Abstract
Teratomas commonly occur in the testis and ovary, whereas in the uterus they are rare. The authors report findings for a mass detected in the uterus of a 26-week-old mouse in a colony of C57BL/6 bred in their laboratory. The mass was located in the endometrium and protruded into the lumen. Histopathologically, it consisted of abnormal diploblastic or triploblastic tissues. Bone with a growth plate and myeloid cells, as well as cartilage, was mainly observed. It also included melanocytes, exocrine gland-like cells, striated muscle, and neuron-like cells. While these tissues were accompanied by extensive necrosis, all of them were well differentiated and lacked features of malignancy, such as invasion and metastasis. This mouse had experienced parturition, but fetal tissue was not observed in the lesion. Therefore, the lesion was diagnosed as a benign teratoma, which was spontaneously developed in the uterus.
Keywords
Teratomas are tumors composed of abnormal tissue derived from at least two, and often three, germinal layers (endoderm, mesoderm, ectoderm). Most teratomas are benign, consisting of well-differentiated tissues. Malignant teratomas are rare, but they generally show poorly differentiated tissue elements, and large areas with necrosis and hemorrhage, as well as invasion and metastasis (Maclachlan and Kennedy 2002; Davis et al. 2001). Teratomas can occur in any site on the body but most commonly develop in the testis and ovary, whereas in the uterus they are infrequent. In humans, some cases of uterine teratoma have been reported (Sissons and Foria 2003; Lim et al. 2003; Iwanaga et al. 1990; Martin et al. 1979). In rodents, experimentally induced teratoma have been reported by several researchers (Sobis, Park, and Vandeputte 1978; Sobis and Vandeputte 1979, 1982; Sobis, Verstuyf, and Vandeputte 1991, 1993; Natsuaki 1983; Katsuse et al. 1981), but only one spontaneous case has been documented, in a mouse (Slye et al. 1924). We here describe the histopathological features of a spontaneous case of uterine teratoma encountered in a mouse.
The animal was a 26-week-old female in a colony of C57BL/6 mice bred in our laboratory and included in a study to obtain background data. This animal was individually housed in a suspended aluminum cage with a stainless-steel wire-mesh front and floor under controlled environmental conditions, including a temperature of 22–26°C, a relative humidity of 40–70%, a frequency of ventilation of more than 10 air exchanges/hr, and a 12-hr light/12-hr dark cycle (lighting period, 0800–2000), with free access to solid CRF-1 (Oriental Yeast Co., Ltd., Japan). The experiment was performed in accordance with the Guide for Animal Care and Use of Sumitomo Chemical Co., Ltd. The animal in question had experienced parturition 3 months before the autopsy. No clinical abnormalities were observed during pregnancy, partus, and postpartum. At autopsy, this animal was sacrificed under ether anesthesia along with others in the same group and subjected to a complete necropsy.
At autopsy, a firm mass (5 × 5 × 10 mm) was incidentally found in the lumen of the right uterine horn. No other abnormal findings were observed. The mass was removed with the uterus, fixed in 10% neutral buffered formalin solution, and cut into 5 portions. On the transverse surface, it presented as a firm whitish mass and filled the uterine cavity. Each part of the mass was embedded in paraffin and sectioned for histological examination. After staining with hematoxylin and eosin, Masson-Fontana and immunohistochemical staining using PCNA-monoclonal mouse antibody (DAKO, Clone PC10) were additionally performed. In addition, the liver including the gall bladder, kidneys, lung, spleen, brain, heart, and pancreas were also removed at autopsy and also examined histopathologically.
Histopathologically, the mass showed a continuity with uterine endometrium and projected into the lumen. It mainly consisted of well-differentiated bone and cartilage tissues (Figure 1 ). Growth-plate-like arrangement of cartilage cells, endochondral ossification, and medullary cavity-like spaces containing red blood cells and some myeloid cells were found in such bone. Necrotic areas and infiltration of inflammatory cells such as neutrophils, lymphocytes and macrophages were also observed (Figures 2 and 3).
