Abstract
Eugenol has been shown to induce anesthesia in African clawed frogs (Xenopus laevis). The toxicity of eugenol, administered at anesthetic doses, was evaluated in Xenopus frogs with an average body weight of 28.2 ± 13.7 g. Frogs were immersed in 250 mL of an aqueous solution containing 350 µl/L of eugenol for ten minutes and received a single administration (group 1, twelve animals) or three consecutive daily administrations (group 2, twelve animals). In each group, six frogs were scheduled to be euthanized the following day (subgroup A) and the other six were scheduled to be euthanized after a one-week recovery period (subgroup B). Morphologic changes consistent with renal tubular apoptosis affecting distal tubules in the medulla were observed in all subgroup A animals, ranging from mild to moderate in group 1, and from mild to severe in group 2. In subgroup B, renal tubular regeneration was present in all but one animal examined. These findings suggest that eugenol toxicity in amphibians is first manifested by renal tubular apoptosis. Other eugenol-related lesions were massive hepatic necrosis in group 2 (n = 6), hyaline membranes in the lung (n = 5), and adipose tissue hemorrhages in group/subgroup 2B (n = 4).
Introduction
Eugenol (1-allyl-3-methoxy-4-hydroxybenzene) is the main constituent of the essential oil extracted from the dried flower buds of the clove tree (Eugenia caryophyllata). Clove essential oil contains approximately 88% eugenol; other compounds include eugenyl acetate, humulenol, α-humulene, β-caryophyllene, calacorene, calamenene, 2-heptanone, and ethyl hexanoate (Chaieb et al. 2007). Eugenol and clove oil have been used as local anesthetics in dentistry since the seventeenth century, specifically to reduce the pain associated with dental cavities (Curtis 1990). More recently, clove oil has been shown to be an effective, inexpensive, and easy to use general anesthetic agent in numerous species of fish, crustaceans, and mollusks. Clove oil is used in these species to reduce stress and its associated morbidity during transport and diagnostic or experimental procedures (Grush et al. 2004; Iversen et al. 2003; Keene et al. 1998; Soltani et al. 2004; Soto and Burhanuddin 1995; Walsh and Pease 2002; Waterstrat and Plnkham 2005; Woody et al. 2002). The potential of clove oil and eugenol as general anesthetic agents has also been tested in amphibians with promising results. Immersion of leopard frogs (Rana pipiens) in an aqueous solution containing 300 µl/L of clove oil for fifteen minutes induced surgical anesthesia in all frogs (Lafortune et al. 2001). Eugenol also induces general anesthesia in African clawed frogs (Xenopus laevis), a species used in developmental biology, molecular biology, and toxicity studies; immersion of female Xenopus laevis (90 to 140 g body weight) in an aqueous solution containing 350 mg/L of eugenol for fifteen minutes provided surgical anesthesia for at least thirty minutes in most frogs. Selected tissues (heart, lungs, kidneys, liver, skin, and eyes) were without histologic lesions twenty-four hours after eugenol anesthesia (Guenette et al. 2007). However, little is known about the toxic potential of eugenol in amphibians, especially with regard to multiple administrations and different body weights. The aim of the present study was to evaluate the toxicity of eugenol in medium-sized female African clawed frogs after a single or three consecutive daily administrations of eugenol at anesthetic levels, with or without a one-week recovery period.
Material and Methods
Animals
Thirty female Xenopus laevis frogs with an average body weight of 28.2 ± 13.7 g were used (Xenopus 1; Dexter, MI, USA). Frogs were housed in polycarbonate cages measuring 40 × 20 × 15 cm (Ancare, Bellmore, NY, USA) filled with purified water (at least 2L per frog). The water was obtained from our facility's water treatment system (filtering through activated charcoal and UV treatment). Water and room temperatures were kept at 21.2°C ± 2°C. The following water parameters were recorded, and all were within normal values: pH 6.8 to 7.3, no chlorine or chloramines, ammonia concentration 0.2 mg/mL (normal values 0.4 to 0.6 mg/mL), no nitrites (normal values < 1 mg/mL), no hardness (normal values 70 to 150 mg/mL), and no copper (all-PVC tubing). Water was changed and polycarbonate cages were cleaned every two days. Animals were fed every other day with commercial Xenopus food (Xenopus Express, Brooksville, FL, USA). The experimental protocol was approved by the Institutional Animal Care and Use Committee of the Faculty of Veterinary Medicine of the University of Montreal prior to animal use, in accordance with the guidelines of the Canadian Council on Animal Care.
Experimental Design
Two groups of twelve frogs each were anesthetized with eugenol by bath immersion. Six frogs kept in water from the same source were used as controls. In group 1, animals received a single administration of eugenol, whereas in group 2, animals received three consecutive daily administrations of eugenol. Each group was further subdivided as follows: in subgroup A, animals were scheduled to be euthanized twenty-four hours after the last eugenol bath, whereas in subgroup B, animals were scheduled to be euthanized after a one-week recovery period. Six control animals were used; two were euthanized every week, starting on day 1 of the experimental period.
