Abstract
Methyl eugenol induces neuroendocrine (NE) cell hyperplasia and tumors in F344/N rat stomach. Detailed histopathological and immunohistochemical (IHC) characterization of these tumors has not been previously reported. The objective of this study was to fill that data gap. Archived slides and paraffin blocks were retrieved from the National Toxicology Program Archives. NE hyperplasias and tumors were stained with chromogranin A, synaptophysin, amylase, gastrin, H+/K+ adenosine triphosphatase (ATPase), pepsinogen, somatostatin, and cytokeratin 18 (CK18) antibodies. Many of the rats had gastric mucosal atrophy, due to loss of chief and parietal cells. The hyperplasias and tumors were confined to fundic stomach, and females were more affected than the males. Hyperplasia of NE cells was not observed in the pyloric region. Approximately one-third of the females with malignant NE tumors had areas of pancreatic acinar differentiation. The rate of metastasis was 21%, with liver being the most common site of metastasis. Immunohistochemically, the hyperplasias and tumors stained consistently with chromogranin A and synaptophysin. Neoplastic cells were also positive for amylase and CK18 and negative for gastrin, somatostatin, H+/K+ ATPase, and pepsinogen. Metastatic neoplasms histologically similar to the primary neoplasm stained positively for chromogranin A and synaptophysin. Based on the histopathological and IHC features, the neoplasms appear to arise from enterochromaffin-like cells.
Introduction
Methyl eugenol is a colorless to pale yellow, oily liquid and is a natural constituent of many essential oils such as rose, basil, hyacinth, pimento, citronella, anise, nutmeg, mace, cinnamon leaves, pixuri seeds, and laurel fruits and leaves (National Toxicology Program [NTP] 2000). It belongs to the alkoxy-benzene class of chemicals, which also includes isoeugenol, eugenol, estragole, and safrole. It is produced by methylation of eugenol. Because of the widespread use and structural similarity to known carcinogens such as safrole, the National Toxicology Program (NTP) tested methyl eugenol for toxicity and carcinogenesis in a 2-year bioassay in F344/N rats and B6C3F1 mice (NTP 2000; Johnson et al. 2000). In addition to liver tumors in male and female rats, and renal tubule adenomas, mammary gland fibroadenomas, malignant mesotheliomas in the abdominal cavity, and fibroma/fibrosarcoma of the skin in male rats, methyl eugenol induced rare neuroendocrine (NE) tumors in the fundic region of the glandular stomach of male and female F344/N rats.
The fundic mucosa of the glandular stomach is comprised of 5 major cell types that include the surface mucous cells, mucous neck cells, parietal cells, chief cells, and NE cells (Helander 1981). These cells arise from a self-renewing stem cell in the stomach (Mills and Shivdasani 2011). There are 5 major types of NE cells in gastric mucosa that include serotonin-producing enterochromaffin cells, histamine-producing enterochromaffin-like (ECL) cells, gastrin-producing G cells, somatostatin-secreting D cells, and ghrelin-secreting P cells (Modlin and Nangia 1992; Lawrence et al. 2011).
In rats, 65% of the NE cells are ECL cells, which are located in the basal half of the fundic mucosa (Hakanson et al. 1994). ECL cells regulate secretion of hydrochloric acid by parietal cells. Hypochlorhydria in the stomach causes increased secretion of gastrin, which has a trophic effect on ECL cells in the fundic mucosa (Ryberg et al. 1990). Thus, any significant damage to the parietal cells or the gastric mucosa with subsequent hypochlorhydria can result in increased activation/proliferation of ECL cells. Chemicals such as butachlor and methyl eugenol that cause gastric mucosal atrophy, hypochlorhydria, and hypergastrinemia have been shown to induce gastric NE tumors (Thake et al. 1995; Hard et al. 1995; NTP 2000). In a previous NTP study, methyl eugenol induced NE cell hyperplasia, and benign and malignant NE tumors in the fundic stomach of male and female F344/N rats (Johnson et al. 2000). However, a detailed histopathological and immunohistochemical (IHC) characterization of these lesions has not been reported. In this article, we present a detailed histopathological description of NE cell hyperplasia and NE tumors along with their IHC staining characteristics.
Materials and Methods
The tissues used in this study were from the NTP (2000) 2-year carcinogenicity bioassay of methyl eugenol in F344/N rats. Groups of 50 male and 50 female F344/N rats were administered 0-, 37.5-, 75-, or 150-mg/kg methyl eugenol in 0.5% methylcellulose by gavage, 5 days per week for 105 weeks. In a stop-exposure study, additional groups of 50 male and female rats were administered 300 mg/kg of methyl eugenol for 52 weeks followed by the 0.5% methylcellulose vehicle alone for another 53 weeks. Husbandry and experimental procedures were in compliance with requirements set forth in the U.S. Public Health Service’s Guide for the Care and Use of Laboratory Animals.
Histopathology
For histopathological characterization, the original hematoxylin and eosin (H&E)-stained tissue sections of the fundic stomach with a diagnosis of NE cell hyperplasia and benign and malignant NE tumors reported in the NTP (2000) Technical Report 491 were reviewed microscopically. The incidences of hyperplasia and tumors are presented in Table 1. In addition, H&E-stained slides of metastatic NE tumors in other tissues were reviewed (Table 2).
Incidences of selected nonneoplastic lesions, and tumors of glandular stomach in male and female rats in a 2-year gavage bioassay of methyl eugenol.
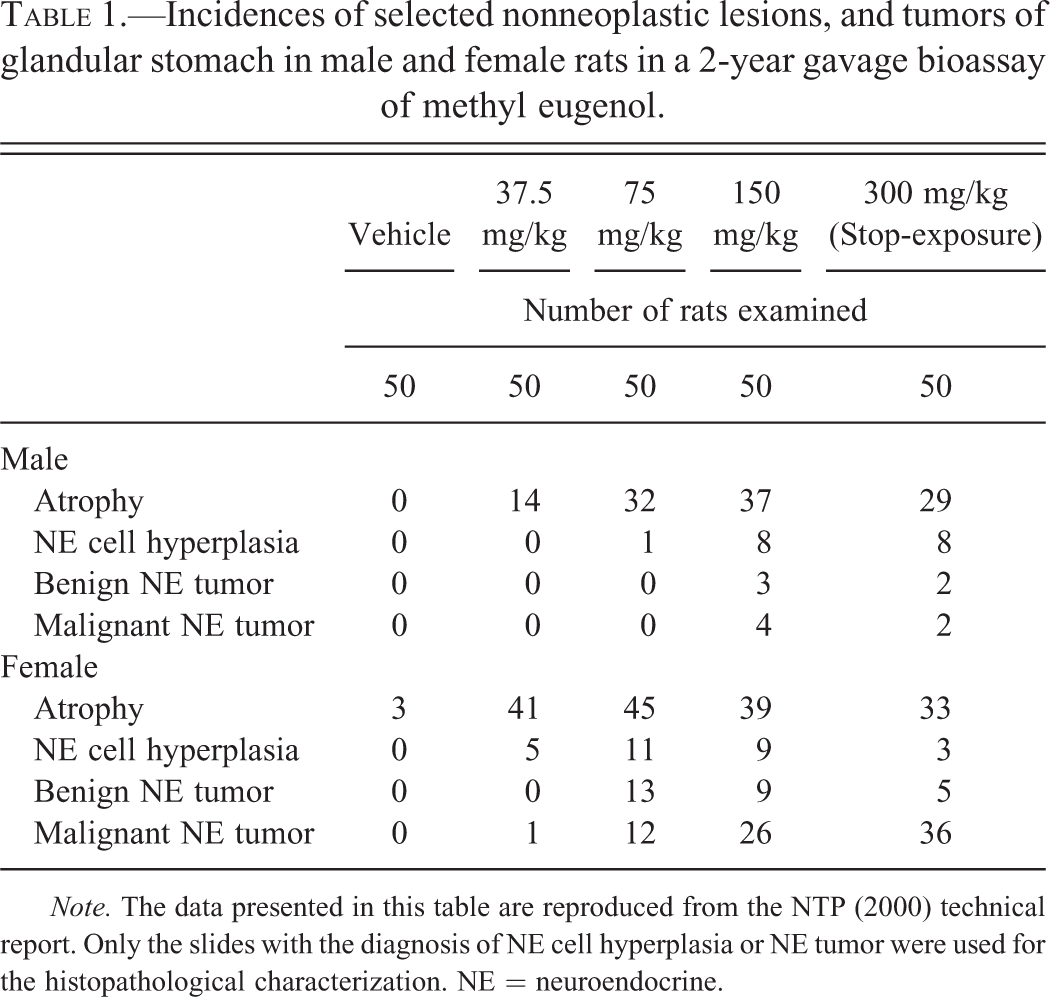
Note. The data presented in this table are reproduced from the NTP (2000) technical report. Only the slides with the diagnosis of NE cell hyperplasia or NE tumor were used for the histopathological characterization. NE = neuroendocrine.
