Abstract
Four groups of ten male and ten female rats each were treated intravenously with saline, 400 mg/kg/day of a commercially available injectable acetaminophen formulation, or 400 mg/kg/day of a new injectable acetaminophen formulation with (aged) or without (fresh) impurities daily for fourteen days. Gross observations of the mucosal surface of the stomachs from treated rats included multifocal to diffuse pale, elevated foci confined to the nonglandular region of the stomach. Treatment-related histologic observations consisted of epithelial hyperplasia and hyperkeratosis of the nonglandular mucosa of the stomach. The epithelial hyperplasia was characterized by a thickened epithelium, frequently accompanied by the development of undulations at the basement membrane zone, resulting in the formation of rete ridgelike structures protruding into the underlying tissue. The submucosa was usually expanded by edema and occasionally contained an infiltrate of neutrophils, eosinophils, and macrophages. The hyperplasia was usually accompanied by hyperkeratosis resulting in thickening of the stratum corneum. The incidence and severity of the gastric changes were similar across all treatment regimens. Although considered clinically irrelevant since humans do not have a forestomach equivalent, these results are significant in that this appears to be the first report of forestomach hyperplasia and hyperkeratosis following intravenous exposure to acetaminophen.
Introduction
A lesion of the nonglandular stomach (forestomach) in rats, consisting primarily of mucosal epithelial hyperplasia with or without hyperkeratosis, inflammation, and/or edema, has been a common finding in repeat-dose toxicology studies. In several instances, these histological changes have preceded forestomach tumor development in chronic studies (Altmann et al. 1985; Ghanayem et al. 1986). According to the Hazardous Substances Data Bank on TOXNET, this forestomach lesion has been associated with approximately 181 chemicals, but only with the oral route of administration. Although acetaminophen was not one of these chemicals, chronic oral exposure to acetaminophen has been reported to induce a similar stomach lesion in rats, specifically, hypertrophied stratified epithelium in the cardiac stomach (Boyd and Hogan 1968). The exact cause of the forestomach lesions has not been determined, but it has been attributed either to the local irritating effects of the chemicals per se (cytotoxicity) or to irritation associated with the gavage procedure itself, leading to a hyperplastic response. However, the presence of this lesion when chemicals were mixed in the feed or water (Hirose et al. 1987; Szabo et al. 1984), the lack of similar lesions in adjacent tissues (esophagus and glandular stomach), and the absence of this lesion in control rats gavaged with vehicle (Nomeir and Uraih 1988) challenge these explanations.
In the present study, acetaminophen was administered intravenously to normal Sprague-Dawley rats daily for fourteen days as part of the qualification process for impurities in new drug products (ICH 2003). Similar lesions of the forestomach were observed, characterized by epithelial hyperplasia, hyperkeratosis, and submucosal inflammation and edema. Although considered clinically irrelevant since humans do not have a forestomach equivalent, the present study is significant in that this appears to be the first report of this type of lesion in rats following intravenous exposure to acetaminophen.
Materials and Methods
The test article was an injectable acetaminophen formulation (paracetamol 1%, 10 mg/mL, Baxter International, Inc.) under development with (aged) or without (fresh) degradation product(s). The reference article was a commercially available injectable acetaminophen formulation (Perfusalgan, 10 mg/mL, Bristol-Myers Squibb). The control article was 0.9% sodium chloride injection, USP. Male and female Sprague-Dawley (HSD: Sprague Dawley SD) rats (Harlan Sprague Dawley, Oregon, WI) were from a colony that is barrier raised and certified by the vendor to be free of specified pathogens as determined by serology, bacteriology, and parasitology testing, were six to eleven weeks old and weighed 166–189 g at study initiation. Only rats showing no signs of clinical illness were used in this study. Animals were housed individually in suspended stainless-steel cages. Rats received water ad libitum, except during dosing, and Teklad Certified Rodent Diet (Harlan Teklad) ad libitum, except during dosing and fasting periods. Water is periodically analyzed for microbial and chemical contaminants, and there are no known contaminants in the feed or water that interfered with this study. All animal care and use procedures were approved by the institution’s animal care and use committee and were conducted in accordance with the Guide for the Care and Use of Laboratory Animals (National Research Council 1996).
