Abstract
Adult male and female rat hepatocytes were individually transplanted into the spleens of adult male and female rats. The recipients were euthanized at either eight, sixteen, thirty, or forty-five weeks following transplantation, at which time hepatic and splenic levels of liver-specific rat albumin mRNA as well as sex-dependent transcript levels of CYP2C11, -2C12, -2C7, -2A1, and -3A2—which accounts for > 60% of the total concentration of hepatic constituent cytochrome P450—were determined. Whereas the pre-infused hepatocytes expressed their expected cytochrome P450 sexual dimorphisms (female-specific CYP2C12, male-specific CYP3A2, and female-predominant CYP2A1), their post-transplantational competence now reflected the sexual dimorphisms of the recipient (as observed in the host’s liver), which supports the concept that the sex-dependent growth hormone circulating profiles are the determinants regulating the expression levels of hepatic cytochrome P450. Also expressed at normal concentrations in the pre-infused hepatocytes, male-specific CYP2C11 and female-predominant CYP2C7 were inexplicably undetectable in the spleens of both recipient males and females, regardless of the sex of the donor hepatocytes, almost one year after transplantation.
Keywords
Introduction
Orthotopic liver transplantation is often the only therapeutic response for the treatment of several liver diseases, for example, hepatic cirrhosis, fulminant hepatitis, and several lethal hereditary enzyme deficiencies. Although the procedure is now routine, it is not without its drawbacks, including post-transplantation complications as well as a shortage of donors. Accordingly, alternative procedures have been investigated to support liver function. Among these procedures is the transplantation of isolated hepatocytes to various systemic sites. Numerous sites have been examined, including fat pads, muscle, subcutaneous tissue, peritoneum, lungs, kidney, liver, and spleen (Balladur et al. 1994; Fuller 1988; Rivas et al. 1992; Sandbichler et al. 1992; Selden et al. 1991). In spite of the disadvantages of the intrasplenic site, such as the need for syngeneic hepatocytes, which would otherwise require immunosuppressive therapy, and its size limitation, which allows the spleen to accommodate hepatic tissue amounting to only about 3–4% of normal liver weight, the spleen has been shown to be a highly effective site, at least in experimental animals (Dixit 1995). The efficacy of intrasplenic hepatocyte transplants in rats with fulminate hepatitis induced by D-galactosamine (Sommer et al. 1979), carbon tetrachloride (Ochenashko et al. 2006), and portacaval anastomosis plus 70% hepatectomy (Cuervas-Mons et al. 1984) was demonstrated by an increase in survival of 20–80%, 100%, and 10–60%, respectively. Moreover, intrasplenic hepatocyte transplants were able to supply catalase in a strain of acatalasemic mice (Sutherland et al. 1980) and reduce both serum bilirubin in the Gunn rat (Vroemen et al. 1986; Woods and Parbhoo 1981) and cholesterol plasma levels in the Watanabe rabbit (Tejera et al. 1992; Wiederkehr et al. 1990).
The liver contains many vital enzyme systems, of which the cytochrome P450 (CYP) system is one. The mammalian CYP superfamily is involved in the synthesis and/or metabolism of prostaglandins, eicosanoids, cholesterol, bile acids, steroid and thyroid hormones, vitamins A and D, biogenic amines, neuroamines, and fatty acids. In addition, the enzymes are the body’s first line of defense in metabolizing pharmaceuticals, foreign chemicals, and pollutants. Whereas humans, rats, and mice each express about fifty to seventy different CYPs in various organs, it is the hepatic isoforms that account for the bulk of the CYP enzymes (Guengerich 1997; Nelson et al. 2004). Moreover, hepatic CYPs are generally expressed at sexually dimorphic levels, although to different extremes depending on the species (Dhir et al. 2006; Shapiro et al. 1995; Waxman and Holloway 2009). In spite of the importance of the CYP enzymes system, there remains a paucity of studies examining its expression in isolated hepatocyte transplants, and in particular, intrasplenic transplants. A few reports have investigated the long-term (approximately one year) expression of CYPs in intrasplenic hepatocyte transplants derived from mixed-sex fetal liver. In these studies, Western blotting, immunohistochemistry, and CYP-dependent monooxygenase activities have indicated the persistent expression of several fetal CYP isoforms, a few minor adult isoforms, and the responsiveness of inducible CYPs to induction by dexamethasone, phenobarbital, and/or β-naphthoflavone (Kato et al. 1994; Lupp et al. 2003; Lupp et al. 2004). In addition, isolated adult hepatocytes transplanted into the spleens of syngeneic Fischer 344 rats survived for at least fifteen months and maintained a responsiveness of their inducible CYP isoform (measured at the mRNA level) to phenobarbital induction (Maganto et al. 1990). To date, however, all of the CYP isoforms measured in the intrasplenic hepatocyte transplants account for only a very small percentage of the total adult constituent pool. In contrast, the expression of CYP2A1, -2A2, -2C6, -2C7, -2C11, -2C12, -2C13, -2E1, and -3A2, which accounts for more than 90% of the total hepatic CYP in adult male or female rats (Ryan and Levin 1993), has not been measured in intrasplenic hepatocyte transplants. In the present study, we have investigated the sexually dimorphic regulation of five constituent hepatic CYPs, which account for > 60% of the total CYP content in male and female rat liver (Ryan and Levin 1993) in intrasplenic hepatocytes derived from adult male and female rats and transplanted into adult male and female rats.