List of rats with gastric neuroendocrine cell hyperplasia and tumor that were used for immunohistochemical characterization.
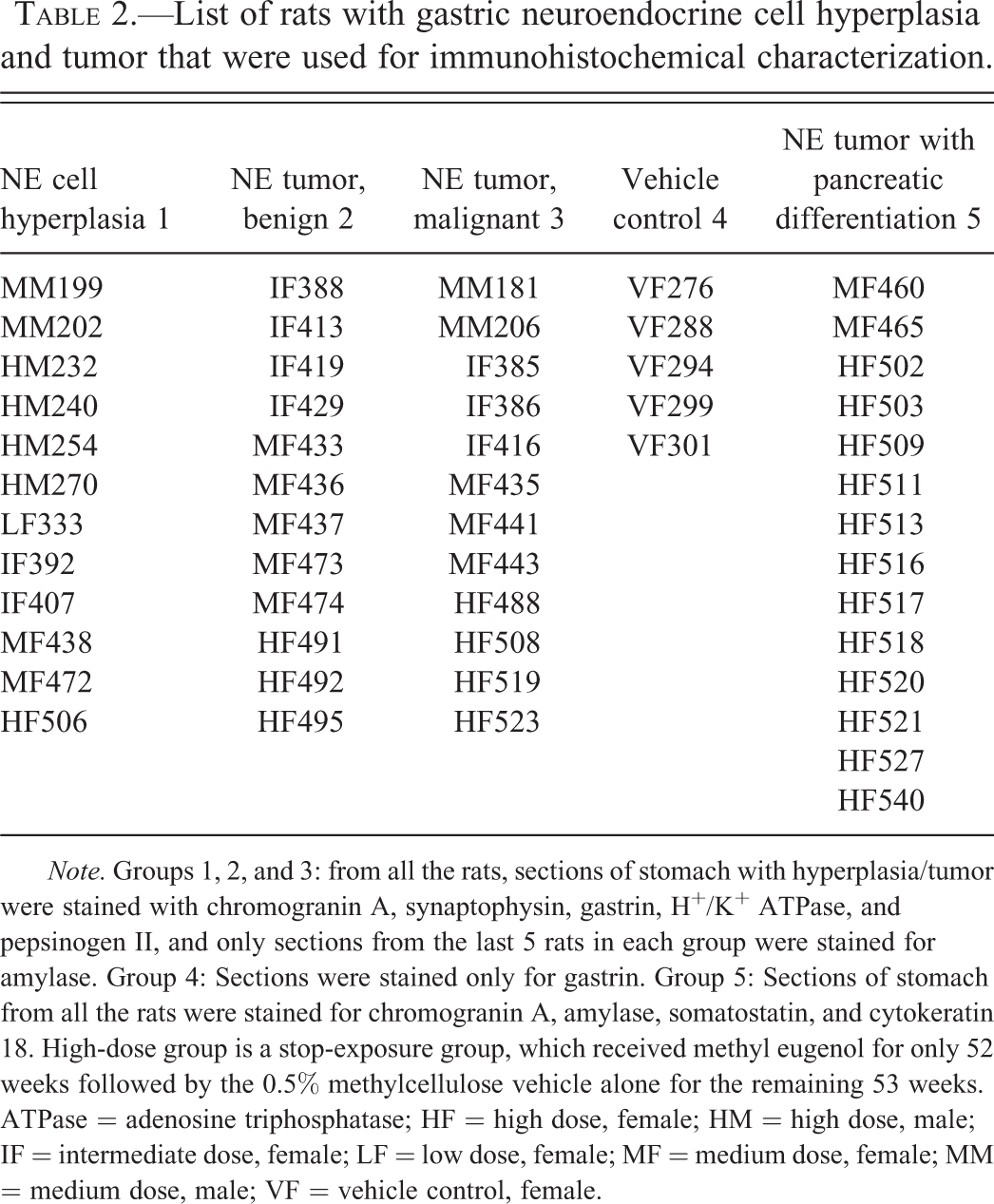
Note. Groups 1, 2, and 3: from all the rats, sections of stomach with hyperplasia/tumor were stained with chromogranin A, synaptophysin, gastrin, H+/K+ ATPase, and pepsinogen II, and only sections from the last 5 rats in each group were stained for amylase. Group 4: Sections were stained only for gastrin. Group 5: Sections of stomach from all the rats were stained for chromogranin A, amylase, somatostatin, and cytokeratin 18. High-dose group is a stop-exposure group, which received methyl eugenol for only 52 weeks followed by the 0.5% methylcellulose vehicle alone for the remaining 53 weeks. ATPase = adenosine triphosphatase; HF = high dose, female; HM = high dose, male; IF = intermediate dose, female; LF = low dose, female; MF = medium dose, female; MM = medium dose, male; VF = vehicle control, female.
Immunohistochemistry
Paraffin blocks of the glandular stomach were retrieved from the NTP Archives and 5-µm thick sections were prepared for staining with chromogranin A, synaptophysin, amylase, gastrin, H+/K+ adenosine triphosphatase (ATPase), pepsinogen, somatostatin, and CK18 antibodies. The details of the individual blocks used for the IHC characterization of NE cell hyperplasias, NE tumors, and metastatic NE tumors are presented in Tables 2 and 3. Summaries of the IHC staining protocols are presented in Table 4. The tissue sections were deparaffinized in xylene and rehydrated through decreasing grades of alcohol followed by Tris Buffered Saline automation wash buffer (Biocare Medical, CA). Endogenous peroxidase was quenched by incubating the tissue sections with 3% hydrogen peroxide. Endogenous biotin and nonspecific binding of avidin/biotin were blocked using an avidin–biotin blocking kit (Vector Laboratories, CA) after which the sections were incubated with the primary antibody (Table 4). For the negative controls, tissue sections were incubated either with normal serum from the species in which the primary antibody was raised or with normal mouse IgG. The concentration of immunoglobulin in the negative control serum was normalized to that of primary antibody. Subsequently, the sections were incubated with a secondary antibody (Table 4) followed by Vectastain Elite ABC kit (Vector Laboratories, CA) for 30 min. For synaptophysin staining, ready-to-use Horseradish peroxidase (HRP)-conjugated streptavidin (Biogenex, CA) was used instead of the ABC kit. Antigen–antibody complex was visualized using 3,3′-diaminobenzidine (DAB) chromogen. Finally, the slides were counterstained with hematoxylin, dehydrated through increasing grades of alcohol, cleared in xylene, and coverslipped. For chromagranin A, synaptophisin, and amylase staining, sections of pancreas were used as positive control tissue. For gastrin, H+/K+ ATPase, and pepsinogen, NE cells in the pyloric stomach, normal parietal cells, and normal chief cells in the fundic stomach were used as positive controls, respectively.
List of rats and tissues with metastatic neuroendocrine tumor of the stomach.
