Abstract
Neonatal exposure to estrogenic chemicals causes irreversible complex damage to the hypothalamus–pituitary–gonadal axis and reproductive system in females. Some lesions are noted after maturation as delayed adverse effects. We investigated the characteristics and dose dependence of delayed effects using female rats neonatally exposed to diethylstilbestrol (DES). Female Donryu rats were subcutaneously injected with a single dose of DES of 0 (control), 0.15, 1.5, 15, 150, or 1,500 µg/kg bw after birth. All except the lowest dose had estrogenic activity in a uterotrophic assay. All rats at 1500 µg/kg and some at 150 µg/kg showed abnormal morphologies in the genital tract, indicating they were androgenized before maturation. Although no morphological abnormalities were noted at 15 µg/kg or lower, onset of persistent estrus was significantly accelerated in the 1.5, 15, and 150 µg/kg groups with dose dependency, and the latest onset was from seventeen to twenty-one weeks of age at 1.5 µg/kg. The neonatal exposure to DES increased uterine adenocarcinoma development only at 150 µg/kg, although uterine anomalies were detected at 1,500 µg/kg. These results indicate that neonatal exposure to DES, which exerts estrogenic activity in vivo, induces delayed adverse effects in female rats in a dose-dependent manner. Early onset of persistent estrus appears to be the most sensitive parameter.
Introduction
Damage to the hypothalamus–pituitary–gonadal axis is the most worrying issue with perinatal exposure to chemicals that have estrogenic activity, because changes caused during the developmental period can last throughout life. Effects of exposure may be qualitatively different from those experienced in adulthood in rodents as well as human beings (Takasugi et al. 1983; Newbold et al. 1999). Although species differences in neonatal effects are known—such as ovary-independent response to the vagina in mice but not rats (Katsuda et al. 2002; Takasugi et al. 1976) or adenomyosis induction by tamoxifen in mice (Parrott et al. 2001) but uterine cancer in rats (Carthlew et al. 2000)—many experimental data on perinatal exposure to estrogens using rodents are considered likely to be relevant to human beings (Herbst and Bern 1981).
Perinatal exposure to estrogens or estrogenic compounds during critical period disrupts hypothalamus function, which results in lower gonadotropin (FSH and LH) production (Katsuda et al. 2000; Yoshida et al. 2002a). The disruption leads to many complicated abnormalities in the hypothalamus–pituitary–gonadal axis and the genital tract, and direct effects of estrogens on the genital system are also added. Although the abnormalities are dependent on exposure period, time, and dose, they are called androgenization and are commonly irreversible. Typical changes in affected females include lower gonadotropins, anovulation, persistent estrus, abnormal organogenesis of the reproductive tract, abnormal expression of estrogen receptors in the uterus, or male-type sexual behavior. Testosterone is known to be converted to E2 by aromatase in the brain, and it has effects that are similar to those of estrogens. These abnormalities are usually detectable immediately after treatment or before puberty. Recently, effects of perinatal exposure that manifest after maturation have attracted attention (Adewale et al. 2009; Yoshida et al. 2002b). Such delayed effects of estrogenic chemicals were previously termed the “delayed anovulatory syndrome” (DAS) (MacLusky and Naftolin 1981), characterized by early onset of persistent estrus as determined by vaginal cytology (Ninomiya et al. 2007). Enhancement of tumor development may also result as well, as has been documented for uterine and vaginal neoplasms after perinatal exposure to diethylstilbestrol (DES) in mice (Newbold et al. 1990) as well as young women (Herbst and Bern 1981). Our previous study also showed increased uterine cancer development in rats with neonatal high-dose p-tert octylphenol (OP) (Yoshida et al. 2002b). Such effects are hypothesized to be caused by some disruption in the hypothalamus (MacLusky and Naftolin 1981), but precise mechanisms and associated characteristics other than persistent estrus (PE) remain to be determined. For risk assessment of chemicals, delayed adverse effects are of particular concern because they might be overlooked by short-term bioassays or developmental toxicity studies. Therefore, the present study was performed to clarify characteristics of delayed effects of neonatal exposure to DES at various doses on the female reproductive tract in rats using multiple retrospective indicators, including morphological parameters, estrous cyclicity, and uterine carcinogenesis. In the present study, we used Donryu rats, which characteristically show a regular four-day estrous cycle as young adults and a high rate of endometrial adenocarcinomas caused by ovarian hormone imbalance (Maekawa et al. 1999; Nagaoka et al. 1990; Nagaoka et al. 1994). To avoid accumulation of DES in the rat body with repeated treatment, single-dose administration was selected in the present study.
Materials and Methods
Animals and Housing Conditions
Nonpregnant adult and pregnant female Crj:Donryu rats were purchased from Charles River Japan (Kanagawa, Japan), maintained in an air-conditioned animal room under constant conditions of 24°C ± 2°C and 55% ± 10% humidity with a twelve-hour light/dark cycle (light, 8:00 a.m.–8:00 p.m.; dark, 8:00 p.m.–8:00 a.m.), and housed three or four per cage. Commercial pellet diet (CRF-1, Oriental Yeast, Kanagawa) and drinking water were available ad libitum throughout the studies. Animal care and use followed the guidelines of laboratory animal welfare and use according to the Sasaki Institute Ethical Committee.
Chemicals
Diethylstilbestrol (CAS number 56-53-1, Sigma, St. Louis, MO) was dissolved in dimethyl sulfoxide (DMSO, Wako Pure Chemical, Osaka, Japan).
Experiment 1
Nonpregnant females underwent uterotrophic assays to examine estrogenic activity of the single doses applied in experiment 2 in vivo. Thirty-seven animals at three or four months of age were ovariectomized, and after complete castration was confirmed by vaginal smears, they were allocated to six groups. Three to eight animals per group were subcutaneously treated with a single dose of DES at 0 (control), 0.15, 1.5, 15, 150, or 1,500 µg/kg body weight in 0.1 mL/100 g body weight of DMSO. Twenty-four hours after treatment, all animals were euthanized under deep anesthesia and uterine weights were measured.
Experiment 2
Experiment 2 was conducted to characterize delayed effects of neonatal exposure to DES on the female reproductive tract and their dose dependence. Dams were assigned to one of six groups (six to eight per group) before delivery. Female offspring were treated with a single subcutaneous injection of DES at 0, 0.15, 1.5, 15, 150, or 1,500 µg/kg body weight in DMSO at 0.04 mL/10 g body weight in their back skin within twenty-four hours after birth. At postnatal days (PND) 14 and 21 (at weaning) and five weeks of age, three or four rats per group at each time point were examined to detect early effects on the female reproductive organs. The animals examined at five weeks of age were at estrous stage by vaginal smear. Remaining animals were checked for clinical signs and body weights throughout their life. After vaginal opening (VO), which is a time point during puberty, all animals were checked for estrous cyclicity by vaginal smear every other week throughout the experiment. Additionally, to examine the effects of the neonatal exposure to DES on uterine carcinogenesis, fifteen to twenty-four rats per group were treated with a single injection of 20 mg/kg N-ethyl-N′-nitro-N-nitrosoguanidine (Nacalai Tesque, Kyoto, Japan) into the uterine horns via the vagina using a stainless steel catheter at seven weeks of age. This treatment is known to cause uterine endometrial adenocarinoma development in the uterine corpus of rats without carcinogenic effects in other sites, with no disruption of estrous cyclicity (Ando-Lu et al. 1994). All survivors were euthanized at ten months of age in the control and 15 µg/kg and higher treated groups, and at eight months of age in the 0.15 and 1.5 µg/kg groups.
