Abstract
The rhesus macaque (Macaca mulatta) is used extensively in translational biomedical research and drug development studies and is an important model of aging. Macaques often develop myocardial fibrosis with age, which can result in the loss of normal cardiac architecture with the expansion of the extracellular matrix and deposition of collagen. The etiology and pathogenesis of this pernicious process is poorly understood. Cardiac fibrosis was assessed using histologic and immunohistochemical techniques in cardiac tissue sections from 34 rhesus macaques. Overall left ventricular and left ventricular mid-myocardial interstitial/perivascular fibrosis were positively correlated with age (r = .6522, p < .0001 and r = .4704, p = .005, respectively). When divided into young (mean = 2.8 years), middle-aged (mean = 17.5 years), and advanced age (mean = 29.2 years) groups, immunophenotypic characterization of antigen presenting cells revealed differential expression of CD163 and DC-SIGN between the young and middle-aged groups compared to the advanced age group (p < .0001). HAM-56 expression decreased significantly in the advanced age cohort (p = .0021). The expression of CD8, CD163, and DC-SIGN correlated positively with age (r = .3999, p = .0191; r = .5676, p = .0005; r = .5245, p = .0014, respectively). These results show the importance of myocardial fibrosis as a common age-related pathology and additionally, alterations in T cell, macrophage, and dendritic cell phenotype in rhesus macaque myocardium are associated with age but unassociated with the fibrosis.
Introduction
Fibrotic cardiac remodeling and increased collagen deposition is strongly associated with aging in humans (Roth et al. 2004). This is also the case in aging rhesus macaques, where there are close parallels to the physiological changes observed in aging humans. Additional similar cardiac pathologies include myocardial infarction, interstitial fibrosis, and congestive heart failure (Roth et al. 2004). Although rodents are extensively used as models for aging and cardiovascular diseases, research using rhesus macaques provides a model more phylogenetically similar to humans, thereby better elucidating the shared pathogenesis of numerous disease processes including cardiac fibrosis (Appt and Ethun 2010; Chamanza et al. 2006; Clarkson et al. 1994; Lowenstine 2003; Park et al. 2009; Pick, Janicki, and Weber 1989; Roth et al. 2004; Vidal et al. 2010; Weber et al. 1988; Zabka, Irwin, and Albassam 2009). Although rhesus macaques are a species used as animal models in safety studies, myocardial fibrosis has been poorly studied in this species (Lammey et al. 2008; Lowenstine 2003; Keenan and Vidal 2006; Chamanza et al. 2006; Zabka, Irwin, and Albassam 2009).
The rhesus macaque is a superior model for many aspects of biomedical research. One important area is normal aging, where the rhesus macaque has been studied from multiple perspectives. In addition, rhesus macaques are frequently used in preclinical pharmacology and toxicology studies for the assessments of cardiovascular safety of drugs intended for use in humans (Lowenstine 2003). Interpretation of the histological changes in the research animals involved in these studies requires that the pathologist be able to differentiate drug-induced histopathologic lesions in the heart from normal histomorphology, incidental, and/or background lesions (Chamanza et al. 2006; Zabka, Irwin, and Albassam 2009; Lowenstine 2003; Keenan and Vidal 2006).
Cardiac myocytes are surrounded and supported by an extracellular matrix (ECM) that is composed of cohesive glycoproteins, proteoglycans, growth factors, and proteases as well as structural proteins such as collagen (de Souza 2002; Krenning, Zeisberg, and Kalluri 2010; Kumar, Fausto, and Aster 2010). The ECM acts to distribute mechanical forces, helps to convey mechanical signals, and electrically separates the atria from the ventricles to provide appropriate cardiac performance (Krenning, Zeisberg, and Kalluri 2010). Excess ECM typically manifests as cardiac fibrosis in which there is robust accumulation of collagen expanding the extracellular matrix (Wei 2011; Wynn 2007; Wynn 2008). The majority of cardiac diseases are associated with some degree of fibrosis in the heart (Krenning, Zeisberg, and Kalluri 2010).
In general, two forms of cardiac fibrosis are described in humans. Reactive fibrosis occurs in response to chronic diseases such as systemic hypertension, myocarditis, and cardiac muscle hypertrophy. It serves as a mechanism to accommodate pressure overload and to maintain cardiac output (Weber et al. 1989; Krenning, Zeisberg, and Kalluri 2010; Anderson, Sutton, and Lie 1979; Allessie et al. 2005). In this phase of repair, interstitial fibrosis progresses without the loss of cardiomyocytes (Krenning, Zeisberg, and Kalluri 2010; Wynn 2008). Replacement fibrosis is the second phase of repair in which disease progression continues and myocytes are replaced by scars of fibrotic connective tissue in an effort to preserve the structural integrity of the heart (Allessie et al. 2005; Wynn 2008; Weber et al. 1989; Moriwaki et al. 2004). As a result of these fibrotic lesions in the myocardium, there is increased stiffness, diastolic dysfunction, reduction of electrical coupling between myocytes, and abnormalities in cardiac conduction (Krenning, Zeisberg, and Kalluri 2010; Lammey et al. 2008; Chen and Frangogiannis 2010; Goumans, van Zonneveld, and ten Dijke 2008).
Macrophages and dendritic cells produce cytokines that influence immune responses, serve as profibrotic mediators, and activate myofibroblasts (Frangogiannis 2008; Wynn 2008; Yearley et al. 2007; Krenning, Zeisberg, and Kalluri 2010). Myofibroblasts are the key cellular mediators of the extracellular matrix and contributors to fibrosis in the heart (Krenning, Zeisberg, and Kalluri 2010). The role that macrophages and other immune cells play in cardiac fibrogenesis in rhesus macaques is unknown. However, studies of fibrogenesis in myocardial infarction, experimentally induced hypertension, myocarditis, and normal aging conducted in humans, rats, mice, canines, and chimpanzees have implicated a number of inflammatory cells including T cells, macrophages, and fibroblasts in contributing to the process (Chen and Frangogiannis 2010; Dobaczewski et al. 2006; Ely et al. 2010; Frangogiannis et al. 2003; Yano et al. 2006; Yu, Horak, and Larson 2006).
