Abstract
Glatiramer acetate (GA), the active ingredient in Copaxone®, is a complex mixture of polypeptides used for the treatment of relapsing remitting multiple sclerosis. Glatiramoids are related mixtures that may differ in some characteristics of the prototype molecule. Our aim is to describe the long-term toxicity studies with protiramer (TV-5010), a new glatiramoid, in comparison with similar studies conducted with GA. The toxicity of twice-weekly subcutaneous injections of protiramer to Sprague-Dawley rats (twenty-six weeks) and cynomolgus monkeys (fifty-two weeks) was compared with similar studies done with daily subcutaneous injections of GA. Daily treatment with GA was safe and well tolerated, without systemic effects or death. Protiramer administration was not as well tolerated as GA and led to dose- and time-related mortalities, probably mediated through severe injection-site lesions both in rats and in monkeys. Bridging fibrosis in the liver and severe progressive nephropathy were seen in rats. A dose-related increase in eosinophils was observed in monkeys. The protiramer toxicity studies show that minor variations in the manufacturing of glatiramoids may lead to significant toxic effects. It is therefore essential that the safety of any new glatiramoid be studied in long-term preclinical studies before exposing humans.
Introduction
The glatiramoid class of compounds is a family of synthetic heterogenous polypeptide mixtures that include four natural amino acids, L-glutamic acid, L-alanine, L-lysine, and L-tyrosine, in a distinct molar ratio of 0.14:0.43:0.09:0.34, respectively, with an average molecular weight (MW) in the range of 5,000–9,000 Daltons (Varkony et al. 2009). The first glatiramoid, and the best studied one, is glatiramer acetate (GA), the active ingredient in Copaxone®, which is approved for the treatment of relapsing remitting multiple sclerosis (MS) in over fifty countries. It has been shown that GA has a unique immunomodulatory mechanism, leading to disruption of the pathogenic process in MS by reinforcing immunoregulatory networks (Arnon and Aharoni 2009; Blanchette and Neuhaus 2008; Liblau 2009; Schrempf and Ziemssen 2007; Van Kaer 2011; Weber et al. 2007). The exact mode of action of GA has not been completely elucidated, but it has been suggested that the heterogeneous nature of the mixture plays an important role in securing activity in such a heterogeneous disease (Johnson 2010; Varkony et al. 2009).
Recently, there has been growing interest in glatiramoids in the pharmaceutical industry (Johnson 2010), and many manufacturers are seeking to develop new glatiramoids (Kovalcin, Krieger, Collins et al. 2010; Kovalchin, Krieger, Genova et al. 2010), and others aim to prepare follow-on (“generic”) versions of GA itself. An additional new glatiramoid product, protiramer (also denoted as TV-5010), was recently developed by Teva Pharmaceutical Industries, Ltd (Petach Tikva, Israel) as a potential follow-on product to GA with a slightly higher MW distribution. The rationale for developing a high-MW glatiramoid was based on results of fractionation studies of GA that showed that the higher the MW of the fraction, the higher potency and immune reactivity the fraction has in pharmacological models. This new glatiramoid was developed to provide better efficacy or less dosing frequency for MS patients.
Protiramer was made by making minor modifications to the GA synthetic process, mainly a slight change in the temperature in which the precursor molecule is cleaved to yield the final mixture. This minor change results in a product with a different MW distribution and different biological properties than GA (De Stefano et al. 2009; Varkony et al. 2009). Based on encouraging preclinical safety results from short-term toxicity studies, protiramer’s safety, tolerability, and efficacy were evaluated in two small phase II clinical trials in MS patients; it demonstrated good general safety and tolerability (De Stefano et al. 2009). Later, long-term preclinical toxicity studies in monkeys and rats revealed marked progression in the severity of injection site reactions, which was also accompanied by kidney and liver damage, and death of some high-dose animals. The findings in animals led to the termination of the drug development program of protiramer (Varkony et al. 2009) and brought about the understanding that even slight changes in the manufacturing process of glatiramoids may have profound safety implications.
The aim of the present report is to describe the clinical and anatomical pathology findings in the long-term preclinical toxicity studies of protiramer and compare these results with the results of similar preclinical chronic toxicity studies in rats and monkeys that were carried out previously with GA. By comparing these two toxicity studies, we show that minor variations in the manufacturing of glatiramoids may lead to significant toxic effects. In addition, we present and discuss the potential mechanistic association between the severe injection site lesions and systemic toxicities seen in animals.
Materials and Methods
Chemicals
Both GA and protiramer were produced by Teva Pharmaceutical Industries, Ltd, Petach Tikva, Israel, at a fully accredited GMP manufacturing site.
Animals and Housing
The toxicology studies were carried out as fully compliant GLP studies in accredited contract research laboratories. All animals used in this research were treated humanely, with due consideration to alleviation of distress and discomfort. For the protiramer studies, male and female Sprague-Dawley rats (approximately eight weeks old) were obtained from Charles River Laboratories (Saint Germain sur l’Arbresle, France), and male and female cynomolgus monkeys (
Experimental Design
The route of administration in all studies was subcutaneous injections. Several injection sites were used in each animal and were alternated on each session to minimize local damage. Doses for the chronic toxicity studies were selected based on tolerability observed in shorter range-finding studies (not shown). The GA rat chronic toxicity study included one control and three treatment groups of twenty animals/sex/group that were treated daily at doses of 0, 3, 10, and 30 mg/kg for twenty-six weeks. Vehicle was sterile physiological saline, and a constant dose volume of 2 mL/kg was used. The protiramer rat chronic toxicity study included four treatment groups and one control group; each group consisted of twenty males and twenty females per dose treated twice a week at doses of 0, 2.5, 40, and 300 mg/kg or once every two weeks at 2.5 mg/kg dose (low-dose, low frequency) for twenty-six weeks. Additional animals (five/sex/group) of the control and high-dose groups were assigned to a four-week treatment-free period. The vehicle was mannitol solution in water for injection, and the dose volume was 5 mL/kg. The GA monkey study included three treatment groups and one control group of four animals/sex/group, treated at doses of 0, 3, 10, and 30 mg/kg for fifty-two weeks. The vehicle was sterile physiological saline, and a dose volume of 1.5 mL/kg was used. The protiramer monkey study included four treatment groups and one control group; each group consisted of seven males and seven females. Protiramer treatment of the monkeys was done twice weekly at doses of 0, 2, 10, and 60 mg/kg for either thirteen weeks (interim group of three animals/sex/dose) or fifty-two weeks (main group of four animals/sex/dose). The fifth group was a low-dose, low-frequency group and was administered protiramer once every four weeks at a dose level of 2 mg/kg (the same number of monkeys for this group). The high dose was lowered from 60 to 30 mg/kg on day 280 of the study because of poor tolerability. Additional animals of the control, high-dose, and low-frequency, low-dose groups (two males and two females per group) were assigned to a four-week treatment-free period after fifty-two weeks of dosing. The vehicle was mannitol solution in water for injection, and the dose volume was 1 mL/kg. All animals were observed at least twice daily to detect any mortality or morbidity, and once daily for ill health or reaction to treatment. Individual body weights were recorded weekly. Blood chemistry, coagulation, and hematology parameters were examined at the end of each rat study and at predose and at several predetermined time points during the monkey studies. Urine samples were also collected on several occasions in both the rat and monkey studies. At the scheduled termination, animals were subjected to a full gross pathology evaluation including weighing of selected organs and histopathological analysis. Moribund animals were also examined for any pathological abnormality.
In the GA studies, the weights of the adrenals, pituitary, brain (including brain stem), prostate, heart, spleen, kidneys, testes with epididymides, liver, ovaries, and thyroids with parathyroids were determined at terminal sacrifice. In the protiramer studies, the weights of the thymus and uterus were also determined. For histological examination, tissues were fixed in 10% neutral buffered formalin, except for the eyes, which were fixed in Davidson's fixative, and the bone marrow smears, which were fixed in methanol (in the GA studies) or air dried (in the protiramer studies). In the protiramer studies, the testes and epididymides were also fixed in Davidson’s fixative. Fixed tissues were embedded in paraffin, sectioned, and stained with hematoxylin and eosin. The following tissues from all high-dose and control groups animals were sampled: adrenals, brain (including brain stem), esophagus, eyes, femur, Harderian gland, heart and aorta, large intestine (cecum, colon, and rectum), small intestine (duodenum, jejunum, and ileum), injection site, kidney, liver, lung (and main stem bronchi), lymph nodes (mandibular and mesenteric), mammary gland, nose, optic nerves, ovary, pancreas, parathyroid gland, pituitary gland, prostate gland, salivary gland, skin, spinal cord and sciatic nerve, spleen, stomach (including forestomach and glandular stomach), testis (with epididymis and seminal vesicle), thigh muscle, thymus, thyroid gland, tongue, trachea, urinary bladder, uterus, vagina, and Zymbal’s gland. In the GA rat study, the following tissues were examined microscopically: all tissues from all animals in control and high-dose groups, all tissues from animals that died or were killed in extremis, injection sites, and abnormalities from the low- and intermediate dose groups. In the protiramer group, histopathological examination was performed for all organs from the following animals: for all animals found dead or sacrificed moribund during the study, and all the control group animals, all intermediate dose group males, and all high-dose group females and all high-dose group males sacrificed after twenty weeks of treatment. Histopathological examination was also performed for kidneys, liver, and injection sites from all other dose groups as well as for the animals that were assigned to the treatment-free “recovery” groups. The severity of microscopic lesions observed was graded based on the following numerical scale: 0 (no lesion), 1 (minimal), 2 (mild), 3 (moderate), and 4 (severe), based on the criteria explained by Shackelford et al. (2002).
Statistical Analysis
In all studies, statistical analysis was performed separately for males and females.