Bath Immersion Protocol
The concentration and time of exposure to eugenol were selected based on a previously published study (Goulet et al. 2010). An aqueous solution of eugenol was prepared by measuring 350 µL of eugenol (purity 99%, Sigma-Aldrich, St. Louis, MO, USA) with a pipette and adding it to 1 L of purified water in a 1-L Pyrex glass bottle (Corning, Lowell, MA, USA). The solution was thoroughly agitated, and 250 mL was immediately poured into a metal container. Each frog was individually placed in the metal container so it was completely immersed in the solution, and the container was covered so the animal was in full darkness. After a ten-minute immersion period, the frog was removed from the solution, thoroughly rinsed with purified water, and placed in a water pan for the recovery period. The frogs were periodically sprayed with purified water during the recovery period to ensure proper skin hydration. The frogs were monitored every five minutes for the presence of the righting reflex, and each frog was returned to its original enclosure when it was able to right itself after being placed on its back (positive righting reflex).
Pathology
At the end of the experimental period, euthanasia was performed on all frogs, including control animals, by decapitation following anesthesia with eugenol as previously described (Goulet et al. 2010). A complete necropsy was performed and the following organs were examined for the presence of gross changes and sampled: adipose tissue (coelomic fat bodies), bone with bone marrow (femur), brain, colon, eyes, gall bladder, heart, kidneys, liver (two sections), lungs, oral mucosa, skeletal muscle (thigh), skin (ventral and dorsal), small intestine (three sections), spleen, stomach, and urinary bladder. Tissues were fixed in 10% neutral buffered formalin, routinely processed for histology, embedded in paraffin, and sectioned at 4 µm. Histological slides were stained with hematoxylin, eosin, phloxin, and saffron (HEPS).
Results
Clinical Observations
Group 1 animals (single administration) were clinically normal and exhibited normal behavior throughout the study. The only observed side effect related to eugenol anesthesia was vomiting in two of the twelve animals during the anesthetic recovery phase. One frog in group 1B (single administration, one-week recovery) was found dead in its aquarium on the fourth day following eugenol immersion. Marked postmortem changes precluded any histologic examination of this animal. In group 2 (three consecutive daily administrations), frogs showed depression, were reluctant to move, and swam less vigorously than control animals from day 3 up to the end of the experimental period. In group 2A (three administrations, no recovery), one animal was found dead on the scheduled date of euthanasia (day 2). In group 2B (three administrations, one-week recovery), two animals were found dead on the scheduled date of euthanasia (day 8). All control frogs remained clinically normal throughout the experimental period.
Gross Lesions
In groups 1A, 1B, and 2A, no significant changes were observed during necropsy. In group 2B (three administrations, one-week recovery), four animals, including the two that had been found dead, had gross lesions in the liver, coelomic fat bodies, and kidneys. In these animals, the liver was diffusely pale and irregularly mottled with yellowish and red areas; it was soft and friable on sectioning. There were multiple petechiae and hemorrhagic foci in the adipose tissue, predominantly in the coelomic fat bodies. There were occasional small red foci, <1 mm in diameter, on the surface of the kidneys. The other two frogs in group 2B did not have any gross changes. All control frogs were grossly unremarkable.
Histopathology
The microscopic changes associated with the administration of eugenol in African clawed frogs are summarized in Table I. Renal lesions were graded as follows : mild, <10% of tubules affected; moderate, 10–25% of tubules affected ; marked, 25–50% of tubules affected; severe, >50% of the tubules affected.
Summary of histologic lesions associated with single or repeated eugenol administrations in African clawed frogs (Xenopus laevis). Number of frogs with histologic lesions (N = 6).

a Marked postmortem changes precluded histologic examination in one frog found dead.
In group 1A (single administration, twenty-four-hour sacrifice), lesions were limited to the kidney. Lesions were morphologically consistent with apoptosis and involved distal tubules in the ventromedial zone (medulla); they were mild in four animals and moderate in two others. Affected tubules were lined with rounded epithelial cells with, hypereosinophilic cytoplasm, and pyknotic, karyorrhectic or inapparent nuclei that were occasionally sloughed in tubular lumina (Figure 1 ).