Although inflammation typically precedes fibrosis, the results from numerous experimental models show that the amount of fibrosis is not necessarily linked with the severity of inflammation and that chronic inflammation does not always induce the deposition of extracellular matrix components such as collagen. These findings suggest that the mechanisms that regulate fibrogenesis are distinct from those that regulate inflammation (Wynn 2004).
The goals of the current study were to identify possible variations in T cell, macrophage, and dendritic cell immunohistophenotypes in myocardial tissue and to determine if these alterations corresponded to the development of cardiac fibrosis in normal aging rhesus macaques.
Materials and Methods
Study Design Groups
Formalin-fixed, paraffin-embedded sections of left ventricle from 34 rhesus macaques (Macaca mulatta) from the pathology archives of the New England Primate Research Center were assessed. Rhesus macaques reach puberty at an age of approximately 3 years, and the expected lifespan is between 25 and 30 years of age. On this basis, the subjects were placed into three separate cohorts. Eleven animals were from a group of juvenile macaques from 2 and a half to 3 years of age (mean = 2.8 years). Twelve animals were middle-aged adult macaques, 14 to 23 years old (mean = 17.5 years). Finally, a group of 11 macaques, ranging from 26.5 to 31.5 years old, comprised the advanced age cohort (mean = 29.2 years). The animals in the young cohort were juveniles that served as control animals in an HIV vaccination study. They were euthanized humanely at the end of the study. There were no significant gross findings. The members of the middle-aged cohort also served as control animals in a simian immunodeficiency virus (SIV) vaccine study or were part of a normal aging study. These animals were also euthanized at the termination of the study. No clinical abnormalities were noted in these animals; however, incidental comorbid findings revealed at necropsy or during the subsequent histological examination included mild myocarditis, Pneumonyssus simicola bronchiectasis and bronchitis, mild vaginitis, mild endometritis, mild hepatitis, typhlocolitis, mild glomerulonephritis, and gastritis. The macaques that composed the advanced age cohort were part of a normal aging study that investigated behavioral and cognitive changes. Comorbidities discovered at necropsy or subsequently at histological examination included benign and malignant neoplasms of the intestine, ovary, uterus, adrenal gland, stomach, or pituitary gland. These animals were not known to have received any compounds or drugs. All subjects were negative for SIV and were confirmed to be tuberculosis negative with quarterly intradermal testing.
All the animals included in this study were cared for in accordance with the National Research Council’s Guide for the Care and Use of Laboratory Animals and the standards of the Harvard Medical School Standing Committee on Animals and the Association for the Assessment and Accreditation of Laboratory Animal Care.
Histological Staining
All animals were necropsied within 24 hours of death; and representative sections of all major organs were collected, fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 5 μm, and stained using hematoxylin and eosin (HE). Left ventricular free wall sections from each subject were examined for this study. Additional sections of left ventricular free wall were stained with Masson’s trichrome stain.
Immunohistochemistry
Standard immunoperoxidase staining for CD3, CD8, CD68, CD83, CD163, HAM-56, and DC-SIGN was performed on formalin-fixed, paraffin-embedded sections of left ventricular free wall from each subject (Table 1) (Yearley et al. 2007). CD3 and CD8 were used to identify T cells. CD68, CD83, CD163, HAM-56, and DC-SIGN were used to identify antigen presenting cells. All sections were deparaffinized and rehydrated, followed by blocking with 3% hydrogen peroxide in phosphate buffered saline (PBS). A wash of tris-buffered saline followed each step. Following pretreatment, an avidin-biotin block (Invitrogen Corporation, Frederick, MD) and then a Dako Protein block (Carpinteria, CA) were conducted on all sections.
Antibody source, dilution, and antigen retrieval for immunohistochemistry protocols.
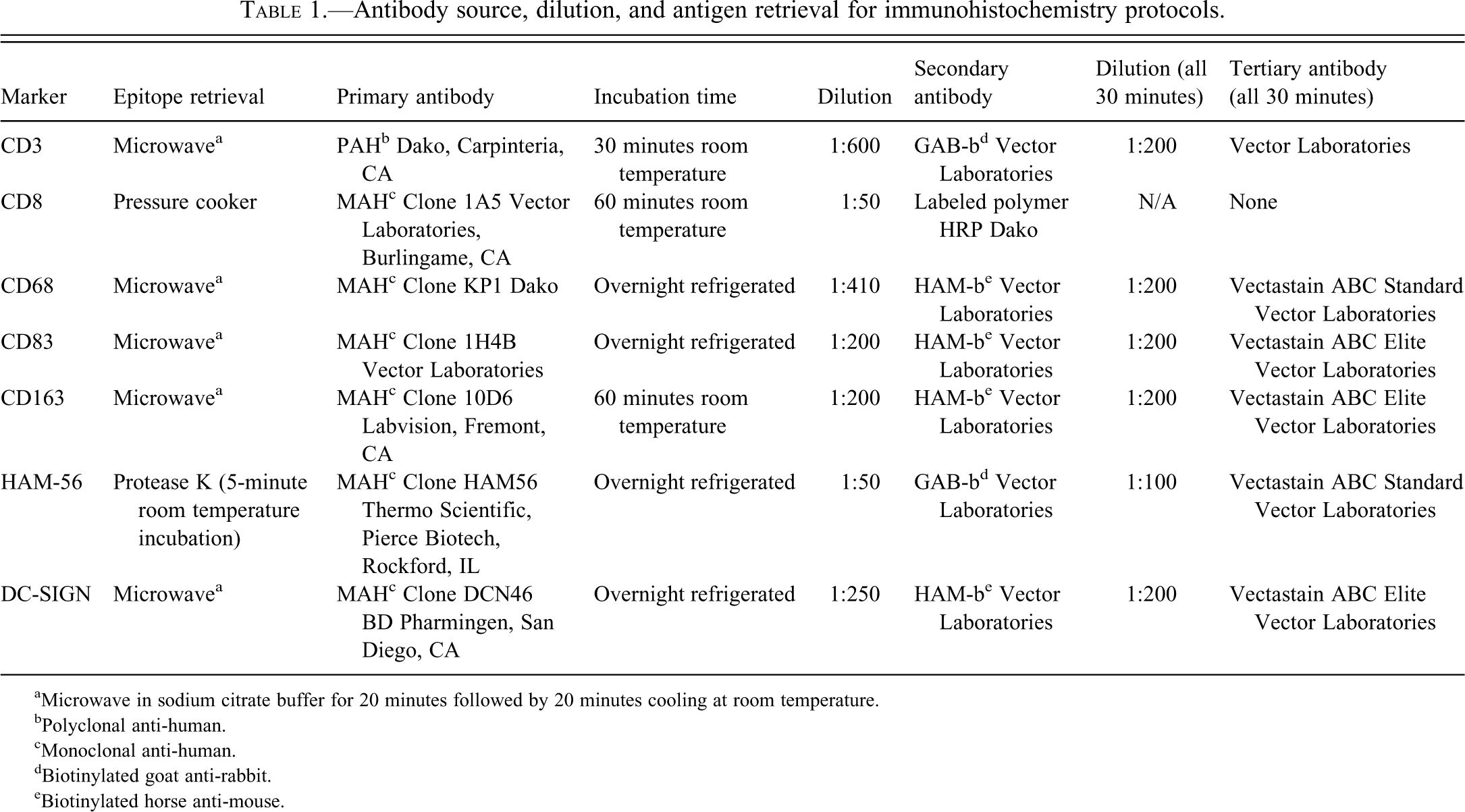
aMicrowave in sodium citrate buffer for 20 minutes followed by 20 minutes cooling at room temperature.