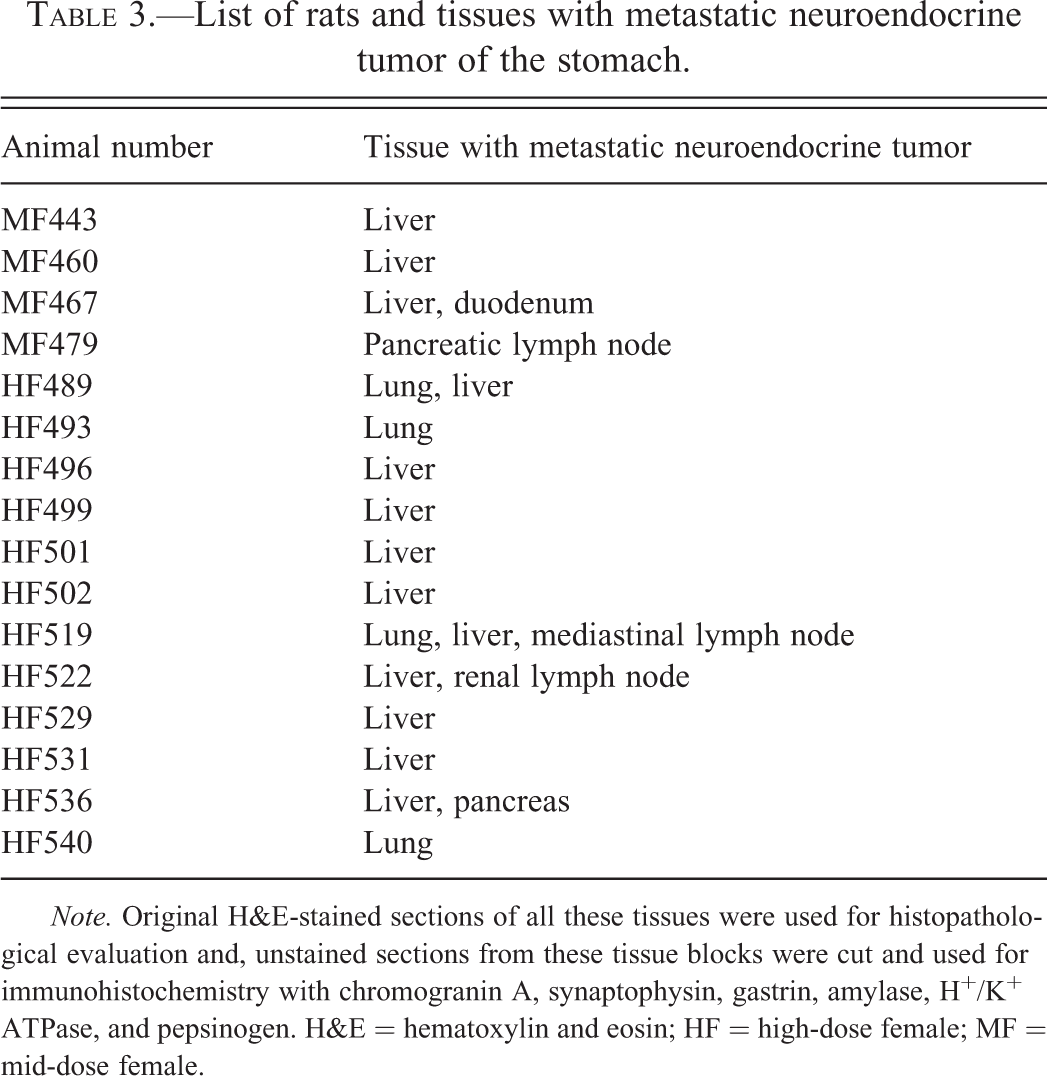
Note. Original H&E-stained sections of all these tissues were used for histopathological evaluation and, unstained sections from these tissue blocks were cut and used for immunohistochemistry with chromogranin A, synaptophysin, gastrin, amylase, H+/K+ ATPase, and pepsinogen. H&E = hematoxylin and eosin; HF = high-dose female; MF = mid-dose female.
Details of immunohistochemistry.
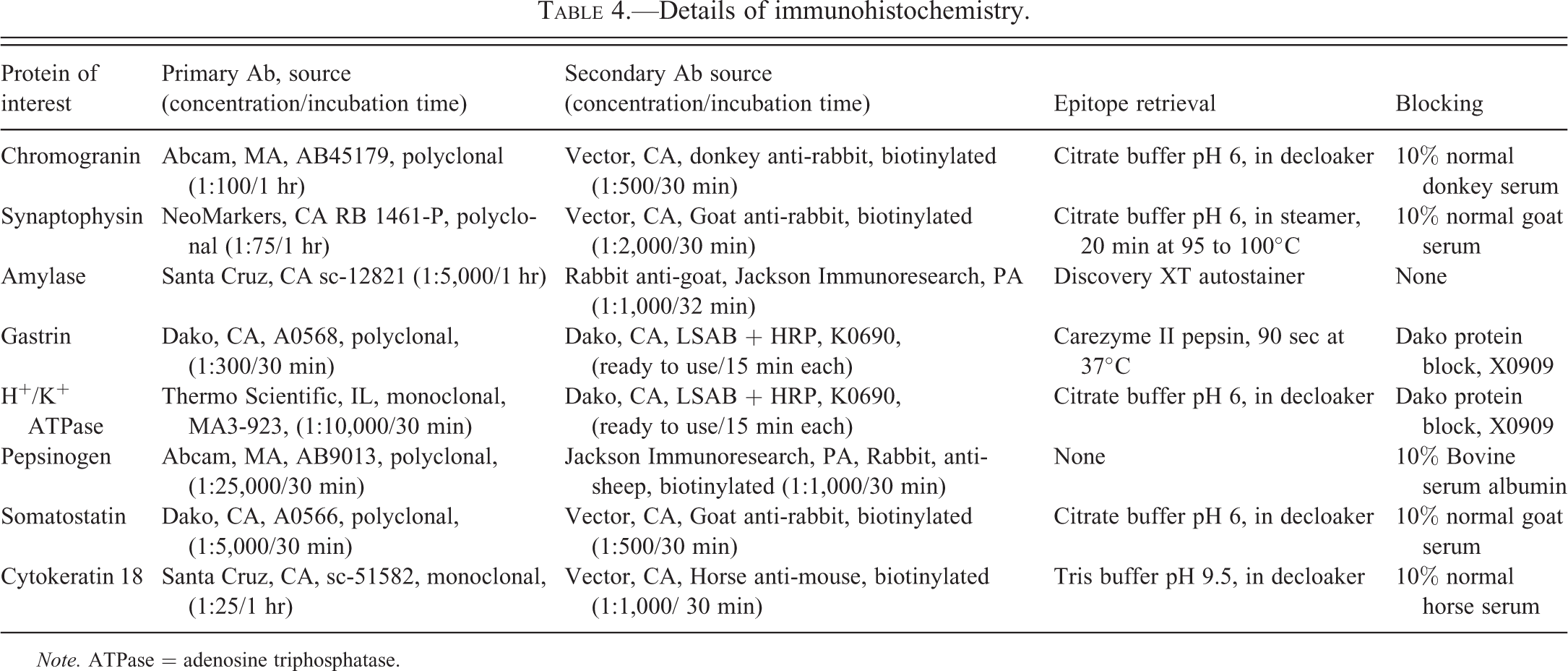
Note. ATPase = adenosine triphosphatase.
Results
Histopathological Characterization
NE cell hyperplasias and primary tumors were confined to fundic stomach in most of the cases. Hyperplasia of NE cells in the pyloric region (G cells) was not observed.
NE cell hyperplasia
H&E-stained sections of 17 males and 28 females from different groups, for which there was a diagnosis of NE cell hyperplasia, were further evaluated to characterize this lesion. The hyperplastic lesions were focal (Figure 1A and B) or multifocal, small clusters of round to polygonal cells confined to the fundic mucosa. The hyperplasia was occasionally focally extensive with the cells occurring in solid sheets or forming acini or both. The cells had indistinct borders, foamy to vacuolated cytoplasm, and a basally placed round nucleus containing finely stippled to vesicular chromatin; mitotic figures were not evident.