For the analysis of the clinical chemistry and hematology parameters, the Levene test for homogeneity of variances and the Shapiro-Wilk test to assess for normality were performed in protiramer studies, and the Levene test was performed in GA studies. Parametric methods were performed in protiramer studies in the case of homogeneous variances and normal distribution in all groups and in GA studies in the case of homogeneous variances between groups. Such analysis consisted of the one-way analysis of variance (ANOVA) followed, if the ANOVA test was significant, by the Dunnett test for pairwise comparisons. Nonparametric methods were performed in protiramer studies in the case of evidence of heterogeneous variances or a non-normal distribution in at least one group and in GA studies in the case that there was a significant group effect or a significant sex–group interaction in the Levene test. Such analysis consisted of the Kruskal-Wallis ANOVA followed, if the Kruskal-Wallis test was significant, by the Wilcoxon rank sum test for pairwise comparisons. The Jonckheere-Tersptra trend test was used to test for an increasing or decreasing trend in response (excluding the low-dose, low-frequency group). In the GA studies, in the case of homogeneous variances between groups, a regression test for post-dose variables was used instead. The ANOVA results are not reported.
For histopathology data, nonparametric methods were used. Such analysis consisted of the Kruskal-Wallis test followed, if the Kruskal-Wallis test was significant, by the Wilcoxon rank sum test for pairwise comparisons. The Jonckheere-Tersptra trend test was used to test for an increasing or decreasing trend in response (excluding the low-dose, low-frequency group). The Kruskal-Wallis results are not reported. Statistically significant results for pairwise comparisons are indicated on the tables next to the appropriate mean value, using the following abbreviations: *
Results
Survival, Clinical Observations, Body Weight Gain, and Food Consumption
Glatiramer Acetate Studies
In the rat chronic toxicity study, treatment was well tolerated at the tested dose levels. Six animals died during the six months of the study, but none of these was considered to be related to treatment, as the deaths were evenly distributed among the dose groups including control animals, and since the pathological examination did not reveal any association to treatment. Two of these animals died accidentally as result of the bleeding procedure. There were no effects on body weight gain or food consumption at any dose level, except for a slight, probably incidental, increase in weight for high-dose females during the interval of weeks 1–4. Clinical observations were restricted to occasional rough hair coat, stained fur, and appearance of sores at the injection sites that were noted occasionally in rats in the 10 and 30 mg/kg/day GA groups. Few dosing sites were abandoned or left to recover because of the above mentioned findings.
In the monkey GA study, one female of the intermediate dose group was removed from the study because of poor food consumption and loss of body weight. Histopathology revealed lymphoid and bone marrow atrophy and adrenal cortical hypertrophy, signs that are consistent with stress. Because of the isolated nature of the mid-dose finding, the condition of this animal was not considered to be treatment related. Clinical signs in the remaining animals were restricted to the sites of injection. Thickening of the skin was noted in all treated groups, with high-dose animals experiencing greater frequency and earlier occurrence, starting generally by week 11. Signs in the low-dose group of 3 mg/kg were milder and noted from week 20. Sores were occasionally accompanied by swelling and abscess. By week 52, thickening was consistently noted in all high-dose animals.
Protiramer Studies
In the rat chronic study, nine out of seventy-two high-dose and one out of fifty-two mid-dose animals died during the study, and these deaths were attributed to treatment with protiramer. Seven other deaths were considered unrelated. Clinical signs consisting of pallor, subdued behavior, thin appearance, stained fur, lacrimation, hair loss, scabs, and piloerection were noted in all treated groups. Local reactions to treatment were dose dependent and included induration associated with erythema and/or hematoma in both the high- and mid-dose groups. There was no obvious local reaction to treatment in animals given the low dose of 2.5 mg/kg (twice weekly or once every four weeks). Most of the local reactions to treatment had disappeared by the end of the four-week treatment-free period. There was a dose-related reduction in mean body weight and mean body weight gain in males given protiramer, with significant differences and even weight loss in males at the dose levels of 40 and 300 mg/kg, but not at 2.5 mg/kg or in the females of any group. The poor tolerance of animals in the high-dose group (300 mg/kg) necessitated the early termination of this group in week 20 of the study.
In the monkey chronic study of protiramer, four unscheduled deaths were noted, two of which were clearly treatment related. The two treatment-related cases were animals in the high-dose group that were diagnosed at necropsy with multiple subcutaneous areas of hemorrhage and vascular necrosis, and lymphoid and eosinophilic infiltrates, which were considered to be factors contributing to death. It should be noted that the first treatment-related death occurred only after more than six months of treatment. The two other deaths that were not treatment related were low-dose animals, one animal that died probably from an accident and one animal that was removed from the study after being diagnosed as a carrier of hepatitis B. Body and organ weights were unaffected by treatment. Occasional animals in the high-dose group were salivating or subdued, or had loss of balance, were retching or circling, had stiff limbs, or were motionless. In the severe cases, there was also a decrease in blood pressure, decrease in body temperature, and increase in respiratory rate. These signs started appearing after seven weeks and were more pronounced and frequent after thirteen weeks. Accordingly, a decision was taken in week 40 to reduce the high-dose level from 60 to 30 mg/kg.
Injection site reactions were observed in all treated monkeys in a dose-dependent manner. In the control animals, the lesions were infrequent and consisted mostly of scabs, and slight erythema and induration at a very low incidence (usually single animals on a very small number of occasions). Skin lesions at the injection site in animals treated at 2 mg/kg once every four weeks were similar to those in controls. In animals treated at 2 mg/kg twice weekly, there was a slight increase in the incidence and severity of the lesions of the type seen in control animals, and in addition, tumefaction was noted. Injection site lesions in animals treated at 10 mg/kg were more frequent and pronounced. The frequency and severity of the injection site lesions were greatest in the animals treated with 60/30 mg/kg. Because of the severity of these lesions, the original injection sites could not be used starting from week 40, and new injection sites were introduced. Lesions at the new sites of injection were similar to those seen in this dose group at the beginning of the study, indicating that these reactions do not involve immunological sensitization.
Clinical Chemistry and Hematology
Glatiramer Acetate Studies
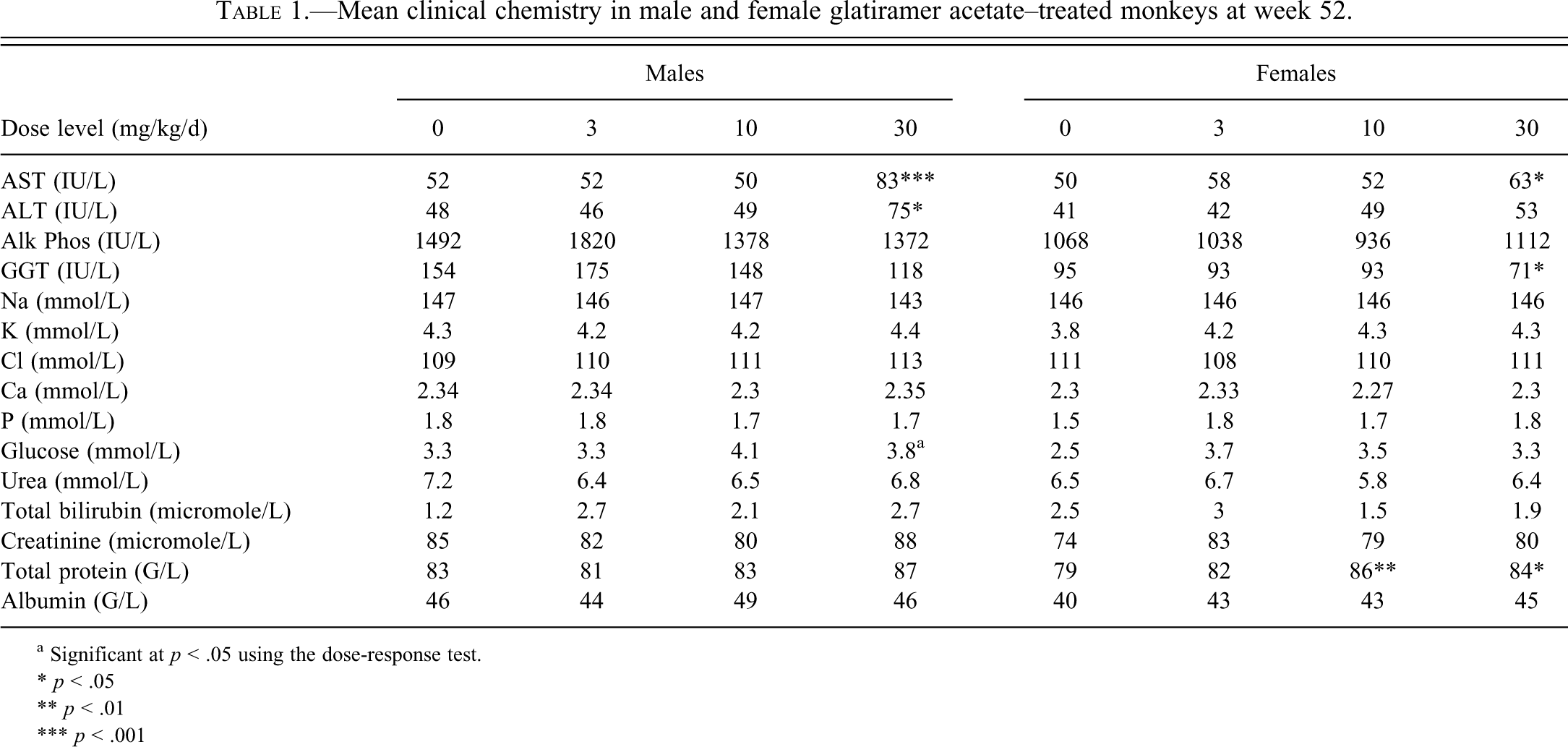
In the rat study, there was no effect of the GA treatment on hematology or clinical chemistry results. In the monkey study, there were occasional minor changes in AST and ALT values at the high dose level (Table 1 ). However, these changes were sporadic, minimal, and without a clear dose- or time-dependent relationship. There were no corroborating histological findings.