bPolyclonal anti-human.
cMonoclonal anti-human.
dBiotinylated goat anti-rabbit.
eBiotinylated horse anti-mouse.
Antigen-antibody complex formation was detected using diaminobenzidine (DAB: DakoCyomation, Carpineteria, CA). Irrelevant, isotype-matched primary antibodies were used in place of the test antibody as negative controls in all immunohistochemical studies. Positive control tissues consisted of rhesus macaque lymph node (CD3, CD8) and spleen (CD68, CD83, CD163, DC-SIGN, and HAM56).
Image Analysis
All immunohistochemically labeled sections were examined with an Olympus BX41 light microscope (Olympus Corporation, Shinjuku, Tokyo, Japan). The total number of DAB stained cells was counted in 10 random high-magnification fields (400×) for each section.
Masson’s trichrome stained whole slides of left ventricular free wall were scanned using a Nikon Super Coolscan 5000 slide scanner (Nikon Inc., Chiyoda-ku, Tokyo, Japan) at a resolution of 4,000 pixels per inch and a color depth of 8 bits per channel. Pixels were classified as fibrosis (blue), cardiac muscle (red), or background (white) by means of a custom image analysis algorithm. Briefly, the RGB color values of each pixel were considered as an absorbance vector originating at <255, 255, 255> corresponding to zero absorbance (white background). Representative absorbance vectors for blue stain wavelengths, <140, 111, 78>, and red stain wavelengths, <42, 206, 126>, were defined by averaging the values of several manually selected pixels of each color. Pixels were classified as fibrosis, muscle, or background based on the least angle between their absorbance vectors and the representative vectors multiplied by a tolerance factor. The correctness of the classification process was confirmed by comparison of an algorithmically generated red/green pseudo-fluorescent image with the original scan. Percentage fibrosis in each section is reported as the number of blue pixels divided by the total of red and blue pixels. Overall fibrosis of the left ventricular sections was calculated for each animal. Overall left ventricular fibrosis was defined as any and all fibrosis evident by Masson’s trichrome staining in the entire tissue section analyzed.
Ten photomicrographs of the mid-myocardium in each of the Masson’s trichrome stained sections were randomly taken at 100× using Olympus DP2-BSW microscope digital camera software (Olympus Corporation). The endocardial, subendocardial, and epicardial areas were avoided to ensure that the photomicrographs captured interstitial and perivascular fibrosis of the mid-myocardial region. Areas of fibrosis were neither intentionally nor unintentionally captured for analysis. Percent fibrosis was determined as above.
Statistical Analysis
The Kruskal-Wallis and Dunn’s multiple comparison tests were used to calculate the significance of fibrosis (overall left ventricular, interstitial/perivascular) among the cohorts. Comparisons between fibrosis (overall left ventricular, interstitial/perivascular) and age were determined using nonparametric Spearman correlation.
The significance of the differences in IHC staining among the three cohorts was determined using one-way ANOVA with Tukey posttest. Correlations between expression of the T cell, macrophage, and dendritic cell markers (CD8, CD163, and DC-SIGN) and age were determined assuming data were sampled from Gaussian populations (Pearson correlation).
For all statistical tests, p ≤ .05 was considered significant. Statistical analysis was performed using GraphPad Prism (GraphPad Software, Inc., La Jolla, CA).
Results
Myocardial Fibrosis Increases with Age
Percentage overall left ventricular fibrosis increased from 0.54% in the youngest cohort (mean age = 2.8 years) to 2.1% in the middle-aged cohort (mean age = 17.5 years) and to 4.6% in the advanced age cohort (mean age = 29.2 years; Figures 1 and 3A). There was a significant increase in fibrosis between the young cohort and both the middle-aged adult cohort and advanced age cohort (p = .0001) but no significant difference in the amount of overall left ventricular fibrosis between the middle-aged cohort and the advanced aged cohort. A strong positive correlation, however, was noted between percent overall left ventricular fibrosis and age (r = .6522, p < .0001).