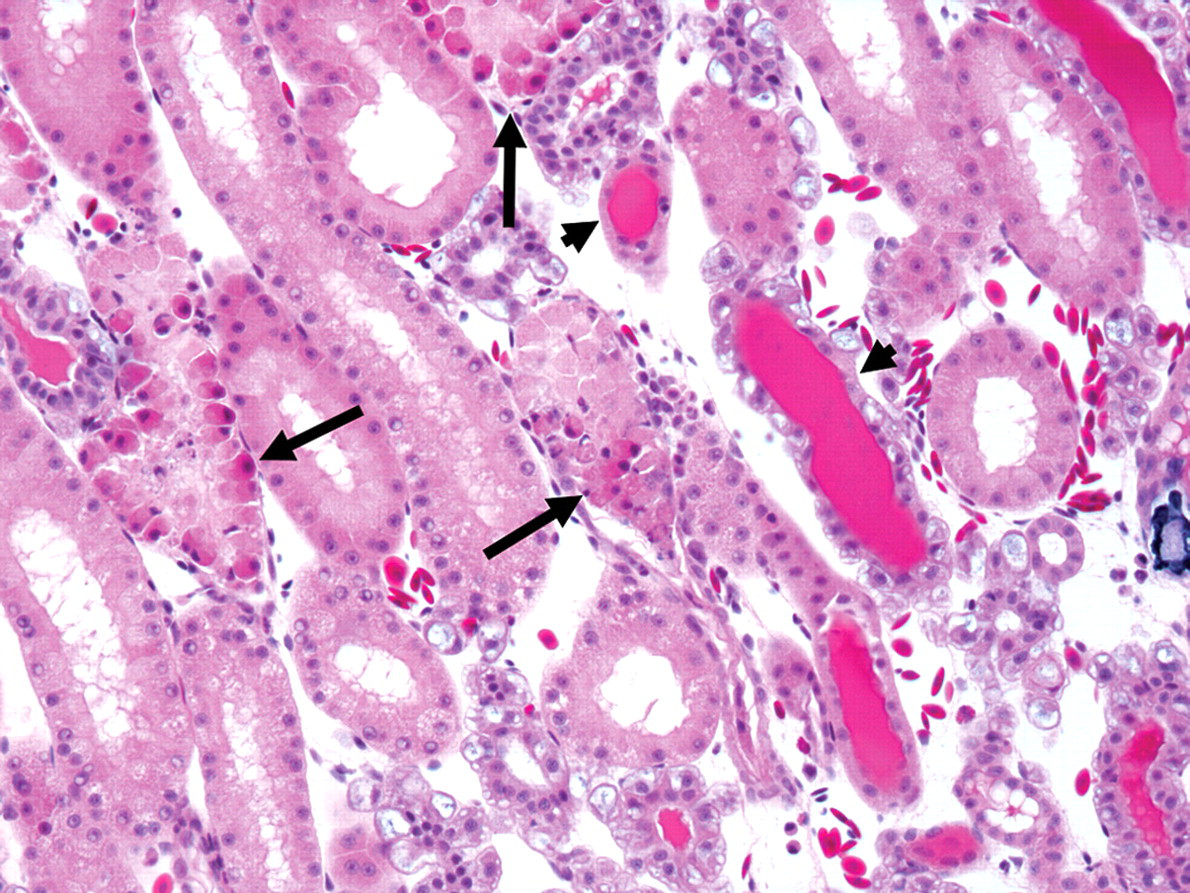
Renal tubular apoptosis in Xenopus laevis that received a single eugenol administration and was sacrificed at twenty-four hours. Apoptotic tubular epithelial cells are rounded with hypereosinophilic cytoplasm and pyknotic, karyorrhectic, or inapparent nuclei. The distal tubules are affected (arrows). Note the tubular proteinosis in less-affected tubules (arrowheads). Hematoxylin, eosin, phloxin, and saffron, 200×.
In group 1B (single administration, one-week recovery), all five animals examined had evidence of renal tubular regeneration. Regenerating tubules were lined by attenuated to large epithelial cells with basophilic cytoplasm, vesicular nuclei with prominent nucleoli, and rare mitotic figures (Figure 2). Interspersed between regenerating tubules, rare to occasional tubules exhibited features consistent with apoptosis. One animal also had a few hyaline membranes lining alveolar sacs and bronchioles. Hyaline membranes were composed of multifocal 20 to 40 µm-thick layers of eosinophilic fibrillar material admixed with rare cellular debris, macrophages and erythrocytes (Figure 3 ).
