Abstract
A retrospective analysis of microscopic evaluation data from control (device and/or saline-treated) animals in intrathecal studies in monkeys, dogs, sheep, and rats was conducted. The studies were performed by multiple testing facilities. All slide preparation and microscopic evaluation were conducted in the laboratory of the author. The data were of observations made at the level of the catheter tip, which typically was located in the intrathecal space near the thoracolumbar region of the spinal canal. The most common microscopic changes in control animals were meningeal infiltrates, catheter track (CT) inflammation, spinal cord compression (at the CT), CT fibrosis, spinal cord gliosis (at the CT), and spinal cord nerve fiber degeneration. Although variable between studies (even within species), in general the average severity of these findings was minimal or less in control animals. CT inflammatory mass/pyogranuloma formation, a known complication following the administration of morphine at higher concentrations/doses, was noted in 3 of 25 control dogs and 2 of 77 control monkeys. These data show that inflammatory mass/pyogranuloma formation may occur in control animals, and this occurrence is most common in dogs as compared to monkeys, sheep, and rats.
Keywords
Introduction
There are numerous ways of delivering drugs, cells, electrical currents, and so forth directly to the brain/spinal cord or the cerebrospinal fluid (CSF). Methods include intrathecal (IT, where “thecal” refers to the subarachnoid space) administration via catheterization or direct injection (Yaksh and Malkmus 1999), intracerebroventricular (ICV) catheterization or direct injection, direct injection into the parenchyma, bolus/continuous infusion (with or without pumps/reservoirs) into the parenchyma, implantation (for example with stem cells), the use of vectors (either implanted/injected or those that gain access to the brain/spinal cord following systemic administration), or simple entrance through the blood-brain/spinal cord barriers.
The direct delivery of pharmaceuticals and other therapeutic systems directly to the central nervous system (CNS) offers many advantages, the main being bypassing the blood-brain/spinal cord barriers. Other benefits include limiting/controlling systemic toxicity, achieving high local concentrations of drug near the intended targets (for example, neurons in the dorsal/posterior horn of the spinal cord gray matter), and using multidose/continuous infusion delivery systems that allow for repeated and/or long-term therapy (Thorne and Frey 2001).
There are also disadvantages, the most obvious being the invasiveness of any technique that seeks to place a drug or other therapy within or immediately adjacent to the brain or spinal cord. In particular, preclinical studies in laboratory species offer challenges to direct CNS delivery. As compared to the common laboratory animal species, humans have relatively large brains and spinal cords, with equally large spaces containing cerebrospinal fluid (CSF) where delivery systems such as ICV or IT catheters can be readily placed. Accessing the intrathecal space or lateral ventricles within a rat, dog, monkey, small ruminant, or other animal brain is a more technically challenging task. Inadequate postoperative care and protection of the device/surgical site in animals may also lead to failure of the system.
From a pathology perspective, the great challenges of any study utilizing direct delivery is separating out the morphologic changes due strictly to the delivery system and or the vehicle from those lesions that have a direct association to the test article. In some studies, reactions directly related to a particular test article may be exacerbated by a reaction to the presence of a catheter or other device. Direct delivery studies involve all of the complexities of a device study and a drug safety study, and possible systemic effects cannot be ruled out without careful examination of all the typical tissues that would be assessed in any drug/device safety study.
Because the evaluation of direct delivery studies is an immense topic, this article limits the information and discussion to intrathecal delivery, which is probably the most common form of direct central nervous system delivery encountered in preclinical laboratories. The data in this article come from the author’s evaluation of intrathecal studies over the past three years where a device and/or saline control group was examined. Except where noted, the data presented were from the spinal canal region at the level of the catheter tip since typically this is the level where most catheter track/spinal cord changes occur. (This is true of control and test article–treated animals.) The objectives of this article are to provide information on what changes might be expected to occur at the catheter tip in control animals in intrathecal studies, offer some insight on the overall incidence and average severity of those lesions, and show the variability that may occur between studies.