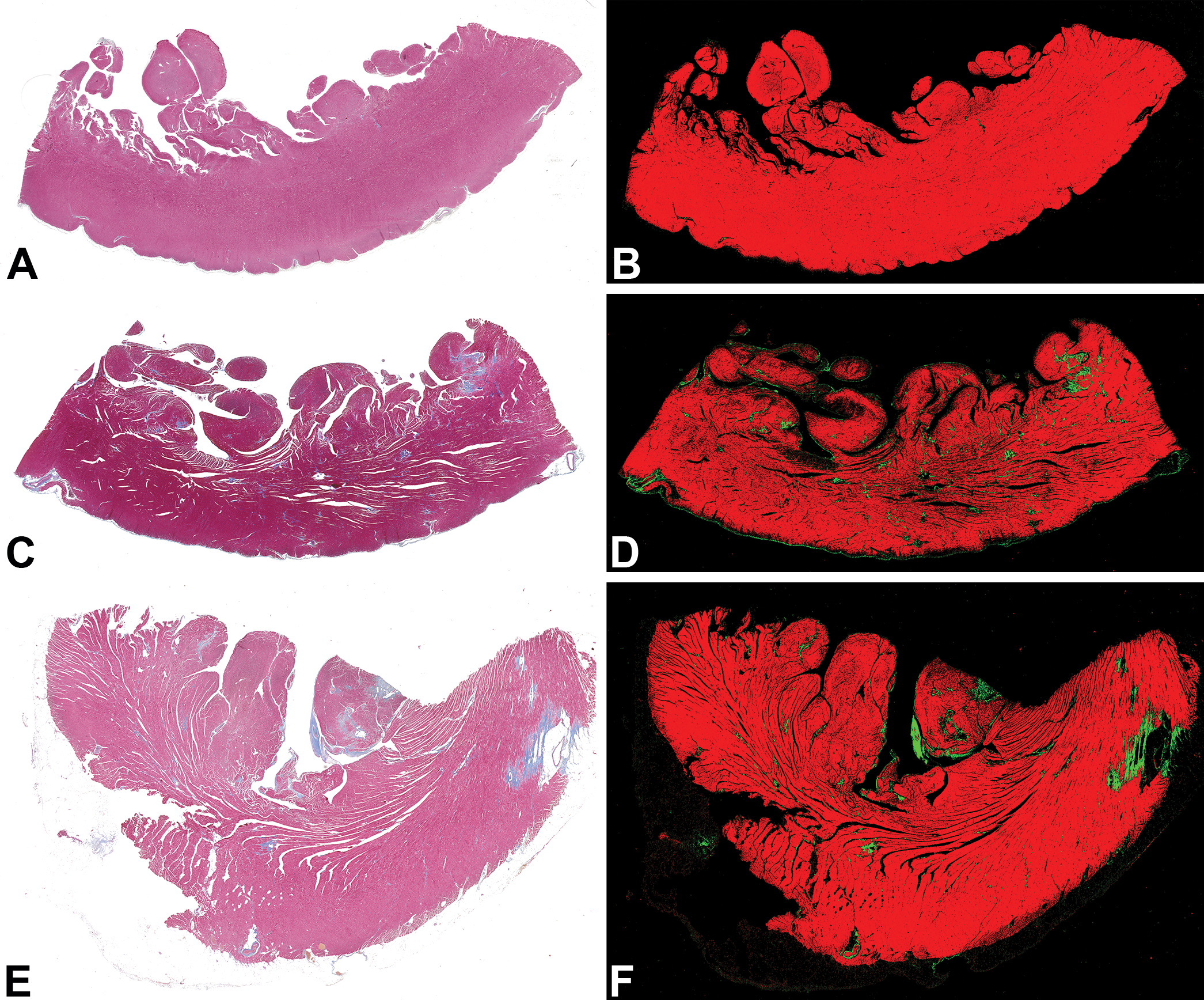
Myocardium. Scanned slides of left ventricular free wall from each cohort. Masson’s trichrome. Algorithmically generated red/green pseudo-fluorescent image of original scan. (A, B) Left ventricular free wall of a subject from the young cohort. (C, D) Left ventricular free wall of a subject from the middle-aged cohort. (E, F) Left ventricular free wall of a subject from the advanced age cohort.
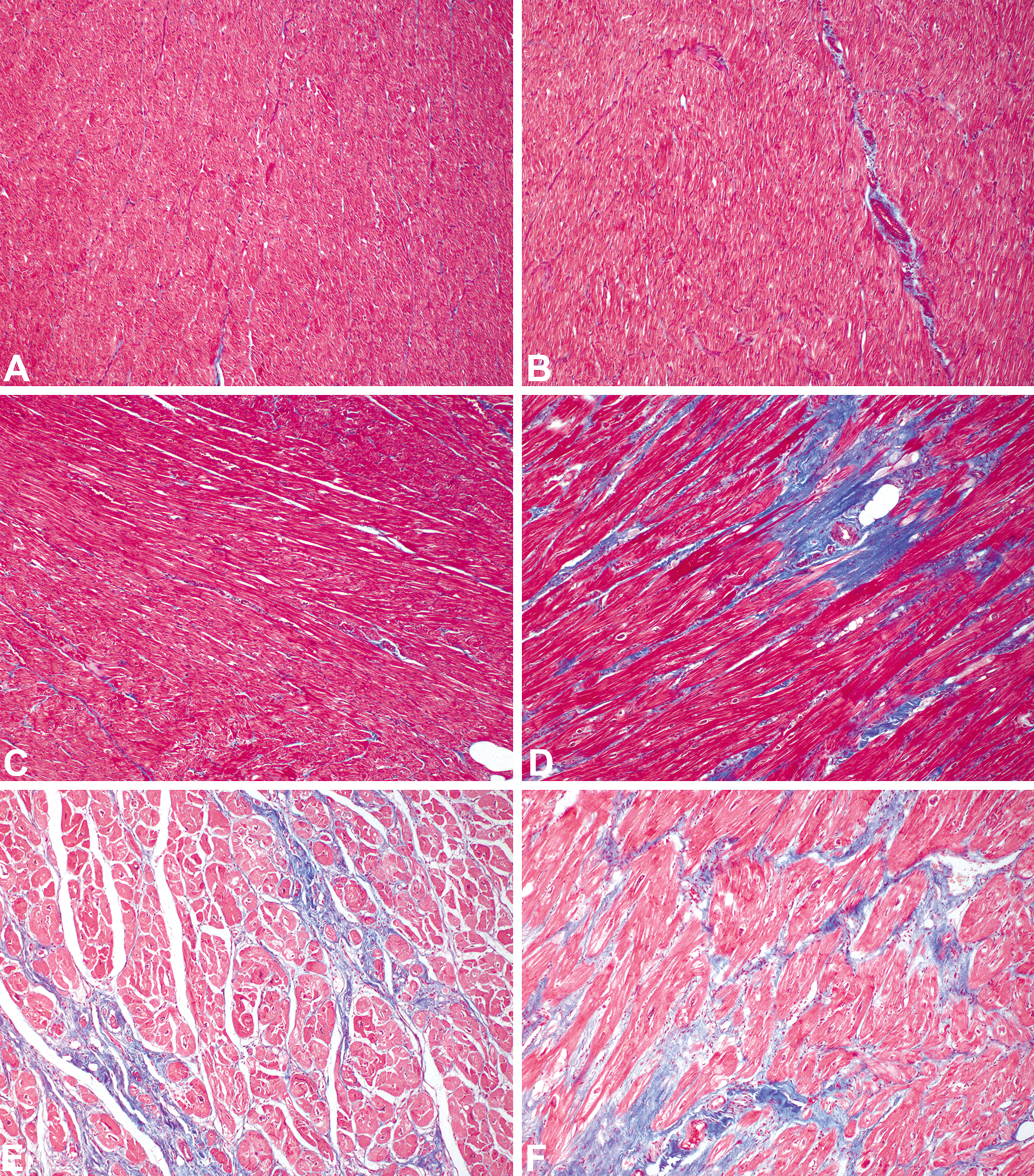
Myocardium; representative left ventricular, mid-myocardial interstitial and perivascular fibrosis photomicrographs at 100× from each cohort. Masson’s trichrome. (A) Interstitial connective tissue from a subject in the young cohort. (B) Perivascular connective tissue from a subject in the young cohort. (C) Interstitial fibrosis from a subject in the middle-aged cohort. (D) Perivascular fibrosis from a subject in the middle-aged