Material and Methods
Animals
Animals were housed in the University of Pennsylvania Laboratory Animal Resources facility, under the supervision of certified laboratory animal medicine veterinarians and were humanely treated according to a research protocol approved by the university's Institutional Animal Care and Use Committee. Rats were housed in plastic cages with corn cob (0.3 cm) bedding changed thrice weekly and maintained at 21°C–23°C and ∼40% relative humidity with a twelve-hour light/twelve-hour dark cycle. Animals were fed commercial rat chow and purified tap water ad libitum. Following ten days of acclimation, all Fischer 344 inbred rats (males and females, recipients and donors) were nine weeks of age at the time of intrasplenic hepatocyte transplantation. Male and female recipients were implanted with viable hepatocytes from either same- or opposite-sex rats and euthanized eight, sixteen, thirty, or forty-five weeks post-transplantation. Each donor rat supplied sufficient cells to implant four rats of the same or opposite sex representing the four euthanasia periods. That is, hepatocytes from the same male donor were infused into the spleens of four different recipient males, of which one was to be euthanized at eight, sixteen, thirty, and forty-five weeks post-transplantation. Cells from another male were infused into four females, one of which was euthanized at one of the four time periods. Hepatocytes harvested from female donors were similarly distributed to male and female recipients. Each treatment group contained eight rats to ensure that at least six rats (the number used in the study) contained viable intrasplenic hepatocytes for analysis. Spleens of sham rats (i.e., controls) were infused with hepatocyte buffer containing no cells. At the appointed times, the rats were decapitated, the livers and spleens were quickly removed and minced, and portions reserved for mRNA determinations were stored in RNA-Later (Invitrogen, Carlsbad, CA). In addition, a few random tissue samples were stored in 10% buffered formalin for subsequent histological preparation. The effectiveness of the procedure was verified at necropsy by the presence of obvious nodules in otherwise normal-appearing spleen, by random histological preparations demonstrating the presence of hepatocytes in the splenic pulp and by the expression of rat albumin mRNA in the treated spleens. All of these markers were negative in the spleens from sham rats.
Hepatocyte Isolation and Transplantation
Isolation of rat hepatocytes was performed with minor modifications (Garcia et al. 2001) by in situ perfusion of collagenase through the portal vein of ketamine/xylazine-anesthetized rats (Seglen 1976). In brief, initial perfusion with a calcium-free buffer was followed by infusion of a solution of collagenase (0.05% w/v). The “softened” liver was excised (rats exsanguinated), and the hepatocytes were separated from connective tissue by filtering through 100-µm macroporous filters (Spectrum Co., Laguna Hills, CA) and from nonparenchymal cells by repeated low-speed centrifugation in wash medium: high-glucose Dulbecco’s modified Eagle’s medium (DMEM) (4.5 g/L) containing streptomycin (100 µg/mL), penicillin (100 U/mL), gentamycin (50 µg/mL), fungizone (0.25 µg/mL), and Hepes (15 mM). The viability of the final cell suspension of hepatocytes was typically between 50% and 60% (trypan blue). (When compared with our previous reports [Thangavel et al. 2004; Thangavel and Shapiro 2007], the lower percent age viability obtained in the present study may be because of our omission of the Percoll gradient known to remove dead as well as small [i.e., immature] hepatocytes; the latter are considered important for colonizing the spleen [Rajvanshi et al. 1998]). An aliquot of isolated hepatocytes from every donor rat was stored for subsequent mRNA analysis of albumin and CYP isoforms.
Hepatocyte transplantation was carried out on laparotomized rats under inhalation anesthesia with isoflurane. The exposed spleen was kept moist throughout the procedure. A hepatocyte suspension containing 4 to 5 × 106 live cells in 0.3 mL of DMEM was very slowly injected into the splenic pulp through a 25-gauge needle. Hemostasis was controlled by compression of the injection site with a sterile cotton tip. Leakage from the site of injection and runoff into the portal vein were minimized by temporary occlusion of the vascular system at the splenic hilum. The entire procedure took no more than ten minutes.
RNA Analysis
Total hepatic and splenic RNA was isolated and prepared as previously described (Thangavel et al. 2004; Thangavel and Shapiro 2007). Basically, 10 µg (liver) or 30 µg (spleen) of RNA was electrophoresed under formaldehyde denaturing conditions on 1% agarose and transferred to Nytran nylon membranes (Schleicher and Scheull, Keene, NH). The Northern blots were probed and occasionally reprobed with 32P-labeled oligonucleotides by using hybridization and high-stringency washing conditions. The nucleotide sequence of oligonucleotide probes for CYP2A1, -2C7, -2C11, -2C12 (Waxman 1991), -3A2 (Ram and Waxman 1991), and rat albumin (Arosio et al. 1993) have been reported. The consistency of RNA loadings between samples was confirmed by ethidium bromide staining of 18S and 28S rRNAs and was verified with an 18S oligonucleotide probe (Ramsden et al. 1993). The hybridized mRNA signals were quantified by scanning the autoradiographs by using an Alpha Innotech FluorChem 8800 (Alpha Innotech, San Leandro, CA) and normalized to the 18S rRNA signals in each lane.
Histology
Light microscopy was performed on hematoxylin and eosin–stained sections of six spleens forty-five weeks after hepatocyte transplantation.
Statistics
All data were subjected to two-way analysis of variance (α = .05) demonstrating statistically significant interactions between groups, and differences were determined with t statistics and the Bonferroni procedure for multiple comparisons.
Results
Morphological Appearance of Intrasplenic Hepatocytes
The presented histological section (Figure 1 ) is that of a spleen from a male rat forty-five weeks after the infusion of donor female hepatocytes. The sex of the hepatocyte donors as well as the sex of the spleen recipients had no observable effect on the morphology of the intrasplenic hepatocytes forty-five weeks post-transplantation. Our limited number of samples showed hepatocytes restricted to the red pulp and often presenting as disoriented and abbreviated cordlike structures, inferior to cords observed in normal liver organized about a central vein. In addition, some areas contained unstructured masses of cells appearing as clumps. Histological sections of liver and control spleens, that is, infused with hepatocyte buffer alone, were unremarkable and have not been presented.
