Abstract
2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD) and structurally-similar dioxin-like compounds affect thyroid function and morphology and thyroid hormone metabolism in animals and humans. The National Toxicology Program conducted eight 2-year gavage studies in female Harlan Sprague-Dawley rats to determine the relative potency of chronic toxicity and carcinogenicity of TCDD, 3,3′,4,4′,5-pentachlorobiphenyl (PCB126), 2,3,4,7,8-pentachlorodibenzofuran (PeCDF), 2,3′,4,4′,5-pentachlorobiphenyl (PCB118), 2,2′,4,4′,5,5′-hexachloro-biphenyl (PCB153), a tertiary mixture of TCDD/PCB126/PeCDF, and two binary mixtures (PCB126/PCB153 and PCB126/PCB118). Administration of these compounds was associated with increased incidences of thyroid follicular cell hypertrophy, variably observed in the 14-, 31-, and 53-week interim and 2-year sacrifice groups. In all studies, the incidences of follicular cell adenoma and carcinoma were not increased. Decreased levels of serum thyroxine were primarily noted in the 14-or-later -week interim groups of all chemicals. Serum triiodothyronine (T3) levels were increased in the TCDD, PCB126, PeCDF, TCDD/PCB126/PeCDF, and PCB126/PCB153 studies, while decreased levels were noted in the PCB153 and PCB126/PCB118 studies. TCDD, PCB126, PCB126/PCB153, and PCB126/PCB118 increased levels of serum thyroid-stimulating hormone almost in a dose-dependent manner in the 14-week groups. These data suggest that although dioxin-like compounds alter thyroid hormones and increase follicular cell hyperplasia, there is not an increase in thyroid adenoma or carcinoma in female Sprague-Dawley rats.
Introduction
Certain polychlorinated dibenzodioxins, polychlorinated dibenzofurans (PCDFs), and coplanar polychlorinated biphenyls (PCBs) have the ability to bind to the aryl hydrocarbon receptor and exhibit biologic actions similar to those of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD). These dioxin-like compounds (DLCs) have similar chemical structural properties to TCDD and may induce a broad array of effects including developmental, endocrine, and immunological toxicity and multiorgan carcinogenicity in animals and/or humans (Agency for Toxic Substances and Disease Registry 1998, 2000; Bertazzi et al. 2001; International Agency for Research on Cancer (1997) Kociba et al. 1978; Steenland et al. 2001).
DLC-induced alterations in thyroid hormone homeostasis have been observed in both humans and rodents. In humans, some studies demonstrate an association between exposure to DLCs and thyroid hormone homeostasis and/or thyroid disease. Exposure to DLCs induces significant increases in thyroid-stimulating hormone (TSH) levels and/or decreases in thyroxine (T4) or triiodothyronine (T3) levels (Bahn et al. 1980; Hagmar et al. 2001; Koopman-Esseboom et al. 1994; Nagayama et al. 1998; Osius et al. 1999; Persky et al. 2001; Pluim et al. 1993). The incidences of thyroid disease were increased in a cohort of factory workers exposed to TCDD due to the explosion in a chemical factory in Germany (Zober et al. 1994). Thyroid cancer risks are increased in a large occupational cohort of pesticide sprayers with possible exposure to TCDD (Saracci et al. 1991). There was also a suggestive increase in the incidences of thyroid cancer in the 20-year period following an accident at a chemical plant in Seveso, Italy, in 1976, which exposed a large residential population to TCDD (Pesatori et al. 1993, 2003). Information on the health effects of PCBs is available from studies of exposure in the workplace, consumption of contaminated rice oil in the Japanese Yusho accident and the Taiwanese Yu-Cheng accident, consumption of contaminated fish, general environmental exposures, and food products of animal origin. Evidence suggests that PCBs can produce several toxicities, such as skin lesions, dental abnormalities, immune deficiency, and/or reproductive abnormalities and increase the risk of developing cardiovascular and/or liver disease and diabetes (Yoshizawa et al. 2007). While PCBs did not increase thyroid cancer risk in individuals exposed to rice oil contaminated by PCBs and PCDFs in these accidents, alteration in thyroid function was seen in some patients (Aoki 2001; Arisawa et al. 2005; Guo et al. 1999).
Exposure to TCDD has been demonstrated to affect thyroid morphology and functionality in rodents. TCDD is thought to induce these alterations by increasing the glucuronidation of T4 via a secondary mechanism involving the induction of hepatic uridine diphosphate glucuronosyltransferase 1. Increased conjugation results in increased biliary excretion and decreased circulating levels of T4, leading to chronic hypersecretion of thyroid-stimulating hormone (TSH), which places the rodent thyroid gland at greater risk of developing tumors (Bastomsky 1977; Capen 1996, 1997; Hill et al. 1989; Potter et al. 1983; Van Birgelen et al. 1994). Chronic oral exposure to TCDD in male Osborne-Mendel rats and female B6C3F1 mice induces thyroid follicular cell adenomas at dose-related incidences (National Toxicology Program [NTP] 1982). In other studies in Sprague-Dawley (SD) rats, no clear increase was observed in the incidence of follicular cell adenomas and adenocarcinoma (Kociba et al. 1978), although TCDD increases the incidences of follicular cell hypertrophy (Nishimura et al. 2002, 2003; Sewall et al. 1995).
The NTP conducted a series of 2-year bioassays in female Harlan SD rats to evaluate the chronic pathology and carcinogenicity induced by DLCs, structurally related PCBs, and mixtures of these compounds, including TCDD; 3,3′,4,4′,5-pentachlorobiphenyl (PCB126); 2,3,4,7,8-pentachlorodibenzofuran (PeCDF); 2,3′,4,4′,5-pentachlorobiphenyl (PCB118); 2,2′,4,4′,5,5′-hexachlorobiphenyl (PCB153); a tertiary mixture of TCDD, PCB126, and PeCDF (toxic equivalency factor [TEF] mixture); and 2 binary mixtures of PCB126 and 153, and PCB126 and 2,3′,4,4′,5-pentachlorobiphenyl (PCB118; NTP 2004a, 2004b, 2004c, 2004d, 2004e, 2004f, 2004g). Although the goals of these studies were broad, we focus here on the functional and morphological changes of the thyroid induced by the individual congeners and the mixtures normalized for TCDD toxic equivalents (TEQs) based on the World Health Organization (WHO) TEFs from the 8 NTP studies. Our investigation is novel in that it compares the long-term chronic effects of various classes of DLCs and in addition evaluates the time-related progression of changes in the thyroid hormones and morphology.
Materials and Methods
Study Design
These studies were conducted by the NTP (http://ntp.niehs.nih.gov) as a series of chronic 2-year rat bioassays designed to evaluate the relative potencies for carcinogenicity of individual congeners and the additivity of carcinogenicity for mixtures of DLCs. The studies were conducted in female Harlan SD rats based on the high potency of TCDD as a hepatocarcinogen in the female SD rat (Kociba et al. 1978). This female strain is more sensitive to TCDD-induced carcinogenic effects (Kociba et al. 1978; Walker et al. 2006). Animals were dosed once daily for 5 days per week by oral gavage using the test compound in corn oil:acetone vehicle (99:1); control animals received vehicle only. Approximately 50 animals per group were dosed for 2 years, except for a group of “stop-study” animals, which were dosed for 31 weeks and then received the vehicle only until study termination. Interim evaluations of 8 to 10 animals per group were conducted at 14, 31, and 53 weeks.