Four groups of ten male and ten female rats each were administered 0 (saline) or 400 mg/kg of the test (aged or fresh) or reference articles daily for fourteen consecutive days via a tail vein by pump programmed to deliver approximately 30 mL/hour. Doses were administered at a volume of 40 mL/kg and adjusted weekly based on body weight. On day 15, fasted rats were anesthetized with approximately 3% isoflurane in oxygen and exsanguinated, and a gross necropsy was performed. Necropsy included examination of the external body surfaces, all orifices, cranial cavity and contents, thoracic cavity and contents, and abdominal cavity and contents. Forty-two tissues, including the stomach, were collected in 10% neutral buffered formalin with the exception of the testes, which were preserved in Bouin’s fixative. The fixed tissues were embedded in paraffin, sectioned, and stained with hematoxylin and eosin for histopathologic evaluation.
Results
Body Weight
Male rats treated with acetaminophen exhibited significant reductions in weight gain compared to the controls (Table 1 ). Female rats given fresh or aged test article gained less weight than the controls during the first week (Table 1). The lower weight gains were consistent with lower feed consumption showing similar group differences (data not shown).
Changes in body weight (g) over time for control rats and rats treated with acetaminophen (mean ± SE).
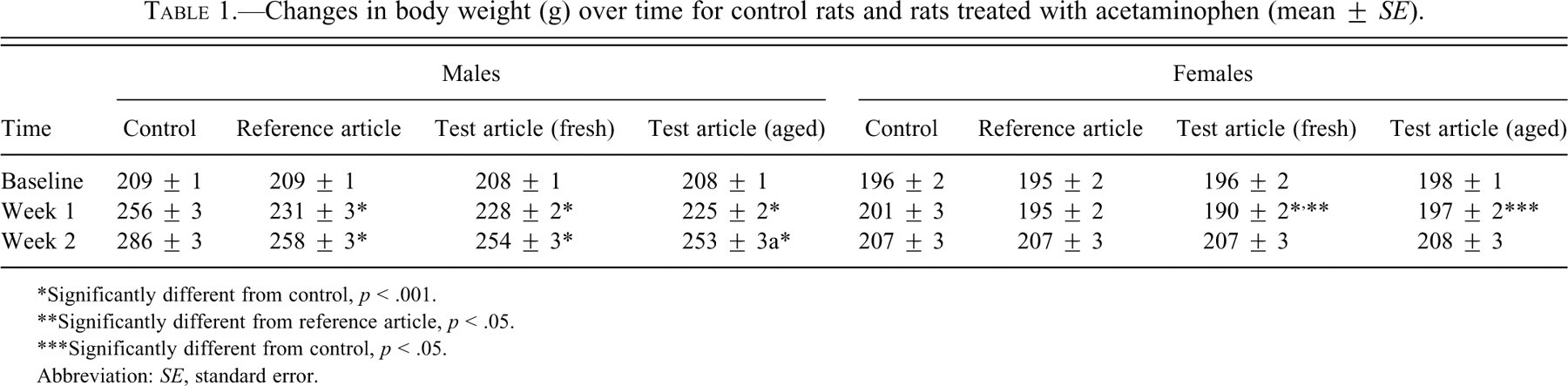
*Significantly different from control, p < .001.
**Significantly different from reference article, p < .05.
***Significantly different from control, p < .05.
Abbreviation: SE, standard error.