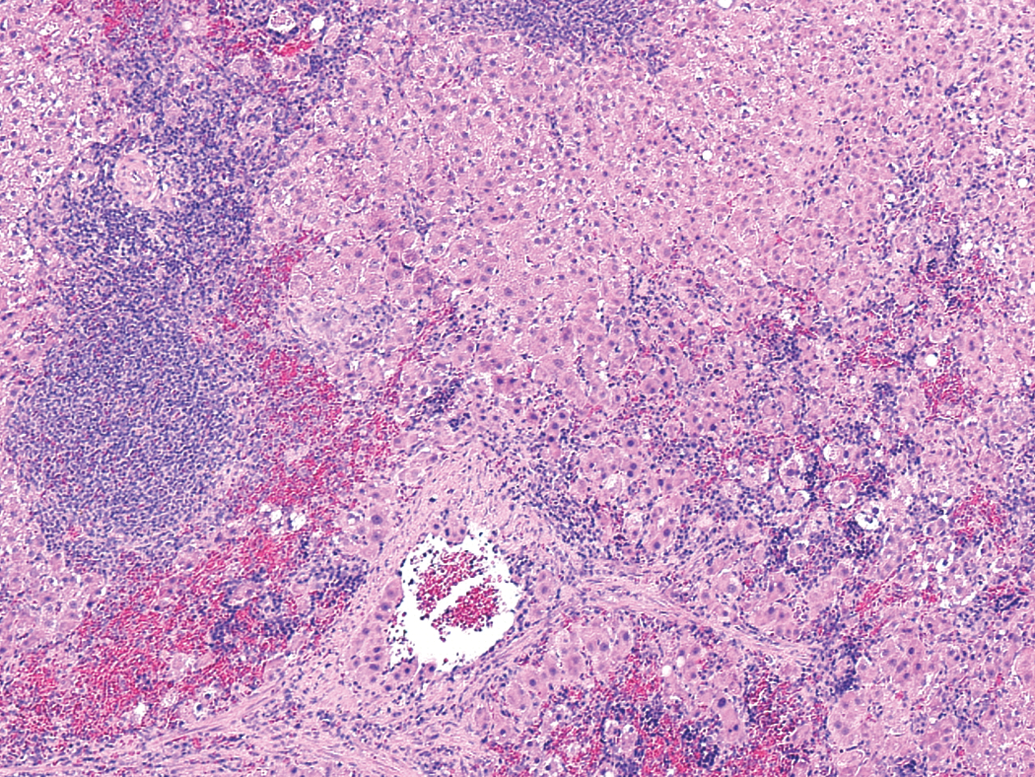
Morphological appearance of intrasplenically transplanted hepatocytes. Light microscopic appearance of intrasplenic hepatocytes forty-five weeks after transplantation (200×). Spleens were fixed in 10% buffered formalin and stained with hematoxylin and eosin. Large masses of hepatocytes within the red pulp occupy approximately 70% of the figure.
Isolated Hepatocyte CYP mRNAs
To determine whether the hepatocytes expressed normal transcript levels at the time of transplantation, an aliquot of isolated cells from every donor rat was stored for analyses at the time the remaining hepatocytes were infused into recipient spleens. We found that the isolated hepatocytes expressed the expected sexually dimorphic levels and concentrations of CYP2A1, -2C7, -2C11, -2C12, and -3A2 mRNAs as observed in the same-sex recipient livers (see subsequent tables), and accordingly the mRNA values for the pre-infused hepatocytes have not been presented.
Albumin
There were no statistical differences in the concentrations of albumin mRNA in the livers of any treatment group. That is, regardless of sex, the presence of splenic hepatocytes, or the number of weeks following transplantation, the livers of rats in all groups contained similar levels of the transcript (Figure 2 ).
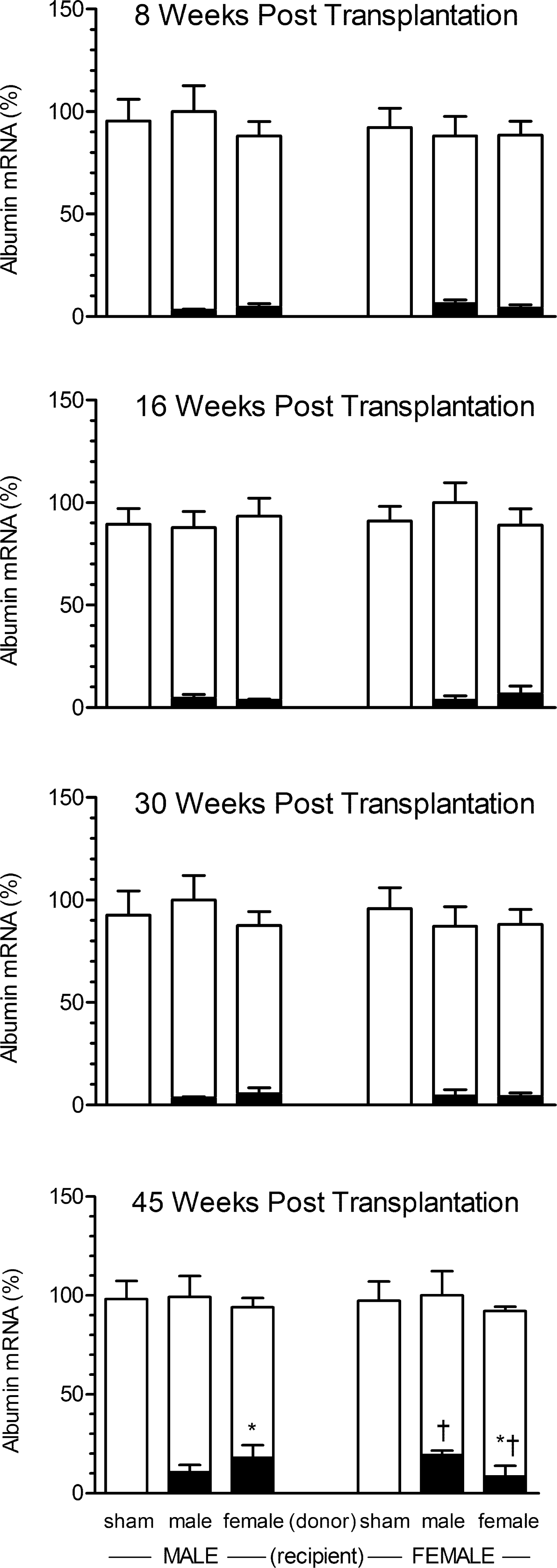
Rat albumin mRNA levels in liver and spleen of recipient male and female rats bearing intrasplenic hepatocyte transplants from same-age male or female donor rats. Spleens from sham rats were infused with hepatocyte medium containing no cells. Recipient rats were euthanized at eight, sixteen, thirty, or forty-five weeks post-transplantation. Each data point is a mean ± SD from six rats. The open bars represent hepatic albumin mRNA expressed as a percentage of the mean value of the group with highest concentration (designated 100%) of the transcript. The black bars represent the concentration of splenic albumin mRNA expressed as a percentage of their autologous liver albumin mRNA. *, p < .05 compares the effects of donor sex in same sex recipient; † p < .05 compares the effects of recipient sex containing hepatocytes from same-sex donors.
