Abstract
The continuing education course “Non-Invasive Imaging as a Problem-Solving Tool and Translational Biomarker Strategy in Toxicologic Pathology” provided a thorough overview of commonly used imaging modalities and the logistics required for integration of small animal imaging into toxicologic pathology. Non-invasive imaging (NIN) is gaining acceptance as an important modality in toxicologic pathology. This technology allows nonterminal, time-course evaluation of functional and morphologic endpoints and can be used to translate biomarkers between preclinical animal models and human patients. NIN can support drug development as well as basic research in academic or industrial environments. An initial overview of theoretical principles was followed by focused presentations on magnetic resonance imaging (MRI)/magnetic resonance microscopy (MRM), positron emission tomography (PET)/single proton emission computed tomography (SPECT), ultrasonography (US, primarily focused on echocardiography), optical (bioluminescent) imaging, and computed tomography (CT). The choice of imaging modality will depend on the research question and the needed resolution.
Keywords
The potential value of performing non-invasive, nonterminal, time-course evaluations in rodents used in toxicology and drug discovery studies cannot be overemphasized. The impact of quantitative imaging data collected at multiple time points from the same animals during preclinical rodent studies can identify subtle functional changes as well as document the temporal development of structural changes. Imaging data can be correlated with routine terminal anatomic pathology as well as terminal and/or intrastudy clinical pathology findings to give a truly integrated data set that may be used to help define mechanisms of action and follow lesion development (functional and/or structural) over time and might allow a more sensitive measure of subtle functional changes. In addition, by using non-invasive imaging, the effect of drug-induced toxicologic changes can be monitored longitudinally during the course of treatment and after withdrawal of dosing.
Imaging data from preclinical toxicity studies is potentially translatable to patients in the clinic depending on the toxicity and the animal model used. Translational biomarkers utilized today tend to be somewhat limited in number and scope. Useful translatable biomarkers that are well recognized by toxicologic pathologists include alanine aminotransferase (ALT), total bilirubin (TBIL), cardiac troponin I (TnCi), and so forth, which are primarily serum-based. Non-invasive imaging is an innovative structural or combined structural/functional approach that has the potential to revolutionize the drug discovery/development paradigm as well as speed the characterization of animal models (natural and/or genetically engineered).
Dr. Kathleen Gabrielson of the Johns Hopkins University School of Medicine (Baltimore, MD) presented an overview of non-invasive imaging that included the introduction of multiple non-invasive imaging modalities used to characterize disease in rodent models, examples of applications for each, and positive and negative features about each modality that figure into selection of a technique for a given research purpose (Table 1 ). Major imaging modalities are based on one of four main physical principles: magnetic fields (magnetic resonance imaging [MRI] and magnetic resonance microscopy [MRM]), radioactivity (conventional radiography, computer-assisted tomography [CT], positron emission tomography [PET], and single photon emission computed tomography [SPECT]), fluorescence (optical imaging), or sound wave transmission (ultrasonography [US]). The choice of imaging modality depends on several factors including the research question, availability of experienced technical staff, and ability to acquire and maintain the imaging systems and any equipment necessary to generate probes.
Comparison of common imaging modalities.