Gross Pathology
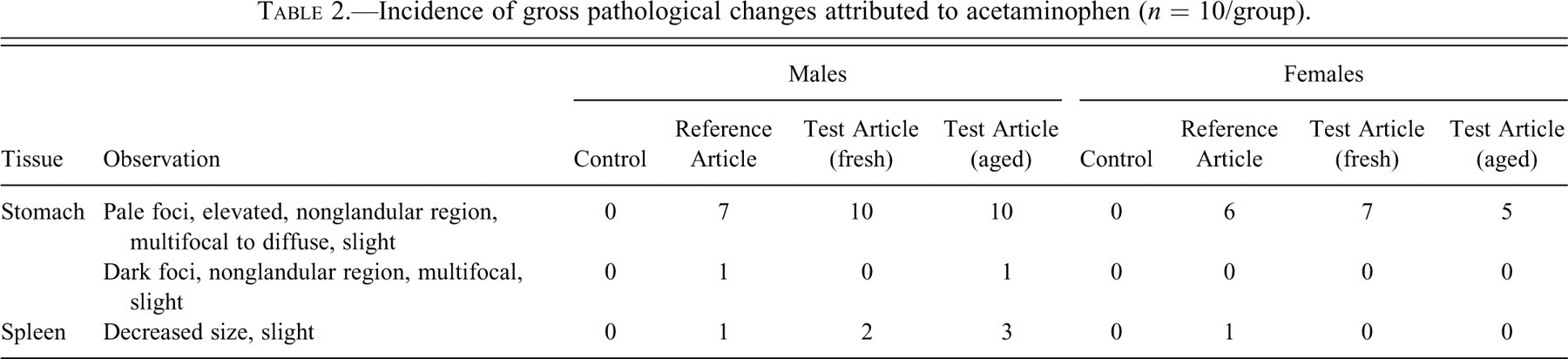
Acetaminophen-related gross observations were limited to the stomach and spleen (Table 2 ). The mucosal surface of the stomachs from rats in the test and reference article groups had multifocal to diffuse, pale, elevated foci confined to the nonglandular region. The pale, elevated foci were grossly detectable in seven of ten males and six of ten females given the reference article. The lesion was detected in all males given the test article (aged and fresh), seven of ten females given the fresh test article, and five of ten females given the aged test article, and tended to be more extensive (diffuse) in males given the aged test article. In addition to the pale elevated foci, two males, one given reference article and the other given aged test article, had dark foci on the gastric nonglandular mucosa. Decreased splenic size was grossly apparent in one male and one female given reference article, two males given fresh test article, and three males given aged test article.
Incidence of gross pathological changes attributed to acetaminophen (n = 10/group).
Histopathology
Acetaminophen-related histologic observations were limited to epithelial hyperplasia and hyperkeratosis of the nonglandular mucosa of the stomach, and a constellation of subtle changes in the thymus, spleen, and bone marrow consistent with a minor down-regulation of hematopoiesis.
Acetaminophen-related gastric histologic observations consisted of epithelial hyperplasia and hyperkeratosis of the nonglandular mucosa of the stomach (Table 3, Figure 1 ). The incidence and severity of the gastric changes were similar across all treatment regimens, reference and test (aged and fresh). The epithelial hyperplasia in the nonglandular stomach was characterized by a thickened epithelium frequently accompanied by the development of undulations at the basement membrane zone resulting in the formation of rete ridge-like structures protruding into the underlying tissue. The underlying submucosa was usually expanded by edema and occasionally contained an acute inflammatory infiltrate of neutrophils, eosinophils, and macrophages. The hyperplasia was usually accompanied by hyperkeratosis resulting in thickening of the stratum corneum. Occasionally, the keratin laminations in the stratum corneum separated, resulting in the formation of intracorneal bullae containing proteinaceous material or blood (Figure 1). These bullae were considered responsible for the dark foci detected grossly in two rats. In addition to these changes, gastric erosion was detected in one male given the aged test article and one female given the fresh test article. The incidence and severity of the gastric epithelial hyperplasia and accompanying changes were slightly more prominent in males relative to females, but they were similar among groups given reference and test article (aged and fresh). Gastric epithelial hyperplasia was detected in nine of ten males and eight of ten females given reference article and all males and nine of ten females per group given test article (aged and fresh).