Materials and Methods
The studies included in this retrospective examination were of at least two weeks' duration, that is, intrathecal catheters were in place during a dosing period of at least two weeks. The studies were conducted at multiple, GLP-compliant testing facilities experienced in the conduct of drug safety studies. All studies were approved by the IACUC of the laboratories.
Many species and animal models have been utilized for spinal cord delivery (Yaksh et al. 1999). For the dog/monkey/sheep studies reported here, catheter implantation was via a laminectomy/hemilaminectomy procedure in the caudal lumbar region. Following implantation, the catheter tip was advanced to the thoracolumbar region of the spinal canal. Multiple surgeons experienced at placing IT catheters performed the procedures. The catheter was typically attached to an implanted pump device from multiple manufacturers. Details regarding specific catheter types must be determined empirically, although options for experimental design may be gleaned from literature reports.
For the rodent studies, the catheter was inserted into the cisterna magna (atlanto-occipital space) and advanced caudally to approximately the thoracolumbar region of the spinal canal.
At necropsy, all animals were fixed via whole body intravascular perfusion using a saline flush (either containing heparin or into pre-heparinized animals) followed by a fixative solution that was either neutral buffered 10% formalin or a 4% formaldehyde/1% glutaraldehyde mixture.
All tissue specimens for evaluation were sent to the author’s laboratory for processing. At the author’s laboratory, sectioning varied slightly according to the specifics of the study protocol. In general, the spinal cord with attached dura was (typically) trimmed to produce sections at the mid to caudal cervical, mid thoracic, level of the catheter tip (thoracolumbar region), lumbar region/level of the catheter insertion (for the nonrodents), and cauda equina. A transverse and oblique section was produced at the cervical, thoracic, and lumbar levels. At the cauda equina, only a transverse section was produced. At the level of the catheter tip, three transverse sections were typically produced: a section just rostral to the tip, a section at the tip, and a section 1 cm caudal to the tip (distances were less in the rat). Additionally, an oblique section was prepared from the tissue between the transverse sections taken at the tip and caudal to the tip. Figure 1A shows an example of the sections taken at the level of the catheter tip in a control monkey.