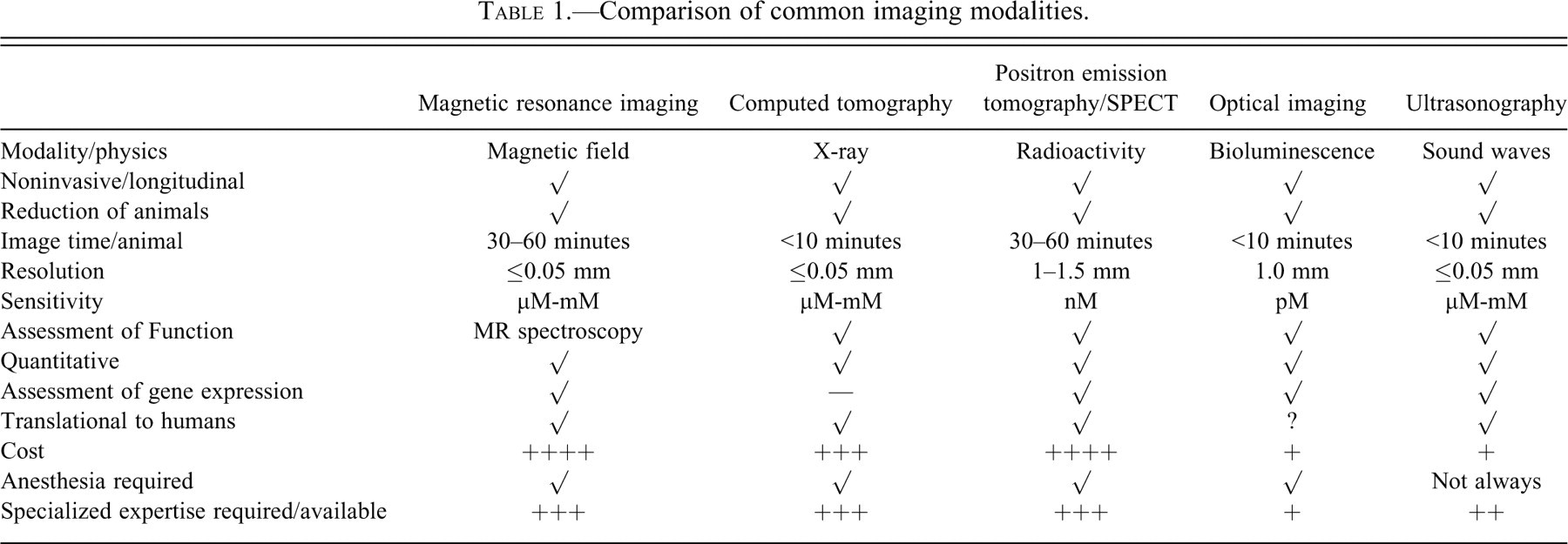
MRI and MRM may be used on fixed specimens or live animals for many purposes. Examples include phenotyping genetically engineered mouse (GEM) adults and embryos (Badea et al. 2007; Maronpot, Sills, and Johnson 2004), identifying toxicities (Lester et al. 2000; Maronpot, Sills, and Johnson 2004), rodent teratology examinations (Maronpot, Sills, and Johnson 2004), and evaluation of carcinogenesis and disease processes by serial imaging in live animals (Freimuth et al. 2010). CT is a 3-dimensional X-ray technique where contrast is generated by differences in tissue absorption (bone vs. soft tissue); by comparison, conventional X-ray machines (e.g., Faxitron X-Ray Corporation, Lincolnshire, IL) produce radiographic images in 2 dimensions. Contrast agents can be given to enhance tissue features on CT, such as perfusion of Microfil® into adult rodent heart or brain vasculature (Virtual Histology™ Neurology Imaging Services, Numira Biosciences, Salt Lake City, UT). Nuclear imaging modalities like PET or SPECT can also be combined with CT to provide greater soft tissue detail. For example, the PEG-Prom-mediated imaging system with a reporter gene/probe has the potential to be a general cancer-specific SPECT imaging agent (Su et al. 2005). The promoter of the progression-elevated gene 3 (PEG-3) drives expression of firefly luciferase (pPEG-Prom-FLuc-TK), which, when luciferin is injected, results in a bioluminescence signal or a radioactively labeled reporter in areas of tumor development when [125I] 1-(2-deoxy-2-fluoro-1-D-arabinofuranosyl)-5-iodouracil (FIAU) is injected (unpublished data). An example of SPECT imaging for cardiac myocyte apoptosis with Tc99m Annexin-V labeling of rodent heart after treatment with doxorubicin was discussed (Gabrielson et al. 2008) in which the SPECT signal correlated strongly with tissue apoptosis (as indicated by TUNEL staining) of the left ventricular myocardium. PET provides functional assessment by tracking labeled molecules in a live animal (e.g., transport mechanisms, metabolism, excretion, pharmacokinetic/pharmacodynamic time course, tumor metabolism, gene expression, etc.). For example, tumor metabolism may be evaluated with the glucose analog 2-deoxy-2-[18F]fluoro-D-glucose (18F-FDG) after treatment with a fatty acid synthase inhibitor molecule in animal xenograft models of primary human tumors (Lee et al. 2007).