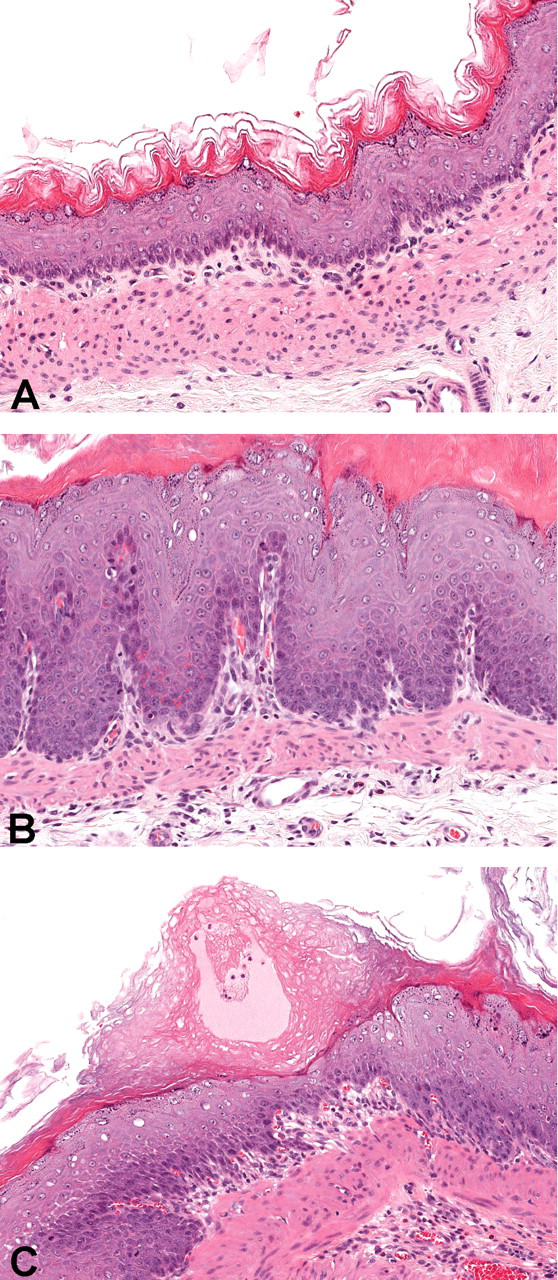
The nonglandular region of the stomach from rats exposed to saline or 400 mg/kg acetaminophen intravenously daily for fourteen days. (A) Saline-treated control animal. (B) Acetaminophen-treated animal demonstrating hyperkeratosis and epithelial hyperplasia with the development of undulations at the basement membrane zone resulting in the formation of rete ridge-like structures. (C) Stomach from acetaminophen-treated animal demonstrating separation of keratin laminations in the stratum corneum resulting in the formation of intracorneal bullae containing proteinaceous material.
Incidence of histopathological changes in the stomach attributed to acetaminophen relative to other parts of the gastrointestinal tract (n = 10/group).

In contrast to the acetaminophen-induced lesion of the nonglandular mucosa of the stomach, no gross or histologic changes in any other part of the gastrointestinal tract were attributable to acetaminophen (Table 3).
Other than the lesion in the nonglandular mucosa of the stomach, acetaminophen-induced changes included minor changes in the thymus, spleen, and bone marrow. Specifically, the observations were decreased splenic extramedullary hematopoiesis, increased adipose cells in the bone marrow, and early atrophy in the thymus. These changes were considered subclinical, since they were not reflected in detectible alterations in the peripheral blood hemogram (data not shown).
Extramedullary hematopoiesis (EMH) in the spleen and, to a lesser extent, the liver is a common histologic finding in rodents. Decreased EMH in the splenic red pulp, relative to controls, was detected in approximately half (four to six/group of ten) the males given reference article or test article (fresh or aged). This finding was not apparent in female rats. Although inconclusive, it should be noted that EMH in the liver was detected in three of ten control males but was detected in only one of thirty males given the reference or test articles (fresh or aged).
The presence of adipose tissue (inactive marrow) in the bone marrow suggests a relative decrease in active, hematopoietic marrow. Increased adipose cells in sternal bone marrow was detected in five of ten males and nine of ten females given the reference article, nine of ten males, and eight of ten females given fresh test article, and nine of ten males and six of ten females given aged test article. This finding was also present in one of ten control males.