Pathology
After complete necropsy of all rats in experiment 2, the ovaries, uterus, vagina, adrenals, liver, pituitary, thymus, brain, and sites with macroscopic abnormalities were fixed in 4% neutral buffered formaldehyde. All organs were routinely processed and stained with hematoxylin and eosin for histopathological examination. Number of uterine glands in the uterine horn were counted using four or five cross-sections (Katsuda et al. 2000). For the uterus at termination of eight or ten months of age, the main target organs of carcinogenic effects of DES—the upper, middle, and lower parts of each uterine horn and the cervix—were cut into three pieces each in cross-section. Preneoplastic or neoplastic lesions detected in each uterus were classified into three degrees of atypical hyperplasia (slight, moderate, or severe) and adenocarcinomas, according to the criteria described in our previous studies (Ando-Lu et al. 1994). Lesions composed of glandular-structured epithelial cells with atypia showing invasive proliferation to the muscle layer or serosa were diagnosed as endometrial adenocarcinomas.
Immunohistochemistry
Serial sections of the uterus, vagina, and ovary at PND14, PND21, and five weeks of age were stained immunohistochemically with the antibody of estrogen receptor (ER)-α (SC7027, Santa Cruz, CA, USA). The organs of three or four rats per group were analyzed, and immunoreactive complexes were detected by the avidin-biotin complex method (LSAB kit, Dako Japan, Kyoto, Japan) and visualized with 3-3′-diaminobenzidine tetrahydrochloride (Wako Pure Chemicals, Osaka, Japan) as the chromogen. Immunohistochemical intensity of ERα expression in these organs was classified into four categories: 0, negative; 0.5, several positive cells in higher magnification field (40×); and 1.0, 2.0, or 3.0 represents about 10–30%, 30–50%, or 50% and higher positive cells in the high magnification, respectively.
mRNA Expression of Estrogen Receptors α and β and Progesterone Receptor (PR) in the ovary and uterus
At five weeks of age, total RNA samples obtained from samples of uterine tissues (about 100 mg) and unilateral ovary of three females at estrous phase in each group except the highest dose were isolated using RNeasy Midi Extraction Kit (QUIAGEN, Germany) and stored at −80°C until reverse transcriptase polymerase chain reaction analysis. Estrogen receptors α and β and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) mRNA transcription were examined using 400-ng aliquots of total RNA. The primers were synthesized and purified by Takara Bio Inc. (Shiga, Japan) based on previous reports (Azabo et al. 2000; Pillai et al., 2002). Expressed levels of ERα, ERβ and PR mRNA relative to GAPDH mRNA expression were compared among the control and the treated groups.
Statistical Analysis
Values for incidences were statistically analyzed using the Fisher exact probability test. Other data were assessed using analysis of variance (three groups) and t test (two groups), and post hoc comparisons between the treated and control groups were made with the Dunnett t test. Statistical significance was considered as p < .05.
Results
Estrogenic Activity in Experiment 1
The results of uterotrophic assays are summarized in Table 1. Both the absolute and relative uterine weights were significantly increased at 1.5 µg/kg and above with dose dependence, except at the highest dose. The uterine weights at the lowest dose were comparable to those in the control group.
Uterine weights in uterotrophic assay.
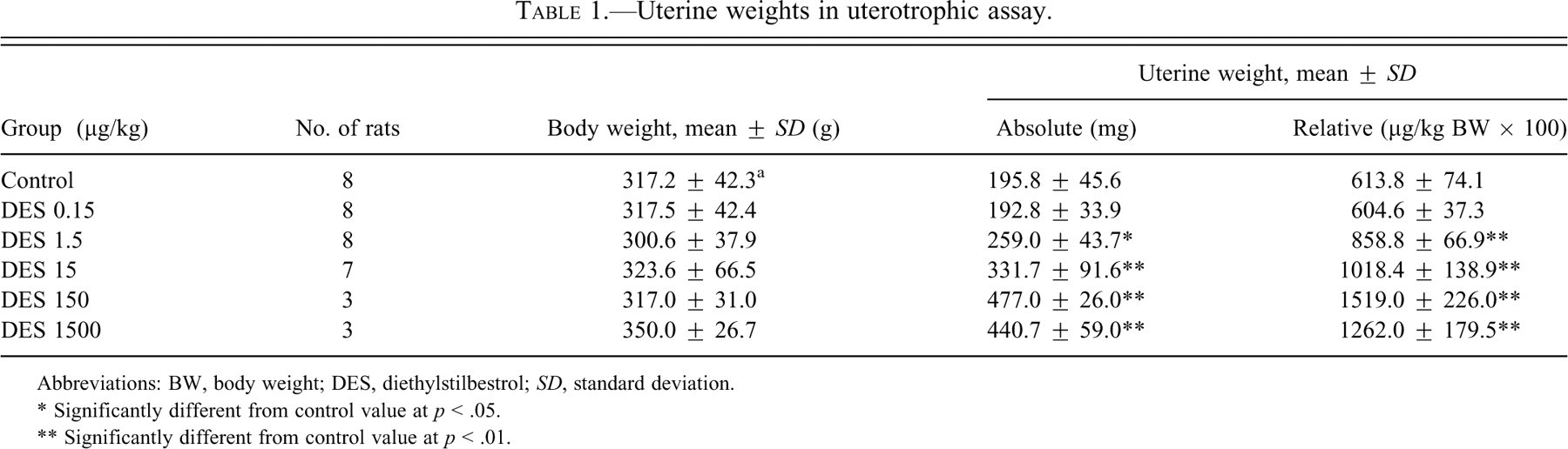
Abbreviations: BW, body weight; DES, diethylstilbestrol; SD, standard deviation.
* Significantly different from control value at p < .05.
** Significantly different from control value at p < .01.
Clinical Observation in Life and Estrous Cyclicity in Experiment 2
No treatment-related clinical signs were observed at any time point. Body weight changes were comparable in all groups, including the controls (data not shown). The timing of VO was not different between the treated groups and control. The age at VO was 28.0 ± 1.0, 28.0 ± 0.0, 28.2 ± 0.4, 28.5 ± 0.5, 28.9 ± 1.2, 28.8 ± 1.3 in the 0, 0.15, 1.5, 15, 150, and 1,500 µg/kg groups, respectively.