Optical imaging, which measures fluorescence or bioluminescence as a measure of a biologic endpoint in live animals, may be used to determine gene expression patterns, infectious disease processes, gene delivery, stem cell biology, tumor growth, and response to therapy over time. The technology uses luciferase-tagged genes, cells, or bacteria (i.e., reporter genes), a CCD camera, a light-free chamber, and imaging software to analyze the fluorescent signal. Examples of where optical imaging can be used include evaluation of cancer relapse with drug failure in an orthotopic prostate cancer model, use of an IkB-luciferase reporter gene to evaluate induction of IkB expression in mice treated with 2 mg/kg lipopolysaccharide (LPS), and monitoring apoptosis with caspase-3-specific cleavage of recombinant luciferase (Laxman et al. 2002).
Dr. Gabrielson concluded with a review of ultrasonography (US), where she highlighted its utility in evaluating embryonic mouse development (Foster et al. 2002), evaluation of umbilical artery mean velocity, and development of visceral organs and the eye in the embryo. She also described the phenotypic characterization of mouse models (e.g., heart valve and aortic imaging in a mouse model of Marfan’s syndrome with or without Losartan treatment, which correlated well with macroscopic and histopathology data [Ng et al. 2004; Habashi et al. 2005]). Examples were given for cancer research (e.g., tumor volume estimation [Feldmann et al. 2007], determination of pre-palpable mammary tumors in adult mice, evaluation of surrounding structures for metastasis/invasion, image-guided needle injection, vascularity and blood flow determination, and determination of necrosis). Contrast agents for use with US include micro-bubbles and targeted micro-bubbles, where a molecular tag will attach the contrast agent to a specific site (e.g., targeted micro-bubbles are coated with biotin and incubated with a streptavidin-labeled anti-vascular endothelial growth factor-receptor 2 antibody will bind to the luminal surface of endothelial cells, thus allowing labeling of vascular structures).
Dr. Allan Johnson from the Duke University School of Medicine (Durham, NC) presented on “Preclinical Imaging in Discovery and Safety Assessment.” The goals of his presentation were to review the “man to mouse” translation problem, types of imaging available, the necessity of integration, and examples of imaging in preclinical studies. Dr. Johnson illustrated the use of digital subtraction angiography (DSA) and biological pulse sequence imaging to produce DSA perfusion maps that illustrate relative blood volume, relative blood flow, and relative mean transit time in normal and tumor vascularization in mouse xenotransplant tumor models (Ostergaard et al. 1996). He next highlighted the use of respiratory and cardiac gating dual micro-CT for small animal imaging (Badea, Hedlund, and Johnson 2004; Badea et al. 2008) to capture high-resolution images at the high heart rates of rodents, liposomal/blood pool contrast agents (Mukundun et al. 2006; Badea et al. 2005) to highlight the vasculature, and evaluation of pulmonary function with hyperpolarized gas MRI (Driehuys et al. 2007; Chen, Yordanov, and Johnson 2005; Viallon et al. 1999; Middleton et al. 1995; Gewalt et al. 1993). Dr. Johnson reviewed MRM and his group’s equipment, which includes a 7T EXCITE MR Microscope (Varian Inc., Palo Alto, CA), as well as showing examples of the utility of MRM in the rodent (i.e., The Visible Mouse™ [Johnson et al. 2002], examples of control and uricase knockout mice [Kelly et al. 2001], identification of altered hepatic foci in the live rodent [unpublished data], and highlights on the resolution possible with MRM (≤0.05 mm) [Maronpot, Sills, and Johnson 2004]).