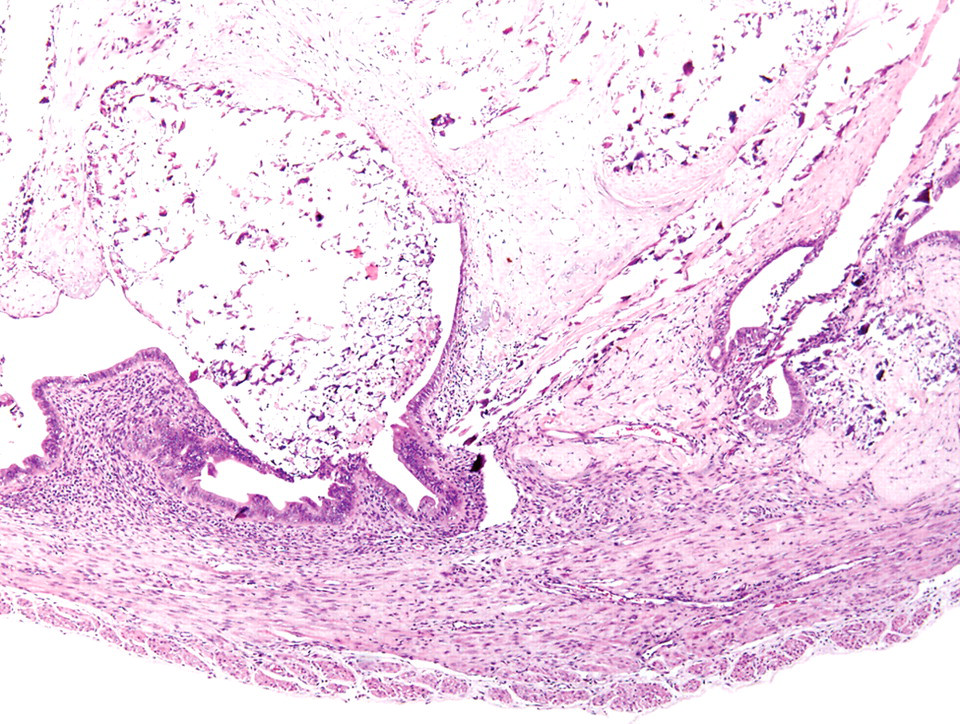
The uterine mass; mouse. The mass showing continuity with the endometrium and projection into the lumen was found to mainly consist of abnormal bone and cartilage. H&E.
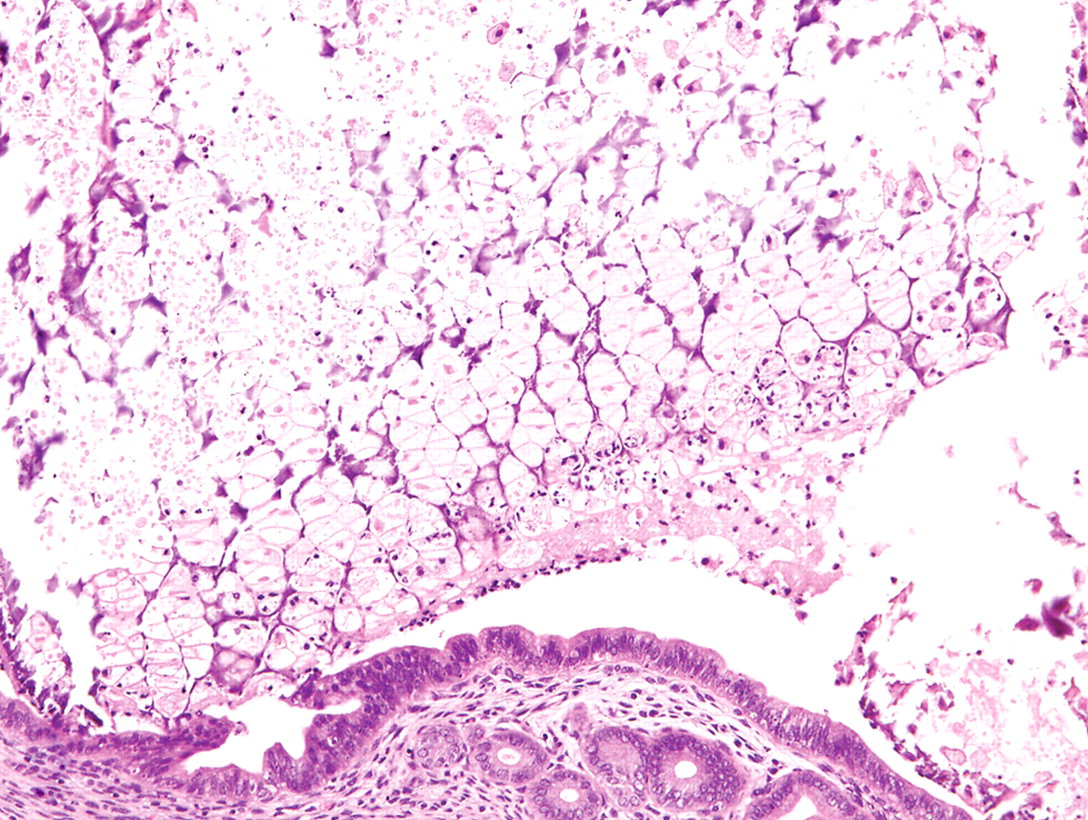
The uterine mass; mouse. Bone tissue consisting of growth plate like arrangements of cartilage