Mean clinical chemistry in male and female glatiramer acetate–treated monkeys at week 52.
a Significant at
*
**
***
Protiramer Studies
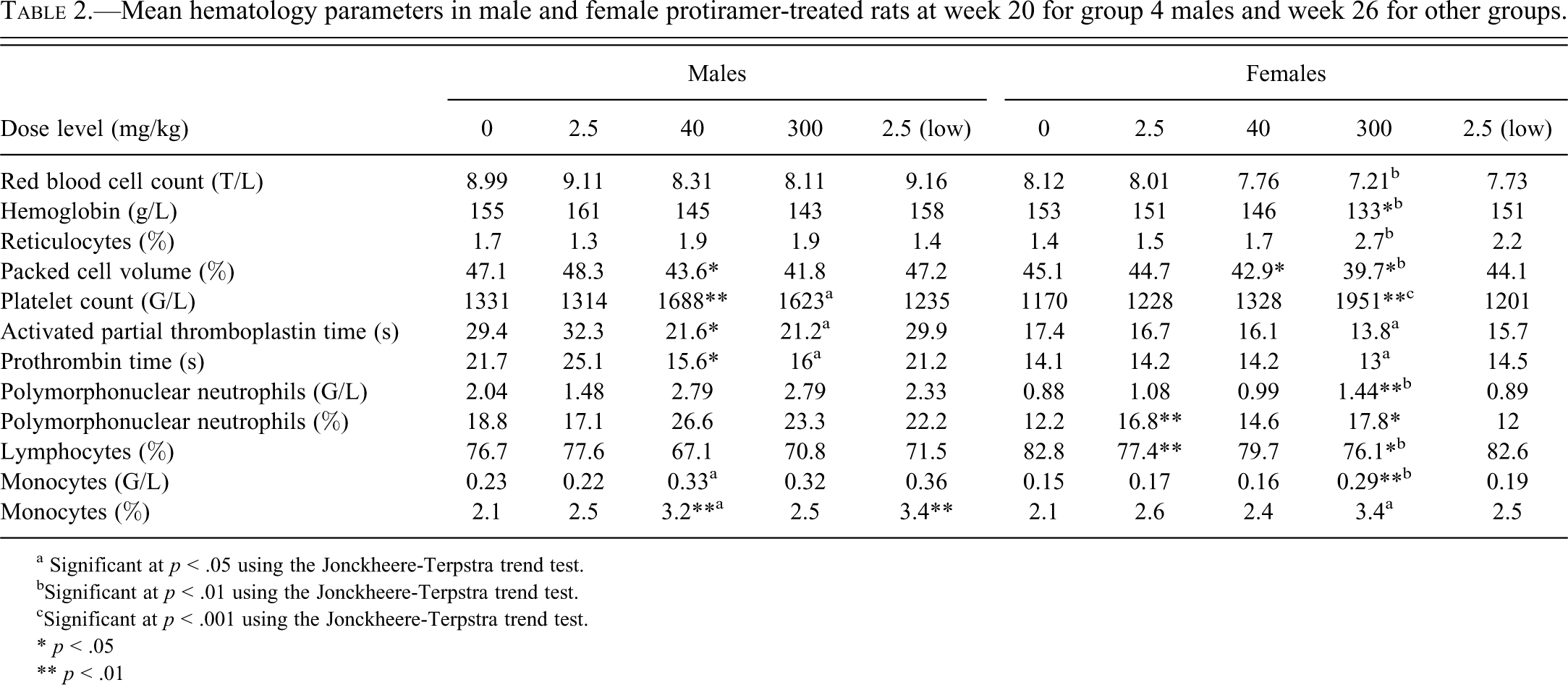
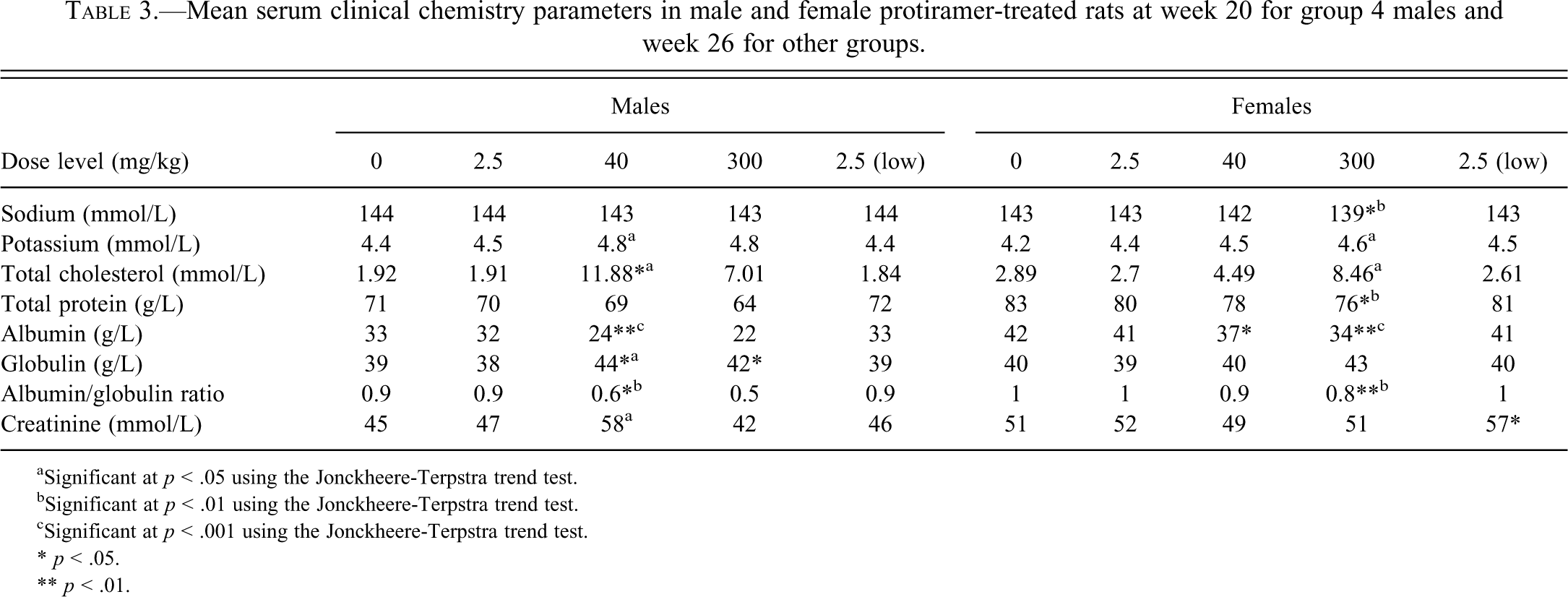
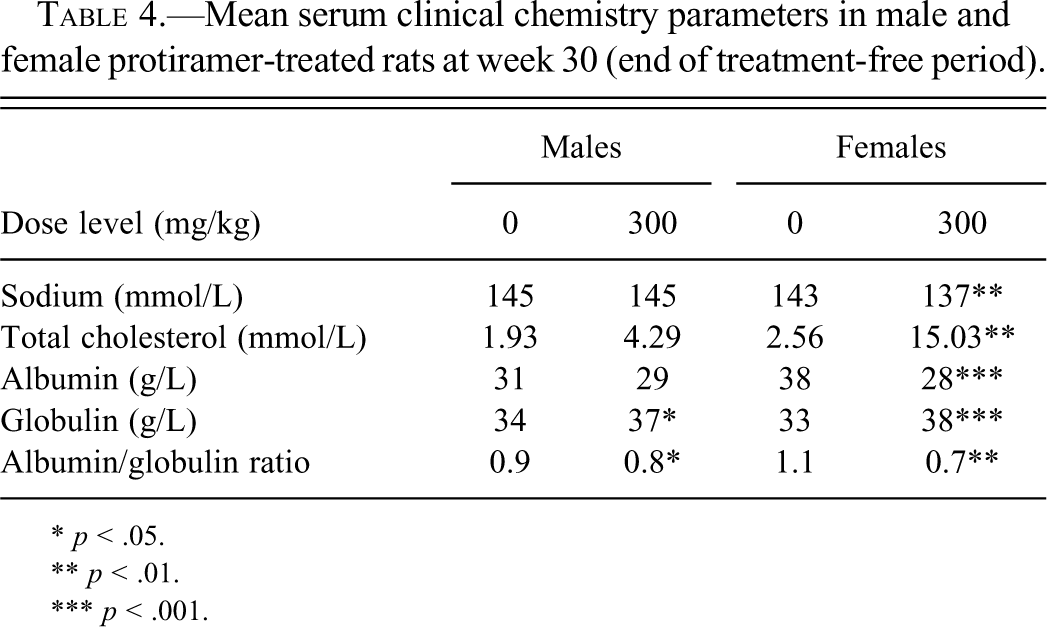
In the rat study, hematology analyses revealed a minor decrease in activated partial thromboplastin time (APTT) and prothrombin time (PT) and a small increase in platelet counts in the males and females given 40 and 300 mg/kg (Table 2 ). A small decrease in red blood cell count and hemoglobin concentration was evident in females given 40 and 300 mg/kg. This effect was accompanied by a slightly higher percentage of reticulocytes in these groups of females. Red blood cell parameters were also slightly affected in males at both dose levels, but to a lesser extent than in the females, with a slightly increased reticulocyte percentage at 300 mg/kg. The absolute and relative polymorphonuclear neutrophil counts were increased in females given 40 mg/kg and in males and females given 300 mg/kg, associated with reduced relative lymphocyte counts. There was also an increase in the absolute and relative monocyte counts at the end of the treatment-free period in females given 300 mg/kg (Table 2). The above treatment-related changes in hematology and coagulation parameters were no longer evident at the end of the four-week treatment-free period, except for the decreased PT time and increased monocyte and platelet counts in females given 300 mg/kg. There were several treatment-related changes in serum clinical chemistry parameters in males and females given 40 and 300 mg/kg (Table 3 ). These parameters included decreased total protein and albumin concentrations, increased globulin concentrations, increased total cholesterol levels, and slightly decreased serum sodium. Most of the above treatment-related changes in serum clinical chemistry were still evident after the treatment-free period (Table 4 ).