Dr. Martin Pomper from the Russell H. Morgan Department of Radiology and Radiological Sciences at Johns Hopkins University School of Medicine (Baltimore, MD) presented on PET and SPECT modalities and provided examples comparing optical imaging with nuclear imaging. Dr. Pomper highlighted the importance of translational research and the utility of small animal imaging including: evaluation of a single rodent in longitudinal studies where it can act as its own control, use of fewer animals, the ability to image transgenic/knockout rodent models of human disease, dedicated imaging systems for use with rodents that obviate competition with the clinic, and finally the ability with PET and SPECT to evaluate protein-protein interactions and gene expression in the live animal model. Dr. Pomper listed the main requirements for starting/setting up a preclinical molecular imaging center, which include a scientific need (justification), availability of qualified personnel, access to or a budget to purchase the imaging equipment (i.e., SPECT = $500,000 versus PET = $2 million), capability needed to answer research questions of interest, a dedicated and appropriately engineered space for imaging, and most significantly strong institutional support (i.e., financial and philosophical [preclinical imaging capability is a priority]). A dedicated imaging group requires a long-term investment in equipment as well as highly skilled people from the academic or industrial institution. Major differences between SPECT and PET are as follows: SPECT equipment is less expensive, requires generator isotopes (i.e., 99mTc, 111In, 123I), is qualitative, provides less resolution (minimum of 1.5 cm), and utilizes chelation chemistry for labeling. PET equipment is significantly higher in price, requires cyclotron isotopes (i.e., 15O, 13N, 11C, 18F, 124I), is quantitative, provides more resolution than SPECT (i.e., minimum of 4 mm), and utilizes physiologic tracers. Potential uses for PET include receptor/enzyme/transporter mapping, evaluation of metabolism, glucose metabolism, oxygen extraction fraction, blood volume, calculation of drug receptor occupancy, and biomarkers for therapy/patient segregation. Dr. Pomper reviewed several examples of translational imaging projects (PET, SPECT, and bioluminescence imaging), including SPECT imaging of prostate specific membrane protein (PSMA) on prostatic tumor cells with 11C-DCMC, a radiolabeled urea-based PSMA inhibitor and a direct probe for PSMA (Foss et al. 2005; Banerjee et al. 2008); primary xenograft and metastatic cancer imaging utilizing 125I-labeled monoclonal anti-human CX-chemokine receptor 4 (CXCR4) antibodies (Nimmagadda, Pullambhatla, and Pomper 2009); imaging hedgehog signaling and ABCG2/BCRP expression with bioluminescence using a reporter gene/probe system (D-luciferin as the substrate) (Zhang et al. 2007, 2009; Zhang, Laterra, and Pomper 2009); imaging of infection (e.g., measuring bacterial burden in a C3HeB/FeJ mouse model of infection with Mycobacterium tuberculosis using [125I] FIAU-SPECT, a nucleoside analog substrate for bacterial thymidine kinase [Davis et al. 2009; Bettegowda et al. 2005], and measuring hypoxia in C3HeB/FeJ mouse tissues infected with Mycobacterium tuberculosis using [64Cu]ATSM-PET [unpublished data]); and imaging effects of bacteriolytic tumor therapy (Clostridium novyi–lethal toxin inactivated labeling of xenograft tumors in mice where the bacteria colonize the hypoxic/anaerobic areas of the tumors and cause tumor lysis [unpublished data]).