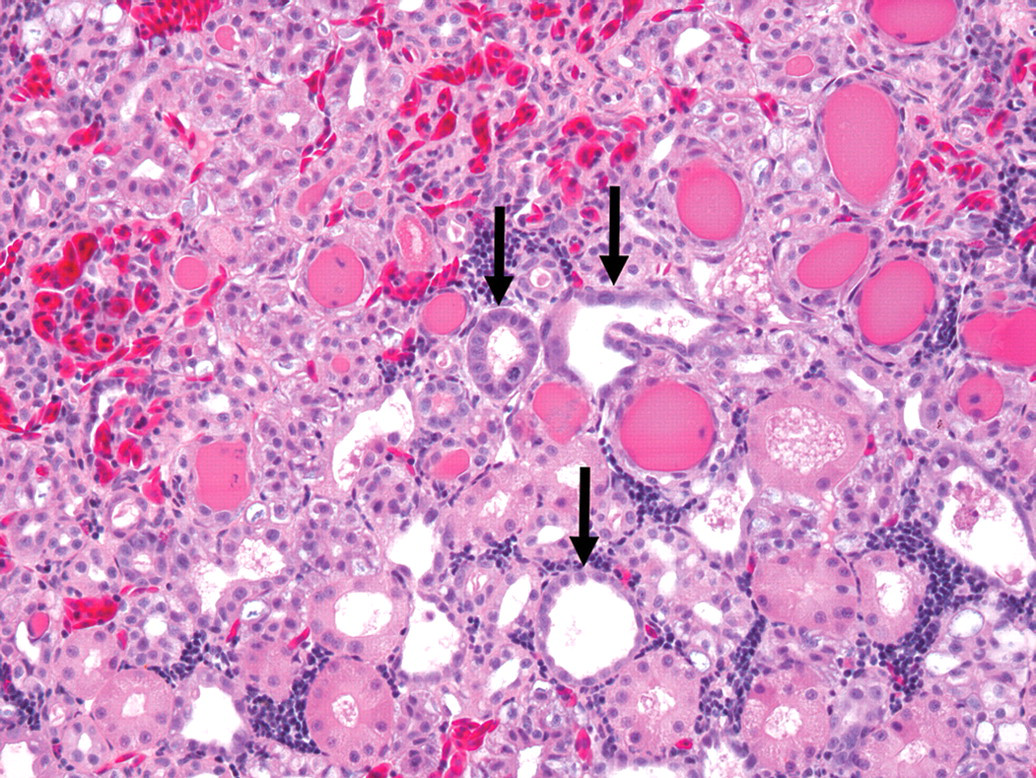
(a) Renal tubular regeneration in Xenopus laevis that received a single eugenol administration and had one week of recovery period. Regenerating epithelial cells are attenuated to large, with basophilic cytoplasm and vesicular nuclei (arrows). Tubular proteinosis is present. HEPS, 200×.
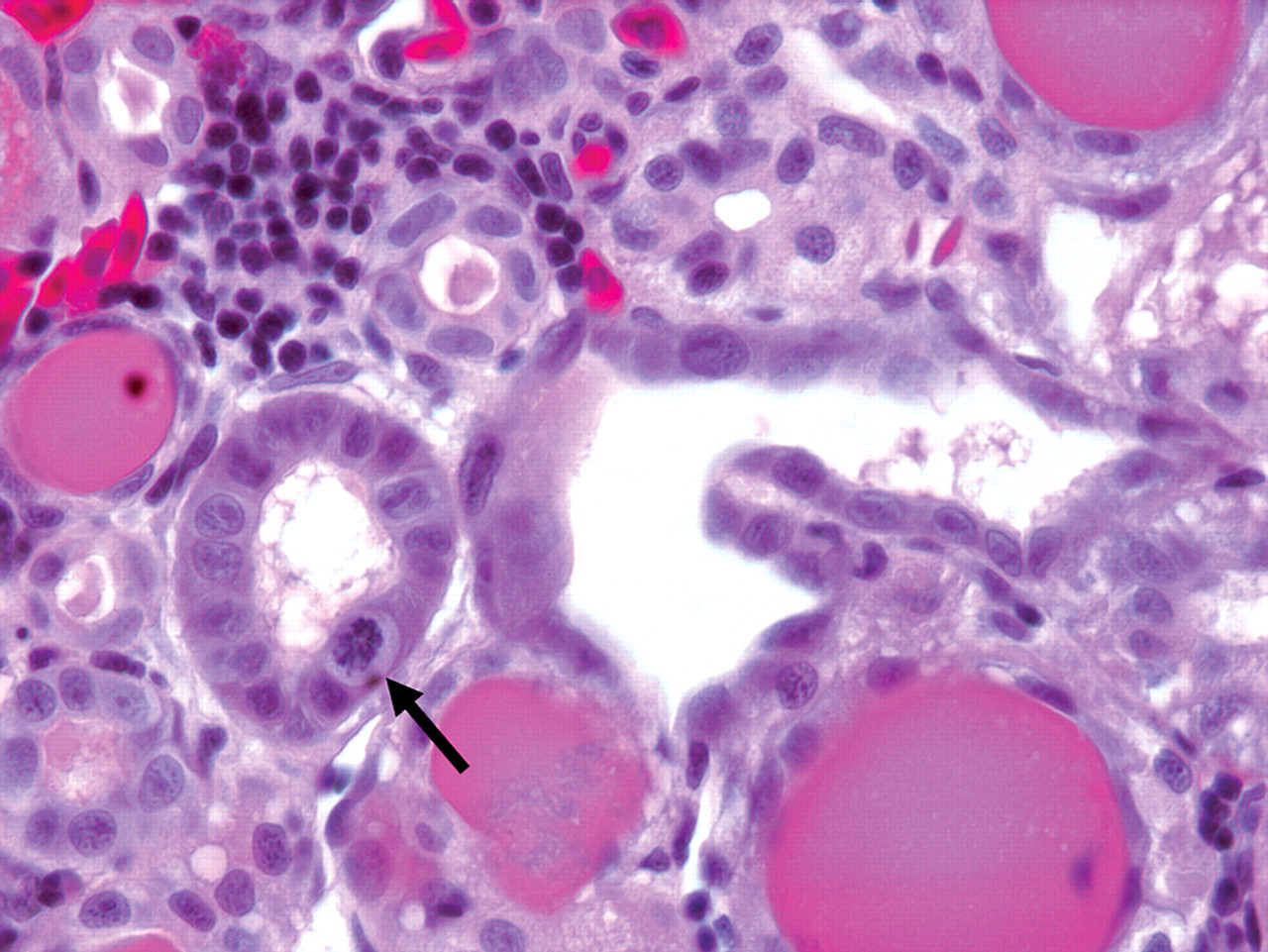
(b) Higher magnification of Fig 2a. Note vesicular nuclei with prominent nucleoli. A mitotic figure is present (arrow). Hematoxylin, eosin, phloxin, and saffron, 630×.