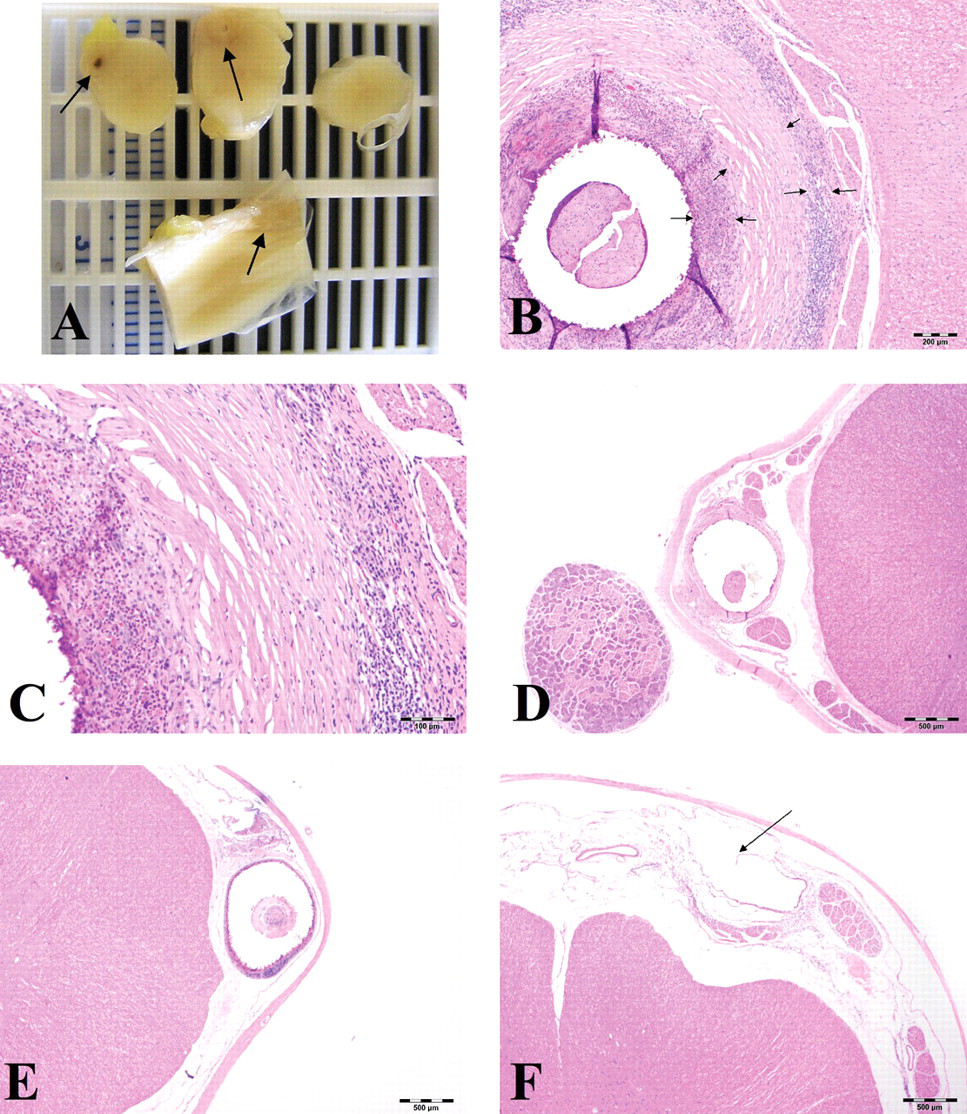
Spinal cord, young cynomolgus monkeys. Figures A, B, and C are from the same animal. Figures D, E, and F are from separate animals. (
For most studies, the spinal cord sections were stained with hematoxylin and eosin (H&E) and anti-glial fibrillary acidic protein (GFAP). The latter is an immunohistochemical stain for an intermediate filament protein mostly unique to astrocytes. When available, the GFAP-stained sections were used to assess the degree of gliosis/astrocytosis at the catheter track. For those specimens where a stain for GFAP was not conducted, an assessment for gliosis was performed using the H&E stain. Typically, astrocytes were identified as cells with round, open-faced nuclei, often with visible eosinophilic cytoplasm (activated astrocytes) or as GFAP-positive cells.
The data in this article are limited to reactions at the catheter tip. All morphologic observations were made by the same pathologist. Various morphologic changes were graded at the level of the catheter tip for all animals. Table 1 describes the most commonly encountered morphologic changes, a description of those changes, and the scheme used to grade those changes. A six-level grading scheme was used in all studies. The general grading scheme used by the author is: 1 = slight, 2 = minimal, 3 = mild, 4 = moderate, and 5 = severe. A grade of 0 indicates the change was not present.
Grading schemes a used for morphologic changes associated with intrathecal catheters.

a Grades: 1 = slight, 2 = minimal, 3 = mild, 4 = moderate, and 5 = severe.
Results
Tables 2A through D provide incidence and average severity data for various morphologic changes associated with intrathecal delivery in monkeys, dogs, sheep, and rats. The data are limited to control animals. The control animals were either device controls (with nothing administered through the catheter) or saline controls (saline only administered through the catheters). Different necropsy times for an individual study are listed with separate Study IDs in Tables 2A through D.
Summary of microscopic evaluation data for controls: Intrathecal monkey studies.