The doses of all compounds were based on the 1998 WHO TEF values (Van den Berg et al. 1998) relative to TCDD, the most potent DLC. The TEF values, structures, and doses for the selected DLCs evaluated in these studies are presented in Table 1. While the WHO TEF values were reevaluated and in some cases changed (Van den Berg et al. 2006), in this article, to avoid confusion with other publications from these studies, we use the TEQ dose values based on the WHO 1998 TEF values that were in use at the time the studies were designed. The compounds selected for evaluation were TCDD, PCB126, PCB153, PeCDF, and PCB118. PCB126, a non–ortho-substituted PCB, is considered the most potent dioxin-like PCB congener present in the environment, accounting for 40% to 90% of the total toxic potency of PCBs exhibiting dioxin-like activity. PeCDF, a dioxin-like polyhalogenated aromatic hydrocarbon, represents the most potent PCDF present in human tissues. PCB118 is a mono–ortho-substituted PCB with partial dioxin-like activity. In contrast, PCB153, which is a di–ortho-substituted nonplanar PCB, exhibits no dioxin-like activity and therefore merits no inclusion in the TEF methodology.
Associated doses used in the carcinogenic studies of dioxin and dioxin-like compounds.
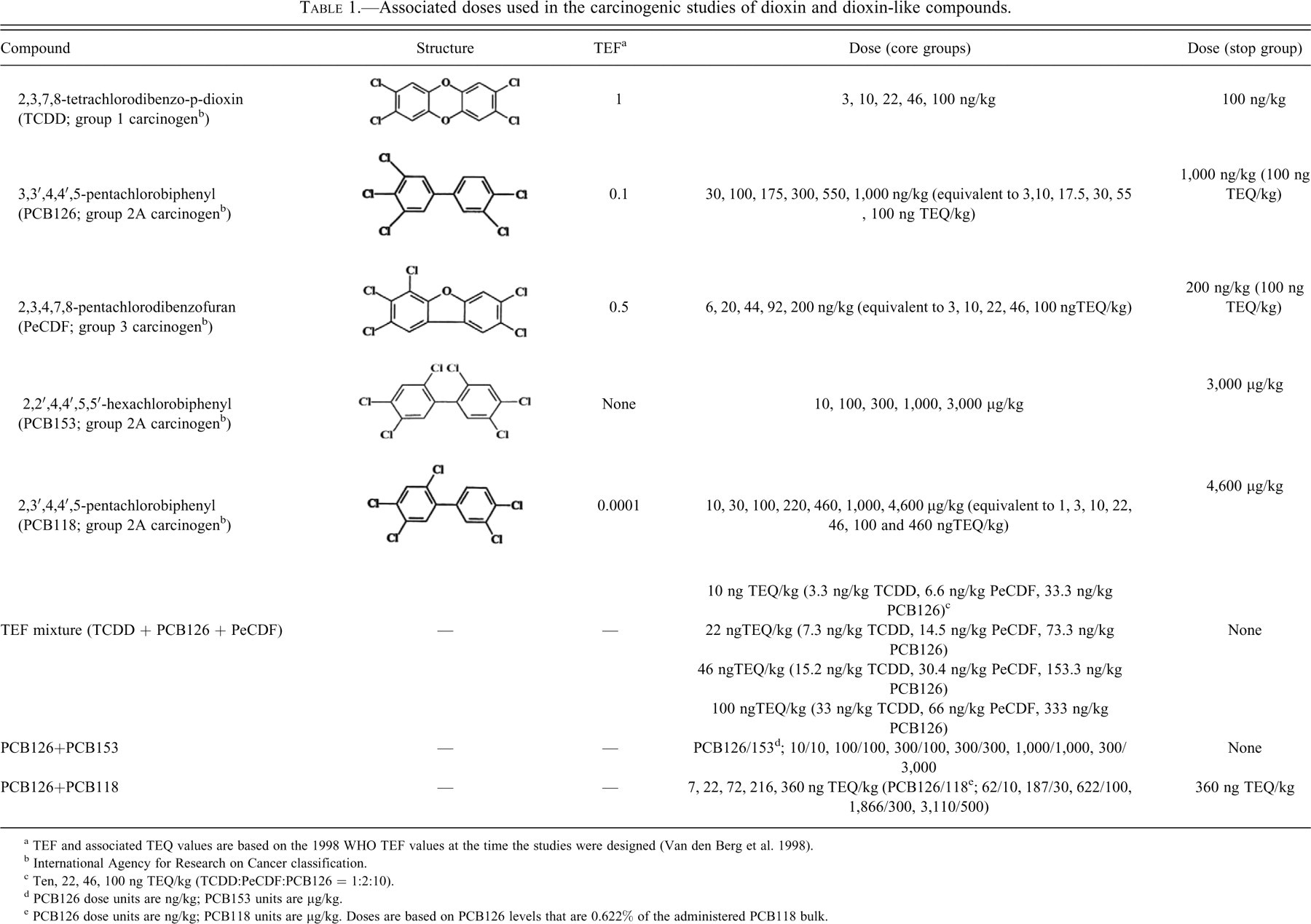
a TEF and associated TEQ values are based on the 1998 WHO TEF values at the time the studies were designed (Van den Berg et al. 1998).
b International Agency for Research on Cancer classification.
c Ten, 22, 46, 100 ng TEQ/kg (TCDD:PeCDF:PCB126 = 1:2:10).
d PCB126 dose units are ng/kg; PCB153 units are µg/kg.
e PCB126 dose units are ng/kg; PCB118 units are µg/kg. Doses are based on PCB126 levels that are 0.622% of the administered PCB118 bulk.
Chemicals
TCDD, PCB126, and PeCDF were supplied by IIT Research Institute (Chicago, IL), AccuStandard, Inc. (New Haven, CT), and Cambridge Isotope Laboratories (Cambridge, MA), respectively. PCB153 and PCB118 were supplied by Radian International LLC (Austin, TX). Each chemical was received in one lot that was used for the entire study.
Animals
The animal studies were conducted in accordance with the Good Laboratory Practice guideline at Battelle Columbus Laboratories (Columbus, OH). Female Harlan SD rats, approximately 6 weeks of age, were obtained from Harlan Sprague-Dawley, Inc. (Indianapolis, IN). All animals were quarantined for approximately 2 weeks for health screening and were approximately 8 weeks old at the start of the study. Animals were housed in solid-bottom polycarbonate cages (Lab Products, Inc., Maywood, NJ) suspended on stainless-steel racks with 5 animals per cage. Filtered room air underwent at least 10 changes per hour. Animal rooms were maintained at 69°F to 75°F with 35% to 65% relative humidity and 12 hours of light and 12 hours of dark. Irradiated NTP-2000 pelleted feed (Zeigler Bros., Inc., Gardners, PA) and water were available ad libitum. All animals were observed twice daily for morbidity and once a month for clinical signs of toxicity; moribund animals were euthanatized and necropsied. The health status of the animals was monitored by serological analysis of serum samples collected from the study animals and male sentinel rats that were placed in the study rooms. Serum samples remained negative for any significant rodent pathogen. Animal husbandry and handling were conducted in accordance with the National Institutes of Health guidelines (Grossblatt 1996).