The early thymic atrophy was characterized histologically by an increased number of tingible-body macrophages in the thymic cortex without noticeable architectural change or involution. This was the finding in three to five males/group of ten given the reference article or the test article (fresh or aged) and in seven of ten females given the reference article. The incidence in females given the test article (fresh or aged) was comparable to female controls (two of ten).
Discussion
In this study, acetaminophen-induced histologic changes in rats included epithelial hyperplasia and hyperkeratosis of the nonglandular mucosa of the stomach, and a constellation of subtle changes in the thymus, spleen, and bone marrow. Collectively, the histologic observations in the thymus, spleen, and bone marrow were interpreted as an indication of a physiologic, minor down-regulation of hematopoiesis and are consistent with a previous report on the toxicity of acetaminophen in rats (Boyd and Hogan 1968). The epithelial hyperplasia and hyperkeratosis of the nonglandular mucosa of the stomach, however, is the novel finding in this study, as this appears to be the first report of this type of proliferative lesion induced in rats following intravenous exposure to acetaminophen.
Two other chemicals have been reported to induce similar proliferative lesions of the forestomach following intravenous exposure. A single intravenous dose of alloxan, a nongenotoxic diabetogenic chemical, induced proliferative lesions of squamous epithelium in the tongue, esophagus, and forestomach of male and female WBN/Kob rats; these lesions progressed to squamous cell carcinoma in 20% of the rats (Kodama et al. 2006). WBN/Kob rats are an inbred strain that spontaneously develops long-lasting diabetic symptoms. Three years later, the same group reported similar findings in non-diabetic–prone F344 rats treated with a single intravenous dose of alloxan (Sano et al. 2009). However, based on the earliest necropsy time point of fifty and twenty-five weeks, respectively, it could not be determined whether the lesions were a result of the direct effects of the alloxan injection or caused by secondary factors owing to the pathophysiological changes associated with the long-lasting diabetic condition. Aristolochic acid administered intravenously to both rats and mice caused superficial ulceration of the forestomach followed by hyperplasia and hyperkeratosis of the squamous epithelium within fifteen days of exposure (Mengs 1987). The exact mechanism underlying the proliferative responses observed in these citations was not determined.
The literature suggests that the rat forestomach is a target tissue for many xenobiotics. The nature of this tissue specificity may be explained, in part, by biochemical changes or differences unique to the forestomach. The daily oral administration of dimethyl hydrogen phosphite (DMHP) to Fischer 344 rats at 200 mg/kg by gavage for six weeks induced histological changes in the forestomach, including thickening of the mucosa, epithelial hyperplasia, hyperkeratosis, subepithelial inflammation, and occasional submucosal intercellular and intracellular edema (Nomeir and Uraih 1988). No histological changes were observed in the liver, lungs, kidneys, or glandular stomach. The forestomach lesion did not appear to be caused by irritation associated with the gavage procedure since there were no forestomach lesions in the control group gavaged with vehicle. Tissue levels of cytochrome P450 and the activities of microsomal P-nitroanisole demethylase, soluble glutathione S-transferase, and soluble superoxide dismutase were comparable, which suggests these enzymes were not involved in the induction of the lesion. The level of soluble nonprotein sulfhydryls (NPSH), however, was significantly elevated in the treated rats as compared with the control rats and was specific to the forestomach. In addition, carboxylesterase activity was significantly reduced in the forestomach of treated rats as compared with the control rats. A significant reduction in carboxylesterase activity was also demonstrated in the forestomach after a single intravenous administration of 1,000 mg/kg, which indicates that reduced carboxylesterase activity was not a localized phenomenon caused by oral administration and can be affected by other routes of exposure.