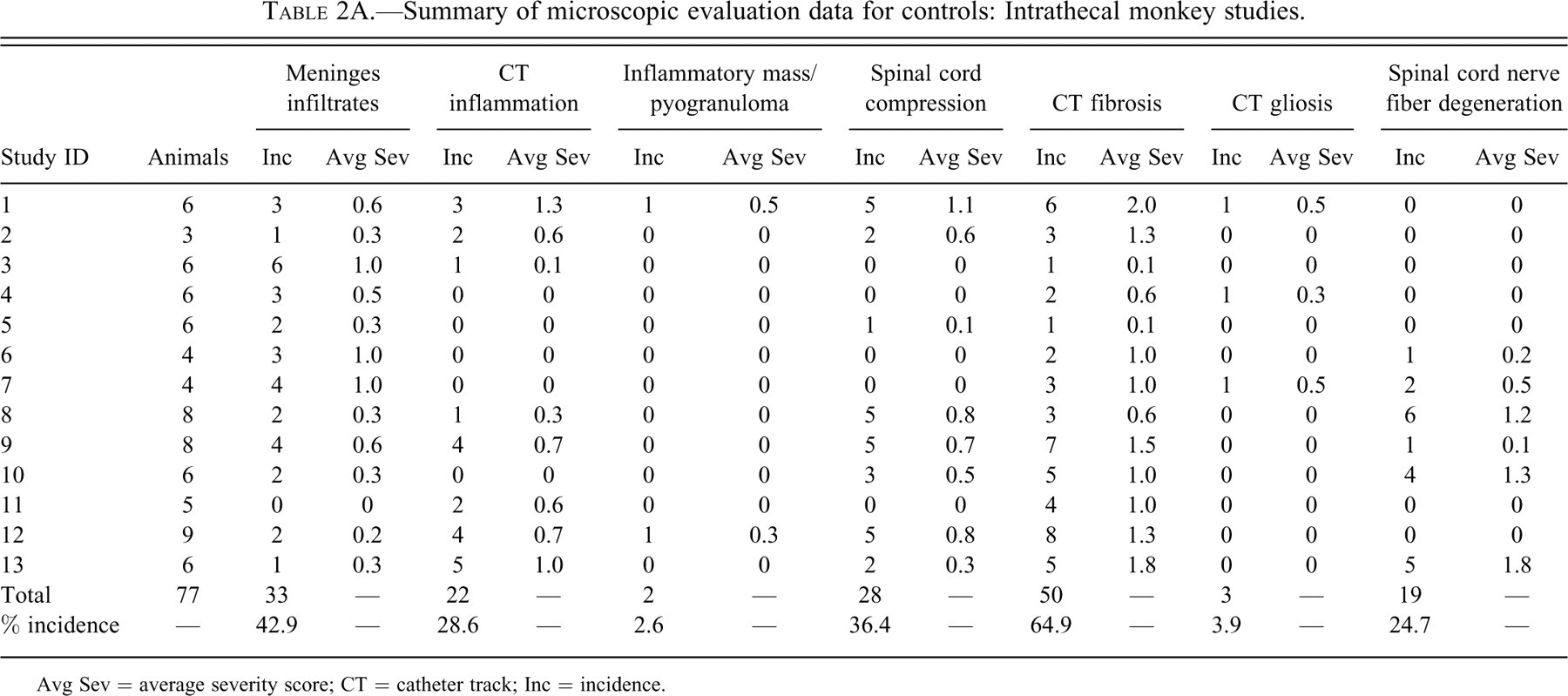
Avg Sev = average severity score; CT = catheter track; Inc = incidence.
Summary of microscopic evaluation data for controls: Intrathecal dog studies.