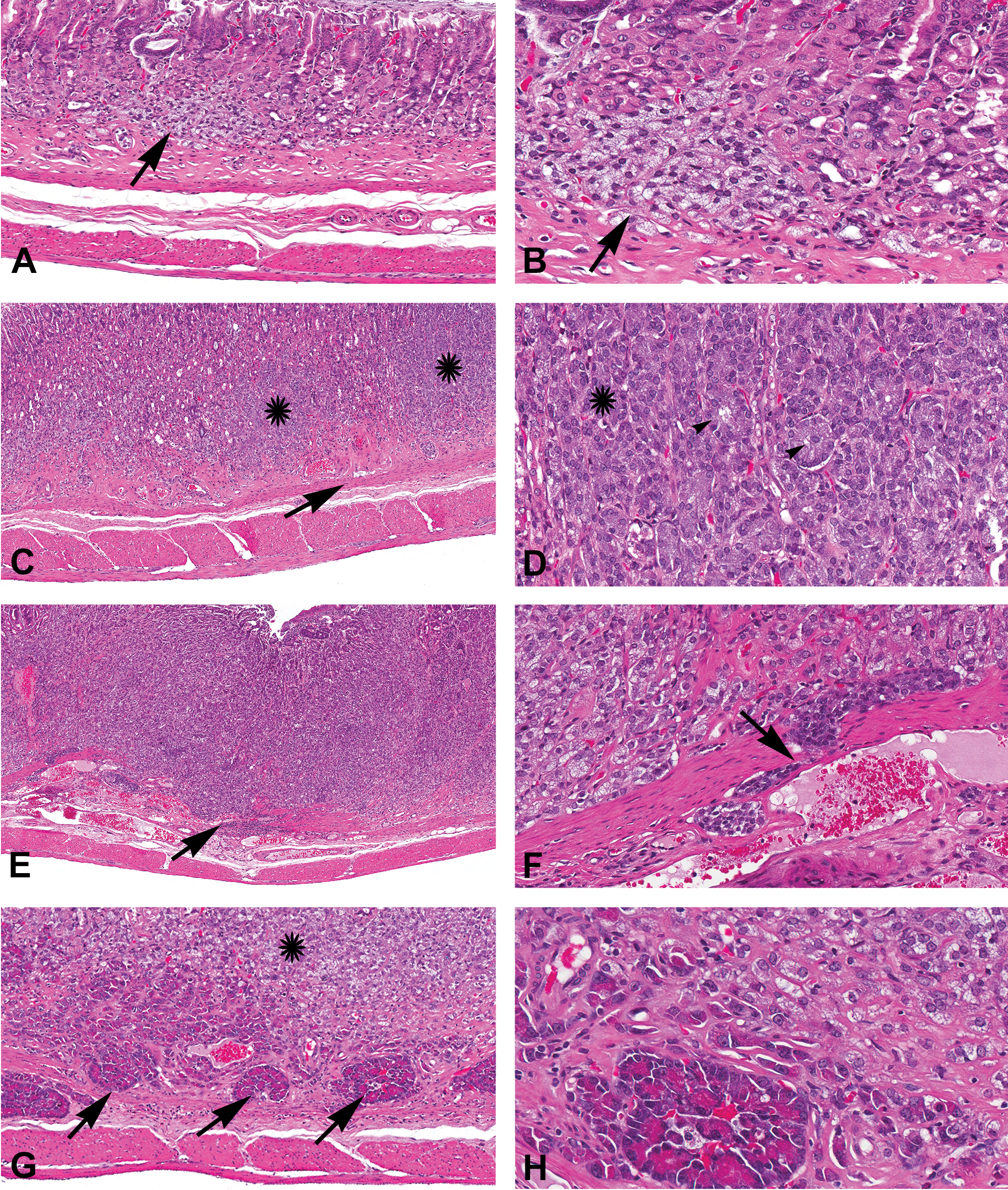
Histopathology of neuroendocrine cell hyperplasia and neoplasia in the fundic region, hematoxylin and eosin (H&E) staining. (A) Histomicrograph showing neuroendocrine (NE) cell hyperplasia (arrow), which is confined to the mucosa. There is not much effacement of the mucosal architecture. (B) Higher magnification of “A” with NE cell hyperplasia (arrow). (C) Benign NE tumor. The neoplastic cells (asterisks) efface the mucosa but do not extend beyond the muscularis mucosa (arrow). (D) Higher magnification of “C” showing the neoplastic cells in clusters (asterisk) and acini (arrowheads) separated by a delicate fibrovascular stroma. (E) Malignant NE tumor; the neoplasm is invasive and neoplastic cells efface the normal architecture and infiltrate through the muscularis mucosa (arrow). (F) Higher magnification of “E” showing the neoplastic cells invading the muscularis mucosa (arrow). (G) Malignant NE tumor with areas of pancreatic acinar-like differentiation (arrows). Also observe the neoplastic cells with typical NE cell morphology above the areas of pancreatic acinar-like differentiation (asterisk). (H) Higher magnification of “G” showing the detailed morphology of cells in the acini. In these acini, the cells contain basal nuclei and apical bright eosinophilic granular cytoplasm.
Benign NE tumor
Slides from 32 rats (5 males and 27 females) with a diagnosis of benign NE tumor (NTP 2000) were retrieved and evaluated for characterization of benign tumors. They were confined to the fundic mucosa and did not invade or extend beyond the muscularis mucosa, but caused moderate to marked thickening of the fundic mucosa, and occasionally occurred as nodular expansions. The neoplasms were well to poorly defined and composed of cells arranged in nests or acini separated by a delicate fibrovascular stroma (Figure 1C and D). Occasionally, the cells were arranged in a trabecular pattern or formed rosettes. Neoplastic cells had indistinct borders, moderate amounts of foamy pale amphophilic cytoplasm, and a basally located nuclei with finely stippled chromatin and variably prominent nucleolus. Mitotic figures were rare. The neoplastic cells effaced the normal architecture and variably contained entrapped gastric pit cells, parietal cells, and chief cells. In 1 female, there was a focal area in which the neoplastic cells resembled pancreatic exocrine cells. In this focus, the cells formed acini and were characterized by basal nuclei and brightly eosinophilic granular apical cytoplasm. In 1 male and 7 females, NE cell hyperplasia was also present in the adjacent mucosa. One female had multiple benign NE tumors. In 2 cases, there was mucous neck cell hyperplasia in the associated mucosa. Except for 1 case, there was no gastric mucosal ulceration associated with the tumor.
Malignant NE tumor
Slides from 80 rats (6 males and 74 females) with a diagnosis of malignant NE tumor (NTP 2000) were retrieved and evaluated to characterize the malignant tumors. Malignant NE tumors were generally large, diffuse, poorly or well demarcated, nodular, masses that effaced the normal mucosal architecture and frequently caused marked mucosal thickening (Figure 1E and F). Invasion through the muscularis mucosa and infiltration of the submucosa and tunica muscularis was common. One tumor extended into the nonglandular and pyloric stomach. The neoplastic cells were predominantly arranged as solid sheets admixed with neoplastic cells forming acini. The neoplastic cells had indistinct borders, scant amount of eosinophilic to foamy cytoplasm, and a round nucleus with finely stippled chromatin and prominent nucleolus. Mitotic figures were rare in most of the neoplasms. However, in the large and poorly differentiated neoplasms, mitotic figures were more frequent. In 22 (30%) of the 74 females, the malignant tumors had small or large areas where the neoplastic cells resembled pancreatic acinar cells (Figure 1G and H); pancreatic acinar cell differentiation occupied most of the tumor in 2 females, approximately 50% to 70% of the tumor in 5 females and a small area in the remaining cases. Of these 22 females with pancreatic acinar differentiation, 18 (82%) were from the 300-mg/kg (high-dose, stop-exposure) group. Tumor emboli were present in the stomachs of 4 (66%) of the 6 males and 35(41%) of the 74 females; tumor emboli were predominantly present in the lymphatic vessels and occasionally in the veins. Metastatic lesions were not present in any of the 6 males; however, 16 (21.6%) of the 74 female rats with malignant NE tumors had metastatic lesions. Sites of metastasis included liver (13/16; Figure 2A), lung (4/16), lymph node (3/16), duodenum (1/16), and pancreas (1/16). Of the 16 tumors with metastases, 11 metastasized to only 1 tissue (8 to liver, 2 to lung, and 1 to pancreatic lymph node). The histological features of the neoplastic cells in the metastatic locations were similar to the primary tumors in the stomach, except for the lack of pancreatic acinus-like differentiation. Of the 22 malignant NE tumors with areas of pancreatic acinus-like differentiation in the primary tumor, 4 had metastasis; however, with the exception of the duodenal metastasis, pancreatic acinus-like differentiation was not evident in the metastatic lesions.