cohort. (E) Interstitial fibrosis from a subject in the advanced age cohort. (F) Perivascular fibrosis in a subject from the advanced age cohort.
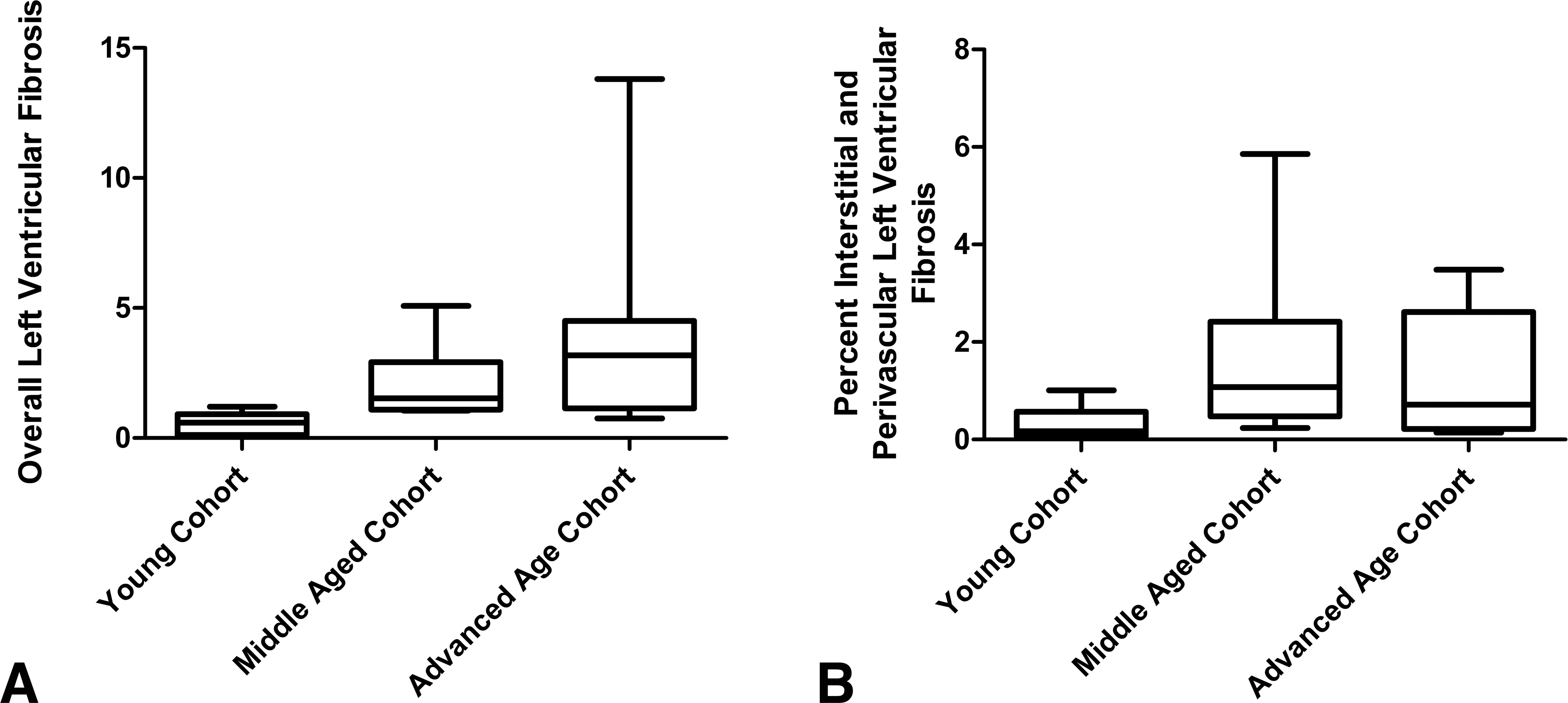
Fibrosis among study cohorts Kruskal-Wallis test with Dunn’s posttest: (A) The degree of overall left ventricular fibrosis between groups was statistically significant (p = .0001). (B) The degree of interstitial and perivascular left ventricular fibrosis between groups was statistically significant (p = .0059).
There was a significant increase in perivascular and interstitial fibrosis in the mid-myocardial region of each left ventricular section between the young cohort (0.33%) and both the middle-aged adult cohort (1.7%) and the advanced age cohort (1.5%; p = .0059; Figures 2 and 3B). Furthermore, the percent interstitial and perivascular fibrosis was positively correlated with age (r = .4704, p = .005).