A role for NPSH in the development of forestomach lesions was supported by Hirose et al. (1987). Pretreatment of rats with diethylmaleate (DEM), a glutathione (GSH)-depleting agent, completely inhibited butylated hydroxyanisole (BHA)–induced forestomach hyperplasia when administered in the feed for four weeks. These findings are consistent with Ghanayem et al. (1991), who evaluated the effects of sulfhydryl modulation on ethyl acrylate (EA)–induced forestomach lesions in rats. Because EA and its metabolite undergo glutathione conjugation in vivo, which may result in their detoxification, it was hypothesized that modulation of endogenous sulfhydryls may affect EA-induced forestomach toxicity. To the contrary, it was demonstrated that the intraperitoneal co-administration of sulfhydryl-containing compounds (cysteamine and cysteine) potentiated, whereas sulfhydryl depletion by subcutaneous DEM decreased forestomach edema caused by a single oral administration of EA by gavage. Cysteamine and cysteine also potentiated the severity of forestomach mucosal hyperplasia produced by daily oral administration of EA for fourteen days. Szabo et al. (1984) observed a prominent elevation of NPSH of the gastroduodenal mucosa after chronic administration of acrylonitrile, linking the change in NPSH to proliferative changes observed in the gastric mucosa. The mechanism by which NPSH exacerbates chemically induced forestomach lesions in rats may be similar to the carcinogenicity induced in the rat forestomach by ethylene dibromide (ED). Whereas conjugation with glutathione usually detoxifies chemicals and facilitates excretion by increasing their water solubility, ED is metabolized by glutathione S-transferase to glutathione S-ethylbromide, a known carcinogen that alkylates DNA at the N 7 position of guanine (Koga et al. 1986). In addition to the glutathione conjugates, cysteine S-conjugates also have been shown to be mutagenic (Monks et al. 1990). This characteristic does not explain, however, why epithelial hyperplasia, hyperkeratosis, and submucosal inflammation and edema caused by many chemicals are primarily localized to the forestomach when NPSH are ubiquitous.
The major metabolites of acetaminophen involve non-oxidative metabolism to the glucuronide and sulfate conjugates. In rats, glucuronide and sulfate conjugates accounted for, on average, 46% and 31%, respectively, of the dose excreted in the urine zero to forty-eight hours following an intravenous dose of 2 mmole/kg (approximately 500 mg/kg) of acetaminophen (Hart et al. 1982). However, a portion of the acetaminophen is oxidized to N-acetyl-benzoquinoneimine (NAPQI) by cytochrome P450. Although several isoenzymes (CYP2E, CYP1A, CYP3A) contribute to the formation of NAPQI, it is widely considered that CYP2E1 is the major source of NAPQI in humans (Manyike et al. 2000). This assumption is based on the conclusion that disulfiram, through its active metabolite diethyldithiocarbamate, is a selective inhibitor of CYP2E1 and decreased the urinary excretion of the metabolites of NAPQI by about 70% (Manyike et al., 2000). Cytochrome P450 is also responsible for the oxidation of acetaminophen to NAPQI in rats (Lee et al. 1991). N-acetyl-benzoquinoneimine is a cytotoxic electrophile that binds covalently to cellular macromolecules including proteins and DNA and also causes production of reactive oxidative species. The formation of NAPQI is believed to be responsible for the characteristic hepatotoxicity following acetaminophen overdose. This toxic metabolite is normally conjugated to GSH by glutathione S-transferase and in the process detoxified, but NAPQI accumulates when GSH levels are inadequate. Inadequate cellular concentrations of GSH, therefore, would be expected to promote acetaminophen toxicity. Indeed, chemical-induced depletion of GSH has been shown to potentiate acetaminophen-induced hepatotoxicity in mice (Kaplowitz 2005).
The present study suggests that the squamous epithelial cells of the forestomach may be more sensitive or susceptible to acetaminophen relative to other epithelial cell types in the other parts of the gastrointestinal (GI) tract. The squamous epithelial cells of the forestomach may preferentially uptake acetaminophen (or its metabolites) and/or may differ in their metabolism of acetaminophen. It is also possible that the effect on squamous epithelial cells of the forestomach is mediated through an extra-epithelial or indirect mechanism (e.g., paracrine signaling from neighboring cells), though there is no evidence in the literature to support such a mechanism.