cells and endochondral ossification, medullary cavity-like spaces containing red blood cells and some myeloid cells, necrosis, and infiltration of inflammatory cells. H&E.
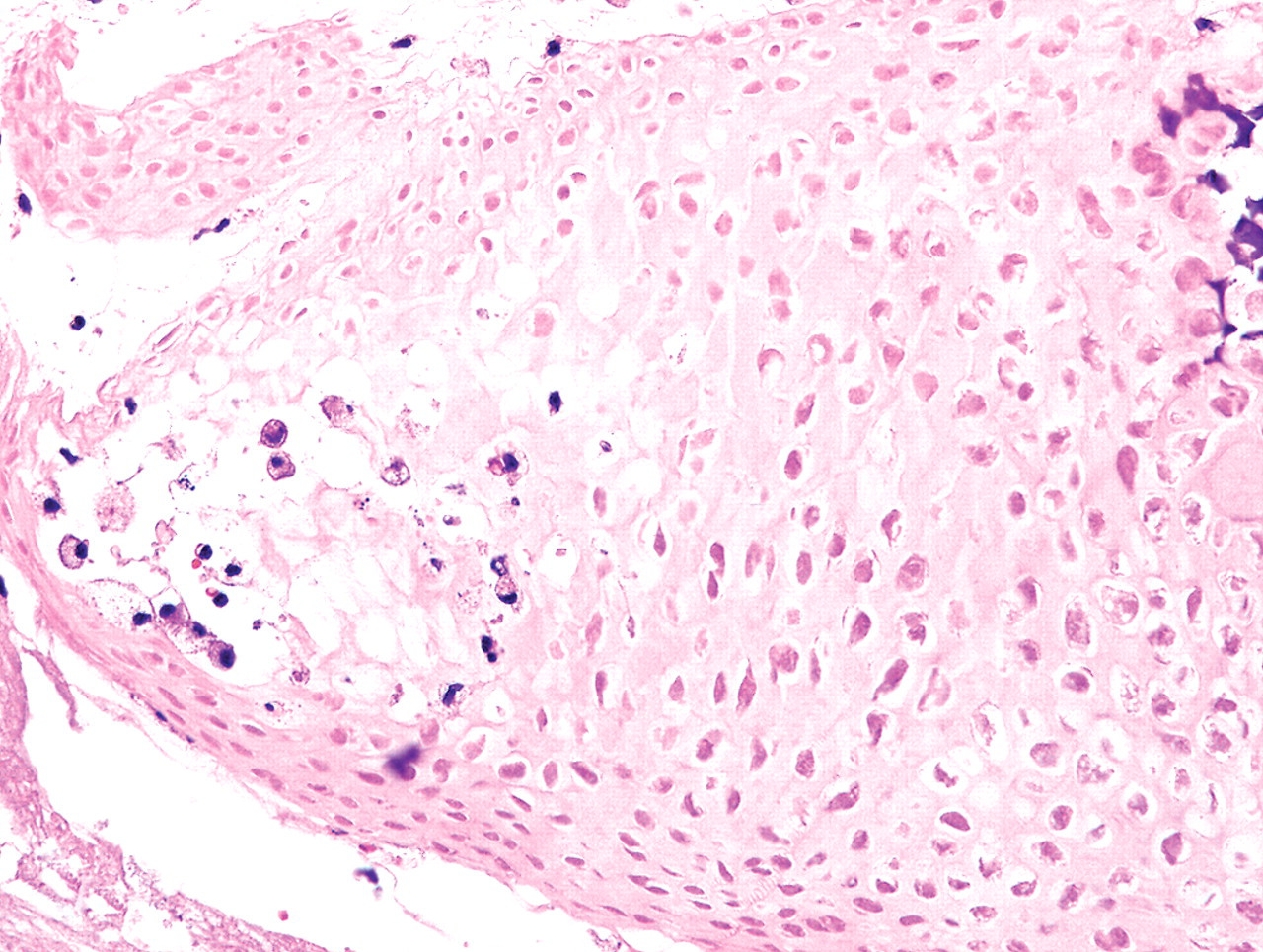
The uterine mass; mouse. Cartilage tissue accompanied by necrosis and infiltration of inflammatory cells. H&E.
