubules in males compared to females (Rins de David et al. 1990), is observed in adult as well as in immature rodents (White and Mudd 1975). Estrogens and androgens are both involved in SSG postnatal morphogenesis, acting on the differentiation of proacini into mature acini and on the development of striated ducts that are the precursors of GCT cells in adult SSG (Jacoby and Leeson 1959). By six weeks of age (i.e., when the mammary gland reaches maturity), striated ducts decrease in size and become highly convoluted to further differentiate the GCTs (Gresik 1980).
In our study, because SSG morphogenesis was in process at PND35; striated ducts were less abundant than acini, as also previously reported (White and Mudd 1975); and the caspase-3 apoptose biomarker, which is highly expressed in PND21 SSG, normally disappeared at PND35 as a result of the normal decrease of apoptosis (Hayashi et al. 2000). Interestingly, genistein and vinclozolin acted similarly on immature female SSG development, decreasing the numbers of striated ducts and increasing their areas, and their combined exposure had a cumulative effect. The decrease in the number of striated ducts was coupled with a concomitant decrease of the Ki-67 proliferative marker. This decrease was particularly expressed in the GEN+VIN group. It was reminiscent of disorders in the maturation of the prepubertal salivary gland, confirming a disturbance in SSG development that seems to appear as a sex hormone–dependent response.
Submandibular salivary gland morphogenesis was similarly disrupted by genistein and vinclozolin, decreasing the numbers of striated ducts and increasing their areas, as estradiol does to adult submandibular GCTs (Flynn et al. 1983). Moreover, as an anti-androgenic response, a study in which male rats were castrated (Miyaji et al. 2008) found sexual dimorphism of SSGs abolished by decreasing both the number and area of GCTs. As a consequence, the histological aspect of SSG became similar to that of normal females, but in our study, vinclozolin did not decrease the area of striated ducts, suggesting a non-anti-androgenic effect of vinclozolin exposure. However, in young mice, neonatal castration abolished sexual dimorphism, and neonatal sex hormone substitutions showed the main involvement of testosterone, but not 17-β estradiol, in GCT development in both male and female SSG (Sawada and Noumura 1991). The functionality of both estrogen and androgen in female SSG development remains unclear, since estradiol, like androgens, could increase adult GCT areas in the case of high chronic 25 mg estradiol exposure (Flynn et al. 1983). In the same way, the synthetic estrogen-like compound Estren produced androgen-like effects on SSG morphology in ovariectomized adult female mice as well as in DERKO mice, indicating an independent ER pathway (Islander et al. 2005). These data support the hypothesis that female SSG could be sensitive to both estrogenic and anti-androgenic chemicals through the AR pathway. In addition, because uterine and lactational exposure to vinclozolin gives rise to two main metabolites (M1 and M2) that have a high affinity for the AR and to a lesser extent the ER (Molina-Molina et al. 2006; Wong et al. 1995), and since androgens stimulate perinatal GCT development in both male and female adult rats (Sawada and Nomoura 1991), our results could also reveal an androgenic response to vinclozolin. This hypothesis could explain the additive effect of the exposure to the genistein+vinclozolin mixture and the similar histologic defects observed in genistein and vinclozolin-treated rats.
In addition, our finding of stronger expression of AR than ERα in SSG of immature female rats and our failure to detect ERβ suggests a greater susceptibility of SSG development to androgen and anti-androgen chemicals than to estrogenic compounds. This must be considered as a consistent result indicating SSG as a singular AR-regulated organ in females.
Granular convoluted tubule maturation is strongly linked to growth factor secretions. Both TGFα and EGF are ligands of the EGF receptor that is involved in the regulation of branching morphogenesis, and growth factor secretions change with age. However, TGFα is highly secreted at birth and decreases over time, becoming very low at thirty-five days of age (Moji et al. 1995), and EGF and NGF secretions increase exponentially until 50 days of age, that is, when estrogenic and androgenic pubertal signals are taking place and GCTs reach maturity (Gresik 1980; Mogi et al. 1995). In our study, NGF and TGF-α were poorly expressed in the SSG of the control animals, confirming that maturation was approaching. Surprisingly, we found no effect of genistein on EGF, NGF, or TGFα mRNA expression, although estrogen greatly down-regulated EGF mRNA expression in SSG of adult ovariectomized mice (Tuomela et al. 1990). This lack of effect on growth factors could be a result both to the weak expression of ERs in the SSG of PND35 female rats and to the window and dose of exposure (gestation/lactation – 1mg/kg BW), since a three-day prepubertal exposure to 500mg/kg BW TEN down-regulated EGF mRNA expression in the mammary gland of fifty-day-old female rats (Brown et al. 1998).