Data for the estrous cycle are shown in Figure 1. At 1,500 µg/kg, most rats already showed PE at the start of checking estrous cycles, and all were affected by seven weeks of age. At 150 µg/kg, a few females demonstrated PE at five weeks of age, and most by seventeen weeks of age. At 15 or 1.5 µg/kg, PE rates began to increase at thirteen or fifteen weeks of age, and most of them reached PE by nineteen or twenty-one weeks of age, respectively. In all these groups, the numbers of rats exhibiting PE were significantly increased compared to that of controls at fifteen to thirty-one weeks and seventeen to twenty-one weeks of age, respectively. At twenty-three weeks of age, PE began to increase in the control group, and most animals were affected by thirty-nine weeks of age. In the 0.15 µg/kg group, the lowest dose group, the onset of PE was similar to that in controls.
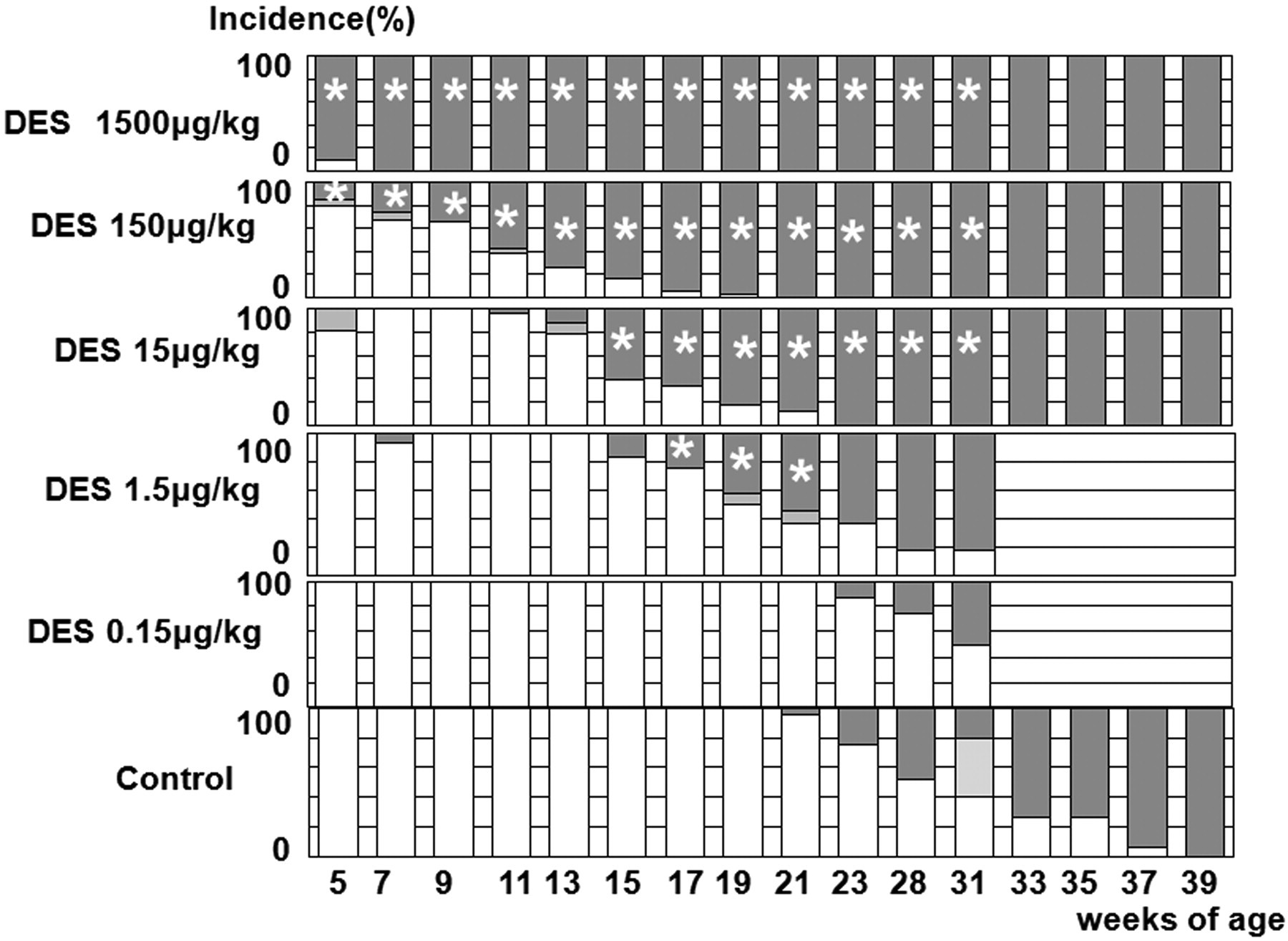
Estrous cycling in experiment 2. White, light grey, and dark grey columns, respectively, represent regular (normal) cycle, irregular cycles, and persistent estrus. Incidences are the percentages of animals showing each estrous stage per group. *, significantly different from the control value at p < .05 and p < .01, respectively.
Morphological Changes up to Five Weeks of Age in Experiment 2
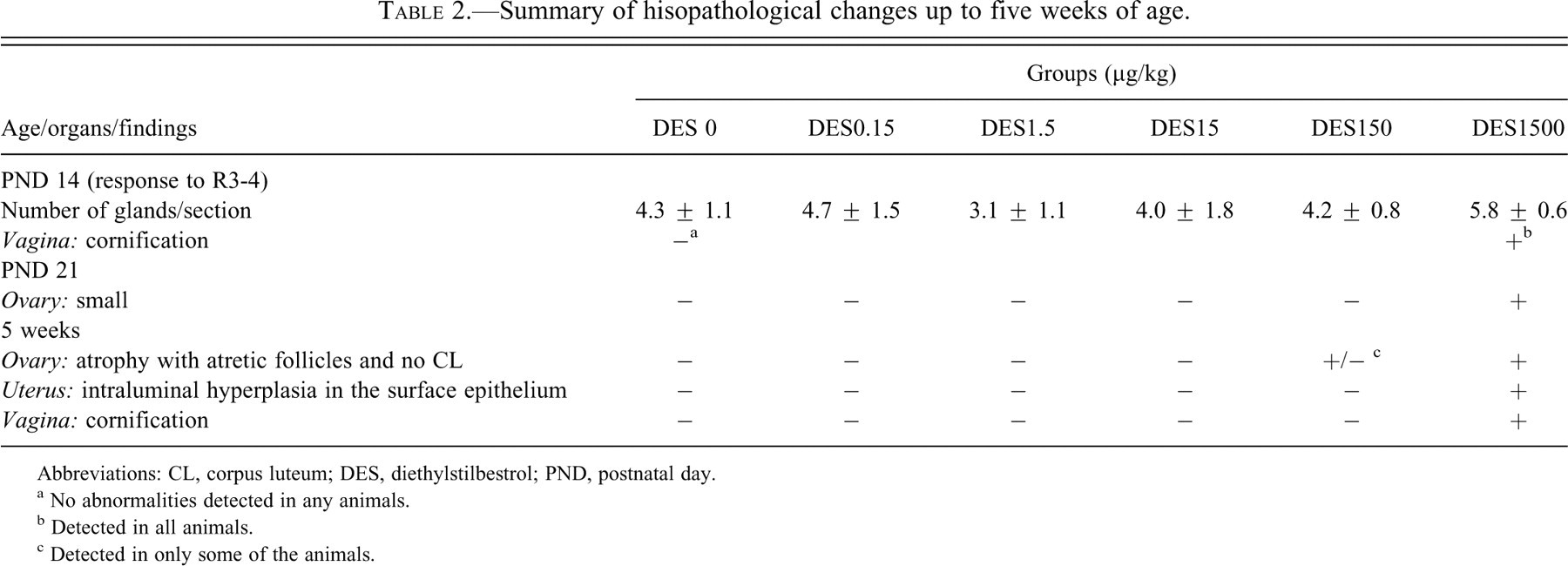
Changes in the ovary, uterus, or vagina at PND14, PND21, or five weeks of age are summarized in Table 2, and characteristic lesions are shown in Figures 2, 3, and 4, respectively. Although no abnormalities were detected in the ovary at PND14, those at 1,500 µg/kg were clearly small at PND21 (Figure 2 ). At five weeks of age, the ovaries of all rats at 1,500 µg/kg and some of rats at 150 µg/kg were macroscopically small in size and microscopically composed of cystic atretic follicles, and they lacked corpora lutea. No hyperplasia or hypertrophy of the interstitial cells was found in the ovaries of all treated groups. In the uterus, there were no differences until PND21, with normal gland genesis at PND14. At five weeks of age, the uteri at 150 µg/kg and higher doses showed intraluminal hyperplasia (Figure 3 ). The uteri in the other groups showed normal morphology at estrus. In the vagina, cornification was detected in the highest dose group at PND14 (Figure 4 ). At PND21, this change was recovered to several layered squamous cells similar to those of control animals. At five weeks of age, it was a feature in all rats at 1,500 and most of those in the 150 µg/kg group. The vaginas in the 15 µg/kg and lower groups showed morphology similar to those in the control group. In the other organs, no morphological features were different between treated groups and controls.