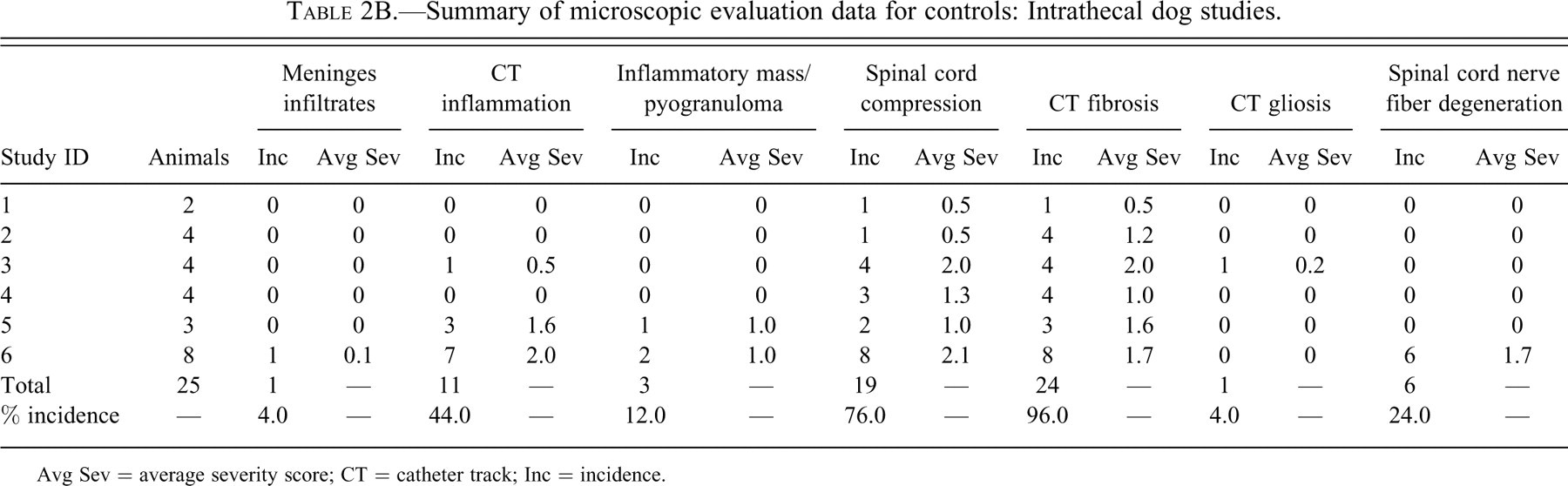
Avg Sev = average severity score; CT = catheter track; Inc = incidence.
Summary of microscopic evaluation data for controls: Intrathecal sheep studies.
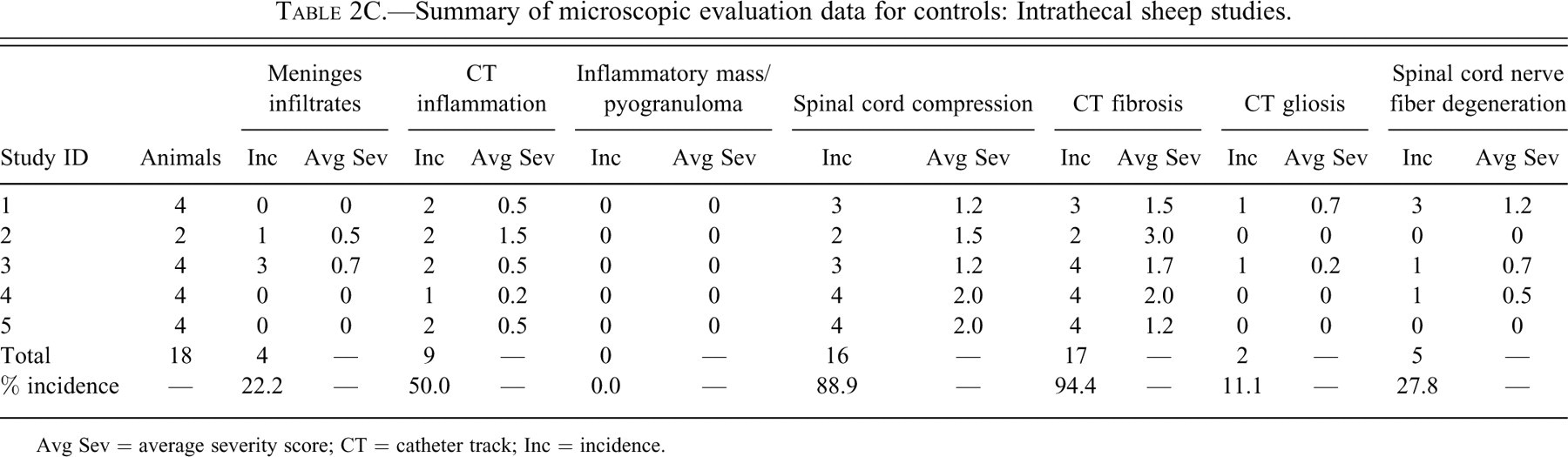
Avg Sev = average severity score; CT = catheter track; Inc = incidence.