In the connective tissue, a focus of dark brown pigmented cells was present (Figure 4A ). Melanin pigments were confirmed by Masson-Fontana staining (Figure 4B).
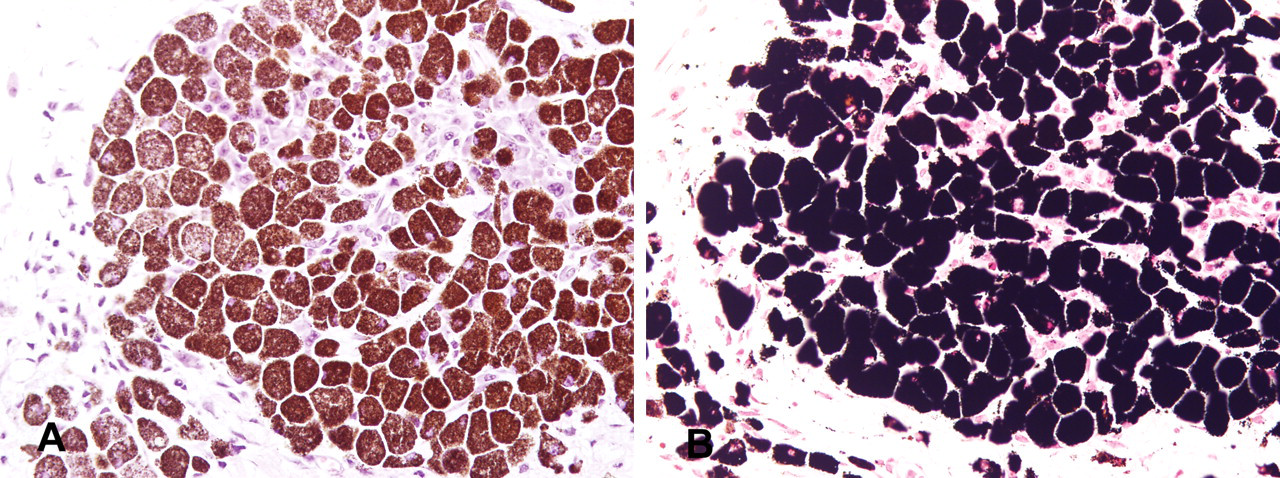
The uterine mass; mouse. A focus of dark brown pigment containing cells confirmed to be melanin by Masson-Fontana staining. A. H&E. B. Masson-Fontana staining.