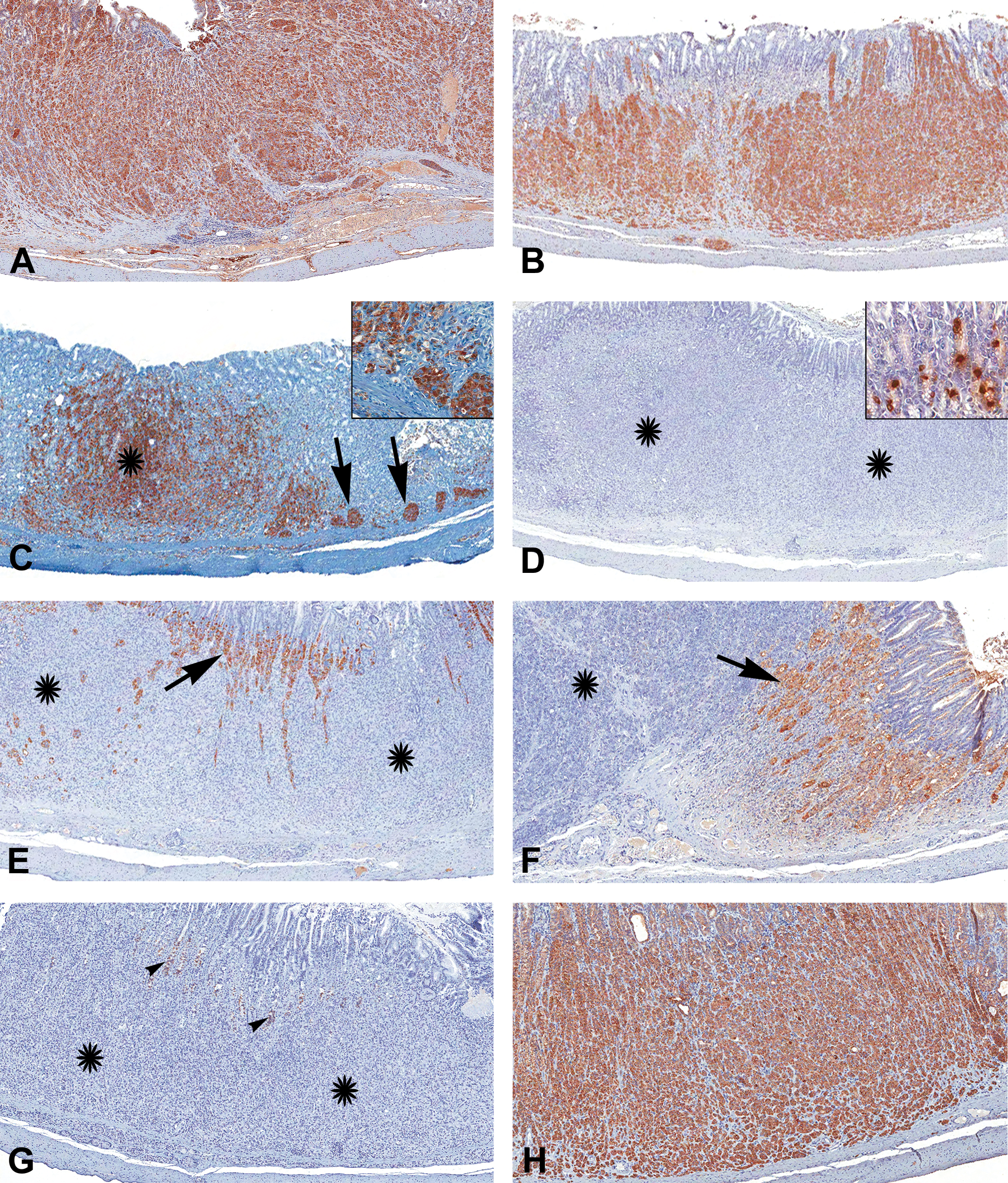
Immunohistochemical characterization of malignant neuroendocrine (NE) tumor in the fundic stomach. 3,3′-diaminobenzidine (DAB) chromogen with hematoxylin counterstain. Neoplastic cells showing strong immunoreactivity to (A) chromogranin A and (B) synaptophysin. (C) Malignant tumor with areas of pancreatic acinar differentiation; there is strong amylase immunoreactivity in the neoplastic cells with pancreatic acinar differentiation (arrows and inset), and in the cells with typical NE morphology (asterisk). (D) The tumor (asterisks) is negative for gastrin; normal NE cells (G cells) in the pyloric region in the same section are positive (inset). (E) The neoplastic cells are negative for H+/K+ ATPase (asterisks); the adjacent parietal cells are positive (arrow). (F) The neoplastic cells are negative for pepsinogen (asterisk); the adjacent chief cells are positive (arrow). (G) There was no immunoreactivity for somatostatin (asterisks); some nonspecific staining is present in a few of the entrapped normal cells (arrowheads). (H) Strong immunoreactivity for cytokeratin 18 is present in the neoplastic cells.
Atrophy of the fundic mucosa
Concurrent mucosal atrophy occurred in the fundic stomach of treated rats in all dose groups. The incidences for the 2-year bioassay are presented in Table 1. Mucosal atrophy frequently involved the entire fundic mucosa and was characterized by decreased height of the fundic mucosa due to the loss of parietal cells and chief cells, and condensation of the lamina propria (Figure S1). Surface mucous neck cells and the mucosa of the pyloric region were not affected.
Immunohistochemistry
Immunohistochemistry was performed on the tissue sections that were recuts from selected archived paraffin blocks of the glandular stomach and tissues that had metastases. In a few cases, the recut sections did not contain the entire tumor that was present in the original H&E-stained tissue sections. However, in those recuts, at least some part of the tumor was present for IHC staining and evaluation. For all the IHC stainings, appropriate negative controls were used as detailed under Materials and Methods section Nonspecific staining was not present in the negative control slides.
Of the 16 cases of metastasis, 4 rats had metastasis to the lung; recuts from 3 of these cases did not contain neoplastic cells and hence the IHC features of the metastatic neoplasms were determined in only 1 lung section. In 13 of the 16 cases of metastasis to the liver, 1 paraffin block was not available and hence the IHC features were analyzed for 12 cases. Of the 14 selected cases of malignant NE tumors that contained areas of pancreatic acinar-like differentiation, 5 cases had no such areas in the recut sections and hence the IHC features of these areas were evaluated in 9 of the selected cases.
Chromogranin A
In sections of the pancreas used as positive control tissue, the antibody specifically stained the islets. All sections with NE cell hyperplasias (Figure S2A), benign NE tumors (Figure S3A), and malignant NE tumors (Figure 3A) showed positive immunoreactivity for chromogranin A. The staining was cytoplasmic and finely granular and in most cases, the intensity of staining was strong. One case of hyperplasia and 2 malignant tumors had weak immunoreactivity. In 3 of the 9 malignant tumors, the areas of pancreatic differentiation were negative, while the reminder of the tumor was positive for chromagranin A. In 6 of the 9 malignant tumors, a few cells in areas of pancreatic differentiation were weakly positive while the other areas of the neoplasm were strongly positive for chromagranin A. Of the 12 hepatic metastases, 11 were strongly positive for chromagranin A (Figure 2B). Metastases in the pancreatic (1/1) and renal (1/1) lymph nodes, and pancreas (1/1) were positive, whereas metastases in a mediastinal lymph node (0/1) and lung (0/1) were negative. In duodenum (1/1), approximately 10% of the neoplastic cells were weakly positive.