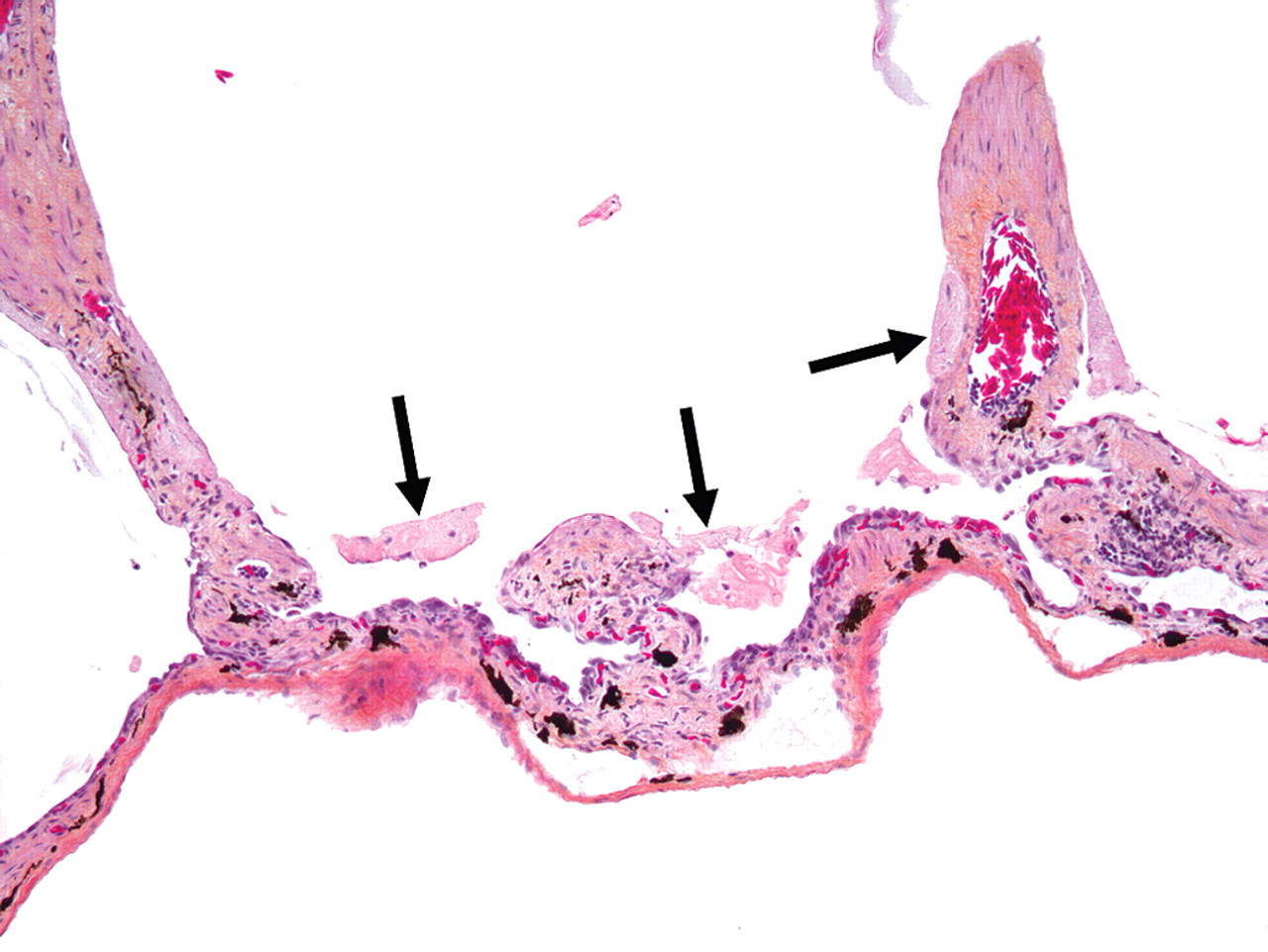
Pulmonary hyaline membranes from Xenopus laevis that received a single eugenol administration and had one week of recovery prior to euthanasia. There is a hyaline membrane lining the wall of an alveolar sac (arrows). Hematoxylin, eosin, phloxin, and saffron, 100×.