Pathological Evaluation
At necropsy, all tissues were examined grossly, any lesions observed were recorded, and tissues were removed and fixed in 10% neutral buffered formalin for microscopic evaluation. After fixation, the tissues were trimmed, processed, embedded in paraffin, sectioned at a thickness of 5 µm, stained with hematoxylin and eosin (H&E), and examined microscopically. The severity of lesions was graded on a four-point scale of 1 = minimal, 2 = mild, 3 = moderate, and 4 = marked. According to the Guides for Toxicologic Pathology (Botts et al. 1991), the following criteria for the diagnosis of thyroid follicular lesions were applied: Hypertrophy: decreased diameter of follicular lumens with increased height of follicular epithelium from cuboidal to tall columnar Hyperplasia: the above-mentioned changes with papillary infoldings of or stratification of follicular epithelium Adenoma: nonencapsulated well-demarcated proliferation of follicular epithelium with compression of adjacent follicles Carcinoma: solid or multiple layers of proliferation of follicular epithelium with cellular pleomorphism
The pathology findings from all studies were subjected to a full NTP peer review. For ensuring the consistency of the histopathological diagnoses among all of the TEF dioxin studies, the same study pathologist, quality assurance pathologist, pathology working group (PWG) chairperson, NTP pathologist, and members of the PWG served in all studies (Boorman et al. 2002), with the exception of the PCB118 studies.
Thyroid-related Hormone Analysis
At 14, 31, and 53 weeks in all 8 studies, blood was taken from the retro-orbital sinus of up to 10 rats per group (except the stop-exposure group) and processed to serum for thyroid hormone determinations. Radioimmunoassays were performed for TSH and T3 using a Packard Cobra II gamma counter (Packard Instrument Company, Meriden, CT). The assay for total T4 was performed on a Hitachi 911R chemistry analyzer using an enzyme immunoassay test system (Boehringer Mannheim, Indianapolis, IN). Thyroid hormone data were summarized using the XYBION system (XYBION Medical Systems Corporation, Cedar Knolls, NJ).
Statistical Analysis
Incidences of lesions in the study animals were evaluated statistically by the Poly-3 test of Portier and Bailer (1989) and Bailer and Portier (1988), which adjusts for survival differences among groups. The incidences of lesions in animals from each of the interim evaluations and from the 2-year study were analyzed separately. For animals in the 2-year studies, the total lesion incidences, including findings from animals that survived until study termination and from early death animals, were included in the analysis. Thyroid-related hormones were analyzed using nonparametric multiple-comparison methods of Shirley (1977) as modified by Williams (1986), or of Dunn (1964), depending on whether a dose-related trend was present (Shirley 1977; Williams 1986) or absent (Dunn 1964).
Results
Thyroid Histopathology
Overall, increased incidences of follicular cell hypertrophy in the thyroid were observed to some extent in all of the studies for the DLC compounds and the non–dioxin-like PCB153 (Tables 2 and 3). These lesions consisted of a localized to diffuse change, characterized by follicles that were decreased in size and contained decreased amounts of colloid in which aggregates of amphophilic, flocculant-appearing material were often present (Figure 1A, B). The affected follicles were lined by large, prominent cuboidal follicular epithelial cells that were approximately two to three times normal size, usually with abundant pale cytoplasm containing small, clear vacuoles. Since some degree of this change can occur spontaneously, the severity grade of minimal was recorded when 50% to 60% of the follicles were involved, mild severity when 60% to 75% of the follicles were involved, moderate when 75% to 90% of the follicles were involved, and marked when more than 90% of the follicles were involved. The severity was minimal to mild in nearly all affected animals in all groups, including the vehicle control and treated groups. In all studies, the incidences of follicular cell adenoma and carcinoma were not increased.
Incidences and average severities of thyroid follicular cell hypertrophy in rats treated with TCDD, PCB126, PeCDF, or the tertiary mixture of TCDD/PCB126/PeCDF.
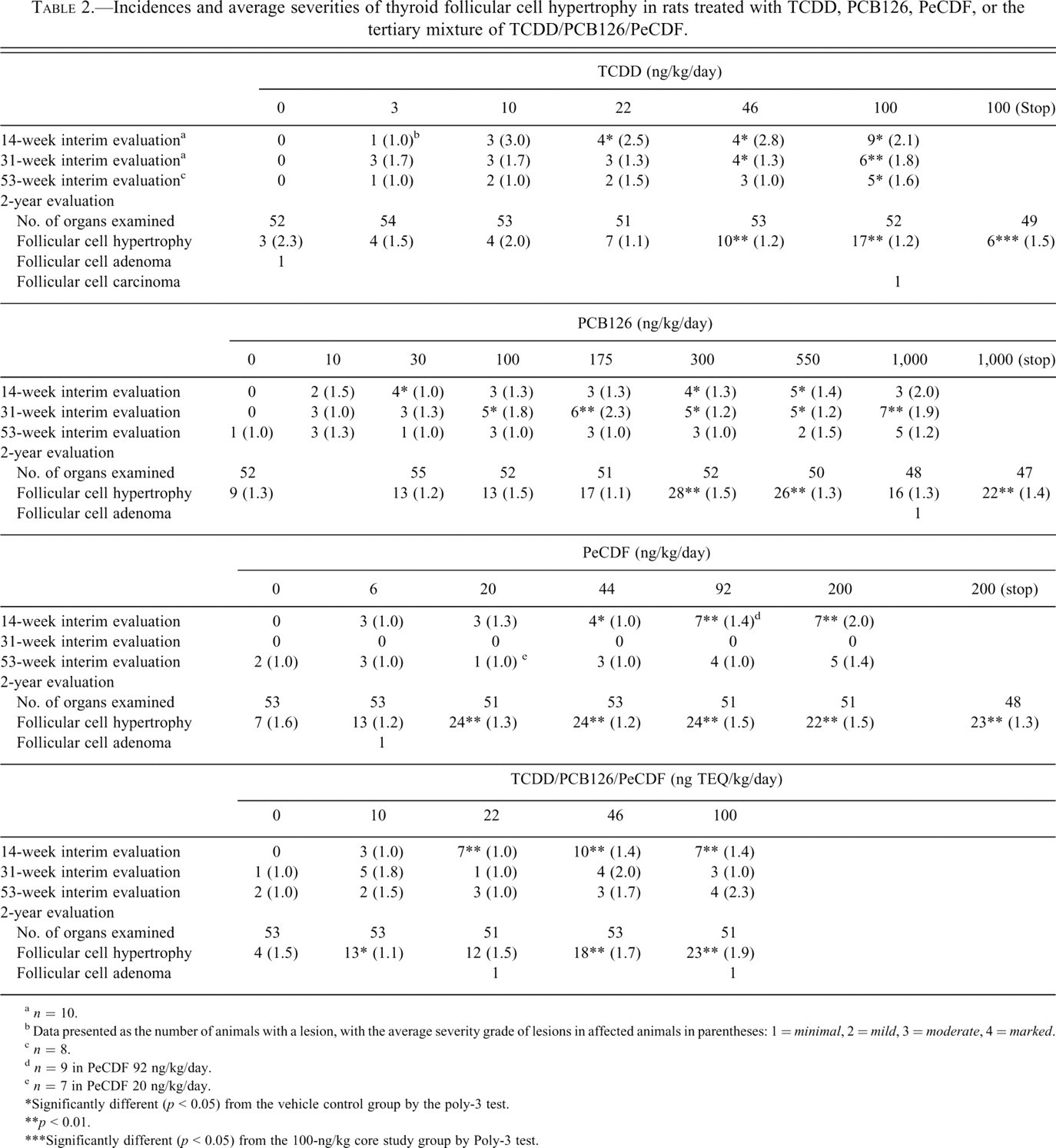
a n = 10.