However, evidence does exist to suggest that the forestomach lesion observed in the present study may be caused by metabolic or biochemical differences in resident cells that favor acetaminophen toxication versus detoxication. Based on previous discussion, it is reasonable to assume that the toxicity of acetaminophen in various tissues would depend, in part, on the relative expression of cytochrome P450 and other relevant enzymes, as well as the relative concentrations of GSH. The balance between these toxication and detoxication processes will ultimately determine the relative toxicity of acetaminophen among different tissues. Cytochrome P450 is constitutively expressed in tissues of the upper alimentary tract of rats including the esophagus, forestomach, and glandular stomach (Farinati et al. 1989). They also demonstrated that cytochrome P450 expression was significantly induced with ethanol and was observed only in the forestomach. More notable was the observation that, as early as two hours after administration and at doses of 500 mg/kg and above, acetaminophen itself induced the expression of three acetaminophen-metabolizing enzymes (CYP1A2, CYP3A11 and GSTPi). GSTPi is a glutathione S-transferase isoform that is particularly effective at inactivating NAPQI. Farinati et al. (1989) also evaluated GSH concentrations in the upper alimentary tract tissues of rats and noted that the forestomachs from controls exhibited the lowest levels of GSH of the three tissues evaluated (i.e., esophagus, forestomach, and glandular stomach). This observation is in agreement with Potter and Tran (1993), who investigated the apparent first-order rate constants for GSH turnover in various rat tissues and determined that the forestomach had the lowest rate of GSH synthesis (<0.1% of the total) for all sixteen tissues evaluated, which included the glandular stomach, duodenum, small intestine, cecum, large intestine, and colon. The synthesis of GSH in the forestomach, expressed in µmol/g/hour, was approximately nine times lower as compared with the glandular stomach and fifty-one times lower as compared with the liver. It has also been shown that the forestomach of untreated rats expressed lower levels of UDP-glucuronic acid transferase and glutathione transferase as compared with the glandular stomach (Farinati et al. 1989). The former observation suggests that acetaminophen in the forestomach would be less likely to be conjugated and would be more available for oxidation. The latter observation, along with the lower GSH levels, suggests that oxidative metabolites such as NAPQI would be less likely to be conjugated with glutathione and be more available for interaction with cellular macromolecules. In fact, radiolabeled acetaminophen administered to mice, at doses (10–400 mg/kg) equivalent to those commonly used therapeutically in humans, formed covalent adducts with hepatic and renal DNA (Rogers et al. 1997). Furthermore, the addition of radiolabeled NAPQI to purified DNA produced measurable levels of DNA alkylation.
The exact cause of the proliferative forestomach lesion induced by acetaminophen in the present study is not known. Perhaps the cell proliferation was the result of low-grade, non-necrogenic cytotoxicity, as cellular necrosis was not a histologically detectable feature. This is a well-documented pathophysiological response. For example, it has been shown that a low, non-necrogenic dose of carbon tetrachloride (CCl4) administered to rats produced a rapid proliferation in hepatocytes, which was thought to be instrumental in the prevention of necrosis. When the early proliferative response to CCl4 was blocked with chlordecone, the non-necrogenic dose of CCl4 caused significant necrosis (Soni and Mehendale 1998).
In conclusion, this is the first report of a forestomach lesion, characterized by epithelial hyperplasia and hyperkeratosis of the nonglandular mucosa, in normal rats following intravenous exposure to acetaminophen. The exact cause of the acetaminophen-induced forestomach lesion is not known, as the purpose of the present study was to qualify impurities following ICH Guideline Q3B(R1) (2003). Other works discussed herein, however, suggest that the ability of acetaminophen to induce the expression of enzymes promoting its own oxidation, coupled with an inherently lower capacity of the rat forestomach to detoxify acetaminophen and its toxic metabolite NAPQI, may be causative factors and serve as a starting point for future mechanistic investigations.
Footnotes
Abbreviations
Acknowledgments
We would like to thank Dolores Fischer and Amy Romano for their support in the generation of this manuscript.