In five out of six animals in group 2A (multiple administrations, twenty-four-hour sacrifice), lesions were limited to the kidney and were morphologically consistent with tubular apoptosis. Renal lesions were mild (n = 1), moderate (n = 2), marked (n = 1), or severe (n = 2). In addition to severe tubular apoptosis, one animal (found dead) also had rare hyaline membranes in the lung and massive hepatic necrosis. Hepatic necrosis was characterized by diffuse disruption of hepatic cords with parenchymal collapse, and necrosis and loss of hepatocytes; necrotic hepatocytes were rounded, with vacuolated and pale cytoplasm and pyknotic, karyorrhectic, or karyolytic nuclei (Figure 4).
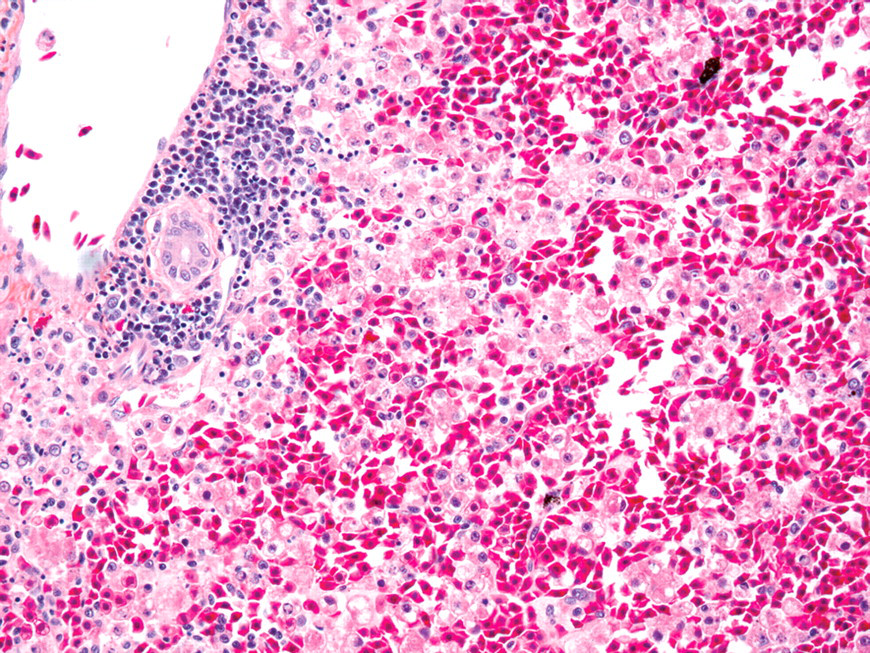
Massive hepatic necrosis from Xenopus laevis that received 3 consecutive daily eugenol administrations and was sacrificed at 24 hr after last administration. There is diffuse disruption of hepatic cords with stromal collapse. Necrotic hepatocytes are rounded, dissociated and admixed with red blood cells. HEPS, 200×.
In group 2B (multiple administrations, one-week recovery), renal tubular regeneration and hepatic necrosis were present in five animals. Hepatic necrosis was massive except in one frog in which large areas of necrosis were adjacent to unaffected areas. Mild to marked bile duct hyperplasia was present in all five frogs with hepatic necrosis. Consistent with the gross findings, hemorrhagic foci were seen histologically in the coelomic fat bodies of four animals. Hemorrhages were multifocal and random in distribution. Hyaline membranes were seen in the lungs of half the frogs from this group and varied from minimal to prominent. In affected animals, alveolar sacs were multifocally lined by cuboidal epithelial cells with intracytoplasmic eosinophilic globules. One frog had neither gross nor histologic lesions.
Two of the six control animals and four of the twenty-three treated animals examined had one to multiple visceral granulomas. Most granulomas measured 50 to 100 µm but were occasionally up to 300 µm in diameter, and they consisted of discrete aggregates of histiocytes with fewer lymphocytes and rare heterophils and multinucleated giant cells. The most common locations were the liver and adipose tissue (coelomic fat bodies, retrobulbar and pericardial fat). Periodic acid-Schiff, Ziehl-Neelsen, and Gram stains did not reveal any microorganisms in the granulomas.
Discussion
In this experiment, eugenol was shown to be toxic to African clawed frogs when administered at anesthetic dosage. The earliest lesion of eugenol toxicity was morphologically consistent with tubular apoptosis in the kidney, affecting the distal tubules of the medulla. The severity of the renal lesions increased with repeated administrations. These lesions appeared to be reversible, as evidenced by the presence of renal tubular regeneration after a recovery period (subgroup B). Furthermore, they were not associated with evidence of clinical disease in group 1 (single administration); in the latter, the only other lesion was minimal hyaline membrane formation, observed in the lung of a single subgroup B (recovery) frog. One frog from group 1B was found dead; the cause of death could not be established. As the recovery period for frogs in group 1B was only one week, we cannot extrapolate on long-term effects. Tubular apoptosis involved distal tubules and was mild to moderate; the lack of overt adverse reaction in affected frogs from group 1 suggests that the functional reserve of the kidney was not compromised (Sebastian et al. 2007). Pulmonary hyaline membranes were also observed in four frogs in group 2 (multiple administrations), more frequently in frogs with a recovery period. Other lesions associated with eugenol administration were generally massive hepatic necrosis and multifocal hemorrhages in adipose tissues; they occurred only in frogs with multiple administrations. These frogs had clinical signs and, in three cases, the lesions likely caused death. Granulomas were present in six animals, including two control and four treated animals. Since these lesions were present in both treated and control animals, they were interpreted as incidental findings and not related to treatment.