Myocardial T Lymphocytic Infiltrates Increase with Age
The number of CD3 cells increased significantly from the young cohort as compared to the middle-aged cohort (p = .0105; Figure 4A). Expression of CD8 increased most significantly between the young cohort as compared to the advanced age cohort (p = .0199; Figure 4B). Cells expressing CD8 also significantly correlated with increasing age (r = .3999, p = .0191; Figure 6A).
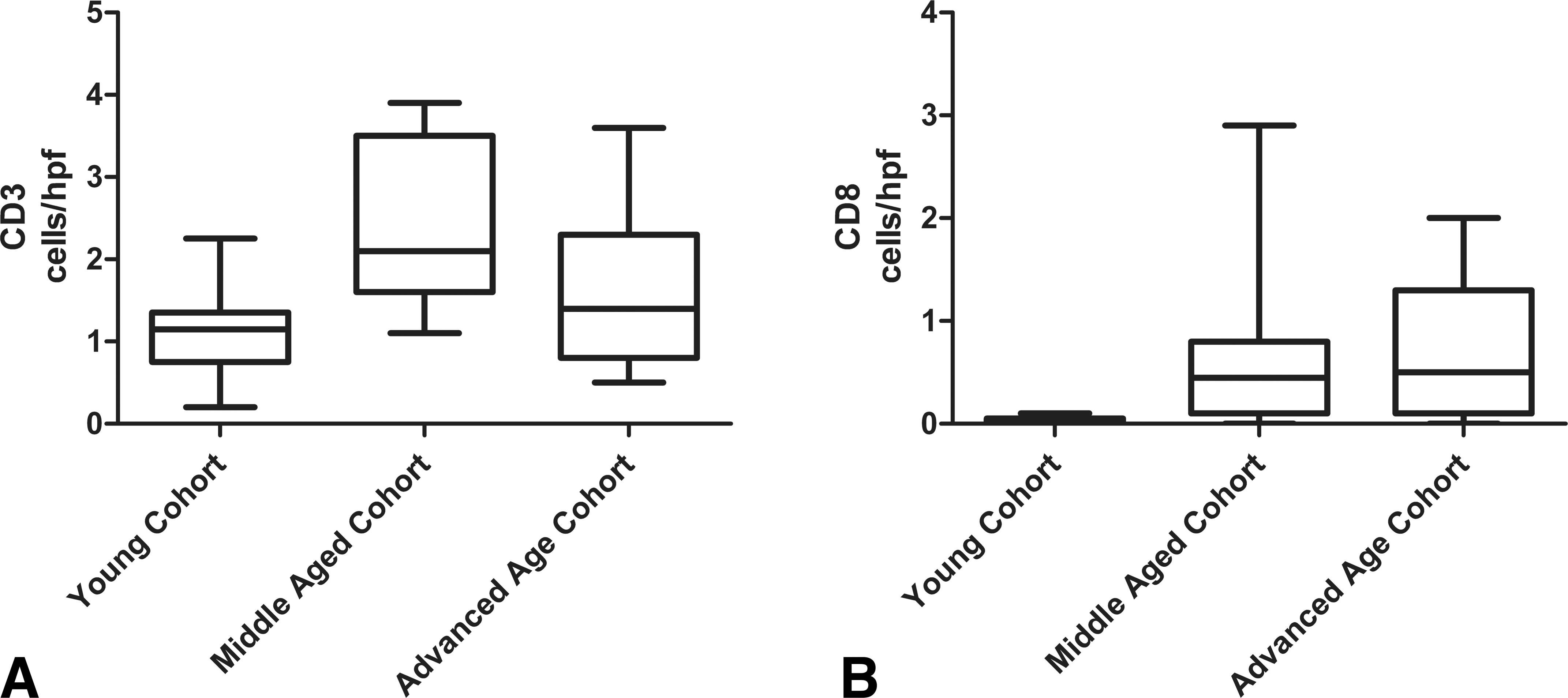
T cell counts across cohorts. (A) CD3+ T cell counts in the young cohort were significantly different from those in the middle-aged cohort (p = .0105). (B) CD8+ T cell counts were also significantly different between the young cohort and the middle-aged cohort. The advanced age cohort also had significantly higher numbers of CD8+ T cells when compared to the young cohort (p = .0199).