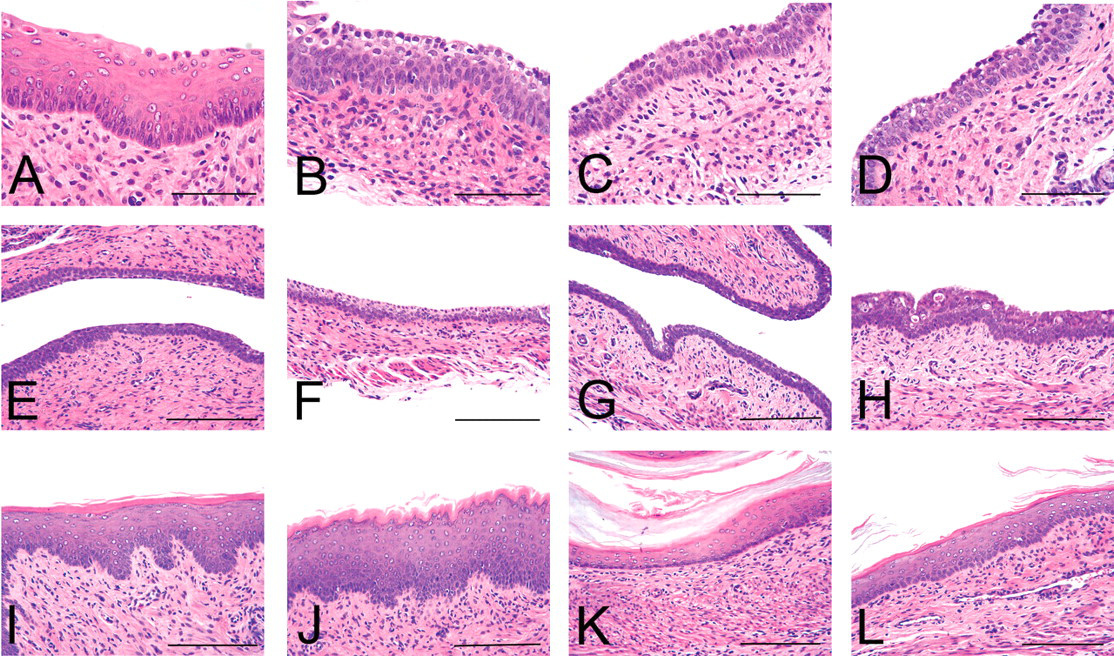
Sequential morphological assessment of the vagina. A–D, E–H, and I–L illustrate changes at PND14, PND21, or five weeks of age (A, E, I; B, F, J; C, G, K, and D, H, L are for the 1,500, 150, 15, and 0 µg/kg groups, respectively). At five weeks of age, the vaginas of A and B were obtained from persistent estrus at 1,500 and 150 µg/kg, and the others were from rats at estrus with normal cyclicity. Hematoxylin and eosin stain. Scale bars are 200 µm and 400 µm in A–D and E–L, respectively.
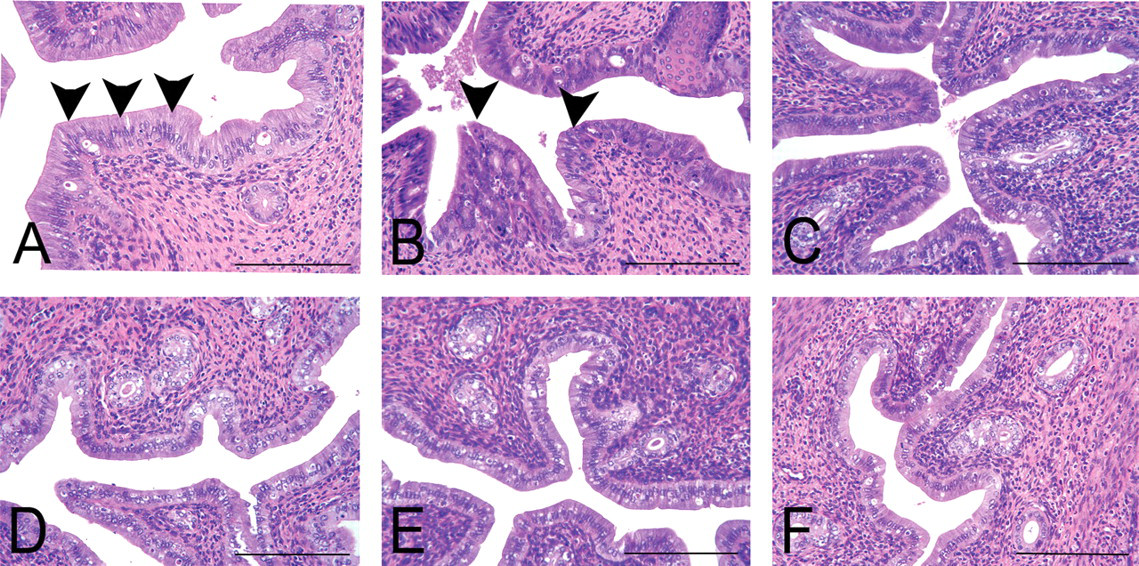
Morphological changes in the uterus at five weeks of age. (A–F) The changes in the 1,500, 150, 15, 1.5, 0.15 and 0 µg/kg group, respectively. The uteri of A and B were obtained from animals demonstrating persistent estrus and that were given 1500 and 150 µg/kg, and the remainder featured estrus with normal cyclicity. (A) Arrowheads indicate high columnar epithelial cells in the luminal surface at 1,500 µg/kg. (B) Arrowheads indicate intraluminal hyperplasia in the luminal epithelium at 150 µg/kg. (C–F) A number of apoptotic cells in the luminal epithelium, which is a typical feature in the uterus at estrus. Hematoxylin and eosin stain. Scale bars are 200 µm.