In addition, neuron-like cells arranged in layered lines, striated muscle, and acinar structures of exocrine gland-like cells were observed. Each of them was well differentiated and accompanied by extensive necrosis (Figures 5–7 ). Immunohistochemical staining for PCNA revealed that the surviving cells showed little cell-proliferative activity (data not shown).
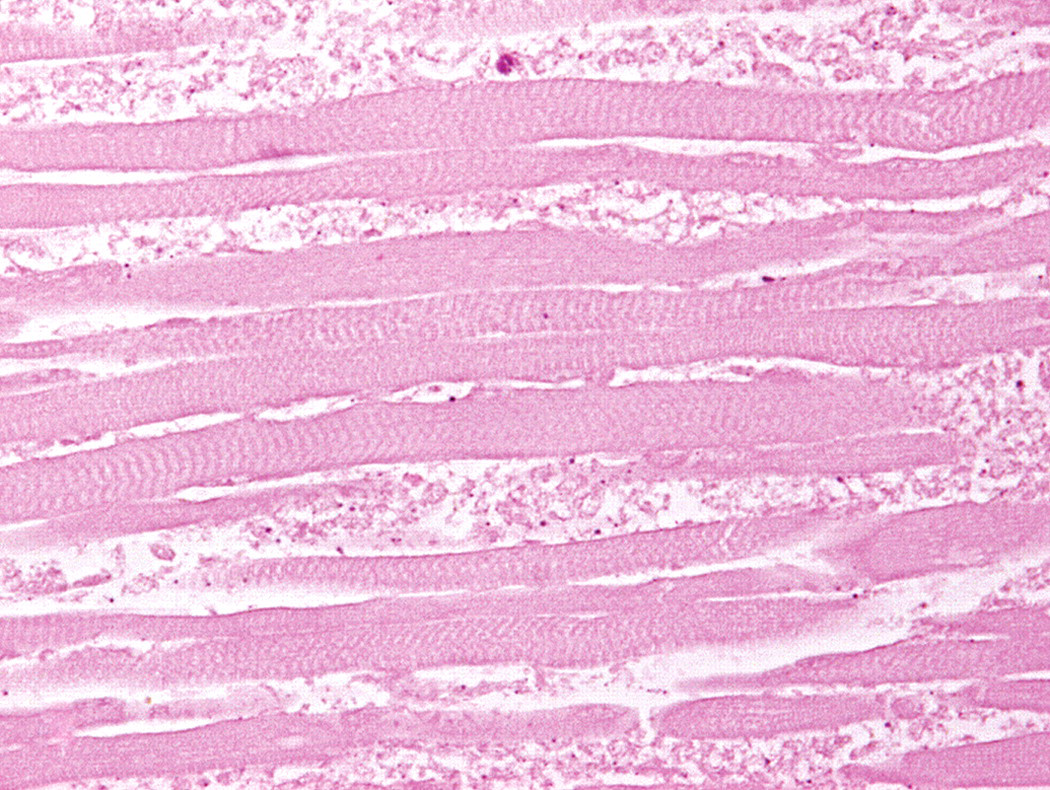
The uterine mass; mouse. Striated muscle with necrosis. H&E.