Dr. Robert Coatney from the Laboratory Animal Sciences Department at GlaxoSmithKline (King of Prussia, PA) delivered a presentation titled “Ultrasound Imaging in Drug Discovery: The Good, the Bad, and the Promising” that reviewed the use of ultrasonography in drug discovery/development with examples of translation to clinical trials. Dr. Coatney’s goals were to highlight where ultrasound is used in the drug discovery paradigm, advantages and challenges of the technology, examples of ultrasonography used in efficacy and safety assessment studies and medicines differentiation, and finally an overview of strain imaging in the heart as a powerful emerging technology. The advantages of ultrasonography in drug discovery are quantitation of structure and function simultaneously, serial assessment, highly translational (man to mouse), complementary nature of the data relative to other approaches, techniques/parameter collection have been well validated, versatility and portability of the method, temporal and spatial resolution, the possibility for molecular and contrast-enhanced imaging, and relatively low cost. Challenges of ultrasonography include the relatively inexpensive approach allows “novices” to purchase and use the equipment inappropriately, producing a “pretty” picture rather than useful quantitative data; difficulties in consistently getting the same image within an animal over time or between animals and between studies; and the multiplicity of imaging techniques that are available to achieve the same parameter (e.g., stroke volume). For consistent and meaningful data collection a robust protocol, validation of the imaging procedure and quantitation techniques and an experienced sonographer are essential. Interobserver variability is a major problem with ultrasonography and is most apparent when comparing novice and highly experienced sonographers. Ultrasound is a Doppler-based imaging modality that allows non-invasive spatial resolution (i.e., in axial and lateral direction), which is based upon the pulse frequency of the transducer. With increasing frequency there is a decrease in the depth of penetration. Temporal resolution is determined by the image acquisition frame rate (i.e., frames/minute or Hz), which must be high because rodents have very rapid heart rates (i.e., 400+ beats/minute). Ultrasound biomicroscopy uses ECG gating/timing, higher spatial resolution, and higher transducer frequency to allow efficient ultrasound evaluations in rodents, but with limited translatability to human patients due to limitations in ultrasound penetration of internal organs. Ultrasound imaging modes include two-dimensional grey-scale (B-mode), one-dimensional grey-scale for evaluating motion/rate (M-Mode), and 3- and 4-dimensional imaging (ultrasound biomicroscopy). These modes can be applied in evaluation of the heart (i.e., echocardiography) where the following parameters can be evaluated: left ventricular (LV) end diastolic volume, LV end systolic volume, stroke volume, cardiac output, ejection fraction, fractional shortening, wall motion abnormalities, LV mass, and local and global wall thickness (Weyman 1994). Echocardiography has been used to evaluate compound efficacy (e.g., in efficacy animal models of hypertension, tumor development, etc.) and compound safety (e.g., structural and functional valvular lesion development, drug-induced lesions in several different organs, and cardiac structure and/or function in the face of cardiac stress/strain). The evaluation of systolic function and strain as well as strain rate with test article treatment in the face of dobutamine stress or in a model of transaortic constriction in the rat allows a very sensitive evaluation of compound safety effects in animal models that have more similarities to human patients, thus allowing more effective translation to the clinics (Mercuro et al. 2007; Jurcut et al. 2008; Dandle et al. 2009). In conclusion, Dr. Coatney illustrated the “Good” (i.e., structure and function, good resolution, translatable endpoints, non-invasive, serial evaluations, good body of literature, and validated protocols), the “Bad” (i.e., high interobserver variability, use of unvalidated techniques, and the dependence on operator level of experience), and the “Promising” (i.e., strain and strain-rate imaging, 3D and 4D imaging, and use of echocardiography in assessing cardiovascular safety endpoints).
Dr. Christopher Winkelmann from Merck Research Laboratories (West Point, PA) reviewed X-ray CT and its use in reproductive and general toxicology. CT systems for animals are optimized to provide spatial resolution in the 1–200 micron range (i.e., micro CT). CT is an X-ray-based technology that is particularly suited for evaluation of bone, including fetal skeletons, and lung due to the natural physiological contrast of these anatomical regions. Developmental and reproductive toxicity (DART) studies are used in safety assessment to evaluate developmental toxicity of compounds after administering to pregnant rats and rabbits. One component of DART studies is the evaluation of the fetal skeleton, which routinely is evaluated grossly after Alizarin Red S staining and clearing (which takes 1–2 weeks to complete). MicroCT can be used to replace alizarin red S staining to provide for faster turnaround times, semiautomated analysis, a reduction in the amount of hazardous waste, and long-term cost savings (unpublished