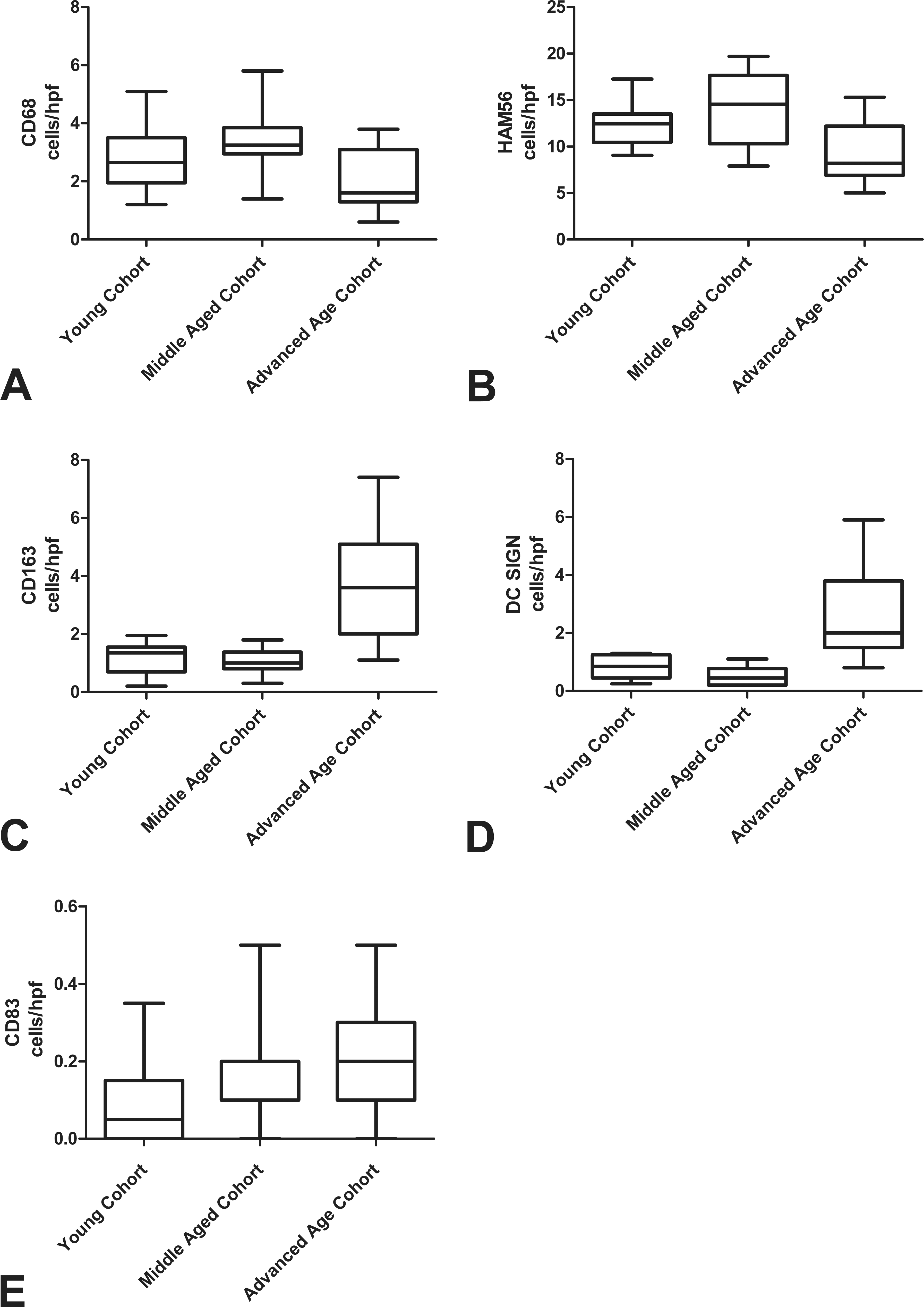
Antigen presenting cell counts across cohorts. (A) The middle-aged cohort had significantly higher numbers of CD68+ cells than the advanced age group (p = .0116). (B) The number of HAM56+ cells decreased significantly in the advanced age cohort as compared to the middle-aged cohort (p = .0045). (C) A significant increase in CD163+ cells occurred in the advanced age cohort compared to the two younger cohorts (p ≤ .0001). (D) DC-SIGN+ cells occurred in low numbers in the two younger cohorts as compared to the advanced age cohort (p ≤ .0001). (E) There was no significant variation in the numbers of CD83+ cells across all three cohorts.
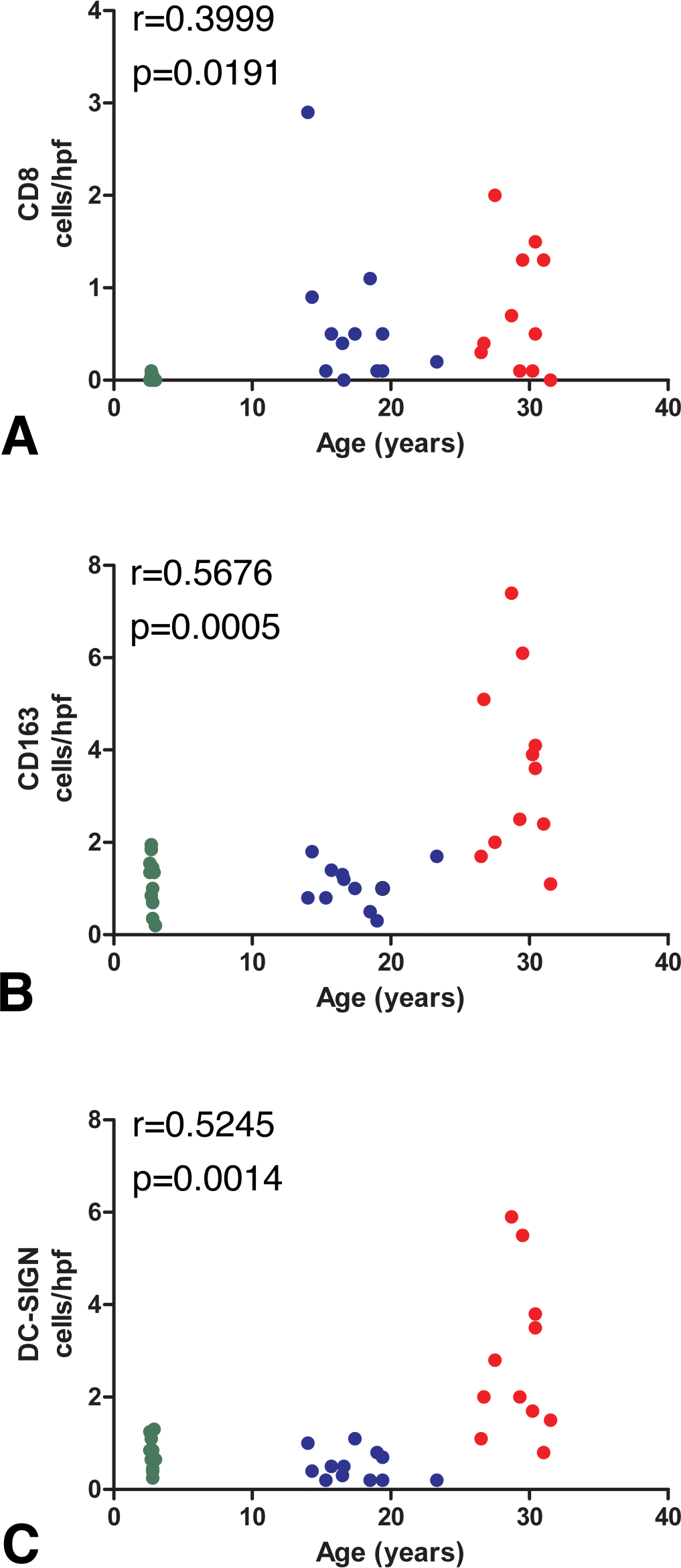
Pearson correlation of CD8+, CD163+, and DC-SIGN+ cell numbers with age in the myocardium of normal rhesus macaques (A, B, C). Age was positively correlated with CD8, CD163, and DC-SIGN.