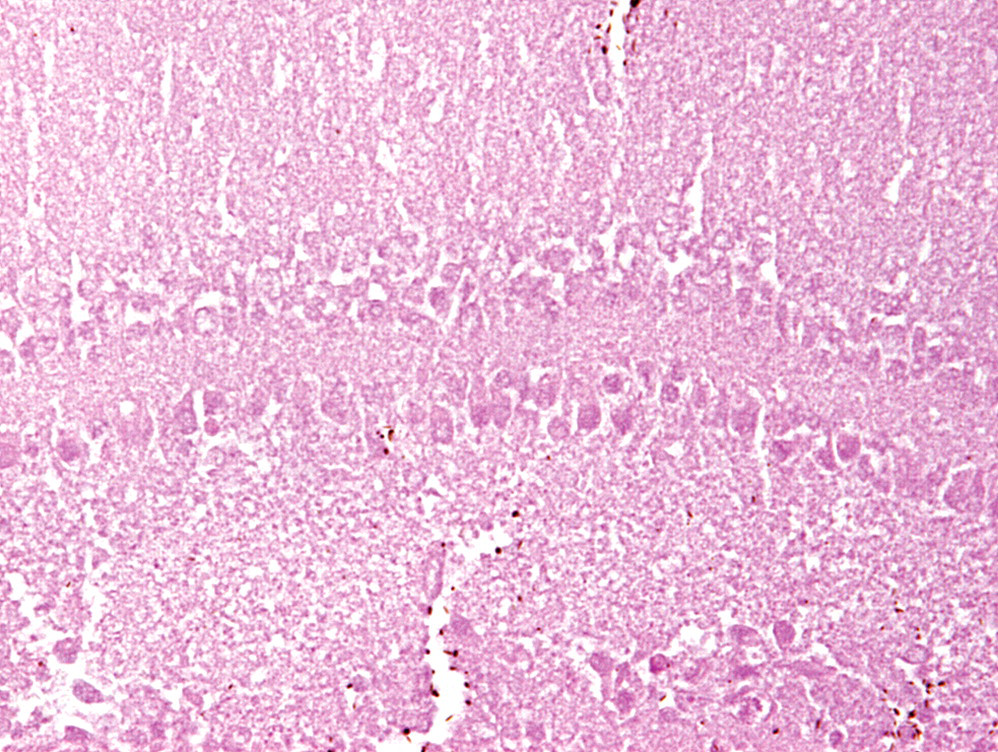
The uterine mass; mouse. Layered structure of neuron-like cells with necrosis. H&E.
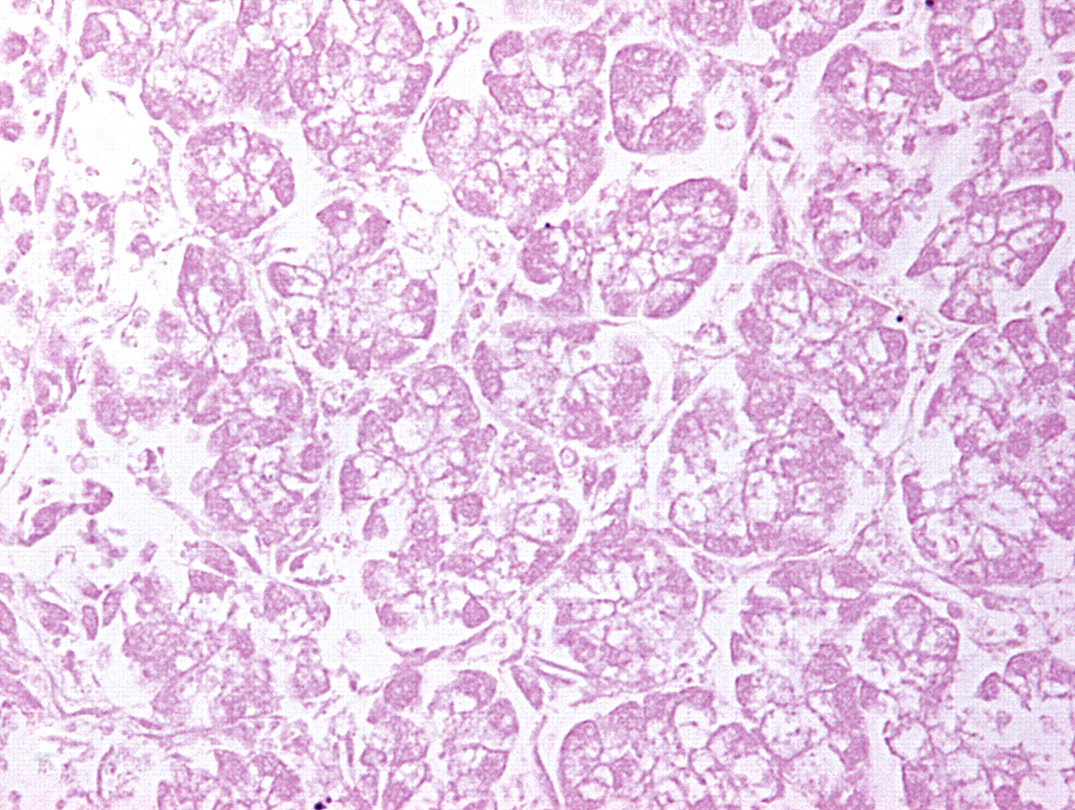
The uterine mass; mouse. Exocrine gland-like acinar cells with necrosis. H&E.
In the examination of the other organs, slight age-related changes, like slight nephropathy in the kidneys and increased interstitial glands in the ovaries, were observed.
This mass consisted of well-differentiated mesodermal tissues (bone, cartilage, and striated muscle), ectodermal tissues (melanocytes and neuron-like cells), and ectodermal or endodermal tissue (exocrine-gland-like cells). Although necrosis was often observed, there was no evidence of anaplasia and malignancy, such as invasion and metastasis. Therefore, it was regarded as a benign teratoma. Since the mouse had experienced parturition 3 months before autopsy, other pregnancy-related tumors should be considered in making a diagnosis. However, neither yolk sac membranes nor cytotrophoblast cells were observed, so that yolk sac and chorionic and trophoblastic tumors could be excluded. It could be also distinguished from a fetus itself since it lacked anatomical components and locations of the fetal mouse and showed continuity with the uterine endometrium. The lesions were therefore concluded to be an extremely rare case of a spontaneous uterine teratoma in a mouse.