data). To optimize this approach, in vivo and ex vivo pilot studies were undertaken in rats and rabbits, fetal holders were developed to increase imaging throughput (i.e., 21 rat fetuses and 10 rabbit fetuses per holder, allowing approximately 400 rat or 200 rabbit fetuses to be scanned per hour), and protocols to visualize and evaluate specific skeletal and soft tissue structures were developed (e.g., contrast administration to label placenta and maternal vasculature to help isolate individual fetal skeletons, and adjustment of colormap parameters to optimize evaluation of different skeletal structures). Colormap setting adjustment allowed differential visualization of skeletal structures of different densities and thickness (e.g., membranous skull bones = 25–200 Hounsfield Units (HU), vertebrae and skull sutures = 300–600 HU) (Winkelmann and Wise 2009). In the validation of the microCT technique for DART studies, evaluation of positive control compound studies was performed (i.e., boric acid in rats, hydroxyurea in rabbits, and retinoic acid in both species [Wise and Winkelmann 2009a, 2009b, forthcoming]). MicroCT identified most skeletal lesions identified by the Alizarin Red S staining in the boric acid study (i.e., hypoplastic rib, cervical vertebrae fusion, and incomplete ossification of the fifth sternebra); the only exceptions involved the small abnormalities (e.g., hypoplastic rib) that are below the resolution capacity of the particular microCT system. MicroCT in rabbits treated with hydroxyurea showed abnormalities such as hypoplastic ribs and incomplete metacarpal ossification, and was able to provide additional information such as femur length measurements. Rats and rabbits treated with all trans-retinoic acid had comparable findings between the Alizarin Red S and microCT (i.e., extra vertebrae and supernumerary ribs). Overall, the positive control studies showed that there were comparable findings with Alizarin Red S staining and microCT techniques. Other microCT endpoints that are not possible with the fetal staining technique include evaluation of bone density and volume. Any skeletal abnormalities that were not detected with microCT were very small and near or below the resolution of the CT. Benefits of microCT over Alizarin Red S staining in DART studies include decreased turnaround time; elimination of hazardous materials; reduced costs of supplies, archiving, and laboratory space needed for the staining method; elimination of staining artifacts; enhanced ability to share images electronically; and easy evaluation of multiple stages of skeletal development. Disadvantages of microCT include costs of microCT equipment, image storing, need for extensive training, and validation and potential affect of equipment malfunction, although that risk can be mitigated by either freezing or fixing the specimens for later evaluation. Dr. Winkelmann and his group are currently working on automated fetal image analysis (i.e., bone volume/density and ultimately automated qualitative evaluation of bones with development of rule sets followed up by reviewer verification), soft tissue evaluation with contrast-stained specimens (i.e., where contrast uptake allows visualization of soft tissues in the fetal specimens), validation of the process to allow GLP compliance (i.e., microCT data is now sent to FDA as non-GLP accessory data with no agency comments to date), and in vivo longitudinal imaging (i.e., to examine the question: do minor abnormalities resolve?). In addition, this technology has the potential to combine embryofetal developmental toxicity and prenatal and postnatal developmental toxicity studies to decrease animal usage. Concerns regarding the use of multiple rounds of anesthesia and radiation dose for interim time points need to be addressed as both anesthetic agents and radiation are potentially teratogenic, although interim evaluations would certainly be useful in other areas of safety assessment.
A question-and-answer session with the course speakers and participants occurred at the conclusion of the session. The questions primarily were regarding regulatory issues and GLP validation of non-invasive imaging techniques. The overall assessment of the speakers was that GLP validation was possible, as Dr. Winkelmann and his group have shown with their work on validating the microCT for developmental toxicity study use. Kathleen Gabrielson introduced the Small Animal Imaging Resource Programs (SAIRP), which are funded by the National Cancer Institute (NCI) and are established at multiple localities around the United States (http://imaging.cancer.gov/programsandresources/specializedinitiatives/SAIRP). The goal of SAIRP is to facilitate the use of imaging methods to track cancer therapy responses and to better understand cancer biology.
In conclusion, this continuing education course on non-invasive imaging provided a venue for the exposure of toxicologic pathologists to the utility and power of non-invasive imaging modalities in animal model evaluation and drug discovery/development, and for interactions with imaging experts. The use of non-invasive imaging as a translatable biomarker strategy as well as unique techniques to evaluate such processes as apoptosis, cancer, cardiovascular toxicity, and bacterial infection in living animal models are technical advances that we hope will soon be incorporated routinely into our toxicologic pathology projects.