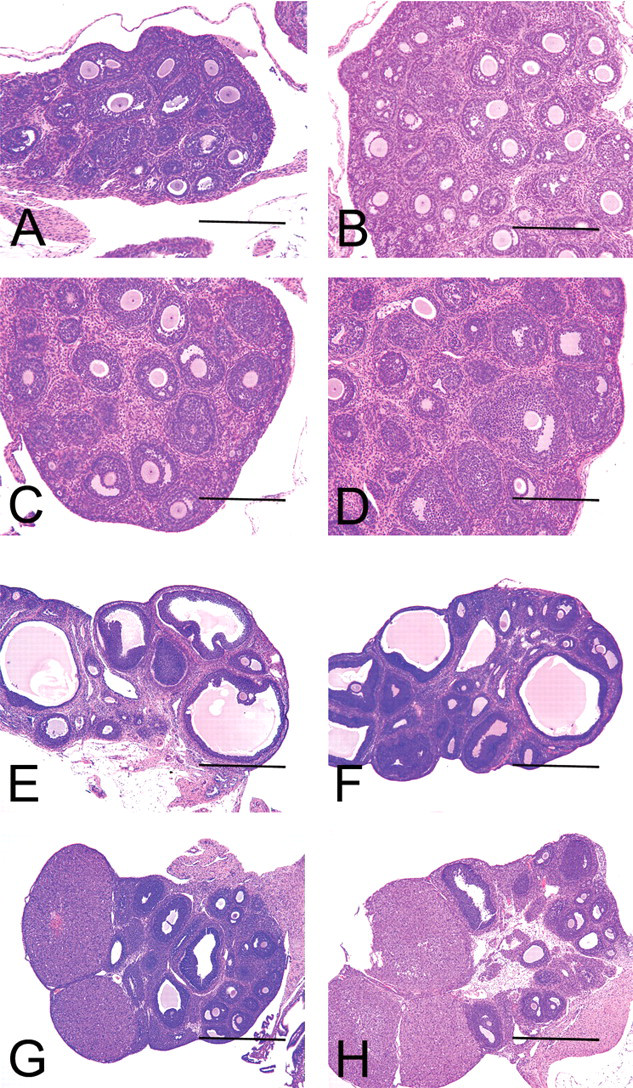
Sequential morphological assessment of the ovaries. A–D and E–H illustrate changes at PND21 and five weeks of age (A, E; B, F; C, G; and D, H are for the 1,500, 150, 15, and 0 µg/kg group, respectively). At five weeks of age, the ovaries in A and B were obtained from animals demonstrating persistent estrus and that were given 1,500 and 150 µg/kg, and the remainder featured estrus with normal cyclicity. Hematoxylin and eosin stain. Scale bars are 1 mm and 2 mm in A–D and E–H, respectively.
Summary of hisopathological changes up to five weeks of age.
Abbreviations: CL, corpus luteum; DES, diethylstilbestrol; PND, postnatal day.
a No abnormalities detected in any animals.
b Detected in all animals.
c Detected in only some of the animals.
Immunohistochemical Intensity of ERα Expression in the Uterus, Vagina, and Ovary
The immunohistochemical expression of ERα in the uterus and vagina is summarized in Figure 5 . The intensities were slightly higher in the uterus and vagina at PND14 and PND21 than those at five weeks of age. Estrogen receptor-α did not express in the granulosa cells, theca cells, or stromal cells of the ovary. Treatment-related changes were not detected in any other organs or at any examined time points.
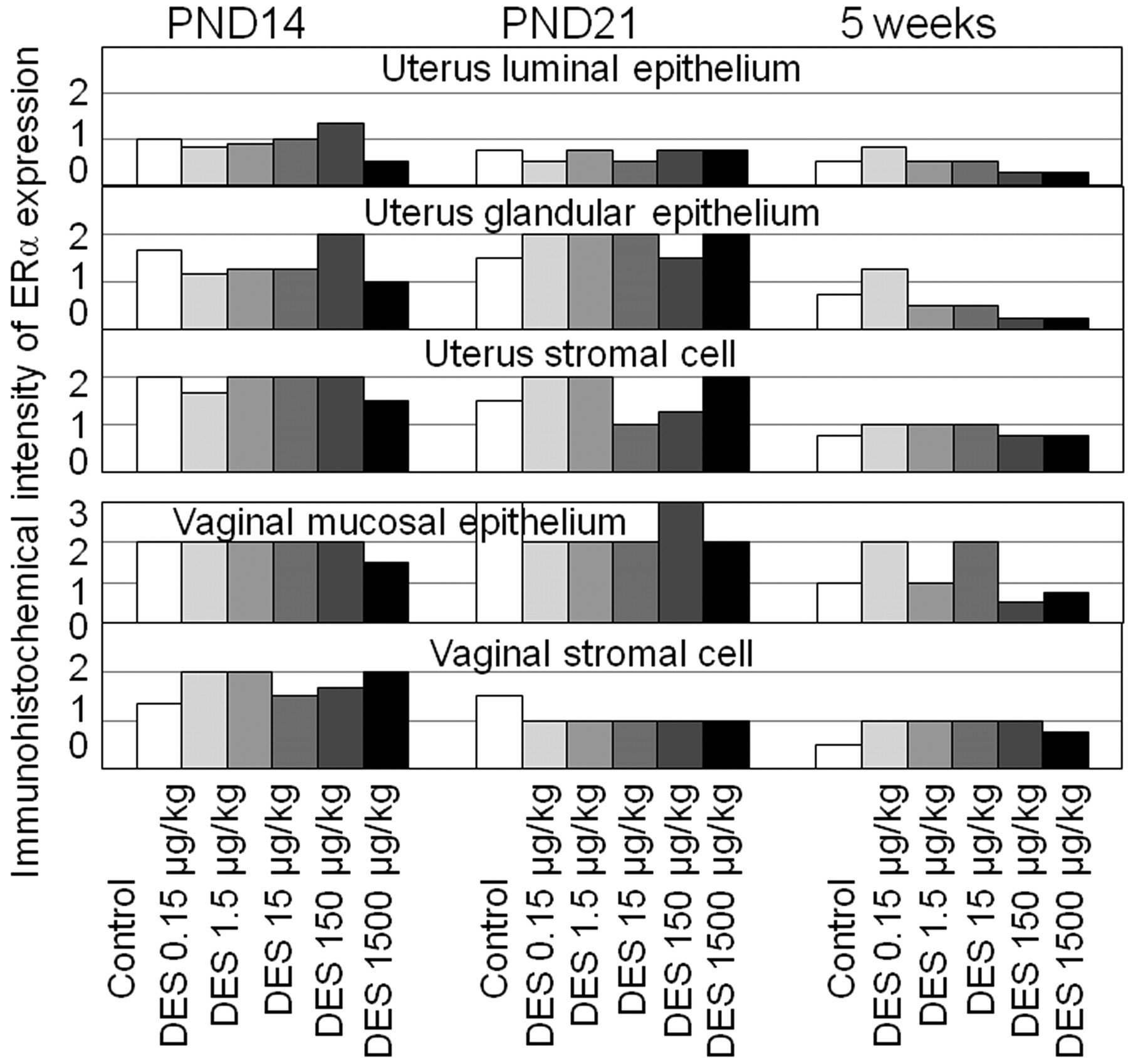
Immunohistochemical intensity of ERα expression in the uterus and vagina at PND14, PND21, and five weeks of age. Treatment-related changes were not detected in any organs. The intensities of these organs at PND14 and PND21 were similar, but overall, expression had declined at five weeks of age.