Not unexpectedly, having been infused with only hepatocyte medium, the spleens of both male and female sham rats contained no measurable rat albumin mRNA. In general, splenic hepatocytes in male and female recipients, regardless of donor sex, contained statistically similar concentrations of rat albumin mRNA, albeit at only 4–6% of the concentrations in their autologous livers. Only at forty-five weeks post-transplantation, however, did we observe a sex-dependent effect in which albumin content was statistically greater in the spleens of recipients that received hepatocyte implants from opposite-sex donors (Figure 2). That is, at forty-five weeks post-transplantation, female-derived hepatocytes expressed higher levels of the transcript than male-derived cells when implanted in the spleens of male recipients. Correlatively, hepatocytes obtained from males contained greater concentrations of albumin mRNA than those from females when maintained in the spleens of females.
CYP in Sham-treated Rats
Although we analyzed the livers and spleens of the sham-treated rats for all the measured CYP transcripts at the four post-transplantation ages, our findings have not been presented. Since the sham spleens contained neither hepatocytes nor albumin, we expected to observe no detectable concentrations of CYP2A1, -2C7, -2C11, -2C12, and -3A2 mRNAs. The livers of the sham-treated rats expressed the same sexually dimorphic levels of CYP transcripts as we report for the livers of the recipients, thus generating no new findings.
CYP2C12
The liver findings clearly agree with the dozens of earlier reports, many of them our own (Shapiro et al. 1995; Waxman and Holloway 2009), demonstrating female-specific expression of hepatic CYP2C12. That is, male liver, regardless of age or splenic implant, had no detectable levels of CYP2C12 mRNA (Table 1 ). On the other hand, female liver expressed robust levels of the enzyme transcript (approximately 40% of its total hepatic CYP [Guengerich 1997; Nelson et al. 2004]) that were uneffected by either the sex of the splenic transplant or the age of the rat.
Sexually dimorphic expression of CYP2C12 mRNA in liver- and spleen-containing hepatocyte transplants.
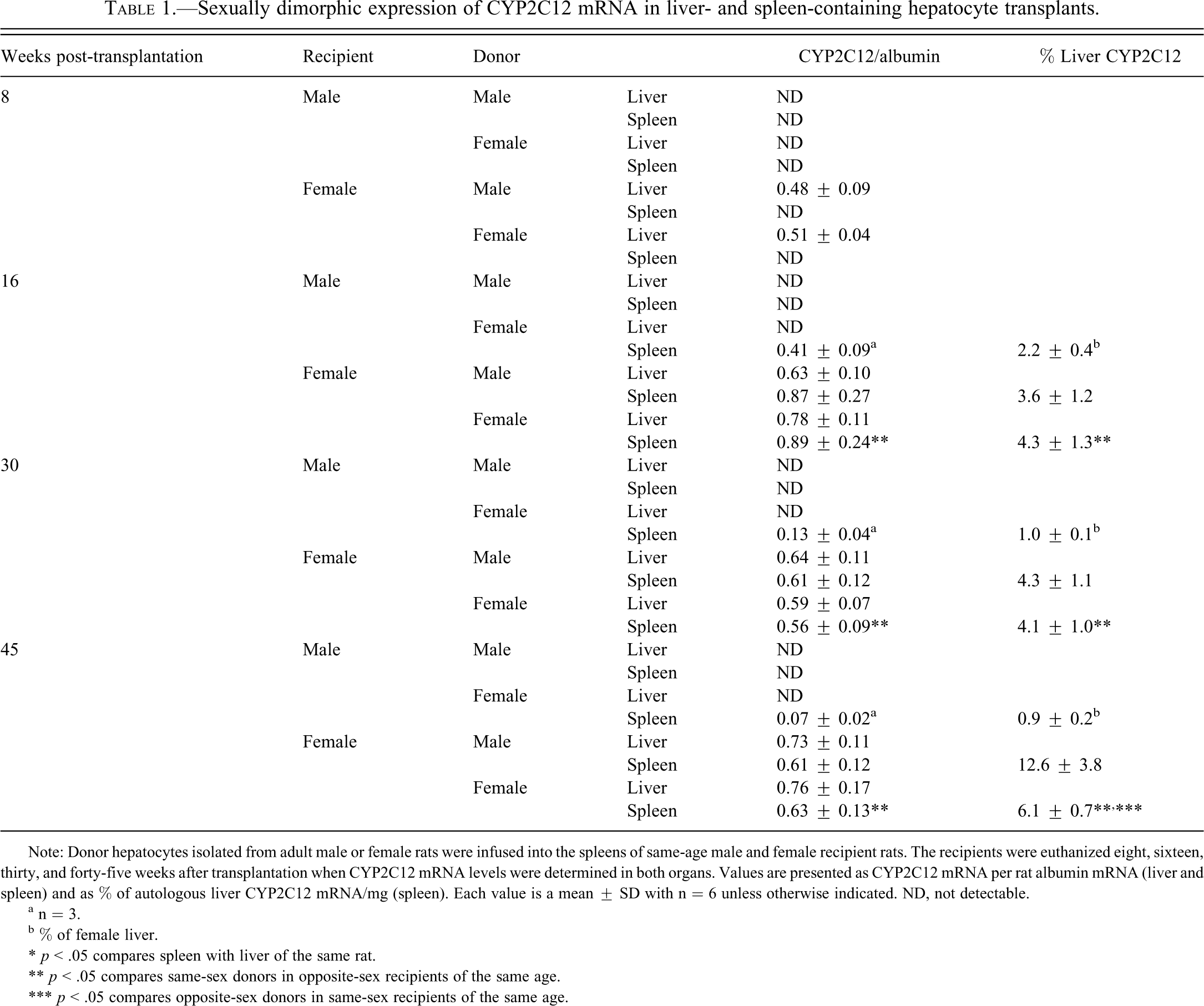
Note: Donor hepatocytes isolated from adult male or female rats were infused into the spleens of same-age male and female recipient rats. The recipients were euthanized eight, sixteen, thirty, and forty-five weeks after transplantation when CYP2C12 mRNA levels were determined in both organs. Values are presented as CYP2C12 mRNA per rat albumin mRNA (liver and spleen) and as % of autologous liver CYP2C12 mRNA/mg (spleen). Each value is a mean ± SD with n = 6 unless otherwise indicated. ND, not detectable.