b Data presented as the number of animals with a lesion, with the average severity grade of lesions in affected animals in parentheses: 1 = minimal, 2 = mild, 3 = moderate, 4 = marked.
c n = 8.
d n = 9 in PeCDF 92 ng/kg/day.
e n = 7 in PeCDF 20 ng/kg/day.
*Significantly different (p < 0.05) from the vehicle control group by the poly-3 test.
**p < 0.01.
***Significantly different (p < 0.05) from the 100-ng/kg core study group by Poly-3 test.
Incidences and average severities of thyroid follicular cell hypertrophy and adenomas in rats treated with PCB153, PCB118, a mixture of PCB126/PCB153, or a mixture of PCB126/PCB118.
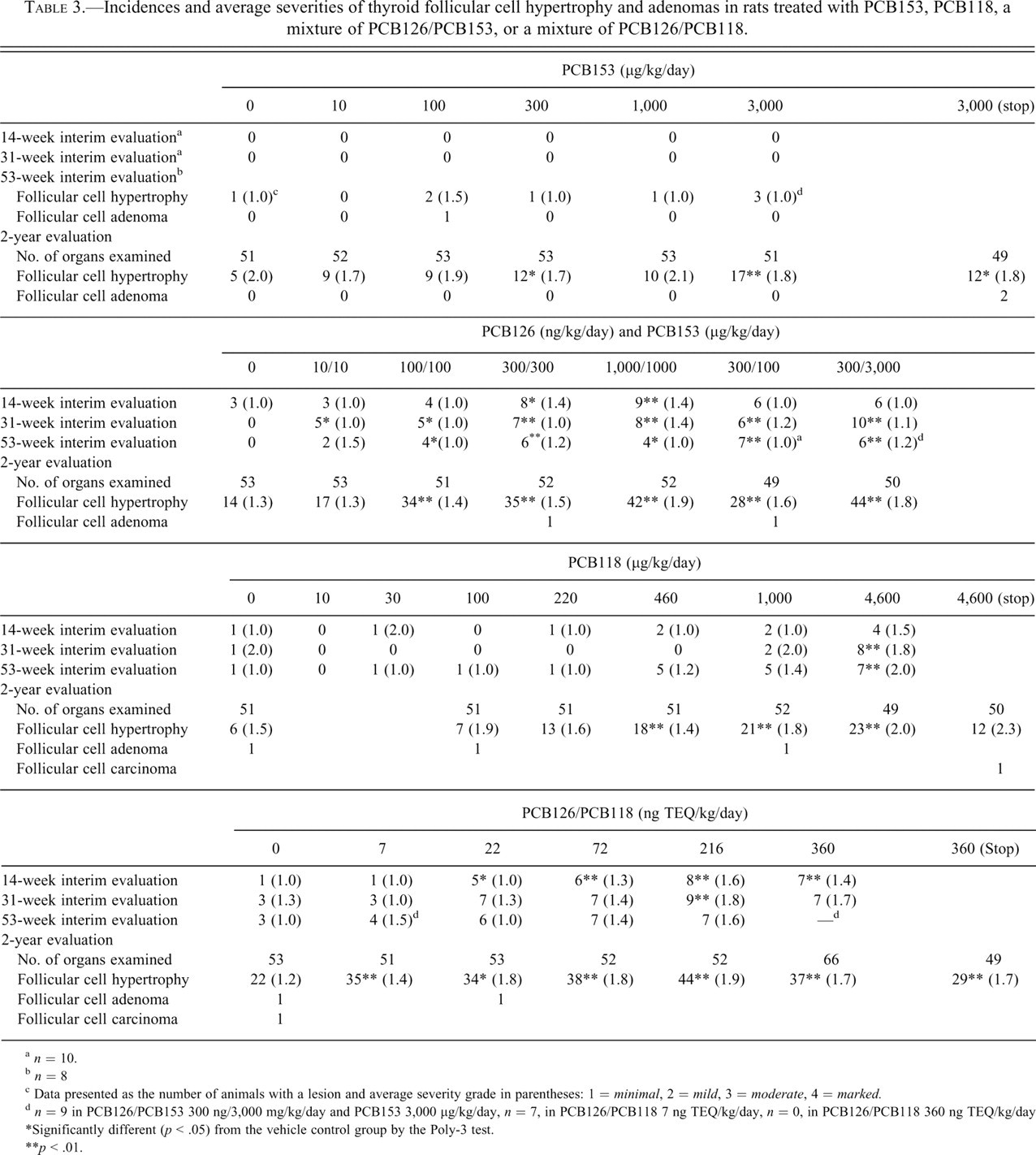
a n = 10.
b n = 8
c Data presented as the number of animals with a lesion and average severity grade in parentheses: 1 = minimal, 2 = mild, 3 = moderate, 4 = marked.
d n = 9 in PCB126/PCB153 300 ng/3,000 mg/kg/day and PCB153 3,000 µg/kg/day, n = 7, in PCB126/PCB118 7 ng TEQ/kg/day, n = 0, in PCB126/PCB118 360 ng TEQ/kg/day
*Significantly different (p < .05) from the vehicle control group by the Poly-3 test.