Rare cases of clove oil intoxication have been reported in humans, mainly in children under two years of age. Clove oil is sold over the counter for the relief of toothache or as an aromatherapy agent, and intoxications occur in children following accidental ingestion. Intoxication in children is usually manifested by central nervous system depression that can progress to coma, metabolic abnormalities including anion gap acidosis and hypoglycemia, severe hepatic failure, urinary abnormalities, and coagulation deficits (Brown et al. 1992; Eisen et al. 2004; Hartnoll et al. 1993; Janes et al. 2005; Lane et al. 1991).
Urinary abnormalities occur with clove oil toxicity in children and include increased blood urea and creatinine levels, proteinuria, and hematuria (Brown et al. 1992; Hartnoll et al. 1993; Janes et al. 2005; Lane et al. 1991). In one report, these changes were hypothesized to be the result of direct nephrotoxicity of unchanged eugenol (Lane et al. 1991). In our study, renal toxicity was the most consistent eugenol treatment–related lesion. Histological evidence of tubular apoptosis was present in all but one of the animals examined. The severity of the lesion was dose related, and the lesion was likely reversible, as evidenced by the presence of tubular regeneration after a one-week recovery period. A diagnosis of acute tubular necrosis was considered; however, the morphological features of the lesion (cell rounding, hypereosinophilia, pyknosis, and lack of inflammation) were more consistent with apoptosis (Kumar et al. 2010). The mechanism of tubular apoptosis was not investigated and thus remains undetermined, although the apparent preservation of the basement membrane suggests nephrotoxicity more than ischemia (Maxie and Newman 2007). The exact segment of the nephron involved was identified as distal tubules; although, according to Wiechmann and Wirsig-Wiechmann, the affected tubules correspond to the thick limb of the loop of Henle (Wiechmann and Wirsig-Wiechmann 2003), most authors do not recognize the equivalent of this segment in amphibians.
Hepatic failure in human cases of clove oil toxicity is an early event and is evidenced by a marked increase in serum transaminase levels during the first twenty-four hours following ingestion of clove oil (Brown et al. 1992; Eisen et al. 2004; Hartnoll et al. 1993; Janes et al. 2005). This finding is in contrast with the present study, in which hepatocellular necrosis occurred only in animals that received three eugenol administrations. In man, the main pathway of eugenol metabolism is the formation of conjugates with sulfate and glucuronide by the liver (Fischer et al. 1990). With overdose, the sulfate and glucuronide pathways are saturated and eugenol is conjugated with glutathione. Incubation of isolated rat hepatocytes with eugenol results in intracellular glutathione depletion which precedes hepatocellular death. Pretreatment of hepatocytes with N-acetylcysteine, which provides cysteine necessary for glutathione synthesis, significantly reduces intracellular glutathione loss and cell death, whereas pretreatment with glutathione-depleting drugs increases the cytotoxicity of eugenol (Thompson et al. 1991). Moreover, in mice treated with eugenol by gavage, hepatotoxicity, characterized by extensive centrilobular necrosis, is observed only in animals pretreated with an inhibitor of glutathione synthesis (Mizutani et al. 1991). Pretreatment with cytochrome P450 inhibitors in these mice partially or completely prevented hepatotoxicity, whereas pretreatment with inducers of cytochrome P450 promoted hepatotoxicity (Mizutani et al. 1991). These results suggest that the hepatotoxicity of eugenol involves depletion of intracellular glutathione and loss of its protective antioxidant effect, and that subsequent hepatocellular damage is at least partly a result of a cytochrome P450-dependant eugenol metabolite, probably a quinone methide (Thompson et al. 1998). This mechanism of toxicity is similar to what is observed in cases of acetaminophen toxicity, and a treatment regimen with N-acetylcysteine, similar to what is advocated in cases of acetaminophen poisoning, has been proposed in cases of eugenol intoxication (Hartnoll et al. 1993; Janes et al. 2005). In our study, hepatic toxicity was seen only in animals with multiple administrations, especially those allowed a recovery period. The distribution was massive in all but one case, in which large areas were affected. Again, the exact mechanism of hepatotoxicity was not investigated.