Several cases of uterine teratomas in humans have been reported, the majority being of mature type. Regarding the pathogenesis, activated germ cell or residual fetal tissue origins have been suggested (Sissons and Foria 2003; Lim et al. 2003; Iwanaga et al. 1990; Martin et al. 1979). The single spontaneous uterine teratoma in mouse described earlier was also of mature type (Slye et al. 1924). It was observed in the neck of uterine accompanied by distended uterine horns with fluid. The case was similar to our case in that there were many islands of cartilage, small islands of bone with bone marrow, striated muscle fibers, and melanin pigments. However, the case also showed islands of squamous epithelium, lymphoid tissue, and areas suggesting neuroglia, while none of them were found in our case. In addition, some researchers reported that externalization of the visceral yolk sac after fetal resection induced the development of benign teratomas in rodents. Such experimentally induced teratomas were histologically characterized by the presence of a variety of well-differentiated tissues such as columnar epithelium with goblet cells, smooth and skeletal muscle, cartilage and bone with endochondral ossification, and bone marrow (Sobis, Park, and Vandeputte 1978; Sobis and Vandeputte 1979, 1982; Sobis, Verstuyf, and Vandeputte 1991, 1993; Natsuaki 1983; Katsuse et al. 1981). These histological features of well-differentiated patterns were similar in our case. The pathogenesis was unclear, but there was thus a possibility that the teratoma originated from activated germ cells or a yolk sac that had abnormally developed during pregnancy and remained after parturition.
Footnotes
Acknowledgments
We thank Ms. M. Yamaguchi-Katsura and Ms. Keiko Maeda for expert assistance with histotechnology.