**p < .01.
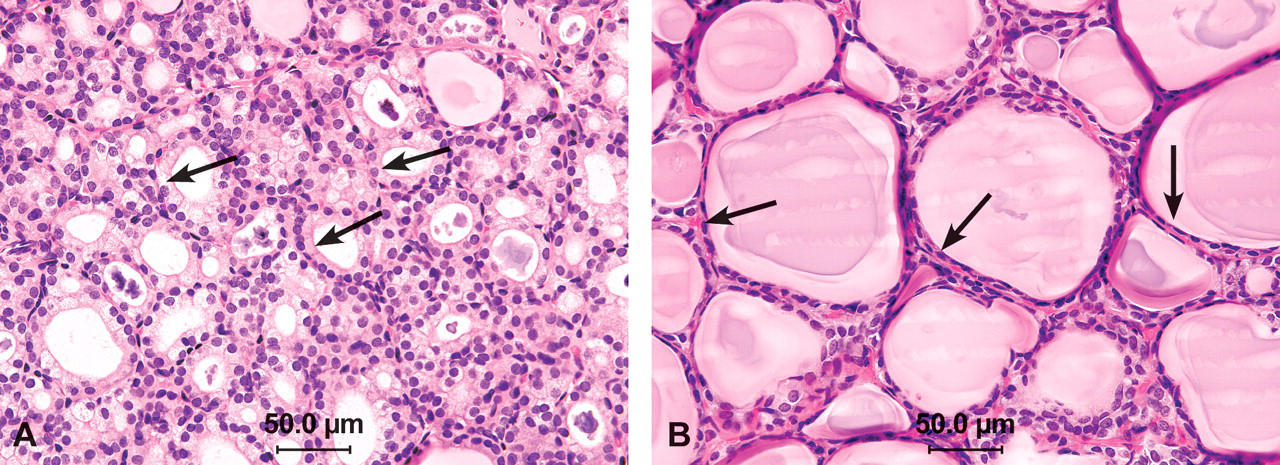
(A) Follicular hypertrophy in the thyroid in a female rat exposed to a high dose of a binary dioxin mixture (126/118) in feed for 31 weeks. The follicles are smaller in size, lined by cuboidal epithelium (arrows), containing decreased amounts of colloid in which aggregates of amphophilic, flocculant-appearing material are present (compare with B). Hematoxylin and eosin (H&E). (B) Normal aspect of thyroid follicles in a control female rat from the same dioxin mixture (126/118) 31 weeks. Note the follicles are distended with homogeneous colloid and the lining epithelium is flattened (arrows). H&E.
The incidences of follicular cell hypertrophy were generally higher in all TCDD-dosed groups at the interim time points compared with controls. Significant dose-related increases were observed at >22 ng/kg/day at 14 weeks, >46 ng/kg/day at 31 weeks, and at 100 ng/kg/day at 53 weeks. In animals treated for 2 years, the incidences of minimal follicular cell hypertrophy were significantly higher than controls in core study rats administered 46 and 100 ng/kg (Table 2). The incidence of this lesion was significantly lower in the 100-ng/kg stop-exposure group compared with the 100-ng/kg core study group.
In the PCB126 study, the incidences of follicular cell hypertrophy were generally higher in all dosed groups at the interim time points compared with controls (Table 2). Incidences were significantly higher than controls at 30, 300, and 550 ng/kg at 14 weeks and at 100 ng/kg and greater at 31 weeks. No significant differences were observed between PCB126-treated and control rats at 53 weeks. At 2 years, the incidences of follicular hypertrophy were significantly higher than controls in the 300- and 550-ng/kg core studies but not the highest dose group (1,000 ng/kg/day). The incidence in the stop-exposure group was significantly higher than in controls but not significantly different than the core study 1,000-ng/kg/day exposure group, which was exposed continuously throughout the study.
In the PeCDF study, at 14 weeks, there was a dose-related increase in the incidences of follicular cell hypertrophy (Table 2). The incidences were significantly higher than controls at 44 ng/kg and higher, with an increased severity observed at 92 and 200 ng/kg. No significant differences were observed between PeCDF-treated and control rats at 31 or 53 weeks. At 2 years, the incidences of follicular cell hypertrophy were higher in all dosed groups compared with controls; significant increases were observed at 20 ng/kg and higher. The incidence in the 200-ng/kg stop-exposure group was significantly higher than controls, and both the severity and the incidence were similar to the 200-ng/kg core study group.
In the TEF mixture study, the incidences of follicular cell hypertrophy were generally increased in all dosed groups at 14 weeks and significantly increased in groups administered 22 ng TEQ/kg or greater (Table 2). No significant differences were observed between treated and control rats at 31 or 53 weeks. At 2 years, the incidences of follicular cell hypertrophy were significantly increased at 10, 46, and 100 ng TEQ/kg. The incidences (12/51) were higher at 22 ng TEQ/kg than in controls (4/53) but were not statistically significant.
In the 2-year PCB153 study, the incidences of minimal to mild follicular cell hypertrophy were significantly higher than controls at 300 and 3,000 µg/kg and in the 3,000-µg/kg stop-exposure group (Table 3). No dose-related changes were observed in the incidences of follicular lesions at the interim time points. One follicular cell adenoma was observed at 53 weeks in the 100-µg/kg group, and 2 follicular cell adenomas were observed in the 3,000-µg/kg stop-exposure group at 2 years. Although the incidences of this lesion were not statistically different from controls, the incidence of this lesion in control animals from all 8 studies combined was 3/418.
In the binary mixture of PCB126 and PCB153, an increased incidence of follicular cell hypertrophy occurred in the constant ratio groups at 300/300 and 1,000/1,000 at 14 weeks and in all of the constant ratio treatment groups at 31 weeks (Table 3). At 53 weeks and 2 years, the incidences of follicular cell hypertrophy were significantly increased at 100/100 and above. In the varying ratio groups, which received a binary mixture of 300 ng/kg of PCB126 with varying concentrations of PCB153, the incidence of follicular cell hypertrophy was higher than in controls at 31 weeks, 53 weeks, and 2 years. The increase was generally dose dependent with respect to higher concentrations of PCB153 in the mixture.
In the PCB118 study, the incidences of minimal to mild follicular cell hypertrophy were significantly higher than controls at 4,600 µg/kg at 31 and 53 weeks (Table 3). No effects were observed at 14 weeks or at doses lower than 4,600 µg/kg at 31 and 53 weeks. In animals treated for 2 years, the incidences of minimal to mild follicular cell hypertrophy were significantly higher than controls in rats administered 460 ng/kg and higher.
In the binary mixture of PCB126 and PCB118 (PCB126/PCB118), no animals administered 216 or 360 ng/kg survived to the end of the study. Significant decreases in mean body weights for these groups were observed by 13 weeks. The incidences of follicular cell hypertrophy were significantly higher than controls in the 22-ng TEQ/kg and higher groups at 14 weeks and in the 216-ng TEQ/kg group at 31 weeks (Table 3). No significant differences were observed in the incidences of follicular cell hypertrophy at 53 weeks. At 2 years, the incidences of follicular cell hypertrophy were significantly increased in all dosed groups, including the stop-exposure group, which was similar to that in the 360-ng TEQ/kg core group.