As an anti-androgenic response, GCT areas as well as EGF and NGF expression in SSG were decreased by castration of male rodents (Hiramatsu et al. 1994; Humpel et al. 1993). In our study, despite an increase in the GCT areas, vinclozolin alone as well as the genistein+vinclozolin mixture strongly decreased TGFα and EGF/NGF expression, indicating a specific anti-androgenic action on all three growth factor mRNA transcripts by vinclozolin or its metabolites. In addition, androgens are self-producted by SSGs (Katsukawa et al. 1983). There might also be inhibition of 5 α-reductase by vinclozolin or its metabolites; our previous findings suggested this hypothesis, since in the neonatal testis, similar exposure to vinclozolin and genistein+vinclozolin inhibited the production of hydroxytestosterone, whereas genistein did not (Lehraiki et al. 2011).
Our findings reveal that the increase observed in the areas of immature GCT (striated ducts) is not linked to an increase in their secretions. Whereas the striated ducts could be enlarged as an effect of targeted morphogenesis, the reduced EGF and NGF mRNA levels in treated groups could reflect a response of the submandibular secretion process, probably by an independent mechanism. Such a hypothesis about independent regulation pathways in development and in the secretion process has already been proposed by Siminoski et al. (1993), who have shown that NGF and EGF secretions are controlled by a similar molecular mechanism that is independent of the growth of GCT cells. Therefore, we suggest that in our conditions, genistein and vinclozolin could affect growth factor synthesis and GCT morphogenesis by independent pathways. This suggestion could be supported by the fact that an early exposure to low doses of genistein/vinclozolin mixture in the testis has been shown to act specifically on gene clusters that are not affected by the chemicals alone at the same low dose (Eustache et al. 2009).
Thus, the similar decrease in TGFα and EGF/NGF expression in the present study indicates a specific anti-androgenic action of vinclozolin, whereas the similar effect of genistein and vinclozolin on the enlargement of striated ducts appears as an estrogen-like effect on morphogenesis, as also observed in the male reproductive tract.
In summary, this study reveals for the first time the morphogenic and endocrine impacts of gestational and lactional exposure of dams to endocrine disruptors on the SSG development of female offspring and underlines the additive or synergic effects of estrogen-antiandrogen EDC mixtures. These findings were obtained for a low dose of genistein, close to normal human dietary exposure, and for a low dose of vinclozolin, below the NOAEL but notably higher than estimated levels in humans. Since SSGs are the major source of circulating EGF, this repression of EGF may be predictive of a disturbance later in adulthood. In this way, several studies using sialoadenectomy have demonstrated the role of SSG in the development of the mammary glands and lactation, in the development of the female reproductive tract, and in oral and systemic health. However, new investigations are needed to describe the effect of endocrine disruptors on the differentiation mechanisms and signaling pathways of the SSG. Moreover, because of the higher sensitivity of males than females to estrogenic and anti-androgenic–like chemicals, we are now investigating the effects of the same exposure morphology and functions of male SSGs.
Footnotes
Acknowledgments
Thanks are due to Laurence Decocq, Patrick Tassin, and Luc Perepelkine for their technical assistance in the animal facility at INRA Dijon; to Anne Hillenweck (INRA Toulouse) for preparing and providing vinclozolin; to Claire Chabanet for statistical assistance; to Richard Gunton for English revision; and to Pr. Y. Artur for discussions.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the French Programme on Endocrine Disruption (PNRPE; contract MEDD CV 05147), the Tunisian Ministry of higher Education and Scientific Research, INRA, and grants from the Burgundy regional government.