Alterations in Antigen Presenting Cell Immunophenotype Occur across Age Groups
The most significant change in the number of CD68 cells was the decrease between the middle-aged cohort and the advanced age cohort (p = .0116; Figure 5A). Macrophages expressing HAM-56 were significantly decreased between the middle-aged cohort and the advanced age cohort (p = .0045; Figure 5B). Cells expressing the macrophage marker, CD163, significantly increased in number from the young and middle-aged groups to the advanced age group (p < .0001; Figure 5C). CD163 cell counts were positively correlated with age (r = .5676, p = .0005, Figure 6B).
Dendritic cells expressing DC-SIGN increased significantly in number from the young and middle-aged groups to the advanced age group (p < .0001; Figure 5D). DC-SIGN+ cell numbers also significantly correlated with increasing age (r = .5245, p = .0014; Figure 6C). The expression of dendritic cell marker, CD83, was similar across all age groups (Figure 5E).
Immunophenotypic Markers Do Not Correlate with Myocardial Fibrosis
The expression of CD3, CD8, CD68, CD163, HAM-56, and DC-SIGN differed significantly between select cohorts. The correlation of HAM-56+ cells to percent interstitial and perivascular fibrosis approached significance (r = .301, p = .0837); however, none of the other immunohistophenotypic markers showed significant correlations with overall left ventricular or interstitial and perivascular fibrosis.
Discussion
Overall Left Ventricular and Interstitial/Perivascular Fibrosis
Myocardial fibrosis in the aged human population is a chronic and widespread pathological finding (Anderson, Sutton, and Lie 1979; Chen and Frangogiannis 2010; Krenning, Zeisberg, and Kalluri 2010). A number of animal models including mice and rats provide strong evidence that the development of cardiac fibrosis is associated with aging (Cieslik et al. 2011; Boyle et al. 2011; de Jong, van Rijen, and de Bakker 2011; Debessa and de Souza 2001). To our knowledge, this is the first study to assess the degree of cardiac fibrosis over various age groups in rhesus macaques. Because there is often a link between fibrogenesis and alterations in local inflammatory cells, we also used immunohistochemistry to identify and enumerate the local tissue T cell, macrophage, and dendritic cell population variations across three age groups of normal rhesus macaques to determine if there was an association with the fibrosis (Yearley et al. 2007; Wynn 2008; Yearley et al. 2008; Wei 2011).
Our work indicates that cardiac fibrosis increases with age and is a common finding among normal rhesus macaques. Although the evaluated population of myocardial immune cells altered across the age groups, there was no direct correlation with the degree of cardiac fibrosis.
T Cells
CD3 is a cell surface marker that identifies T cells, while CD8 is specific for the cytotoxic phenotype (Leong and Leong 2003). T lymphocytes are thought to affect the ECM by modulating collagen synthesis (Yu, Horak, and Larson 2006). A number of studies involving patients with heart disease as well as those in animal models have shown that infiltration of T lymphocytes in the myocardium is common (Wynn 2007; Kvakan et al. 2009; Yu et al. 2010; Vidal et al. 2010). While no direct correlation was observed between CD3 or CD8 density and measurements of overall left ventricular and interstitial fibrosis, positive correlations were observed between these markers and age. The presence of T lymphocytes indicates chronicity of inflammation. However, given that the degree of fibrosis does not correlate with our measures of CD3 and CD8 expression, this suggests that fibrogenesis may be driven by factors separate from those regulating chronic inflammation (Wynn 2008).
Antigen Presenting Cells
Macrophages, along with T lymphocytes, have been implicated in the alteration of the ECM leading to cardiac fibrosis in humans and animal models (Frangogiannis 2008; Yu et al. 2010; Krenning, Zeisberg, and Kalluri 2010; Wynn 2008; Yearley et al. 2007; Hinglais et al. 1994). CD68 is a classic macrophage marker. HAM-56 is an uncharacterized antigen that labels a large subset of monocyte-macrophage lineage cells and is conventionally used to identify macrophages in tissues (Yearley et al. 2007). CD163 is a marker for macrophages with anti-inflammatory phenotypes (Yearley et al. 2007). Dendritic cells are antigen presenting cells that play a central role in the adaptive immune response and have unique migratory properties that separate them from macrophages (Randolph, Ochando, and Partida-Sanchez 2008). DC-SIGN is a marker for the dendritic cell specific C type lectin that mediates adhesion to T cells (Geijtenbeek et al. 2000).
Our findings show that there is significant alteration of the immune cells expressing CD68, CD163, HAM-56, and DC-SIGN between the different cohorts. These alterations indicate that the local populations of these cells differ depending on the age of the animal and may indicate that the biological role of these cells evolves with age. These alterations may also shape the character of the immune response (Mosser 2003). It has been proposed that activated macrophage populations switch phenotypes over time responding to environmental signals (Mosser and Edwards 2008). This is consistent with our results showing a marked plasticity to the antigen cell populations as rhesus macaques age.
In conclusion, there is a direct correlation between aging in macaques and fibrogenesis in the myocardium. Myocardial fibrosis is a common background lesion in aged rhesus macaques and must be considered when assessing drug-induced cardiovascular lesions in preclinical toxicological studies (Lowenstine 2003; Chamanza et al. 2006; Qureshi 1979). In addition, there are striking differences in the immune cell phenotypes across three age groups with some phenotypes (CD8, DC-SIGN, and CD163) being directly correlated with age. T cells, macrophages, and dendritic cells may play distinct and evolving roles with age, which may alter myocardial disease susceptibility and suggest that the mechanisms that drive fibrogenesis may be distinct from those that are regulating chronic inflammation (Wynn 2004, 2008).
Footnotes
Abbreviations
Acknowledgment
We would like to thank Kristen Toohey for assistance with graphics.