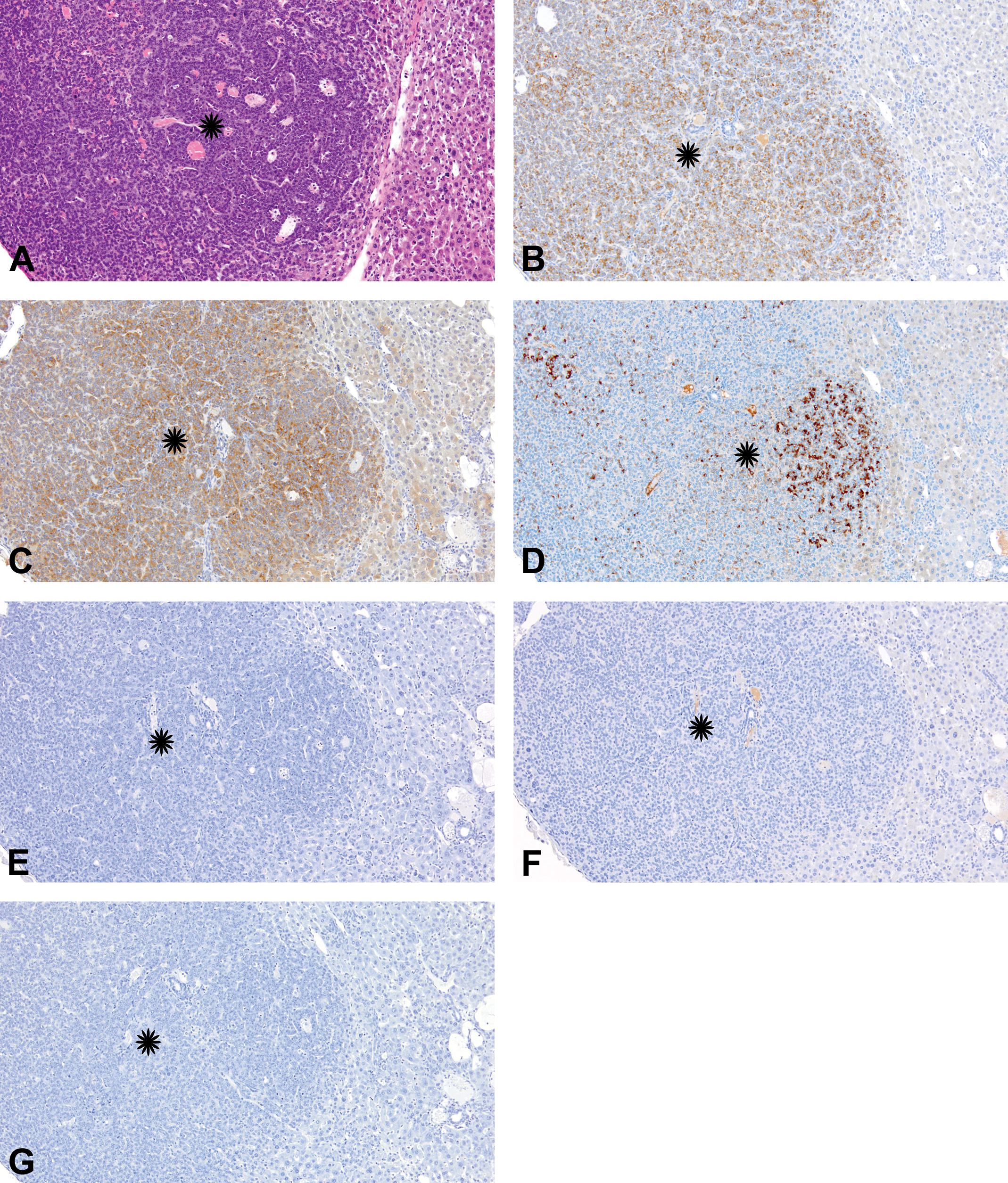
Metastatic gastric neuroendocrine tumor in liver. (A) Hematoxylin and eosin (H&E)-stained section of the metastatic neuroendocrine (NE) tumor (asterisk). The neoplastic cells are effacing the normal architecture of the liver and are forming solid sheets separated by vascular spaces. (B–G) Immunohistochemical staining of the metastatic gastric neuroendocrine tumor in liver. Immunohistochemically, the neoplastic cells were positive for (B) chromogranin A, (C) synaptophysin, and (D) amylase, and negative for (E) gastrin, (F) H+/K+ adenosine triphosphatase (ATPase), and (G) pepsinogen. 3,3′-diaminobenzidine (DAB) chromogen with hematoxylin counterstain.
Synaptophysin
In sections of the pancreas used as positive control tissue, the antibody specifically stained the islets. All NE cell hyperplasias (Figure S2B), benign NE tumors (Figure S3B), and malignant NE tumors (Figure 3B) had positive immunoreactivity for synaptophysin. The staining was cytoplasmic and finely granular. In the liver, 10 of the 12 cases with metastases had weak to moderate immunoreactivity (Figure 2C); 1 of the 12 cases had no neoplastic cells and the remaining case was negative. Metastases in the pancreatic (1/1) and renal (1/1) lymph nodes, and pancreas (1/1) were weakly to moderately positive, while metastases in a mediastinal lymph node (0/1), duodenum (0/1), and lung (0/1) were negative.
Amylase
In the sections of the pancreas used as a positive control tissue, the antibody stained the exocrine pancreas while the islets were negative. In all 9 cases (9/9) of the selected malignant NE tumors that contained areas of pancreatic differentiation, the cells in areas of pancreatic differentiation stained positively with amylase (Figure 3C). In the same cases, neoplastic cells with NE morphology also stained positively with amylase (Figure 3C). Therefore, 5 additional cases each of NE cell hyperplasia, and benign and malignant NE tumors without areas of pancreatic differentiation, were also stained with amylase; all these stained positive for amylase (Figures S1C and S2C), with the exception of 1 NE hyperplasia, which was negative. The intensity of staining varied from weak to strong. Also in some neoplasms approximately 20% to 50% of the neoplastic cells stained positively. In the metastatic neoplasms to the liver, cells were positive in 9 of the 12 (Figure 2D). Two were negative and in 1 the recut section did not contain neoplastic cells. The staining was cytoplasmic. In some of the liver metastases, entire tumor was positive and in the others positivity varied from occasional cells to approximately 10% to 50% of neoplastic cells.
Gastrin, H+/K+ ATPase, and pepsinogen
NE cell hyperplasias, tumors, or metastatic tumors were negative for gastrin, H+/K+ ATPase, or pepsinogen (Figures 2E, F, G; S1, S2 D, E, F; 3D, E, F). Internal controls in the tissues stained positively with these markers; for gastrin, H+/K+ ATPase, and pepsinogen, there was positive staining in the NE cells in the pyloric stomach, normal parietal cells, and normal chief cells in the stomach, respectively.
Somatostatin and CK18
Due to limited number of available tissue sections, staining was performed only on the tumors with pancreatic differentiation (Table 3). For somatostatin, section of pancreas was used as a positive control, and specific staining was present in the islets. In NE tumor samples, neoplastic cells including those in the areas of pancreatic acinar differentiation were negative in all cases (Figure 3G). For CK, the glandular stomach was used as a positive control and specific staining was present in the mucosal epithelial cells. In all tumors, neoplastic cells including those in the areas of pancreatic acinar differentiation were positive for CK18 (Figure 3H).
Discussion
In NTP 2-year rodent bioassays, the most common spontaneous and chemically induced gastric neoplasms in rats and mice are squamous cell papilloma and squamous cell carcinoma of the nonglandular stomach (Chandra, Nolan, and Malarkey 2010). Spontaneous tumors of glandular stomach (adenomas, adenocarcinomas, benign, and malignant NE cell tumors) are extremely rare in laboratory rodents in NTP 2-year bioassays (Chandra, Nolan, and Malarkey 2010); however, spontaneous gastric NE tumors are described in cotton rats, Sprague-Dawley rats, striped field mouse, and Mastomys natalenesis mice (Majka and Sher 1989; Snell and Stewart 1969; Oh, Chae, and Jang 1997; Cui et al. 2000). Among chemically induced glandular stomach neoplasms in rodent bioassays, NE tumors are the most common (Chandra, Nolan, and Malarkey 2010). Similar to various chemicals and drugs such as butachlor (Hard et al. 1995), histamine receptor antagonists (Hirth et al. 1988), hypolipidemic agent ciprofibrate (Spencer et al. 1989), and proton pump inhibitor (Havu 1986), methyl eugenol also induces NE tumors in the glandular stomach of laboratory rats (NTP 2000). Our current experiment provides a detailed histopathological and IHC evaluation of methyl eugenol–induced NE cell hyperplasia and tumors in F344/N rats.
In humans, the gastrointestinal NE tumors are classified based on histological or clinicopathological features. Based on histology, human gastrointestinal NE tumors are classified as NE tumor, NE carcinoma, and mixed adenoneuroendocrine carcinoma (MANEC; Rindi, Arnold, and Bosman 2010). NE tumors were previously referred to as carcinoids and are defined as well-differentiated tumors composed of cells that resemble NE cells of the gastrointestinal tract. NE carcinoma is defined as a poorly differentiated, high-grade, malignant neoplasm composed of small, intermediate, or large cells. MANEC should contain both proliferative NE cells and gastric adenocarcinoma components, with each component representing at least 30% of the tumor (Rindi, Arnold, and Bosman 2010; Ishida et al. 2013). In this study, it was decided to use the terms benign NE tumor and malignant NE tumor based on the location, invasiveness, presence of tumor emboli, and metastasis of the neoplasm. Although there was pancreatic acinar differentiation in some of the neoplasms, none could be classified as MANECs, as they lacked gastric adenocarcinoma component.