a n = 3.
b % of female liver.
* p < .05 compares spleen with liver of the same rat.
** p < .05 compares same-sex donors in opposite-sex recipients of the same age.
*** p < .05 compares opposite-sex donors in same-sex recipients of the same age.
Eight weeks after transplantation, in spite of the presence of albumin mRNA, the female-derived splenic hepatocytes appear to have lost their ability to express CYP2C12 mRNA, whereas the male-derived splenic hepatocytes had not obtained that function (Table 1). However, following sixteen, thirty, and forty-five weeks of transplantation, the splenic hepatocytes in recipient females, regardless of donor sex, had the same concentration (per hepatocyte) of the transcript as their autologous livers. Since the actual number of hepatocytes capable of residing in the spleen is profoundly lower than the number of hepatocytes in the liver, it is not surprising that the spleens expressed only a fraction of the total CYP2C12 mRNA of that measured in the livers. As expected, male-derived splenic hepatocytes in recipient males expressed no detectable levels of CYP2C12 mRNA at all time points measured. The fact, however, that the female-derived splenic hepatocytes expressed CYP2C12 mRNA in male recipients, albeit at much lower and declining levels than female liver, and in only half the recipients, suggests that expression of the female-specific enzyme is to some degree independent of suppressive factors in the male host.
CYP2A1
Hepatic CYP2A1 is a known female-predominant hepatic isoform whose approximately three-fold higher expression in females (Table 2 ) is clearly in agreement with previous reports (Shapiro et al. 1995; Waxman and Holloway 2009). Neither the age of the rats nor the presence of splenic hepatocytes, male or female derived, had any effect on the characteristic sexually dimorphic expression of liver CYP2A1 mRNA.
Sexually dimorphic expression of CYP2A1 mRNA in liver- and spleen-containing hepatocyte transplants.
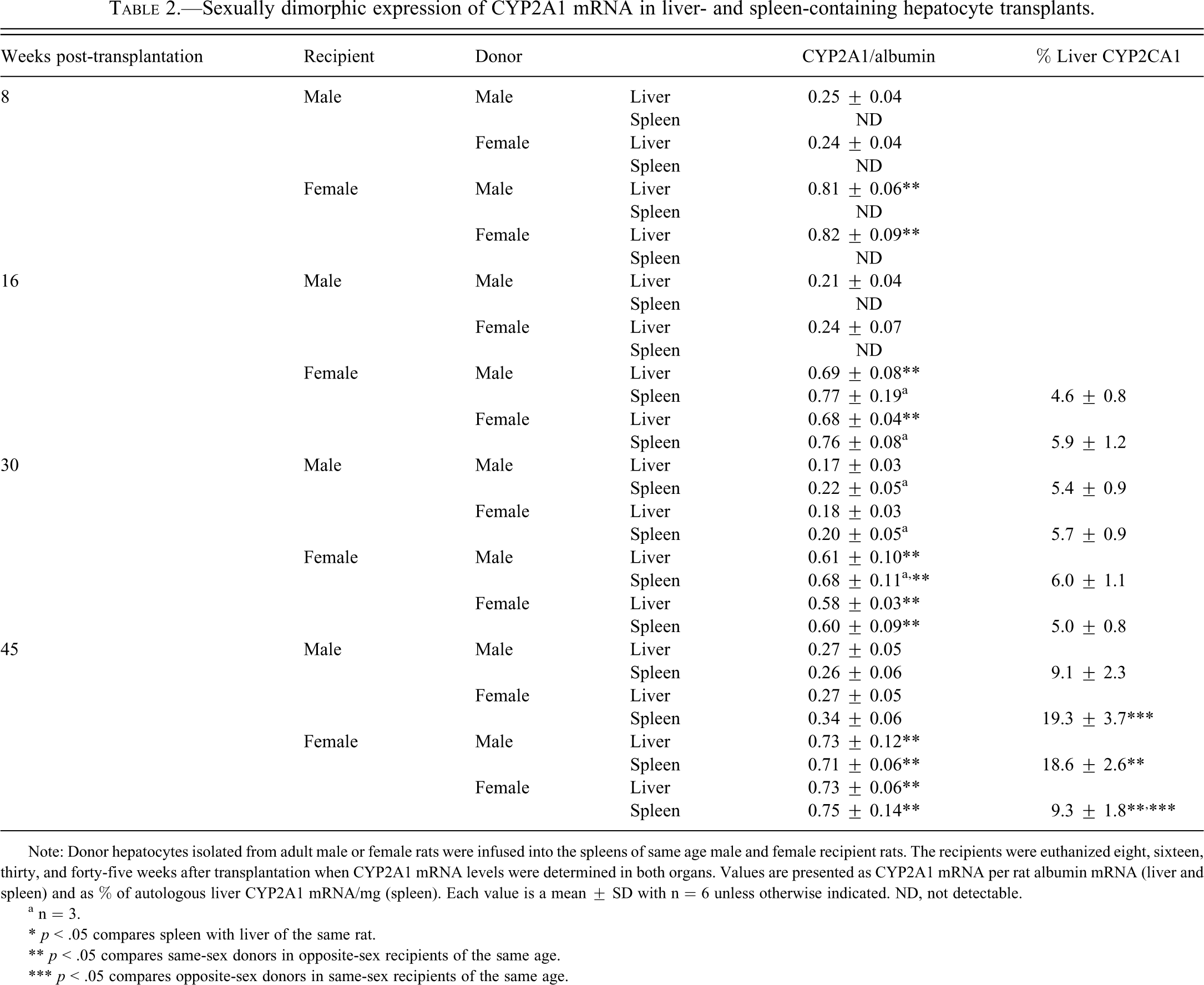
Note: Donor hepatocytes isolated from adult male or female rats were infused into the spleens of same age male and female recipient rats. The recipients were euthanized eight, sixteen, thirty, and forty-five weeks after transplantation when CYP2A1 mRNA levels were determined in both organs. Values are presented as CYP2A1 mRNA per rat albumin mRNA (liver and spleen) and as % of autologous liver CYP2A1 mRNA/mg (spleen). Each value is a mean ± SD with n = 6 unless otherwise indicated. ND, not detectable.