Uterine Carcinogenicity and Histopathology at Termination
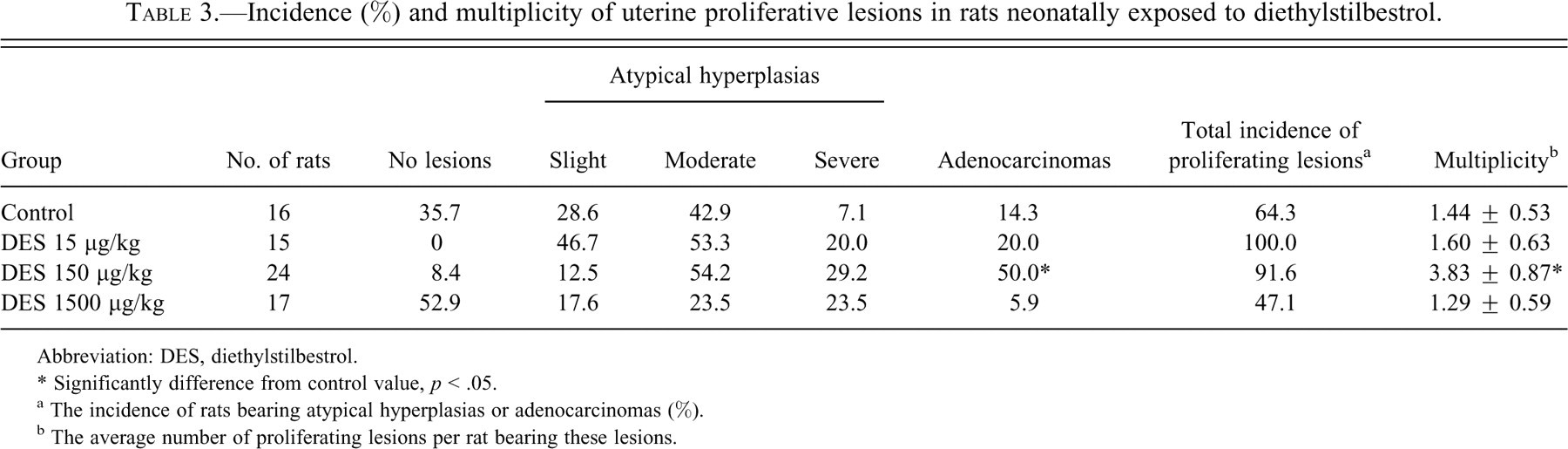
The incidences of atypical hyperplasias and endometrial adenocarcinomas are shown in Table 3 . Values for both were lower in the highest dose group than in the controls. On the other hand, neoplastic lesions showed an increasing trend in the lower groups, and the incidence of adenocarcinomas and total multiplicity of atypical hyperplasias and adenocarcinomas were significantly higher in the 150 µg/kg group than those in the controls. In the 1.5 and 15 µg/kg groups, which were sacrificed after a shorter period than other groups, few atypical hyperplasias were detected, and the incidence was similar to background data at the same age in this strain.
Incidence (%) and multiplicity of uterine proliferative lesions in rats neonatally exposed to diethylstilbestrol.
Abbreviation: DES, diethylstilbestrol.
* Significantly difference from control value, p < .05.
a The incidence of rats bearing atypical hyperplasias or adenocarcinomas (%).
b The average number of proliferating lesions per rat bearing these lesions.
At termination, morphological abnormalities such as atrophic uterine horns, disappearance of lumina (Figure 6 ), or short and straight oviducts were detected in the highest dose group. Such abnormalities were not observed in other treated groups. As common changes in all groups including controls, most of the ovaries were atrophic with several cystic atretic follicles and few or no corpora lutea (Figure 6). Several primary follicles remained, with no obvious variation among the control and treated groups. The vaginal mucosa was similarly composed of several layered squamous cells in the control and treated groups. Neoplastic and non-neoplastic lesions were abundant in the treated and control groups, but their incidence and severity were comparable among the groups. Treatment-related morphological changes were not detected in the genital tracts.