Based on the clinicopathological characteristics, NE tumors in humans can be classified as types 1, 2, and 3. Type 1 is the most common form, is more common in females than in males, often small in size, associated with atrophic gastritis and elevated serum gastrin levels, and has a metastatic rate of less than 10%. Type 2 also has elevated serum gastrin levels, is associated with multiple NE neoplasia-1 and Zollinger–Ellison syndrome, and has a metastatic rate of 10% to 30%. Type 3 is sporadic, is not associated with any nonneoplastic gastric pathology or elevated serum gastrin levels, and has a metastatic rate of 50% to 100% (Scherubl et al. 2010).
Similar to humans, in this study, the incidence of NE tumors was much higher in females than in males. Predominance of NE tumors in females has also been described for Cotton rats (Cui et al. 2000). While no metastasis was observed in males, metastases occurred in 21% of the females. Many of the rats in this study had gastric mucosal atrophy. In a previous NTP short-term study, gastric atrophy along with elevated serum gastrin levels in response to methyl eugenol administration was demonstrated in rats (Abdo et al. 2001). Considering the above-mentioned features, the gastric NE tumors of rats match with the human clinicopathological classification type 1 with the exception of tumor size and rate of metastases.
Mucosal atrophy characterized by the loss of parietal and chief cells, similar to a previous NTP short-term (14-week) study with methyl eugenol in rats (Abdo et al. 2001), also occurred in this study. This mucosal atrophy is in contrast to the nonatrophic gastric mucosa seen after long-term administration of proton pump inhibitors such as omeprazole (Hagiwara, Mukaisho, Ling, Sakano, et al. 2007). Irrespective of the histologic appearance of gastric mucosa, the most important change that determines the development of NE cell hyperplasia and tumors is the development of hypocholrhydria and hypergastrinemia. The exact mechanism of mucosal atrophy in this study was not addressed nor has its mechanism been completely defined in the literature. Autoantibodies against- and CD4+ cytotoxic T-cells targeting H+/K+ ATPase of the parietal cells play a major role in pathogenesis of human and experimental autoimmune gastritis resulting in mucosal atrophy (D’Elios et al. 2001; Faller and Kirchner 2005). Increased apoptosis could play a significant role in the loss of parietal cells (Judd et al. 1999; Faller and Kirchner 2005). However, there is no evidence for the direct targeting of chief cells in the stomach. Thus, it is very likely that loss of chief cells is linked to loss of parietal cells. Parietal cells probably indirectly control the differentiation of gastric chief cells (Li, Karam, and Gordon 1996). In this study, there are 2 possibilities for the loss of gastric parietal and chief cells. Methyl eugenol may (1) affect H+/K+ ATPase, thus mimicking the pathogenesis of autoimmune gastritis or (2) directly cause apoptosis/necrosis of parietal cell or both parietal and chief cells. The first mechanism may not be true in this study as there was abundant H+/K+ ATPase immunoreactivity in the cells surrounding the NE cell tumors. The second mechanism may be more plausible in this study, as the previous NTP 14-week toxicity study in F344 rats showed the presence of degenerate and necrotic parietal cells in the mucosa of the glandular stomach (Abdo et al. 2001; NTP 2000). In a 2-year bioassay with butachlor, which also induces NE tumors in fundic stomach, necrosis was not observed (Hard et al. 1995). In cotton rats with spontaneous ECL tumors, parietal cells were unaffected (Cui et al. 2000). In some instances, there was an increase in the number of parietal cells (Hirth et al. 1988). These variable findings highlight the complexity in the fundic mucosal response to different chemicals. Irrespective of these variations, 1 common finding in these studies is hypergastrinemia.
Increased gastrin secretion can result from loss of parietal cells and resulting elevation of gastric pH. In a previous NTP 14-week toxicity study, gastric pH and serum gastrin levels were elevated (Abdo et al. 2001). In the stomach, gastrin is produced by G cells that are present in the pyloric region (Helander 1981). Gastrin has a trophic effect on gastric mucosa, particularly on the number of ECL cells in the fundic stomach (Hakanson et al. 1986). The trophic effect of gastrin is mediated through its interaction with ECL cells and subsequent release of products such as histamine and Reg protein (Fossmark, Qvigstad, and Waldum 2008). Persistent hypergastrinemia results in ECL cell hyperplasia, which can progress to neoplasia (Fossmark, Qvigstad, and Waldum 2008). Based on the previous NTP study demonstrating hypergastrinemia after administration of methyl eugenol for 14 weeks, it is reasonable to assume that the same mechanism may have also occurred in the 2-year bioassay. Increase in the gastrin level can occur by 2 mechanisms, such as (1) an increase in the number of G cells and (2) increased activation of G cells. In our H&E slides, the number of NE cells was not increased in the pyloric region where G cells are known to be present (Helander 1981). Neither the IHC evaluation of a few representative samples nor the evaluation of H&E-stained slides showed any increase in the number of NE cells in the pyloric region. We also ruled-out production of gastrin by tumor cells as neither the hyperplasias nor tumors were positive for gastrin. Thus, hypergastrinemia in our experiment is probably a result of hyperfunction of G cells. In 1 study in rats, hypergastrinemia was associated with an increase in the G cell numbers (Creutzfeldt et al. 1986), while there was no such increase in the G cells in humans (Creutzfeldt et al. 1989). However, many of the studies that focused on hypergastrinemia mediated ECL cell hyperplasia and neoplasia have not evaluated or reported the numbers of G cells in the pyloric region (Hirth et al. 1988; Thake et al. 1995; Havu 1986; Martinsen et al. 2003; Larsson et al. 1988). Irrespective of G cell hyperfunction or hyperplasia, the effect of hypergastrinemia on gastric fundic mucosa is well documented. While gastrin, irrespective of the cause of hypergastrinemia, has a trophic effect on ECL cells, parietal cells and gastric stem cells, the trophic effect is more pronounced on ECL cells (Ryberg et al. 1990; Koh and Chen 2000). The reason for this variability in the trophic effect on non-ECL cells is not completely understood. Somatostatin from the D cells in gastric mucosa inhibits the secretion of gastrin and histamine (Zaki et al. 1996). In this study, there was neither an increase in the somatostatin positive NE cells in the stained sections nor were the NE hyperplasias or tumors positive for somatostatin.
NE neoplasms can be diagnosed by their typical histomorphology and by using IHC markers. Chromogranin A (a major constituent of NE cell granules) is commonly used to identify endocrine cells (O’Connor, Frigon, and Sokoloff 1982; DeLellis, Shin, and Treaba 2010). However, the expression of chromogranin A in human NE tumors varies depending upon the location of the tumor (DeLellis, Shin, and Treaba 2010). Synaptophysin is a membrane glycoprotein present in the synaptic vesicles of neurons and in the synaptic-like microvesicles in the cytoplasm of NE cells (DeLellis, Shin, and Treaba 2010; Wiedenmann et al. 1986, 1998). Neuron-specific enolase (NSE) can also be used as a marker of NE cells. However, NSE lacks specificity (DeLellis, Shin, and Treaba 2010) and is useful only in combination with other markers such as chromogranin A and/or synaptophysin. We have not used this marker in this study. However, the gastric NE tumors evaluated in this study have been shown to stain positively with NSE in the previous NTP (2000) study. CKs are intermediate filaments of epithelial and NE cells. Gastric NE cells contain several CKs including CK18 (DeLellis 2001).
In addition to the use of NE cell markers, antibodies that identify other gastric mucosal cell types such as parietal and chief cells help to exclude other possible tumor types in the fundic stomach. Gastric parietal cells contain H+/K+ ATPase, which is a proton pump that plays an important role in acid secretion, and antibodies to this protein can be used to identify parietal cells (Smolka, Helander, and Sachs 1983; Franic et al. 2001). Gastric chief cells contain abundant pepsinogen and can be immunohistochemically stained using antibodies to pepsinogen (Helander 1981).