Coagulopathy is often present in clove oil intoxication in infants and is usually manifested by an increase in prothrombin time (Brown et al. 1992; Eisen et al. 2004; Hartnoll et al. 1993; Janes et al. 2005). In one report, blood coagulation deficits were profound and characterized by increased prothrombin ratio and activated partial thromboplastin ratio, thrombocytopenia, increased crosslinked fibrin degradation products (XDP) and hematemesis, among other anomalies. This child also had fulminant hepatic failure and low levels of vitamin K–dependent coagulation factors (factors II, VII, IX, and X), and the authors considered that the hepatic damage was the main cause of the coagulation deficits (Brown et al. 1992). In the present study, we observed multifocal hemorrhages in the coelomic fat bodies in frogs that had repeated eugenol administrations, all of which had concurrent massive hepatic necrosis, so liver lesions were the most likely cause of the hemorrhages in these animals. However, other mechanisms might have played a role in the development of hemorrhages. In vitro studies with rabbit and human platelets have demonstrated the antiplatelet aggregation activity of eugenol, which was at least partly owing to the inhibition of cyclooxygenase-dependant formation of thromboxane A2 (Chen et al. 1996; Raghavendra and Naidu 2009). Eugenol was found to be twenty-nine times more effective than aspirin in inhibiting arachidonic acid–induced platelet aggregation (Raghavendra and Naidu 2009).
The pulmonary toxicity of clove oil and eugenol has been reported in humans and in animals. Noncardiogenic pulmonary edema and acute respiratory distress were observed in a thirty-two-year-old woman who voluntarily administered herself an intravenous dose of clove oil (Kirsch et al. 1990). Intravenous infusion of eugenol in adult male Sprague-Dawley rats caused acute lung injury of rapid onset, characterized by perivascular and interstitial edema and extravasation of erythrocytes and neutrophils in alveolar spaces. Furthermore, intravenous eugenol caused an increase in the protein content of the bronchoalveolar lavage fluid of treated rats, which was decreased with pretreatment of rats with the antioxidant enzymes superoxide dismutase and catalase. These findings suggest that eugenol-induced lung injury is at least in part mediated through an oxidative mechanism (Wright et al. 1995). Pulmonary toxicity possibly associated with smoking clove cigarettes has also been reported to the Centers for Disease Control. Severely affected individuals experienced acute respiratory distress, pulmonary edema, bronchospasm and hemoptysis (CDC 1985). When administered by intratracheal instillation to F344 rats, eugenol was found to be more toxic than other constituents of cloves. Pulmonary changes in these rats included congestion, hemorrhages, acute emphysema, and pulmonary edema. It was also concluded that when administered to rats by intratracheal instillation, eugenol is at least 200 times more toxic than when administered by gavage (LaVoie et al. 1986). In the present study, pulmonary hyaline membranes were always concomitant with renal lesions of at least moderate severity and mostly occurred after repeated administrations. Hyaline membranes were present in only one animal after a single administration and a one-week recovery period, and these lesions were minimal. Although the pulmonary lesions could represent direct damage to alveolar epithelium and/or endothelium induced by eugenol or one of its metabolites, we cannot exclude the possibility that they are secondary to the renal lesions. In mammals, uremia is one of the known causes of diffuse alveolar damage (Husain 2010); we did not find any reference stating renal failure could cause alveolar damage in amphibians. In group 2B, most animals with hyaline membranes had some regions of the lungs where alveolar sacs were lined by cuboidal epithelial cells with intracytoplasmic hyaline globules. We interpreted these changes as being a regenerative attempt from the damaged pneumocytes lining the alveolar sacs. However, this change could not qualify as type II pneumocyte hyperplasia, since ultrastructural studies have demonstrated that amphibians lack type II pneumocytes (Meban 1973).
The only reported eugenol-related toxic lesion in amphibians is a case of cutaneous necrosis in African clawed frogs following topical application using a eugenol-soaked gauze (Ross et al. 2006). In large-size Xenopus laevis female frogs (90 to 140 g), no histologic changes were observed following a single immersion of frogs in a solution containing 350 mg/mL eugenol for fifteen minutes (Guenette et al. 2007). These results differ from those of the present study, in which renal tubular apoptosis was present in all animals and mild pulmonary hyaline membranes were present in one animal after a recovery period, following a single administration of eugenol at a similar concentration and for a shorter period of time (ten minutes instead of fifteen minutes). These results suggest that smaller frogs are more susceptible to the toxic effects of eugenol, and that the therapeutic index of eugenol in amphibians may be related to metabolic maturity. The half-life of eugenol is relatively long in amphibians (four hours), which may allow for the accumulation of this lipophilic compound and the development of greater toxicity after consecutive daily administrations (Guenette et al. 2007). Additional studies are needed to determine adequate concentration and time of exposure to a eugenol solution that are both safe and effective for the anesthesia of African clawed frogs of different body weights. To our knowledge, the present study constitutes the first comprehensive evaluation of the toxicity of eugenol in amphibians.
Footnotes
Acknowledgments
We would like to thank Jacinthe Cardin, Josée Letellier, Nancy Veilleux, Louise Cossette, and Maria Paula Colas Ramirez for their excellent technical assistance, and Marco Langlois for the preparation of the figures.
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article. The author(s) received no financial support for the research and/or authorship of this article.