Thyroid-related Hormones
Administration of all of the individual dioxin-like congeners and the mixtures was generally associated with a decrease in serum T4 levels, with the exception of PCB126 at the 31-week interim time point (Table 4 ). In those groups demonstrating significant decreases in serum T4 levels, there was a 22% to 46% decrease in the TCDD-treated groups, a 24% to 39% decrease in the PCB126-treated groups, a 16% to 40% decrease in the PeCDF groups, a 24% to 98% decrease in the PCB118 groups, and a 17% to 51% decrease in the TEF mixture groups compared with the respective interim evaluation controls (Tables 4 and 5 ). In the PCB126/PCB118 mixture study (Table 6 ), serum T4 levels were significantly decreased by 34% to 79% at doses ≤72 ng TEQ/kg compared with the respective controls for all of the interim evaluations. In the ≥216-ng TEQ/kg groups, serum T4 levels were severely decreased (86%-96%). These results likely reflect the poor health in these animals, as demonstrated by significant decreases in body weight gain throughout most of the study and the 100% mortality rate in these groups. In the PCB126/PCB153 mixture study, serum T4 levels were decreased 22% to 56% compared with the respective controls (Table 6). These lower serum concentrations were similar to those observed in the study in which PCB126 was administered alone. In the study of the non–dioxin-like PCB153, serum T4 levels were significantly decreased by 24% and 37% in the 3,000-µg/kg group compared with controls at 14 and 53 weeks, respectively. Serum T3 levels were also decreased by 30% in the 3,000-µg/kg group compared with controls at 14 weeks. No other significant treatment-related effects were observed in serum T4, T3, or TSH levels at any of the interim time points.
Serum concentrations of thyroid hormones in female rats at the 14-, 31-, and 53-week interim evaluations in the 2-year gavage study of TCDD, PCB126, and PeCDF.
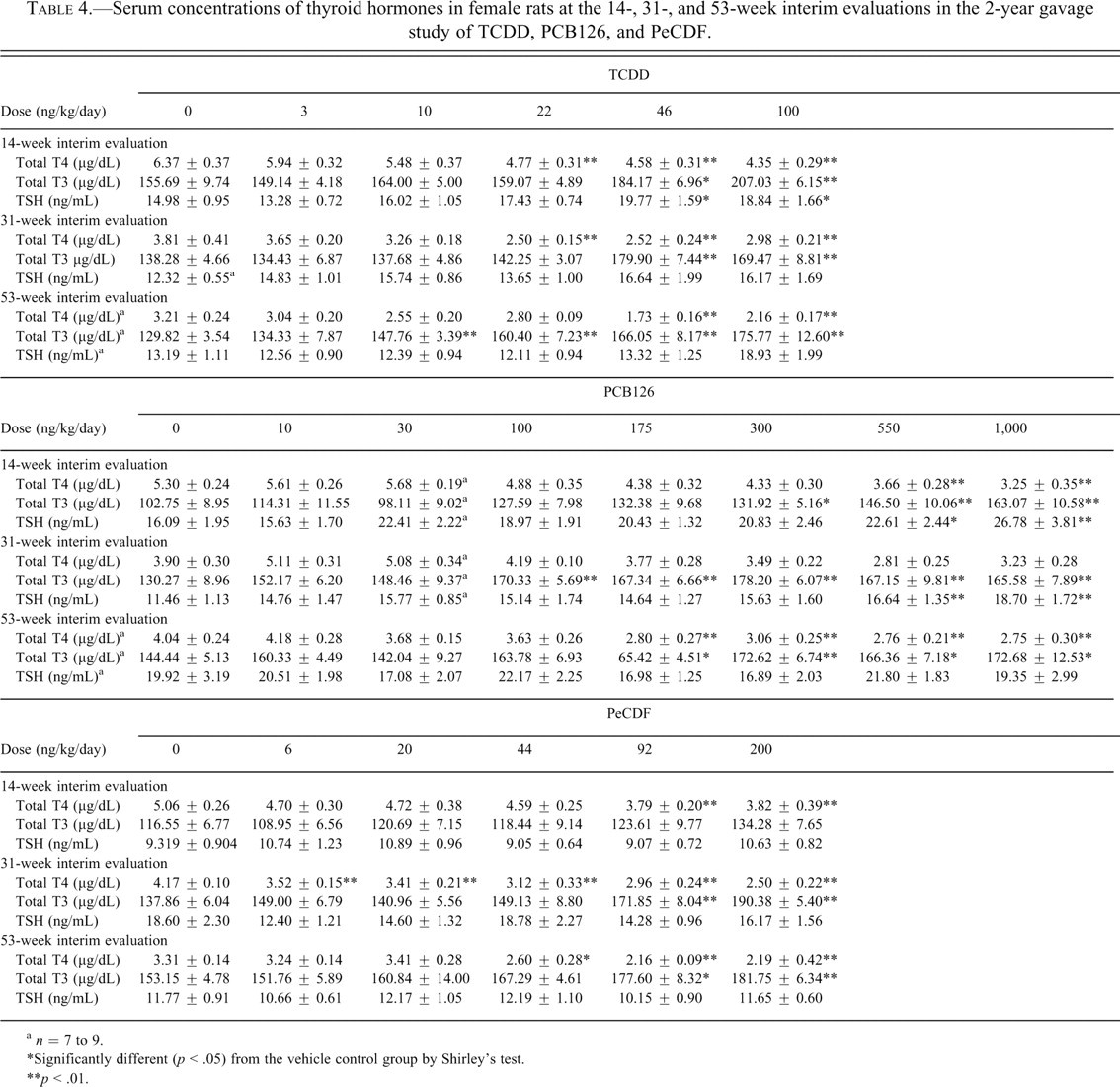
a n = 7 to 9.
*Significantly different (p < .05) from the vehicle control group by Shirley’s test.
**p < .01.
Serum concentrations of thyroid hormones in female rats at the 14-, 31-, and 53-week interim evaluations in the 2-year gavage study of the tertiary mixture of TCDD/PCB126/PeCDF, PCB153, and PCB118.