In this study, the hyperplastic and neoplastic cells comprising the gastric neoplasms stained strongly and consistently with chromogranin A, synaptophysin, and CK18, and were negative for pepsinogen and H+/K+ ATPase. These results along with histomorphology confirm and support the diagnosis of gastric NE cell hyperplasia and NE tumors. Similar positive staining for chromogranin A and/or CK has been previously described in the NE tumors of rat glandular stomach (Hard et al. 1995; Martinsen et al. 2003; Waldum et al. 1999). In an evaluation of 104 human gastric NE endocrine tumors, chromogranin A and synaptophysin stained 100% (72/72 and 6/6, respectively) of the neoplasms (Thomas et al. 1994). In another evaluation of 51 human gastric malignant NE tumors, 86% and 94% of tumors stained positively with chromogranin A and synaptophysin, respectively (Ishida et al. 2013). Based on our results and those reported in the literature, chromogranin A and synaptophysin are sensitive markers of gastric NE tumors. In addition, the lack of staining for pepsinogen and H+/K+ ATPase rules out chief cell or parietal cell as possible cells of origin for these neoplasms. Based on the location of the lesions and pathogenesis, we wanted to prove that the proliferating NE cells in hyperplasia and neoplasms are ECL cells. Since ECL cells secrete histamine, antibodies to histamine or histidine decarboxylase can be used as a marker to specifically identify them. Unfortunately, no good histamine or histidine decarboxylase antibodies were available for immunohistochemistry on formalin-fixed paraffin-embedded sections. Considering the fact that ECL cells are the predominant NE cell type in the fundic region of rat stomach, and there was atrophy of the gastric mucosa and evidence of hypergastrinemia, we speculate that the methyl eugenol–induced NE cell hyperplasias and NE tumors are very likely to be of ECL cell origin.
Metastases of the neoplasms in this study were mainly to the liver, and the same is true for human gastrointestinal NE tumors (Strosberg et al. 2008). The rate of metastasis was 21.6% in females. In the metastatic lesions, as in the primary tumors, the neoplastic cells consistently stained with chromogranin A and synaptophysin, indicating that these markers can be used for IHC detection of metastatic lesions. Similarly, the expression of markers in metastatic gastric NE carcinomas in humans mimics those of the primary tumors (Uchiyama et al. 2012).
Of the 74 female rats with malignant NE tumors, 22 had areas of pancreatic differentiation. Pancreatic acinar metaplasia in the fundic mucosa has been described in humans with autoimmune gastritis, chronic gastritis, and gastric carcinoma (Jhala et al. 2003; Doglioni et al. 1993). Pancreatic metaplasia in the fundic mucosa and hypergastrinemia is also described in rats treated with a proton pump inhibitor, omeprazole, for 6 months (Hagiwara, Mukaisho, Ling, Sugihara, et al. 2007). Also in another study, hypereosinophilia of chief cell granules has been described in rat stomach following long-term administration of omeprazole (Ekman et al. 1985). Similar change was observed in a study involving administration of famotidine, a histamine H2 receptor antagonist (Burek, Majka, and Bokelman 1985), and hypereosinophilia was shown to represent the increased electron density of zymogen granules. In our study, the metaplastic change was within the NE tumors, and the NE tumors stained negative for pepsinogen. Therefore, the change observed in our study is more likely to be pancreatic metaplasia rather than hypereosinophilia of the chief cells. Further, in this study, based on the location of pancreatic acinar differentiation within the tumor, areas of transition from NE cells to pancreatic acini, presence of pancreatic acinar cells in a few emboli, infiltrative nature, and their presence beyond the muscularis mucosa, it is very likely that the NE tumor cells have differentiated into pancreatic acinar cells rather than the NE tumor entrapping areas of the mucosa that have differentiated into pancreatic acinar cells. Although we did not explore the mechanism of pancreatic acinar differentiation in this study, certain mechanisms are described in the literature. In the fundic stomach, parietal cells have a major role in regulating differentiation of gastric progenitor cells (Li, Karam, and Gordon 1996; Judd et al. 1999). Sonic hedgehog (Shh) protein plays a critical role in the development of gastric mucosa and is predominantly expressed in parietal cells (Faller and Kirchner 2005; Dimmler et al. 2003). Pancreatic duodenal homeodomain transcription factor (PDX1) is one of the essential transcription factors involved in pancreatic development (Faller and Kirchner 2005). PDX1 is normally not expressed in the fundic region but is expressed during mucosal atrophy (Buettner et al. 2004) while the expression of Shh is lost in gastric mucosal atrophy (Dimmler et al. 2003). Further, blocking Shh has been shown to promote pancreatic development in embryos (Kim and Melton 1998). Downregulation of Shh and upregulation of PDX1 are possible mechanisms of pancreatic metaplasia in these tumors. In a potassium channel mutant rat, PDX1 expression was not detectable in the areas of pancreatic metaplasia in the stomach (Kuwamura et al. 2008). This suggests that there may be mechanisms other than Shh and PDX1 involved in the pancreatic metaplasia.
Positive amylase staining observed in the neoplastic cells that did not resemble pancreatic acinar cells indicate 2 possibilities: (1) aberrant expression of amylase in neoplastic cells and (2) origin of NE tumors from gastric progenitor cell that could differentiate into multiple lineages.
In conclusion, methyl eugenol induced NE cell hyperplasias and benign and malignant NE tumors in the fundic stomach of male and female F344/N rats. These lesions appear to be of ECL cell origin. Immunohistochemically, the lesions consistently expressed chromogranin A and synaptophysin and not gastrin or somatostatin. Therefore, chromogranin A or synaptophysin can be used as a marker in suspected NE cell lesions in the glandular stomach of rats. Metastatic lesions resembled the primary neoplasms in both histology features and IHC staining characteristics.
Footnotes
Acknowledgment
We thank the histology and immunohistochemistry core laboratories, Cellular and Molecular Pathology Branch for their help. We thank Drs. Amy Brix, Gordon Flake, and Arun Pandiri for reviewing the manuscript.
Author Contribution
Kyathanahalli S. Janardhan contributed to conception or design; Kyathanahalli S. Janardhan, Yvette Rebolloso, Geoffrey Hurlburt, David Olson, Otis Lyght, Natasha P. Clayton, Margarita Gruebbel, Catherine Picut, Cynthia Shackelford, and Ronald A. Herbert contributed to data acquisition, analysis, or interpretation; Kyathanahalli S. Janardhan and Ronald A. Herbert drafted the manuscript. All authors critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Figure S1. Hematoxylin and eosin (H&E)-stained sections of glandular stomach from control (A and C) and methyl eugenol–treated (B and D) rats. Compared to the control (A), in the treated rat (B) there is a significant decrease in the mucosal thickness (solid line). (C) Higher magnification of “A” demonstrates the normal architecture of the fundic mucosa showing the region of the surface mucous cells (black bracket), and the region of parietal cells and chief cells (white bracket). (D) Compared to the control (C), in the treated rat the thickness of the parietal and chief cell region (white bracket) is markedly reduced due to loss of both parietal and chief cells. The thickness of surface mucous cell region (black bracket) is unaffected.
Figure S2. Immunohistochemical features of neuroendocrine (NE) cell hyperplasia in fundic stomach. 3,3′-diaminobenzidine (DAB) chromogen with hematoxylin counterstain. The hyperplastic NE cells (arrows) were positive for (A) chromogranin A, (B) synaptophysin, and (C) amylase, and were negative for (D) gastrin, (E) H+/K+ adenosine triphosphatase (ATPase), and (F) pepsinogen. In E and F, normal parietal cells and chief cells surrounding the hyperplastic cells are positive for H+/K+ ATPase and pepsinogen, respectively.
Figure S3. Immunohistochemical features of benign neuroendocrine (NE) tumors in fundic stomach. 3,3′-diaminobenzidine (DAB) chromogen with hematoxylin counterstain. The neoplastic NE cells (arrows) were positive for (A) chromogranin A, (B) synaptophysin, and (C) amylase, and were negative for (D) gastrin, (E) H+/K+ adenosine triphosphatase (ATPase), and (F) pepsinogen. In E and F, normal parietal cells and chief cells surrounding the hyperplastic cells are positive for H+/K+ ATPase and pepsinogen, respectively.