Mean hematology parameters in male and female protiramer-treated rats at week 20 for group 4 males and week 26 for other groups.
a Significant at
bSignificant at
cSignificant at
*
**
Mean serum clinical chemistry parameters in male and female protiramer-treated rats at week 20 for group 4 males and week 26 for other groups.
aSignificant at
bSignificant at
cSignificant at
*
**
Mean serum clinical chemistry parameters in male and female protiramer-treated rats at week 30 (end of treatment-free period).
*
**
***
In the monkey study, hematology evaluation revealed a marked dose-related increase in absolute and relative eosinophil counts in the groups treated twice weekly at 2, 10, and 60 mg/kg throughout the treatment period (Table 5 ). Animals treated with 2 mg/kg once every four weeks were unaffected. Throughout the treatment period, animals treated at 60 mg/kg tended to have lower mean phosphorus, cholesterol, and albumin concentrations, and from week 13, lower globulin concentrations (and lower A/G ratios). After four weeks without treatment, these differences had essentially returned to normal, although a slight difference in serum cholesterol concentrations remained.
Mean absolute and relative eosinophil counts in male and female-protiramer treated monkeys at week 52.
Abbreviation: nm, not measured.
Pathology
Glatiramer Acetate Studies
Gross pathology examination at the end of dosing of both rats and monkeys did not reveal treatment-related changes in any organ, except for the injection site lesions, and there were no changes in either absolute or relative organ weights.
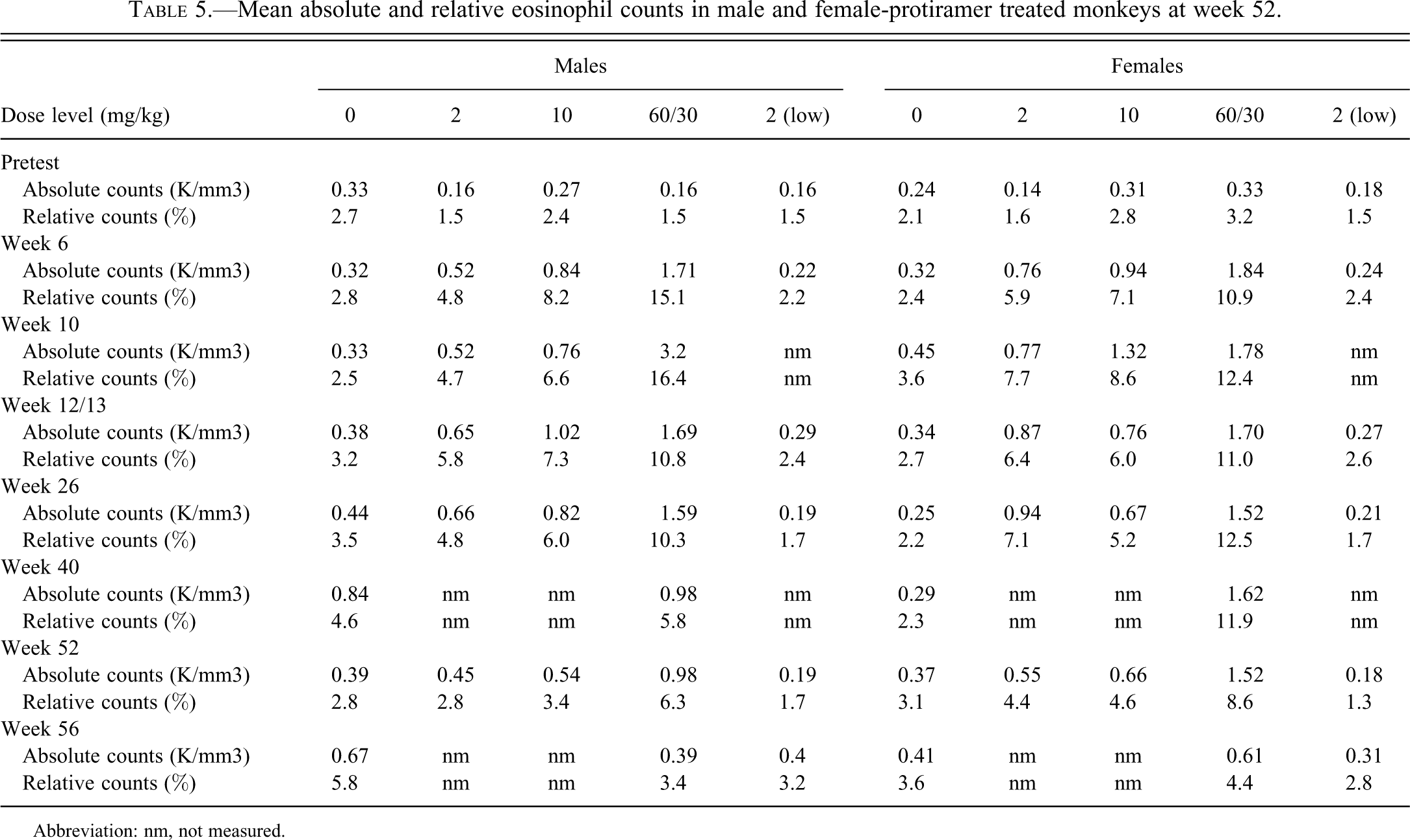
Histopathology examination of tissue sections from the rat study indicated that the pathological changes were confined to the injection sites (Table 6 ). Fibrosis was the most common lesion, with a dose-related increase in incidence and severity. Myositis in the subcutaneous panniculus muscle was less common and was seen mostly at the high dose of 30 mg/kg/day, with a slight increase in males of the 10 mg/kg GA group. There were sporadic cases of cellulitis, with increased severity in the 30 mg/kg dose group. In general, injection site lesions occurred more frequently at the posterior injection sites than in the anterior sites. This difference might be attributed to the presence of multiple layers of fascia at the anterior sites, which may have contributed to increased dispersal of the injected material (Figures 1–3).
Figure 1. Minimal subcutaneous fibrosis (arrows) in a control male rat repeatedly injected with the vehicle, 0.9% NaCl, for twenty-six weeks. Hematoxylin and eosin, 20×.
Figure 2. Slight subcutaneous fibrosis (arrows) in a male rat repeatedly injected with 30 mg/kg/day of GA for twenty-six weeks. Hematoxylin and eosin, 16×.
Figure 3. Moderate cellulitis (arrows) in the subcutis of a male rat repeatedly injected with 30 mg/kg/day of GA for twenty-six weeks. Hematoxylin and eosin, 16×.
Figure 4. Minimal subcutaneous fibrosing chronic inflammation in a control male cynomolgus monkey repeatedly injected with the vehicle, 0.9% NaCl, for fifty-two weeks. The fibrosing chronic inflammation is characterized by areas of fibrosis, in which mononuclear-cell accumulations are seen (arrow). Hematoxylin and eosin, 2.5×.
Figure 5. Minimal subcutaneous fibrosing chronic inflammation in a control male cynomolgus monkey repeatedly injected with the vehicle, 0.9% NaCl, for fifty-two weeks. The fibrosing chronic inflammation is characterized by areas of fibrosis, in which mononuclear-cell accumulations are seen (arrows). A minor diffuse eosinophil infiltration is also present. Hematoxylin and eosin, 16×.
Figure 6. Moderate subcutaneous fibrosis (arrowheads) associated with moderate mononuclear cell infiltration (arrows) and minimal subcutaneous germinal follicles (white arrow) in a male cynomolgus monkey repeatedly injected with 30 mg/kg/day of GA for fifty-two weeks. Hematoxylin and eosin, 2.5×.
Figure 7. Moderate subcutaneous fibrosis (arrowheads) associated with moderate mononuclear cell infiltration (arrows) in a male cynomolgus monkey repeatedly injected with 30 mg/kg/day of GA for fifty-two weeks. Hematoxylin and eosin, 16×.