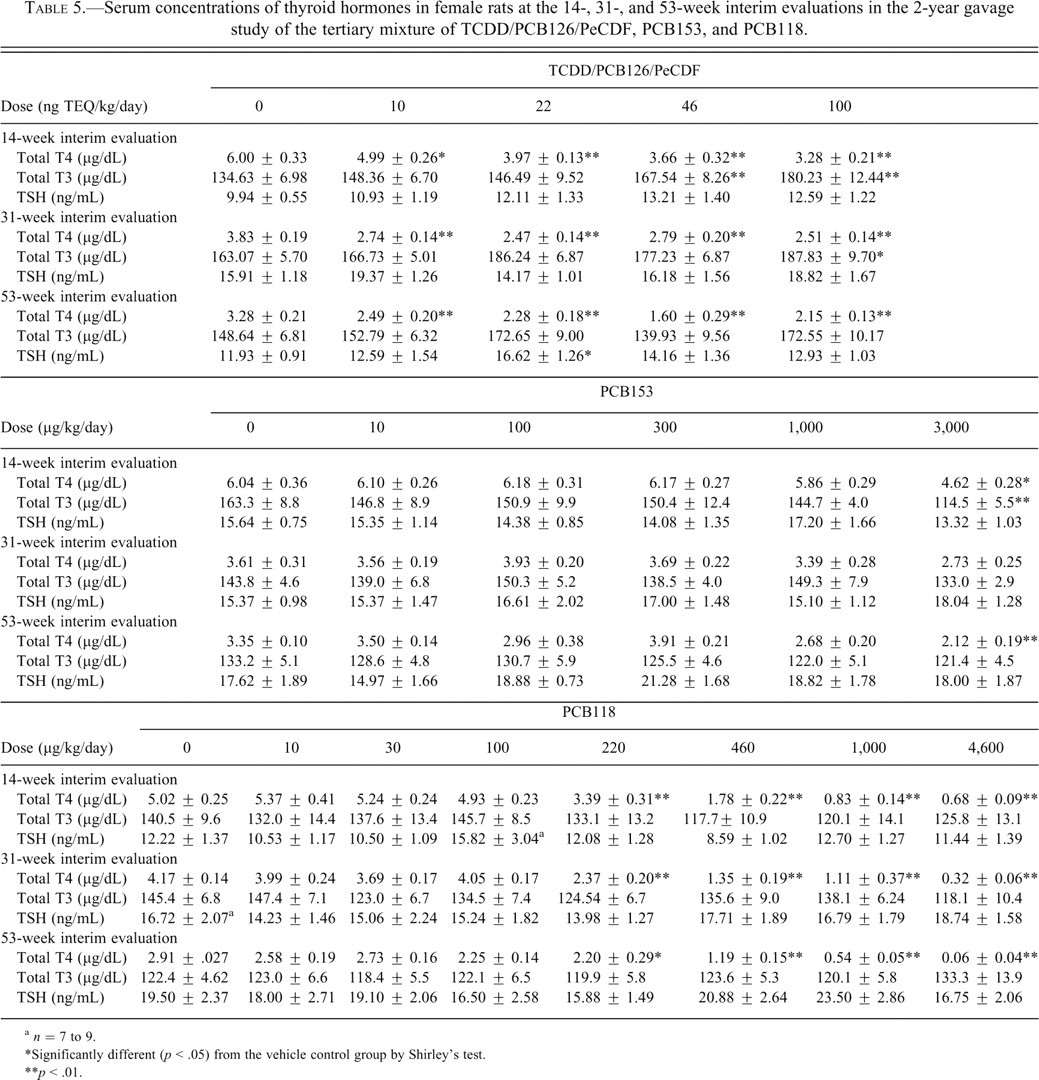
a n = 7 to 9.
*Significantly different (p < .05) from the vehicle control group by Shirley’s test.
**p < .01.
Serum concentrations of thyroid hormones in female rats at the 14-, 31-, and 53-week interim evaluations in the 2-year gavage study of PCB126/PCB153 and PCB126/PCB118.
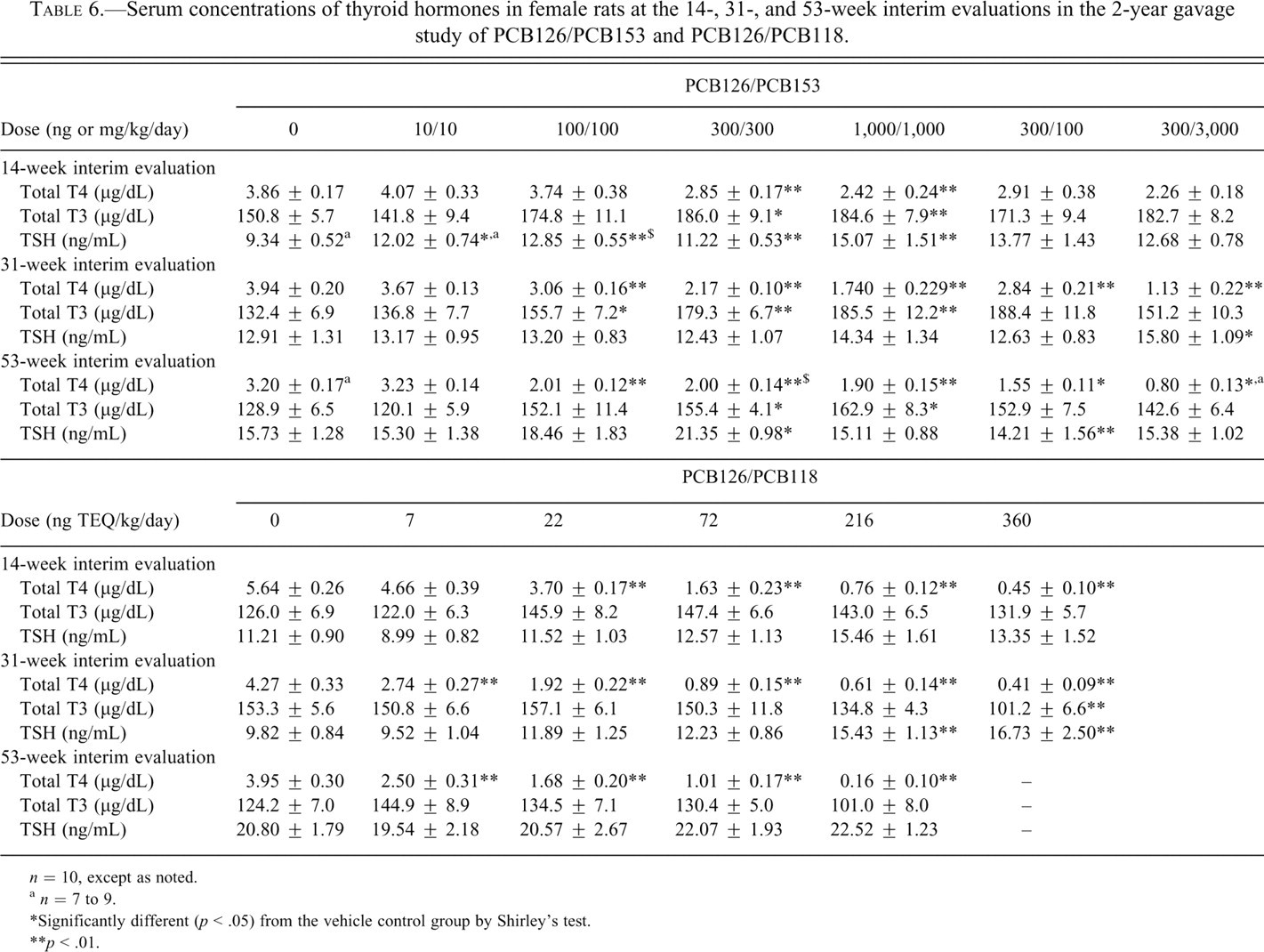
n = 10, except as noted.
a n = 7 to 9.
*Significantly different (p < .05) from the vehicle control group by Shirley’s test.
**p < .01.
Administration of the individual congeners and the mixtures was generally associated with an increase in serum T3 levels, with the exception of PCB153, PCB118 and PCB126/PCB118 mixture at any of the interim evaluations (Tables 4-6). In the groups demonstrating significant increases in serum T3 levels, there was a 14% to 35% increase in the TCDD-treated groups, a 15% to 59% increase in the PCB126-treated groups, a 16% to 38% increase in the PeCDF groups, and a 15% to 34% increase in the TEF mixture groups compared with the respective interim evaluation controls. In the PCB126/PCB153 mixture study, serum T3 levels were increased 18% to 40% compared with the respective controls for all of the time points. These levels were similar to those observed in the study with PCB126 alone.