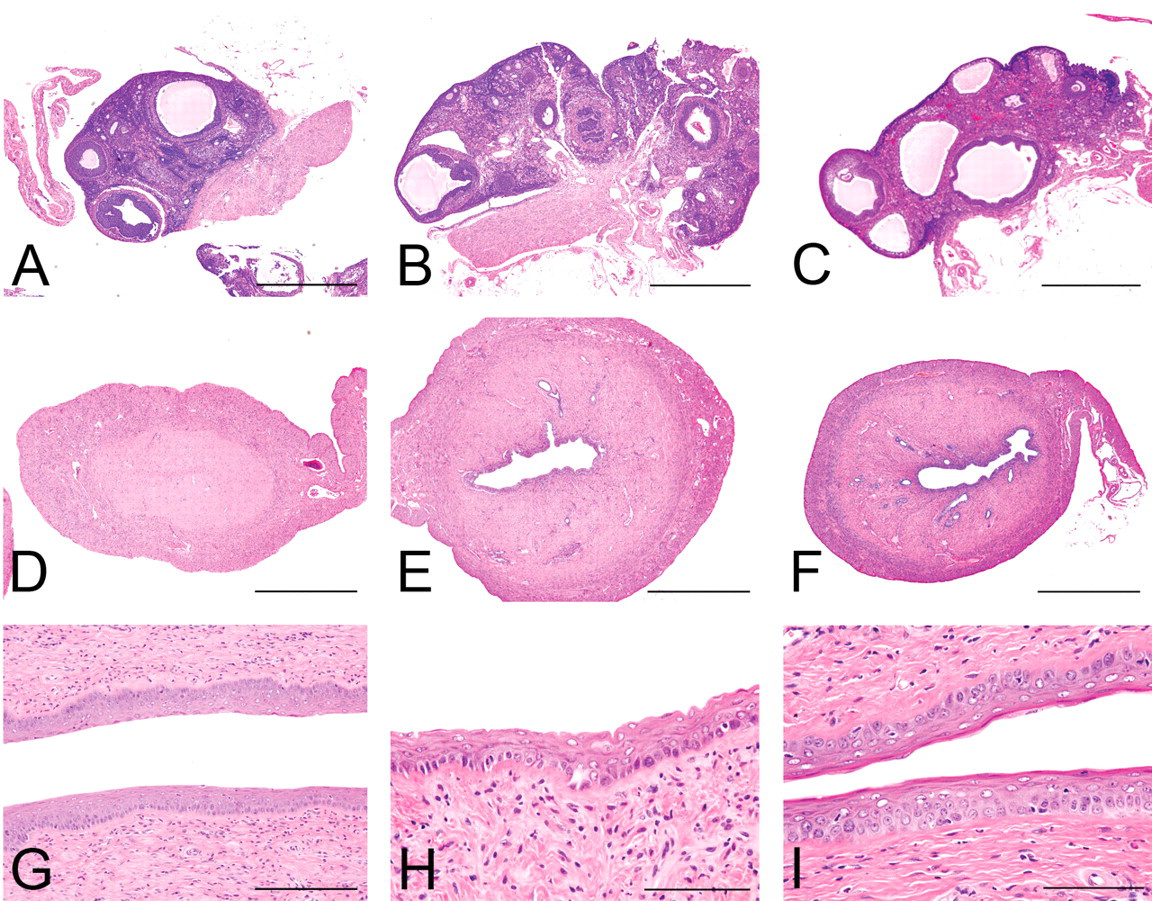
Morphological changes at the termination. A–C, D–F, and G–I are the ovaries, uteri, and vaginas, respectively. (A, D, G) 1,500 µg/kg; (B, E, H) 15µg/kg; and (C, F, I) 0 µg/kg. Hematoxylin and eosin stain. Scale bars are 1 mm, 2 mm, 400 µm, and 200 µm in A–C, D–F, G, and H–I, respectively.
mRNA Expression of the Ovary and Uterus at Five Weeks of Age
Data for mRNA expressions of ER α and β, progesterone receptor (PR), and GAPDH in the ovary are shown in Figure 7 . Glyceraldehyde 3-phosphate dehydrogenase mRNA was constitutive in all animals. Although no differences in ERα or ERβ could be detected in the ovary or uterus, PR in the uterus demonstrated increased expression at the 150 µg/kg dose.
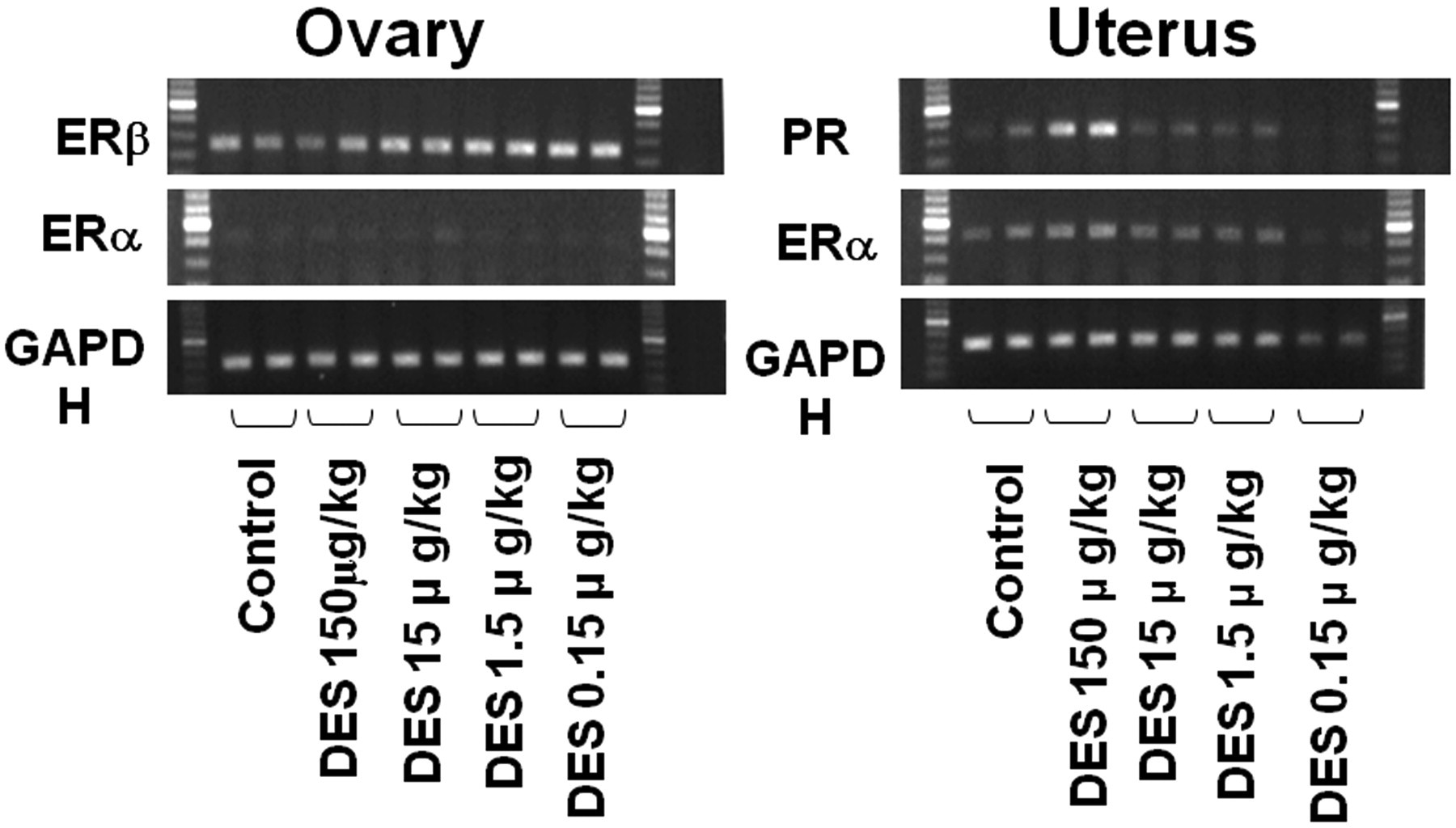
mRNA Expression of estrogen receptors α and β and progesterone receptor in the ovary and uterus in the 150, 15, 1.5, 0.15 and 1.5 µg/kg and control groups at five weeks of age. The ovaries and uteri at 150 µg/kg were obtained from animals in persistent estrus, and others are from rats at estrus with normal cyclicity.
Discussion
The present study demonstrated clear, dose-dependent, delayed effects of a single neonatal treatment with DES at various doses on morphology in the female reproductive tract and estrous cyclicity in Donryu rats. In particular, earlier onset of PE was evident in all but the lowest dose group. Although timing of VO, which represents puberty, and uterine gland genesis was not affected, the ovarian morphology at five weeks of age at the highest dose indicated an abnormal status with anovulation and excess estrogen levels relative to progesterone (E:P ratio).