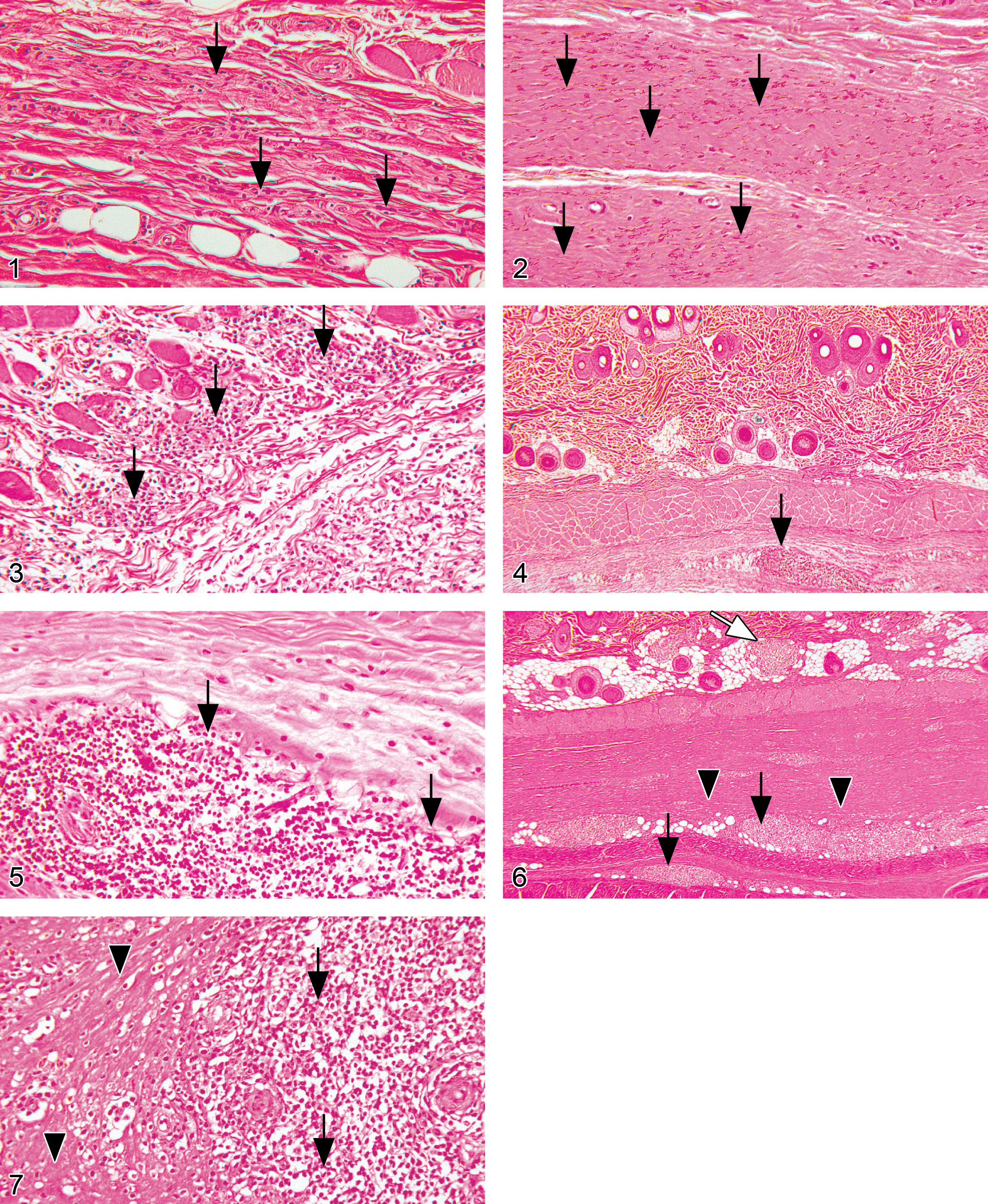
Figure 8. Histological section from an injection site from a control male Sprague-Dawley rat injected twice weekly with the vehicle, 0.9% NaCl, for twenty-six weeks. No abnormality detected. Hematoxylin and eosin, 3×.
Figure 9. Histological section from an injection site from a male Sprague-Dawley rat injected twice weekly with 300 mg/kg of protiramer for twenty-six weeks. Note presence of moderate degree of chronic inflammatory reaction (white arrows) with area of necrosis (arrows) close to a central cavity (arrowhead). Hematoxylin and eosin, 3×.
Figure 10. Higher magnification of the previous photo of Figure 10. Histological section from an injection site from a male Sprague-Dawley rat injected twice weekly with 300 mg/kg of protiramer for twenty-six weeks. Note, moderate degree of chronic inflammatory reaction (white arrows), associated with an area of necrosis (arrow) close to a central cavity (arrowhead). Hematoxylin and eosin, 15×.
Figure 11. Histological section from an injection site from a male Sprague-Dawley rat injected twice weekly with 300 mg/kg of protiramer for twenty-six weeks. Note moderate degree of chronic inflammatory reaction (white arrows) associated with thrombosis (arrows) and formation of a central cavity (arrowhead). Hematoxylin and eosin, 3.4×.
Figure 12. Histological section of a liver from a male Sprague-Dawley rat injected twice weekly with 300 mg/kg of protiramer for twenty-six weeks. Note centrilobular bridging fibrosis (white arrows). Hematoxylin and eosin, 20×.
Figure 13. Histological section of a kidney from a male Sprague-Dawley rat injected twice/week with 300 mg/kg of protiramer for twenty-six weeks. Note changes consistent with chronic progressive nephropathy (i.e., basophilic tubules, hyaline casts, and thickening of basement membrane [arrows]). Hematoxylin and eosin, 10×.
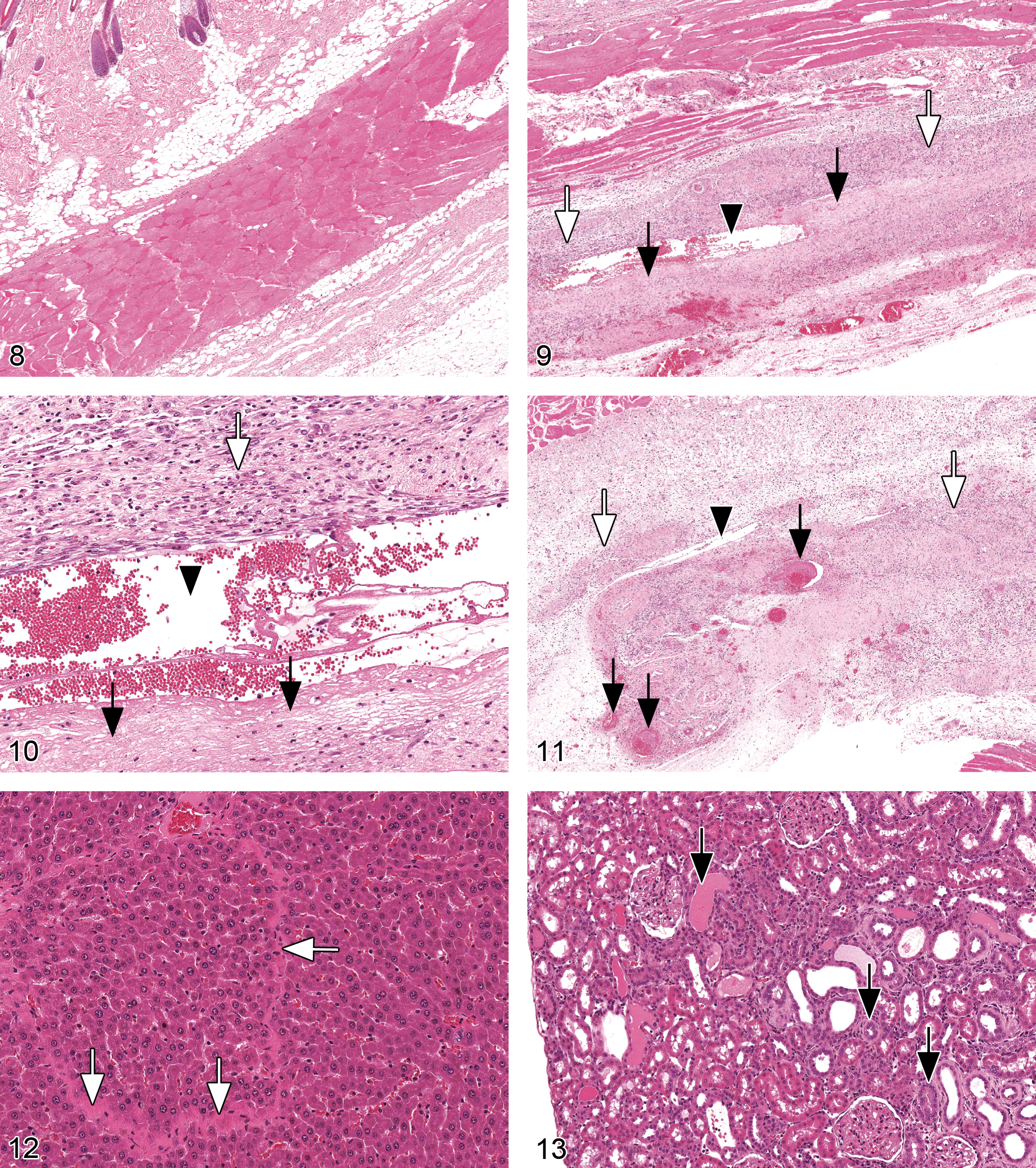
Figure 14. Histological section from an injection site from a control male cynomolgus monkey injected twice weekly with the vehicle, 0.9% NaCl, for fifty-two weeks. No treatment-related changes were noted. Hematoxylin and eosin, 1.5×.
Figure 15. Histological section from an injection site from a male cynomolgus monkey injected twice weekly with 30/60 mg/kg of protiramer for fifty-two weeks. Note presence of subcutaneous fibrosis with neovascularization, presence of lymphoid infiltrates mainly at perivascular locations (arrows), and the area of necrosis surrounding a central cavity (arrowheads). Hematoxylin and eosin, 1.5×.
Figure 16. Histological section from an injection site from a male cynomolgus monkey injected twice weekly with 30/60 mg/kg of protiramer for fifty-two weeks. Note presence of subcutaneous area of necrosis surrounding a central cavity (arrowheads). Hematoxylin and eosin, 20×.
Figure 17. Histological section from an injection site from a cynomolgus monkey injected twice weekly with 30/60 mg/kg of protiramer for fifty-two weeks. Note presence of subcutaneous fibrosis (white arrow) with neovascularization, presence of lymphoid infiltrates mainly at perivascular locations (arrow), and diffuse infiltrates of eosinophils (arrowheads). Hematoxylin and eosin, 20×.
Incidence and mean severity of injection site lesions in rats in the twenty-six–week study with glatiramer acetate.