Summary of microscopic evaluation data for controls: Intrathecal rat studies.
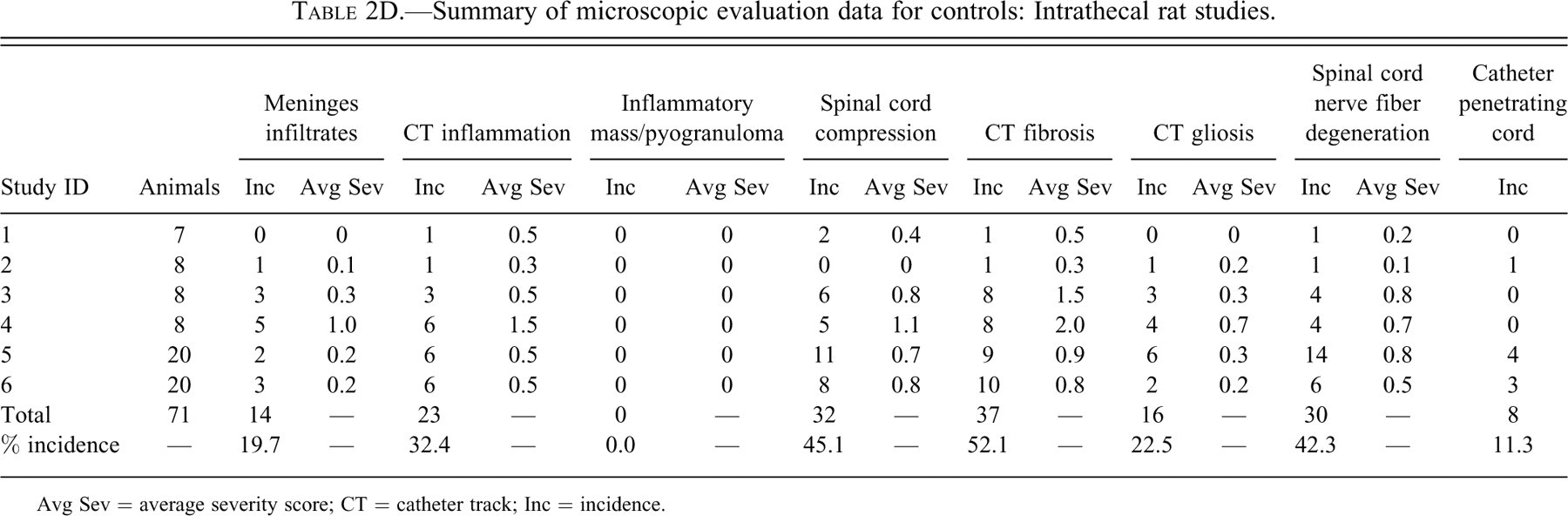
Avg Sev = average severity score; CT = catheter track; Inc = incidence.
Because it is the purpose of this initial retrospective examination to present only the changes that might be expected to occur in control animals in an intrathecal study, the data are presented categorized by species only. Duration of catheter implantation, the catheter material, the specific pump, and other parameters were not considered for data presentation/analysis.
Meningeal Infiltrates
The incidence of meningeal infiltrates was 42.9%, 4.0%, 22.2%, and 19.7% for monkeys, dogs, sheep, and rats, respectively. Regardless of the species, the overall average severity of meningeal infiltrates was very slight (usually less than Grade 1). Infiltrates at this severity would not be considered to be an adverse effect but rather a slight reaction to the presence of the catheter.