In addition, the cornification of vaginal mucosa and uterine intraluminal hyperplasia were consistent with those in rats (Katsuda et al. 2000) or mice (Bern et al. 1987; Yoshida et al. 1999) neonatally exposed to DES or high-dose p-tert octylphenol. The vaginal changes in the present study are considered to be ovary dependent events in rats (Katsuda et al. 2000), but not mice (Forsberg 1979; Takasugi et al. 1962). The present results clearly indicated that the animals at the highest dose were already affected before puberty. Perinatal exposures to high doses of estrogens are known to irreversibly disrupt steroid hormone receptor or related gene expression in the uterus (Nakamura et al. 2008; Yoshida et al. 2000; Yoshida et al. 2002a). The studies on ERα or ERβ mRNA as end points of neonatal exposure to estrogens were limited, whereas PR and AR as well as IGF1 have been shown to be important (Weihua et al. 2002). The present study did not provide proof of the involvement of ERα in both mRNA and protein levels in the uterus. The increased expression in PR in the uterus at 150 µg/kg might be related to fewer corpora lutea, although mechanisms of the increase have not been determined.
Several animals at 150 µg/kg revealed similar PE status and morphology in the female reproductive tract to those in the highest group, indicating that these animals at 150 µg/kg were also affected before puberty. Besides, clear abnormalities including morphological or gene expressions were not detected in the remainder of the 150 µg/kg group and in all rats in the 15 and 1.5 µg/kg groups until five weeks of age. Therefore, the earlier onset of PE in these animals was considered to be a delayed effect. In addition, the onset showed clear dose dependency. The early onset of PE has already been reported as an indicator of DAS (Mobbs et al. 1985), but there are limited data on dose dependency (Bern et al. 1987). Recently, Ninomiya et al. (2007) reported early onset PE with dose dependency in Sprague-Dawley rats exposed to DES at doses of 1–100 µg/rat (approximately equivalent to 17–1,700 µg/kg if the body weights are expected to be 5–6 g at PND0), but not 0.1 µg/rat (approximately equivalent to 1.7 µg/kg) until 150 days of age. Although diversity exists at lower doses, their results showing delayed effect are in line with our data. The estrous cyclicity follows a similar tendency until eight months of age in the 0.15 µg/kg and control groups. Although the lower two groups were terminated two months earlier than the other groups, most of the control rats (80%) reached persistent estrus at thirty-three weeks of age.Thus, the lowest dose would not be likely to affect the estrous cycle after eight months of age.
The results of the uterotrophic assay demonstrated that the single treatments with DES at 1.5 µg/kg and higher were inducible doses of estrogenic activity in vivo with dose dependency. Interestingly, our results demonstrated that the doses showing estrogenic activity in vivo corresponded to the inducible doses of early onset of PE. These results suggest that test substances with estrogenic activity in vivo may induce delayed effects. A concern that twenty-four hours after a single treatment is too soon to detect any estrogenic activity in uterotrophic assay might exist, because uterine weights are usually measured twenty-four hours after a three-day treatment with a test substance in a uterotrophic assay (OECD 2007). However, a previous study that demonstrated, using radioautography, that DES di [35S] sulfate injected intraperitoneally at 1 mg/rat distributed in the whole body within twenty-four hours (Barford et al. 1977) could be support that the concern could be excluded.
It is well known that neonatal exposure to DES can induce malformations in the female reproductive tract not only rodents (Newbold et al. 1983; Rothschild et al. 1987–88), but also in human beings as DES daughters (Jeffries et al. 1984). The present results of uterine carcinogenesis did not show a dose dependency. The reason is likely to be related to the extreme atrophy without the endometrial epithelium apparent in the uteri at the highest dose at termination, because the epithelial cells are the histogenesis of endometrial adenocarinomas. The increase of endometrial adenocarcinoma development at 150 µg/kg of DES observed here is in line with previous studies of neonatal treatment with estrogenic compounds such as DES, tamoxifen, or high-dose p-tert octylphenol (Carthew et al. 2000; Newbold et al. 1990; Yoshida et al. 2002b). Prolonged exposure to excess estrogen relative to progesterone in the blood (increased E:P ratio), which is reflected as PE in a vaginal smear, is an important cause of uterine cancer development in Donryu rats, a high-yield strain of uterine endometrial cancers (Ando-Lu et al. 1994; Nagaoka et al. 1990; Vollmer 2003; Yoshida et al. 2004). Excess estrogens also play a crucial role in cancer of the uterine corpus in other strains of rats (Deerberg and Kaspareit 1987) or mice, as well as women (World Health Organization Classification of Tumours 2003), although androgen or prolactin might influence uterine cancer development in rodents (Nantermet et al. 2005; Yoshida et al. 2009). Our results described above show that early onset of PE might be the most sensitive indicator of delayed effects by neonatal exposure to estrogenic compounds rather than susceptibility to induction of preneoplastic and neoplastic lesions and other morphological changes.
It is still unclear what mechanism is crucial for the early onset of PE. MacLusky and Naftolin (1981) first reported such effects as DAS, and they hypothesized that disruption of the hypothalamus might be the trigger. There is evidence of involvement of loss of noradrenergic neuronal responsiveness in rats (Lookingland and Barraclough 1982) and that DAS is caused by age-related neuroendocrine function rather than ovarian impairment (Mobbs et al. 1985). Recently, Adewale et al. (2009) reported gonadotropin-releasing hormone (GnRH) release following neonatal estrogen exposure to be normal in the organum vasculosum of the lamina terminals in rats displaying early onset of irregular cycles, including PE. One present focus is on the newly discovered neuropeptide in the hypothalamus, kisspeptin, a regulator of gonadtropin secretion via GnRH secretion (Messager et al. 2005), because it plays pivotal roles in the onset of puberty (de Roux et al. 2003; Seminara et al. 2003), sex differentiation (Kauffman et al. 2007), and reproductive activities such as estrous cycling (Adachi et al. 2007) and ovulation (Clarkson and Herbison 2009; Uenoyama et al. 2009). It has been established that neonatal exposure to estrogens or estrogenic compounds decreased hypothalamic Kiss 1 mRNA levels or stimulation of GnRH neurons by kisspeptins, resulting in impairment of female reproductive functions (Bateman and Patisaul 2008; Navarro et al. 2009). Kisspeptin and its signaling pathway in GnRH neurons might be anticipated as key factors in delayed effects.
In conclusion, neonatal exposure to a single dose of DES, which shows estrogenic activity in vivo, induces delayed adverse effects on female rats in a dose-dependent manner. Detection of early onset of PE by vaginal smear appears to be the most sensitive and useful parameter to detect such effects, though a wait of over four months is necessary for significance to become apparent. Its appearance, however, can be considered adverse in rats as well as human beings, because it is irreversible and increases the risk to development of uterine corpus cancer.
Footnotes
Acknowledgment
This study was supported by Health and Labour Sciences Research Grants, Research on Risk of Chemical Subtances, Ministry of Health, Labour and Welfare [H22-Toxicol-003].