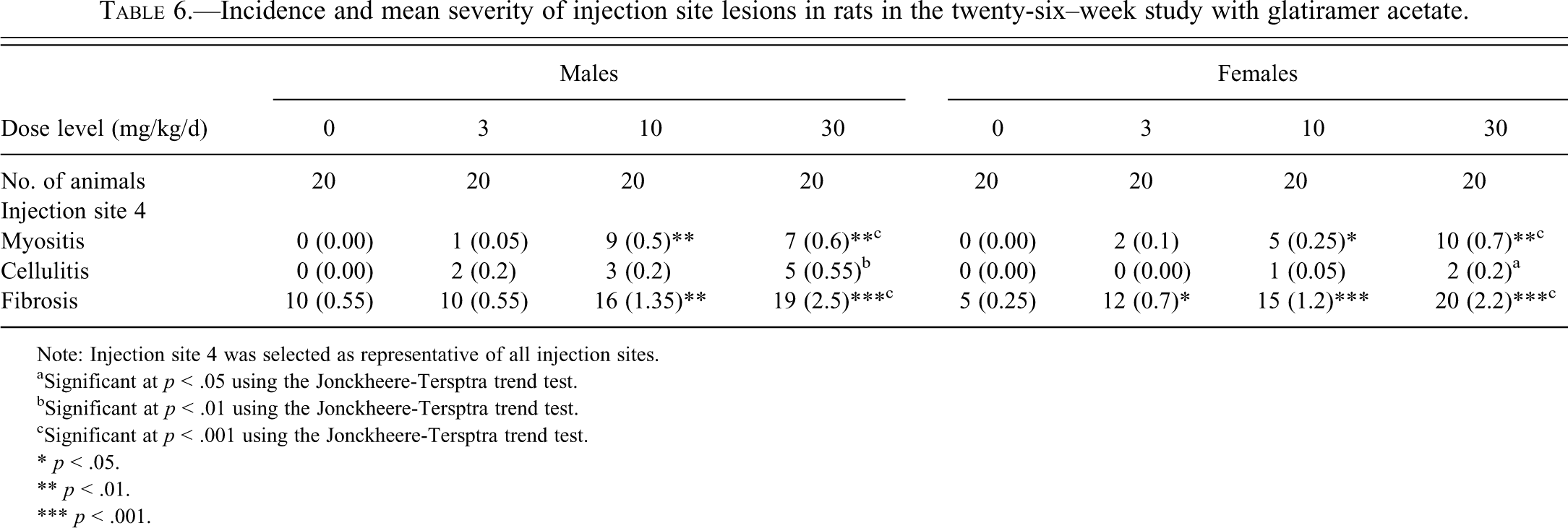
Note: Injection site 4 was selected as representative of all injection sites.
aSignificant at
bSignificant at
cSignificant at
*
**
***
Likewise, in the monkey study, treatment-related lesions were confined to the injection sites (Table 7 ) that were characterized by areas of dermal fibrosis, in which small-mononuclear-cell infiltration, occasionally giant cells, and rarely, lymphoid germinal follicles were present. In most animals, a minor diffuse eosinophilic infiltration was also present. All groups were affected (including controls), but there was a clear treatment-related increase in severity. There were also minor and less prevalent edematous, hemorrhagic, and necrotic lesions at the injection sites, which showed no clear association with administration of GA (Figures 4–7).
Incidence and mean severity of injection site lesions in monkeys in the 52–week study with glatiramer acetate.

Note: Injection site 1 was selected as representative of all injection sites.
aSignificant at
Protiramer Studies
Macroscopic examination at the termination of the rat study revealed enlarged livers and kidneys in the high-dose groups. Weighing of the organs confirmed this macroscopic observation, and the weights of the liver, kidney, and spleen were greater in animals treated at 40 and 300 mg/kg when compared with concurrent controls at the end of the treatment period.
In the histopathological examination of the rat tissues, treatment-related changes were seen in the injection sites, liver, and kidneys.
Injection Sites
Fibrosis was seen in all injection sites from all groups (Table 8 ). In control animals, the fibrosis was graded as minimal to slight, whereas in other treated groups, the fibrosis was graded as minimal to marked. Associated with fibrosis, especially at the high dose level, there were edema, thrombus formation, lymphoid cell infiltration, hemorrhage, neovascularization, and degeneration/regeneration of the panniculus skin muscle. In some animals, particularly at the high dose, these lesions were associated with subcutaneous necrosis, which occasionally formed a central necrotic cavity (Figures 8–11). As each injection site was used several times, the inflammation was a mix of chronic and subacute reactions. The changes were dose related, and the increase in severity in treated animals indicated poor local tolerance to the test item.
Incidence and mean severity of selected lesions in rats in the 26–week study with protiramer.
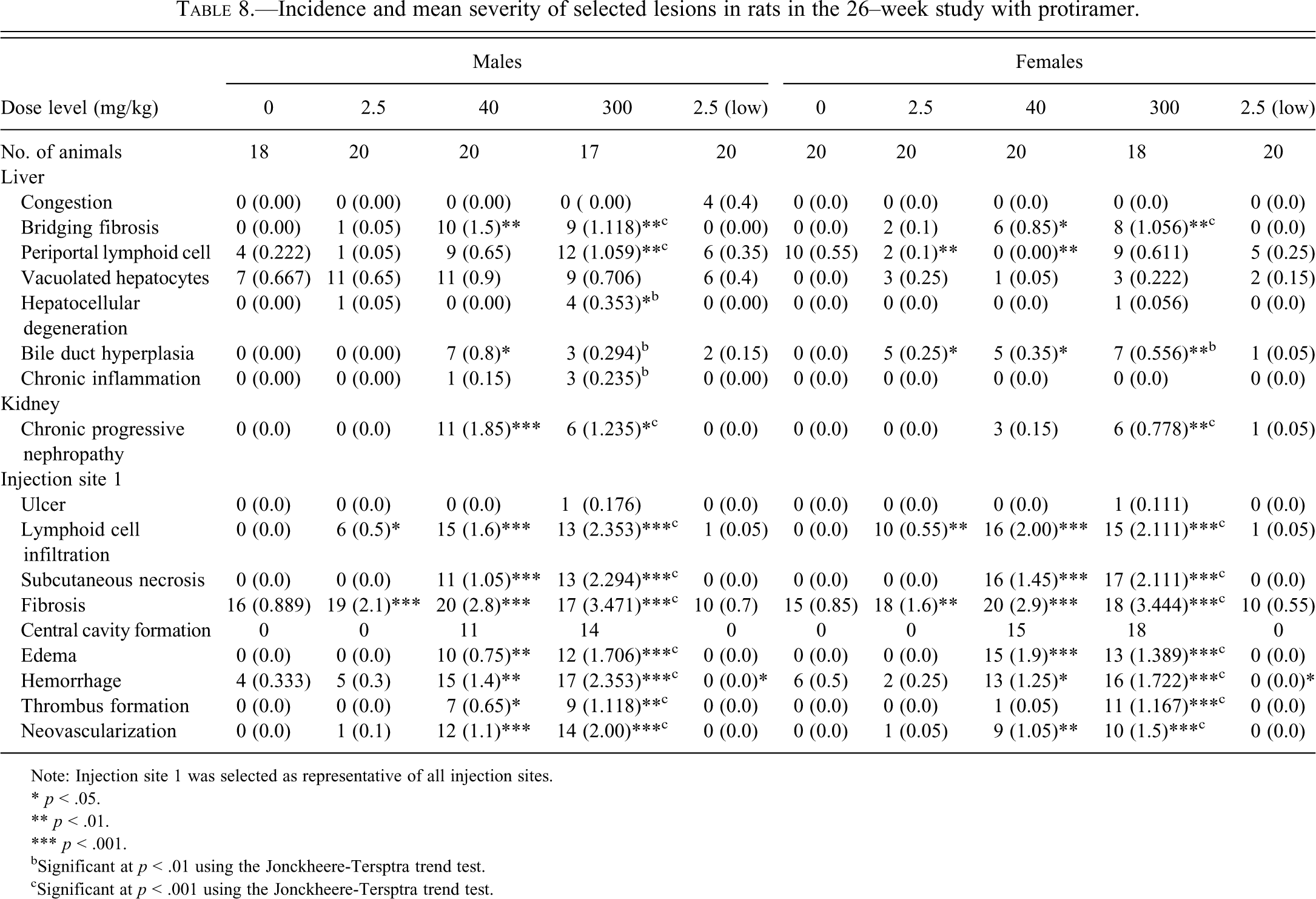
Note: Injection site 1 was selected as representative of all injection sites.
*
**
***
bSignificant at
cSignificant at
Following a four-week treatment-free period, previously treated animals had subacute to chronic inflammation associated with fibrosis, necrosis, and hemorrhage. The incidence and severity of these lesions were lower in recovery animals than in terminal sacrificed animals (data not shown).
Liver
Minimal to marked bridging fibrosis (Figure 12), minimal to moderate periportal lymphoid cell infiltration, minimal to moderate vacuolated hepatocytes, minimal to moderate bile duct cell proliferation, marked lobular necrosis, and/or minimal to moderate chronic inflammation were noted in treated animals with severity and incidence that were dose dependent. Low grade of some findings (periportal lymphoid cell infiltration, vacuolated hepatocytes, sinusoidal lymphoid cell aggregates) were also seen in some control animals, but these were far less pronounced or frequent than in animals treated at 40 or 300 mg/kg. The lesions were still evident at the end of the four-week recovery period, although the severity and incidence were lower.
In the low-dose, low-frequency group, one treated male showed multifocal necrosis in the liver, graded as slight, and two males and one female from the treated group showed minimal to slight bile duct cell proliferation.
Kidneys
Treatment-related findings in the kidneys were minimal to severe chronic progressive nephropathy (CPN) composed of tubular basophilia, tubular dilatation, lymphoid cell inflammation, tubular degeneration, and fibrosis (Figure 13 ). The effect was minimal in the low-dose group and appeared at a higher incidence and severity in the high-dose group and correlated with the larger kidneys observed in the macroscopic examination. At the end of the treatment-free period, there was only partial recovery.