Catheter Track, Inflammation
An observable inflammatory reaction at the catheter track was present in 28.6%, 44.0%, 50.0%, and 32.4% monkeys, dogs, sheep, and rats, respectively. This incidence in dogs was heavily influenced by one of the studies, where inflammation was noted in 7/8 control dogs examined microscopically. There was more variability in the average severity in the control groups between studies within the same species (especially in the dog studies), but in general, total inflammation was still, on average, less than a Grade 1 (slight) in overall severity.
Catheter Track, Inflammatory Mass/Pyogranuloma
Inflammatory masses have been reported at the catheter tip in intrathecal studies with multiple species, including humans, dogs, and sheep (Deer et al. 2008; Gradert et al. 2003; Murphy et al. 2006; Yaksh et al. 2003). These lesions are known to occur in association with intrathecal morphine administration above a dose level of 12 mg/day (when given at fairly high concentrations) (Gradert et al. 2003; Yaksh et al. 2003). Observations (unpublished data) by the author in numerous studies where morphine was delivered intrathecally verify the observations by Gradert and Yaksh. Some of these masses have been reported as pyogranulomatous inflammation or pyogranulomas (terms also used by the author in reporting the observations in study reports), but a term used in the literature, and in many cases more correct (although less descriptive), is inflammatory mass (Deer et al. 2008). Figures 1A, B, and C show a well-formed, but also fairly typical, inflammatory mass/pyogranuloma at the catheter tip in a young adult monkey (all images are of the same mass). The development of an inflammatory mass/pyogranuloma at the catheter tip has the possibility of causing compression of the adjacent spinal cord (Figures 1A and 1B).
Inflammatory masses/pyogranulomas were rare in control animals, but they were present in 2 of 77 monkeys and 3 of 25 dogs. No inflammatory masses/pyogranulomas were noted in any of the control rats or sheep. Although the overall incidence of pyogranulomas was low in all the species, the observations do verify that these inflammatory masses may occur in control animals. Therefore, this lesion is definitely not strictly related to the administration of opiates or other drugs, at least in dogs and monkeys.
Spinal Cord Compression (at the Catheter Track)
Regardless of the reaction at the catheter tip, it is the effect of these reactions on the adjacent spinal cord (and spinal nerve roots) that has the most potential to produce an adverse effect.
Compression was a common finding. The incidence of spinal cord compression was 36.4%, 76.0%, 88.9%, and 45.1% for monkeys, dogs, sheep, and rats, respectively.
Compression of the cord was due to the presence of the catheter, the reaction around the catheter (inflammation and/or fibrosis), or both. This can be noted by the correlation between the average severity of fibrosis, inflammation, and compression in the data tables. Because inflammation and fibrosis at the catheter track varied between the studies, compression also varied.
Examples of spinal compression are shown in Figures 1D, E, and F. It is notable that compression that is mild or of lesser extent generally does not cause other spinal cord pathology (i.e., adjacent gliosis or nerve fiber degeneration). The spinal cords in Figures 1D, E, and F are essentially normal except for the compression. In the author’s experience in both diagnostic and toxicologic neuropathology, compression that takes place slowly (weeks to months) is less likely to produce other changes in the cord (nerve degeneration) that might lead to neurologic dysfunction.
Catheter Track, Fibrosis
Fibrosis at the catheter track was a very common finding, occurring in 64.9%, 96.0%, 94.4%, and 52.1% of monkeys, dogs, sheep, and rats, respectively. Fibrosis at the catheter track is shown in Figures 1B through F.
The connective tissue is nearly always circumferential and connected to/originating from the dura. This tissue sometimes can be observed to grow into the lumen of the catheter either through the open end of the catheter or through a fenestration in the catheter wall (Figures 1B, D, and E). However, the fibrosis is only significant (clinically) when it is of a size that contributes to the formation of a compressing mass at the tip.
In one sheep study, the average severity for fibrosis was a 3 (mild). But in most studies in all the species, the overall severity of fibrosis was generally minimal or less.
Spinal Cord Gliosis (at the Catheter Tip)
An increase in size and/or number of glial cells, predominantly astrocytes (as verified on sections stained immunohistochemically for GFAP), was noted in all species in the spinal cord adjacent to the catheter track.