a n = 3.
* p < .05 compares spleen with liver of the same rat.
** p < .05 compares same-sex donors in opposite-sex recipients of the same age.
*** p < .05 compares opposite-sex donors in same-sex recipients of the same age.
In spite of the expression of albumin mRNA in all of the hepatocyte transplants at eight weeks post-transplantation, none of the cells, regardless of recipient or donor sex, contained detectable concentrations of CYP2A1 mRNA. At sixteen weeks post-transplantation, half of the female recipients with splenic hepatocytes from either sex expressed high autologous liver–like concentrations of CYP2A1 mRNA. The remaining 50% of the female recipients exhibited no measurable splenic CYP2A1. In contrast, CYP2A1 levels in splenic hepatocytes, male and female derived, remained undetectable in all the male recipients at sixteen weeks post-transplantation (Table 2). At thirty and forty-five weeks post-transplantation, donor hepatocytes in female, and now male recipients, expressed CYP2A1 mRNA. The transcript levels, however, reflected the sex of the host and not that of the donor. That is, isoform levels in male-derived splenic hepatocytes in female recipients were now elevated to female-like hepatic concentrations, whereas CYP2A1 mRNA levels in female-derived splenic hepatocytes in male recipients were suppressed to male-like hepatic concentrations. Total splenic CYP2A1 mRNA levels (i.e., percentage of autologous liver CYP2A1 mRNA/mg) generally reflected albumin mRNA levels (Table 2).
CYP2C7
As previously reported (Shapiro et al. 1995; Waxman and Holloway 2009), we found that CYP2C7 mRNA is another female-predominant hepatic isoform whose expression was approximately three-fold greater in female liver than male liver (data not presented). Neither the age of the rats nor the presence of splenic hepatocytes altered the sex-dependent hepatic ratio of the transcript. Regardless of the sex of either the recipients or donors, all of the transplanted splenic hepatocytes lost the ability to express measurable levels of CYP2C7 mRNA at any of the determined time points. (One or two spleens with female-derived hepatocytes in female recipients contained trace amounts of the transcript at sixteen and thirty weeks post-transplantation, which was not observed fifteen weeks later.)
CYP2C11
CYP2C11 is a male-specific hepatic isoform that accounts for > 50% of the total liver cytochrome P450 in adult male rats (Shapiro et al. 1995; Waxman and Holloway 2009). Therefore, although we found robust levels of the transcript in the livers of males at all ages, we were unable to detect CYP2C11 mRNA in female livers at any of the ages analyzed (data not presented). Although the pre-infused male hepatocytes expressed CYP2C11 mRNA at concentrations no different than that observed in the livers of recipient male rats, none of cells expressed measurable levels of the transcript when transplanted into the spleens of recipient males as well as females after eight, sixteen, thirty, and even forty-five weeks. Similarly, the isolated female hepatocytes, containing no detectable CYP2C11 mRNA at the time of infusion, remained so when transplanted into the inducible environment of the male rat.
CYP3A2
Like CYP2C11, CYP3A2 is a male-specific hepatic isoform whose expression is suppressed in female rats (Shapiro et al. 1995; Waxman and Holloway 2009). Therefore, we observed that expression of CYP3A2 mRNA was limited to male liver and that the concentration of the transcript was independent of the male’s age and the presence of splenic hepatocytes (Table 3 ). Unlike CYP2C11 mRNA, CYP3A2 mRNA was expressed, sex-dependently, in splenic hepatocytes. Although none of the female recipients, regardless of donor sex, expressed any detectable splenic levels of the transcript, hepatocytes of either sex, transplanted into the spleens of males, did contain CYP3A2 mRNA thirty and forty-five weeks after infusion. However, the concentrations of the isoform in splenic hepatocytes were only around 30% of that observed in the recipient males’ livers (Table 3). Consequently, the amount of CYP3A2 mRNA in a milligram of implanted spleen was only about 2.5% of that found in a milligram of autologous liver.
Sexually dimorphic expression of CYP3A2 mRNA in liver- and spleen-containing hepatocyte transplants.