In the monkey chronic toxicity study, microscopic changes observed at the injection sites in the two treatment-related decedent animals that died during treatment included fibrosis with neovascularization, slight focal areas of necrosis surrounding a central cavity, perivascular lymphoid infiltrate, eosinophilic infiltrate, hemorrhage, and vascular changes (thrombus formation, vascular necrosis, and endothelial edema/degeneration). Thrombus formation and/or intravascular eosinophilic material (considered to be an early stage of thrombus formation) were seen in various organs of one or both of these decedent animals, including the liver, lungs, heart, and kidneys. These changes were considered to be factors contributing to the deaths of these animals.
Scheduled histological examination of the injection sites was done at week 13 for the interim group, at week 52 for the main group, and at week 56 for the recovery group after four weeks of drug washout (Tables 9 and 10). In the control and low-dose/low-frequency group, there were only minor and infrequent observations of fibrosis and neovascularization at the end of treatment. In the other dose groups, there were more severe findings, typically consisting of minimal to marked diffuse fibrosis with neovascularization in most sites. In the most severe cases, these findings involved the skin muscle and/or the deep dermis, and they were occasionally associated with minimal to moderate focal area of necrosis surrounding a central cavity, particularly at the site of the most recent injection. Minimal to moderate subacute inflammation, composed mainly of lymphocytes with a perivascular distribution and occasionally minimal to moderate eosinophilic infiltrate, were also present. Minimal to moderate hemorrhage and/or edema were observed at all sites, and particularly at the site of the most recent injection. Vascular changes consisting of thrombus formation or stasis in vessels and vessel damage described as vessel necrosis or endothelial edema/degeneration of vessels were often seen in animals treated with 10 or 60 mg/kg (Figures 14–17). Occasionally observed pigmented macrophages were considered to be associated with hemorrhage and foreign body granuloma associated with the technique. The incidence and severity of the injection site lesions were low in animals treated at 2 mg/kg but greater in those treated at 10 or 60 mg/kg. The severity of the findings was generally dose dependent, except that the differences between the two high-dose groups was not always obvious, since the high dose was lowered at week 40 (60 to 30 mg/kg) because of the low tolerability, and new injection sites were introduced exclusively for this dose group. This change in design also obscures the difference between the interim analysis and the final analysis of week 52.
Incidence and mean severity of injection-site lesions in monkeys in the thirteen-week study with protiramer.
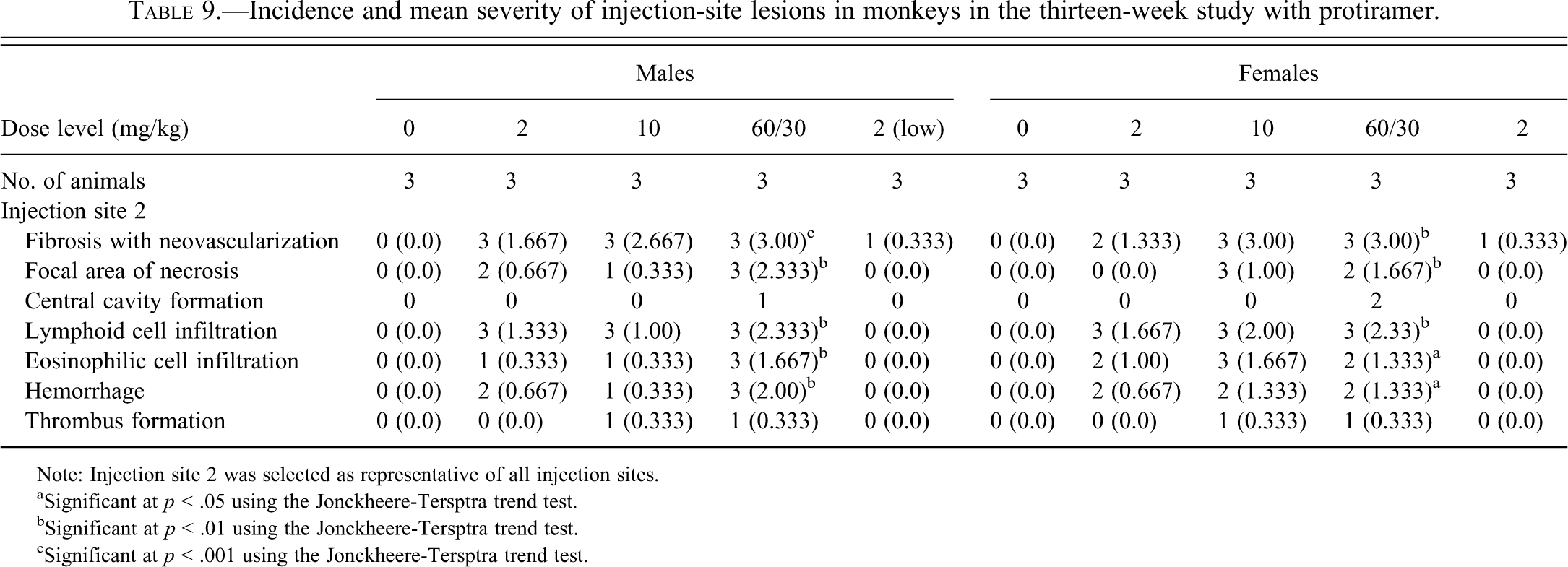
Note: Injection site 2 was selected as representative of all injection sites.
aSignificant at
bSignificant at
cSignificant at
Incidence and mean severity of injection-site lesions in monkeys in the 52–week study with protiramer.
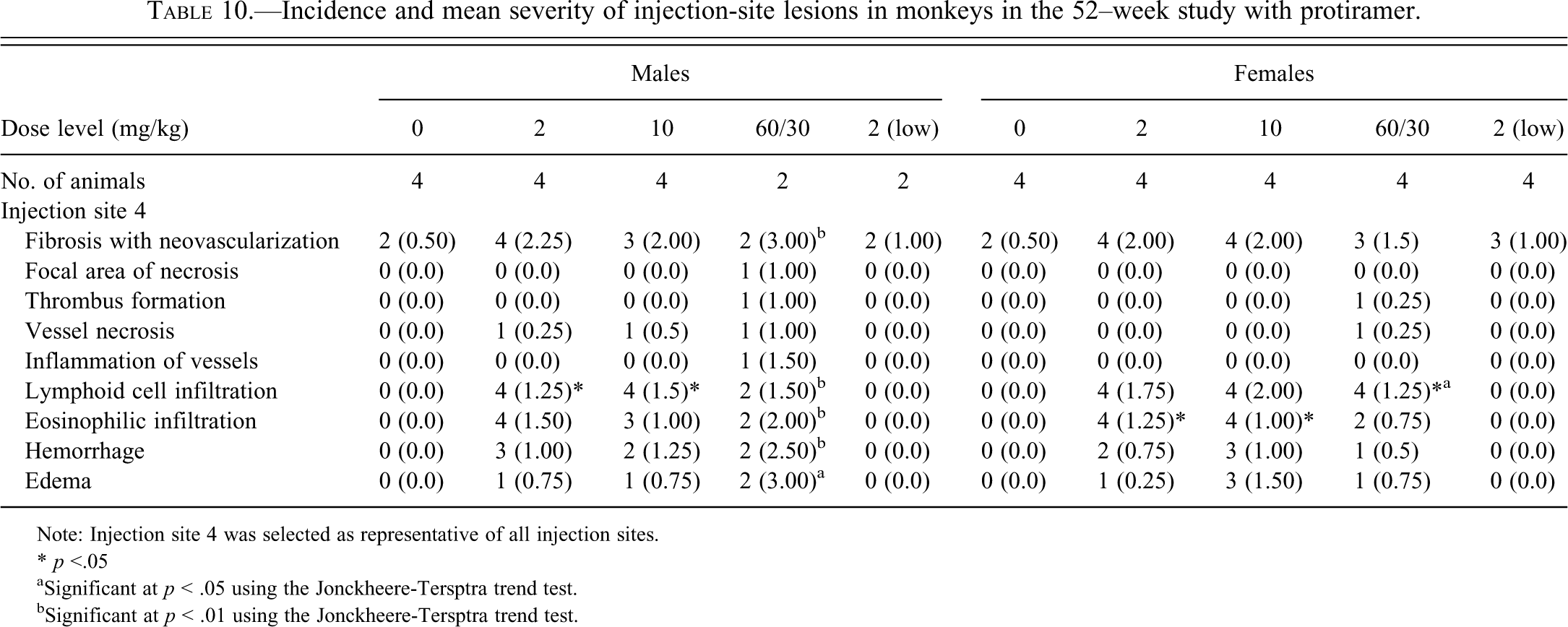
Note: Injection site 4 was selected as representative of all injection sites.
*
aSignificant at
bSignificant at
Following a four-week treatment-free period, previously treated animals had only chronic remnants of inflammation in the injection sites, suggesting progression to recovery (data not shown). In animals previously treated with 60/30 mg/kg, these effects included minimal to moderate fibrosis with neovascularization and minimal lymphoid infiltrates, mainly perivascular. In animals previously treated with 2 mg/kg every four weeks, there was minimal fibrosis with neovascularization at a slightly higher incidence than in controls. Pigmented macrophages were seen in females previously treated with 2 mg/kg every four weeks.
Discussion
In the present study, we aimed to systematically compare the toxicity of two glatiramoid compounds, GA and protiramer, in rats and monkeys. The two compounds share the same polymeric backbone of four amino acids, but they were prepared using a modification in the cleavage conditions of the primary polymer, yielding slightly longer chains for protiramer. Although similar in structure, the two glatiramoid molecules seem to have a different safety profile in animals. The most prominent adverse effect observed for both compounds was the induction of injection site lesions. However, whereas injection of GA was generally well-tolerated, the administration of protiramer caused severe injection site lesions composed of subcutaneous necrosis, vascular necrosis, cavity formation, and inflammation, which were evident after chronic administration for twenty-six to fifty-two weeks of dosing. In the more severe cases, these lesions were associated with mortality in both rats and monkeys, possibly related to vascular damage, hemorrhage, thrombus formation, and septicemia.