Gliosis was uncommon in monkeys, dogs and sheep (incidence 3.9–11.0%) but more common in rats (22.5%). The increased incidence of gliosis in rats may have at least partially been due to the propensity for the catheter to penetrate the spinal cord in that species. Penetration of the spinal cord (at any level) was evident in 11.3% of the rats examined. The average severity of gliosis in all species and in all studies was less than 1 and was not believed to be of any biologic relevance other than to serve as a visible marker of some spinal cord damage/reaction secondary to the catheter track reaction.
Catheter Track, Nerve Fiber Degeneration
Nerve fiber degeneration was common in all species, being present in 24.7%, 24.0%, 27.8%, and 42.3% of monkeys, dogs, sheep, and rats, respectively. Although not specifically reported in the data, the nerve fiber degeneration in the spinal cord at the level of the catheter tip (in dogs, monkeys, and sheep) comes from two main sources: some local damage to the cord caused by the presence of the catheter and/or reaction to the catheter and damage to the cord at the site of catheter insertion. In addition to these two factors, the higher overall incidence of nerve fiber degeneration in the rat was at least partially secondary to the penetration of the spinal cord by the catheter in 11% of rats.
The average severity of nerve fiber degeneration varied among individual studies in the same species. Dogs and monkeys had studies with the highest average severity of nerve fiber degeneration at the catheter tip (1.75 in dogs; 1.83 in monkeys). Sheep were somewhat similar, with one study having an average severity of 1.25. Rats had the most consistent nerve fiber degeneration.
Discussion
Gavage studies are relatively straightforward. The tube goes in, the test article goes in, the tube comes out, eventually the tissues come out; and the pathologist looks at them, comparing control to test article–treated animals and deciding whether the single variable (the presence or absence of the test article) caused any adverse effects. But to date, oral and systemic injectable delivery methods have proven ineffective (or at least suboptimal) in combating some of the scourges of the nervous system, including neurodegenerative diseases, storage diseases, and in some cases even pain. The blood-brain/spinal cord/nerve barrier is particularly effective in preventing protein-based therapies from reaching the brain (Thorne and Frey 2001). Getting the therapy directly to the central nervous system may help.
But when a therapy begins with the implantation of a device into or in proximity to the brain or spinal cord, many variables can (and always do) complicate the morphologic evaluation. These variables include, but are definitely not limited to, the skill of the surgeon, the length of surgery, catheter insertion, catheter placement, catheter materials, pump materials, impurities that may leach out of the delivery system, rate of flow, concentration of the test article, after-care of the surgical site, length of time the device is in place, infection, and so forth. Any of these variables may elicit or exacerbate lesions in control and treated animals.
The purpose of this article was to indicate that changes, even (rarely) the development of inflammatory masses/pyogranulomas, may occur in control animals with an implanted intrathecal catheter. The exact pathogenesis of these inflammatory masses is still unknown. The addition of a test article may worsen these changes. For example, while some degree of meningeal infiltrates and/or catheter track inflammation is rather frequently present in controls, these changes can be exacerbated when a biologic material is introduced into the intrathecal space (unpublished data). This reaction to biologic materials may be a nonspecific immune-mediated response to the presence of a foreign protein, or it may be a direct effect of the test article formulation, or it may be both or neither. Also, the presence of such a reaction in a laboratory animal exposed to a recombinant human protein may not be predictive of what will occur in humans. There is much to learn.
Dealing with direct delivery studies involves embracing a new paradigm: some low level of risk, and some morphologic changes that might be considered adverse or undesirable with more traditional therapies, may be unavoidable when the therapy involves an implanted device. These changes are not necessarily unfavorable, and in the experience of the author, there are seldom clinical signs, neuronal damage, or other adverse events associated with the changes described in this article, unless the spinal cord compression or nerve fiber degeneration is moderate or severe.
Footnotes
Acknowledgment
The author thanks Beth Taylor for her assistance in preparing the data tables for this manuscript.