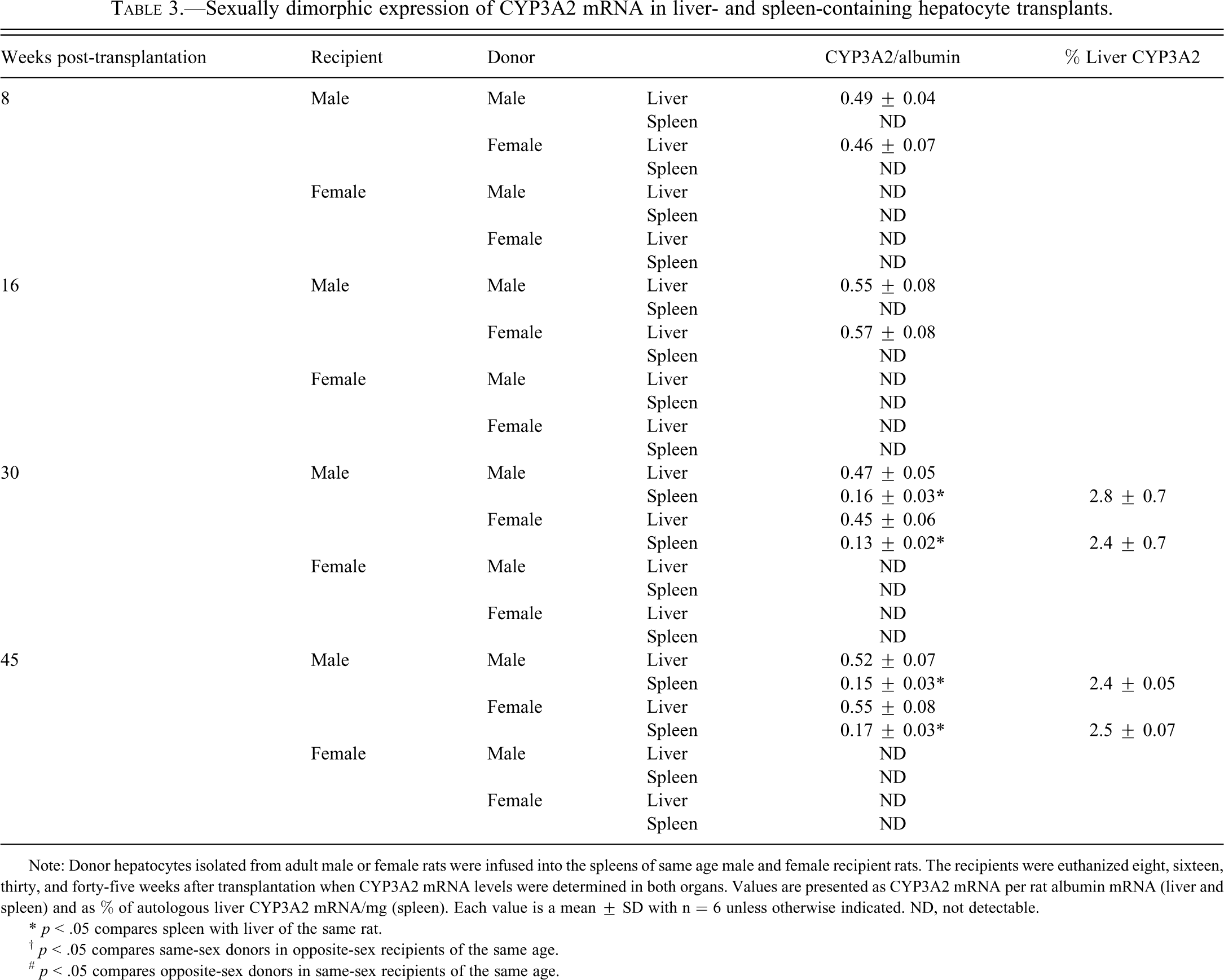
Note: Donor hepatocytes isolated from adult male or female rats were infused into the spleens of same age male and female recipient rats. The recipients were euthanized eight, sixteen, thirty, and forty-five weeks after transplantation when CYP3A2 mRNA levels were determined in both organs. Values are presented as CYP3A2 mRNA per rat albumin mRNA (liver and spleen) and as % of autologous liver CYP3A2 mRNA/mg (spleen). Each value is a mean ± SD with n = 6 unless otherwise indicated. ND, not detectable.
* p < .05 compares spleen with liver of the same rat.
† p < .05 compares same-sex donors in opposite-sex recipients of the same age.
# p < .05 compares opposite-sex donors in same-sex recipients of the same age.
Discussion
Whereas the pre-infused hepatocytes expressed their expected CYP sexual dimorphisms, their post-transplantational competence, depending on the isoform, was profoundly delayed, if not permanently lost, and reflected the sexual dimorphisms of the recipient (as observed in the host’s liver) rather than their pre-infused dimorphisms. This finding that CYP expression in the transplanted hepatocytes takes on the sexual characteristics of the recipient’s liver supports previous observations that the sex-dependent growth hormone (GH) secretory profiles to which the hepatocytes are exposed (in the present case, that of the host) determines which sexually dimorphic CYP isoforms are expressed. Presently, all species examined, including rats, mice, chickens (Shapiro et al. 1995), and humans (Enstrom et al. 1998; Hartman et al. 1993) exhibit sex differences in GH secretory profiles. In the case of the rat, males secrete GH in episodic bursts (about 200–300 mg/mL of plasma) about every three and a half to four hours. Between the peaks, GH is absent. In females, the hormone pulses are more frequent and irregular and are of lower magnitude than those in males, whereas substantial concentrations of GH are always secreted between peaks. Although the sexually dimorphic GH profiles are not exactly the same in all species, there are several commonalities such as the higher pulse amplitudes in the males and greater pulse frequencies with corresponding briefer interpeak periods in the females. In fact, it is now clear that sex differences in the circulating GH profiles, and not GH concentrations per se, are responsible for sexual dimorphisms ranging from body growth, muscle mass, bone density, and fat metabolism, as well as the expression of hepatic isoforms of CYP in all species examined (cf. Thangavel and Shapiro 2007).