Comparison of the toxicity between the two glatiramoids is compromised by the fact that different regimens were employed in the two programs. GA was administered daily, whereas protiramer was administered twice weekly and in some cases even at lower frequency. Protiramer injections were also rotated between a larger number of injection sites than for GA, thus each site was used less frequently in the chronic studies. This low-frequency dosing regimen was compensated by the higher dose levels that were given with protiramer. The dose of 300 mg/kg protiramer that was given twice weekly to rats was clearly poorly tolerated, and a decision was made to terminate the treatment during the study. This dose is not comparable with any of the tested doses of GA, but it demonstrates what might be the toxicological consequences of high dose levels. The mid-dose of protiramer in rats was 40 mg/kg given twice weekly, and injection site lesions seemed to be more pronounced than those in GA animals administered once daily at a dose level of 30 mg/kg, even though the cumulative dose for GA is much higher considering the higher frequency. In monkeys, the doses were more balanced, especially since the high protiramer dose was reduced to the same level as the GA high dose, namely, 30 mg/kg. Taking into consideration both the more frequent dosing regimen of GA and the equivalence of the high dose in the monkeys, it is evident that animals were exposed to a larger cumulative dose of GA than protiramer, and nevertheless, the injection sites were much more pronounced in the protiramer-treated animals. These results demonstrate that the difference in tolerability is a true one, reflecting the greater intrinsic irritancy of protiramer, rather than just a reflection of the higher dose level. Vehicles used in the studies were either saline or an iso-osmotic solution of mannitol, vehicles that are inert and nonirritant. The dose volumes used in the studies were within the acceptable range of volumes typically used in toxicology studies. Severe injection site reactions were observed in an additional investigation carried out in pigs administered once or twice with subcutaneous protiramer (15–50 mg/kg). Two or six days following injection, findings were comparable with the ones observed in rats and monkeys (skin necrosis, cavity formation, and inflammation). It should be noted that in this study, a dosing volume of 1 mL per animal, which is much lower than in routine toxicology studies, was used. The fact that severe skin reactions were also observed with this low dosing volume suggests that the adverse reactions were not the result of the large volumes used in this study.
The data also indicate a progressive development of these lesions and repeat dosing to the same area aggravates the lesions. This finding is evident from the deteriorating tolerability of the animals to treatments, but it is not evident from the histological data of week 13 and week 52. The reason for this discrepancy is the lowering of the dose at week 40 of the study because of poor tolerability and the introduction of new injection sites.
Glatiramoids, both GA and protiramer, are immunogenic, and antidrug antibodies develop in all tested animals as well as in humans. The possibility that the effects are immune mediated was therefore investigated, but results have indicated that this is an unlikely explanation. This finding derives primarily from the observation that new injection sites that were introduced when reactions in the old sites were intolerable behaved as naïve sites, showing mild effects that progressed upon repetitive dosing. The appearance of eosinophilic infiltrates and the marked increase in the numbers of circulating eosinophils in monkeys that were exposed to protiramer is remarkable. The increase in circulating eosinophils was unique to protiramer, and no such systemic response was noted for GA, for which only small tissue eosinophilic infiltrates could be detected in some lesions. Theoretically, emergence of an eosinophilic response may indicate a hypersensitivity response, mediated by secretion of interleukin-5 by drug-specific T-cells (Pichler 2003; Posadas and Pichler 2007). However, measurement of antidrug antibodies of the IgE type did not reveal such a response, and we could not detect an increase in IL-5 or GMCSF secretion from peripheral blood mononuclear cells from these animals when they were stimulated in vitro with protiramer. The eosinophilic response is therefore not likely related to immunological sensitization but may resemble responses to parasites. The possibility of infestation with parasites was also investigated, but all tests were negative and the dose-response nature of the response is not consistent with such an infection.
In the clinic, injection site reactions are the most common adverse effects of Copaxone® administration in humans, and most patients report at least once some local reaction such as erythema (66% of patients), induration (13%), inflammation (49%), or pain (73%; Langer-Gould et al. 2004). However, these adverse effects are generally minor and infrequent, and they disappear spontaneously within hours or a few days (Soares Almeida et al. 2006). Incidence of these effects is reduced by rotation of the injection sites or by applying warm compresses before injection (Jolly et al. 2008). Rarely, panniculitis, mostly lobular, followed by lipoatrophy, appears in the area of injection (Drago et al. 1999; Edgar et al. 2004; Hwang and Orengo 2001; Mancardi et al. 2000; Soares Almeida et al. 2006; Soos et al. 2004).
The administration of protiramer in humans in early clinical trials was also connected to injection site reactions even though injections were done once weekly and not on a daily basis, as with Copaxone®. These adverse effects were observed in 50% of patients in the low protiramer dose (15 mg/wk) and 89% of patients in the higher dose (30 mg/wk; De Stefano et al. 2009).
It is not unusual for foreign injected materials to cause subcutaneous tissue injuries, and especially panniculitides, termed “factitial panniculitis” (Sanmartin et al. 2008). The exact mechanism of the cutaneous changes is not known, and several explanations have been suggested. The pathogenesis of panniculitis that was observed after GA administration was hypothesized to be a consequence of a direct toxic effect on adipocytes, leading to the observed inflammation (Soares Almeida et al. 2006).
According to Engelhardt (2008), the residence time of implants and proteins is a contributing factor in determining the magnitude of a local subcutaneous reaction. Other factors such as physical form of the protein, such as crystalline arrays, protamine incorporation, high sialic acid content, dose volume, and concentration also determine the rate and magnitude of a response. Accordingly, it is suggested that the severe subcutaneous tissue damage seen in the case of protiramer, in both rats and monkeys, is related to local extended permanence of the injected compound as a result of one of the above-mentioned factors. This damage eventually led to significantly more severe local irritation and extensive subcutaneous necrosis, which ultimately resulted in septic shock and death (Brook 2008; Mitchell 2004). It is also possible that the distribution of protiramer away from the injection site is slower than for GA because of the higher MW distribution, leading to a higher residence time and greater tissue damage.
In rats that were administered with protiramer, kidney and liver lesions were observed. These lesions were exclusive for protiramer and were not seen during the GA chronic studies. The lesions consisted of bridging fibrosis in the liver and severe chronic progressive nephropathy (CPN) in the kidneys. These two lesions correlated in severity with the injection site reactions. Similar lesions in the liver were previously described in rats and guinea pigs that underwent tight wrapping as part of a topical drug application process (Cushman et al. 1989; Nyska et al. 1992; Parker and Gibson 1995). This entity, which was termed “wrapping hepatopathy,” was suggested to be analogous to the “corset liver” found in women who wore tight corsets (Popper and Schaffner 1957). It was postulated that the pressure exerted by wrapping leads to systemic vascular congestion, which causes centrilobular hepatic degeneration and fibrosis (Nyska et al. 1992). The extensive subcutaneous inflammation and fibrosis that were observed in the treated rats may mimic this kind of pressure and thus lead to chronic hepatic congestion.
The accompanying CPN that was observed in the rats exposed to high protiramer doses was not reported in other studies describing wrapping hepatopathy, but it may be related to the overload of the kidney with short polypeptide chains that are the degradation products of protiramer. It is well documented that several natural or physiological factors influence the incidence and severity of CPN in rats, most prominently protein and caloric intake and male sex hormones (Hard et al. 2009). Although not seen for GA in the chronic toxicology study, some exacerbation of normal age-related glomerulonephritis was seen in the carcinogenicity study of GA after two years of daily dosing at the high dose level of 30 mg/kg (data not shown), thus indicating that this effect is not unique to protiramer, but that with protiramer it occurs much faster, more extensively, and at lower dose levels.
Protiramer and GA are relatively similar in their peptide size distribution, however they differ in their primary and secondary structures (Johnson 2010). Protiramer was originally designed based on the assumption that its higher molecular mass distribution would have increased immunoreactivity, thus leading to enhanced efficacy and decreased dosing frequency than for GA (Varkony et al. 2009).
GA is the active ingredient of Copaxone®, a drug that has been on the market for more than a decade. Today, after more then one million patient years of exposure, it is evident that this drug has a remarkable safety profile. This result is consistent with the preclinical safety studies of GA that showed that the drug is safe and well tolerated at doses that are sixteen-fold higher than the equivalent human dose.
Our detailed analysis of preclinical studies of protiramer clearly shows that safety is not guaranteed and cannot be extrapolated based on the safety profile of GA. Even slight changes in the manufacturing process of glatiramoids, which lead only to differences in the molecular weight distribution, without changing the ratio and overall composition of the mixture, can have major safety consequences. Although many differences in glatiramoids can be detected by analytical, biological, and immunological methods, the complex nature of GA as a mixture of polypeptides cannot guarantee that every difference is detected, or that the safety impact of every variation in composition can be extrapolated from closely related products.
The studies with protiramer also indicate that safety over a short time may be misleading, as the full extent of the damage was realized only in the chronic toxicity studies. In the case of protiramer, patients were exposed for a short period without manifesting overt adverse effects, but a decision was made to abort the program because of the progressive nature of the injection sites and the concern that long-term administration of this irritant material will lead to intolerable responses.
Footnotes
This work was performed at Teva Pharmaceutical Industries, Ltd, Petach Tikva, Israel. Funds for this project were provided in part by Teva Pharmaceutical Industries, Ltd, Petach Tikva, Israel. Some of the authors are employees of Teva Pharmaceutical Industries, Ltd, Petach Tikva, Israel.