Of nearly a dozen sex-dependent isoforms of CYP in the rat liver, which account for the bulk of constituent activity, some are expressed only in males or females (sex-specific), whereas others are expressed in both sexes, but at considerably different concentrations (sex-predominant). In this regard, we have discovered numerous inherent “regulatory signals” in the sexually dimorphic GH profiles that are individually responsible for either inducing or suppressing each of the sex-dependent CYP isoforms. For example, CYP2C12, the major female-specific rat isoform, requires a continuous (no GH-devoid interpulse of any length) profile for expression (Agrawal and Shapiro 2001; Pampori and Shapiro 1996; Pampori and Shapiro 1999). Accordingly, male hepatocytes, before isolation, were exposed to the episodic GH profile and thus were unable to express CYP2C12. Transplantation of the male-derived hepatocytes into the spleens of females exposed them to the continuous GH profile now inducing CYP2C12 expression. As would be expected, the elevated expression levels of CYP2C12 observed in pre-infused female hepatocytes were completely suppressed when the cells were transplanted into male spleens. In contrast, male-like levels of hepatic CYP3A2 are dependent upon the masculine episodic GH profile, and the feminine continuous GH profile is completely suppressive (Agrawal and Shapiro 2000; Agrawal and Shapiro 2001; Pampori and Shapiro 1996; Pampori and Shapiro 1999). Accordingly, pre-infused female hepatocytes expressed no measurable CYP3A2 mRNA. Following transplantation into the spleens of male rats, the hepatocytes were now exposed to the masculine circulating GH profile, which subsequently induced expression of the isoform in the female hepatocytes. In agreement with the proposed regulatory mechanism, the elevated expression levels of CYP3A2 observed in pre-infused male hepatocytes were completely suppressed when the cells were transplanted into the spleens of female rats. Unlike CYP2C12 and CYP3A2, CYP2A1 is not a sex-specific isoform, but rather a sex-predominant isoform. Whereas the masculine GH secretory profile can induce some CYP2A1 expression, the isoform is more responsive to the feminine continuous profile, resulting in a male-to-female sex ratio of 1:3 (Agrawal and Shapiro 2000; Agrawal and Shapiro 2001; Pampori and Shapiro 1996; Pampori and Shapiro 1999). Accordingly, pre-transplanted male hepatocytes expressed the lower male-like levels of CYP2A1. Following transplantation into the spleens of females, concentrations of the isoform increased three-fold to the characteristic female-like levels. Moreover, the elevated pre-infusion expression levels of CYP2A1 in female hepatocytes were reduced to male-like concentrations when the cells were transplanted into the spleens of males.
Although the CYP2C12, -3A2, and -2A1 findings support the reported mechanism that circulating GH profiles are the regulators of sex differences in expression levels of hepatic CYP isoforms, we have otherwise noted two confounding results. First, while the transplanted hepatocytes expressed albumin mRNA at all measured time points, expression of the three detectable isoforms were delayed until sixteen to thirty weeks post-transplantation. Second, and perhaps more significant, two of the isoforms, the major male-specific CYP2C11 and the female-predominant CYP2C7, remained statistically undetectable almost a year after transplantation. It is possible that had CYP transcripts been measured by real-time quantitative polymerase chain reaction, as we have reported in previous studies (Thangavel et al. 2007), we may have observed an earlier appearance of CYP2C12, -3A2, and -2A1 and possible nominal, but physiologically inconsequential, concentrations of CYP2C11 and CYP2C7.
The fact that the intrasplenic hepatocytes did not express CYP2C12, -3A2, and -2A1 for four to seven months after transplantation and exhibited a permanent loss of CYP2C11 and CYP2C7, in spite of normal expression levels of all the isoforms at the time of infusion, suggests an initial, prolonged, and sometimes permanent period of CYP repression followed by a subsequent re-expression of some isoforms. These findings of an initial suppression of isoform expression somewhat parallel in vitro findings of hepatocytes in cell culture (Wright and Pain, 1992; Niwa et al. 1996). We have reported (Thangavel et al. 2004; Thangavel et al. 2006) that primary hepatocytes in culture initially experience a dramatic decline in the expression levels of their near-dozen sex-dependent constituent CYP isoforms, some of which never recover, as also seen in the intrasplenic hepatocytes. Although careful replication of the species and sex-dependent GH profiles to the culture media can restore physiologic concentrations of many of the constituent isoforms, some remain permanently repressed (Thangavel et al. 2004; Thangavel et al. 2006), again, similar to the present findings with the intrasplenic transplanted hepatocytes.
It may be concluded that although CYP enzymes are both an ancient (about 1.5 billion years old) and an enormous superfamily (made up of many hundreds of isoforms) expressed in nearly every, if not all, phyla and within almost every cell (Guengerich 1997; Nelson 1998; Nelson et al. 2004), they simply do not function well ex situ. In this regard, the hepatic acinus, the structural and functional unit of hepatic parenchyma, is composed of hepatocytes that differ in morphological, biochemical, and functional abilities, depending on the location of the cells in relation to the inflow or outflow of portal blood, that is, periportal or perivenous (Jungermann 1986; Traber et al. 1988). It is now well known that most of the sex-dependent rat hepatic CYP isoforms that account for the bulk of constituent activity exhibit a particularly prominent zonation within the acinus, which then determines both their constituent and inducible levels (Buhler et al. 1992; Lindros 1997). These isoforms include CYP2A1, -3A1, -3A2, -2C11, -2C12, and -2E1. Although the intrasplenic hepatocytes exhibit some cord-like structures, their general architecture does not resemble that of a normal hepatic acinus and might explain the inability of the transplanted liver cells to express CYP2C11 and CYP2C7. However, primary hepatocytes in cell culture whose architecture in no way resembles an hepatic acinus are capable of expressing physiologic-like concentrations of CYP2C11, but no detectable CYP3A2 (Thangavel et al. 2006). Clearly, the loss of architectural integrity of the transplanted intrasplenic hepatocytes as well as primary hepatocytes in cultures is not sufficient to explain the inability of ex situ hepatocytes to express their normal complement of constituent CYP isoforms. Normal expression of some of the constituent isoforms outside the confines of the normal acinus structure apparently require yet to be identified factors. (Differences in the source of arterial blood are unlikely to explain abnormalities in CYP expression of intrasplenic hepatocytes since both the liver and spleen are perfused by branches of the celiac artery.) Although the likely sex-dependent expression of CYP isoforms of human intrasplenic hepatocyte transplants is speculative, it is noteworthy to mention that the major human CYP isoforms are also expressed at sexually dimorphic levels regulated by sex differences in circulating GH profiles (Dhir et al. 2006; Jaffe et al. 2002).
Footnotes
All of the authors declare no conflict of interest or financial disclosures. This work was supported in part by National Institutes of Health Grant HD-061285 and the Chutzpah Foundation.