Significant treatment-related effects in serum TSH levels were observed only in the TCDD, PCB126, and PCB126/PCB153 studies (Tables 4 and 6). In the TCDD study, serum TSH levels were significantly higher at 14 weeks in the 46- and 100-ng/kg groups. No differences were observed between control and treated groups at 31 or 53 weeks. In the PCB126 study, serum TSH levels were significantly higher in the 550- and 1,000-ng/kg groups (which correspond to 55 and 100 ng/kg on a TEQ basis) at 14 and 31 weeks compared with controls. Serum TSH levels were significantly higher in all groups treated with a mixture of PCB126/PCB153 at 14 weeks. No treatment-related effects were observed in TSH at 31 or 53 weeks, except for a statistically significant increase in the 300/300 PCB126/PCB153 group.
Discussion
Serum TSH levels were transiently elevated in the TCDD, PCB126, and PCB126/PCB153 studies, but these alterations were observed primarily at 14 weeks, and there was no treatment-related effect in the other studies. Comparison between the four studies of individual dioxin-like congeners and the tertiary TCDD/PCB126/PeCDF mixture suggests that there was no specific threshold TEQ-adjusted dose that was consistently associated with alterations in serum thyroid hormones. Similar to the DLCs, administration of the non–dioxin-like PCB153 induced follicular cell hypertrophy and decreased T4 both alone and in combination with PCB126. However, PCB153 decreased serum T3 levels and dose dependently increased TSH when administered in combination with PCB126.
There were differences between all of the congeners and the mixtures with respect to the TEQ-adjusted doses in each study that induced follicular cell hypertrophy at the interim time points. The underlying premise behind the use of TEQ-adjusted doses is to be able to compare exposures to different dioxin-like congeners or mixtures normalized based on the relative potencies of the compounds with respect to TCDD, the most potent congener. Therefore, similar TEQ-adjusted doses would be expected to result in similar effects regardless of the dioxin-like congener or the components of a mixture of congeners. In the TCDD study, TCDD-induced increases in the incidence of follicular cell hypertrophy were observed at all time points, but higher doses were required to induce this effect with longer exposures, such that significant increases were observed at ≥22 ng/kg/day at 14 weeks, ≥46 ng/kg/day at 31 weeks, and only in the 100-ng/kg/day group at 53 weeks. The PeCDF and the tertiary mixture (TCDD/PCB126/PeCDF) studies were consistent with the TCDD study in that the increased incidences of follicular cell hypertrophy were observed at ≥44 ng PeCDF/kg/day (22 ng/kg/day on a TEQ-adjusted basis) and ≥22 ng TEQ/kg/day, respectively. However, no treatment-related increases were observed in these lesions at 31 or 53 weeks in these studies as were observed for TCDD. PCB118 induced follicular cell hypertrophy at 31 and 53 weeks; however, this occurred at 4,600 µg/kg/day (equivalent to 460 ng/kg/day on a TEQ-adjusted basis), which exceeds doses at which this was observed in the TCDD study. Although the induction of follicular cell hypertrophy was not strictly a function of TEQ-adjusted dose and duration of exposure, the general trend was similar between all of the studies of the individual congeners, mixtures of DLCs, and the mixture of PCB126 and PCB153.
At the end of the 2-year study, treatment-related increases in the incidences of thyroid follicular cell hypertrophy were observed in all of the studies, including the non–dioxin-like PCB153. Similar to the observations at the interim time points, there was not a consistent TEQ-adjusted dose between the studies that induced follicular cell hypertrophy at 2 years. TCDD-induced increases were observed at ≥46 ng/kg/day. Similarly, PCB118 induced incidences at ≥460 µg/kg, which is equivalent to ≥46 ng/kg on a TEQ-adjusted basis. For all of the other studies, significant treatment-related increases were observed at lower TEQ-adjusted doses. These increases occurred at 7 ng TEQ/kg for PCB126/PCB118; 10 ng TEQ/kg for PeCDF, the tertiary mixture, and the PCB126/PCB153 mixture; and 30 ng TEQ/kg for PCB126. PCB153 also induced increases in the incidences of this lesion at the end of the 2-year study, but it cannot be compared on a TEQ basis since as a non–dioxin-like compound, it is not included in the TEF methodology.
The effect of stop-exposures on the incidence of follicular cell hypertrophy differed between the individual congeners. In the TCDD study, the incidence of follicular cell hypertrophy in the 100-ng/kg stop-exposure group was significantly lower than the incidence in the 100-ng/kg group receiving TCDD continuously for the duration of the 2-year study. These results suggest that TCDD-induced development of thyroid follicular cell hypertrophy is a reversible effect. The effect of terminating exposure in the PCB126, PeCDF, and non–dioxin-like PCB153 were not consistent with the effects observed for TCDD. In these studies, the incidences of follicular cell hypertrophy were significantly higher than 2-year controls and not significantly different from the groups receiving the same doses for the duration of the study without cessation. These results suggest that the induction of these lesions by the other congeners is not reversible. The congener-specific differences and the mechanistic basis for these findings are not clear.
In this series of studies, administration of individual dioxin-like congeners and various mixtures of DLCs was generally associated with decreased serum T4 levels, increased serum T3 levels (Table 7 ), and increased incidences of thyroid follicular cell hypertrophy. TCDD induces thyroid gland follicular cell adenomas in female B6C3F1 mice and male Osborne-Mendel rats and elicits a nonsignificant increase in the incidence of follicular cell adenomas in female Osborne-Mendel rats (Huff et al. 1991; Mann 1997; NTP 1982). The proposed mechanism for the development of thyroid neoplasms by TCDD and other hepatic microsomal enzyme inducers involves the disruption of thyroid hormone homeostasis via alteration of T4 metabolism, leading to increased serum TSH levels. TCDD induces hepatic UDP-glucuronosyltransferase, a phase II microsomal enzyme that increases clearance of T4 through hepatic glucuronidation and consequently decreases serum T4. Alteration of serum T4 levels increases serum TSH levels by decreasing the negative feedback inhibition and disrupting the hypothalamic-pituitary-thyroid axis (Potter et al. 1983, 1986; Sewall et al. 1995; Van Birgelen et al. 1995). In general, overstimulation of the thyroid gland by TSH is thought to play a major role in the mechanism of follicular cell carcinogenesis (Hill et al. 1989). Chronic hyperstimulation of the thyroid follicular cells contributes to the development of follicular cell hypertrophy, which is considered reactive activation secondary to the increased levels of TSH. Persistent follicular cell hypertrophy can result in follicular cell proliferation and progression to follicular cell hyperplasia, and it ultimately leads to the development of follicular cell adenomas. Kohn et al. (1996, 2000) developed a mathematical model for the effects of TCDD on UDP-glucuronosyltransferase expression and thyroid hormone homeostasis that is consistent with this mechanism.
Summary of thyroid function and induction of follicular hypertrophy.
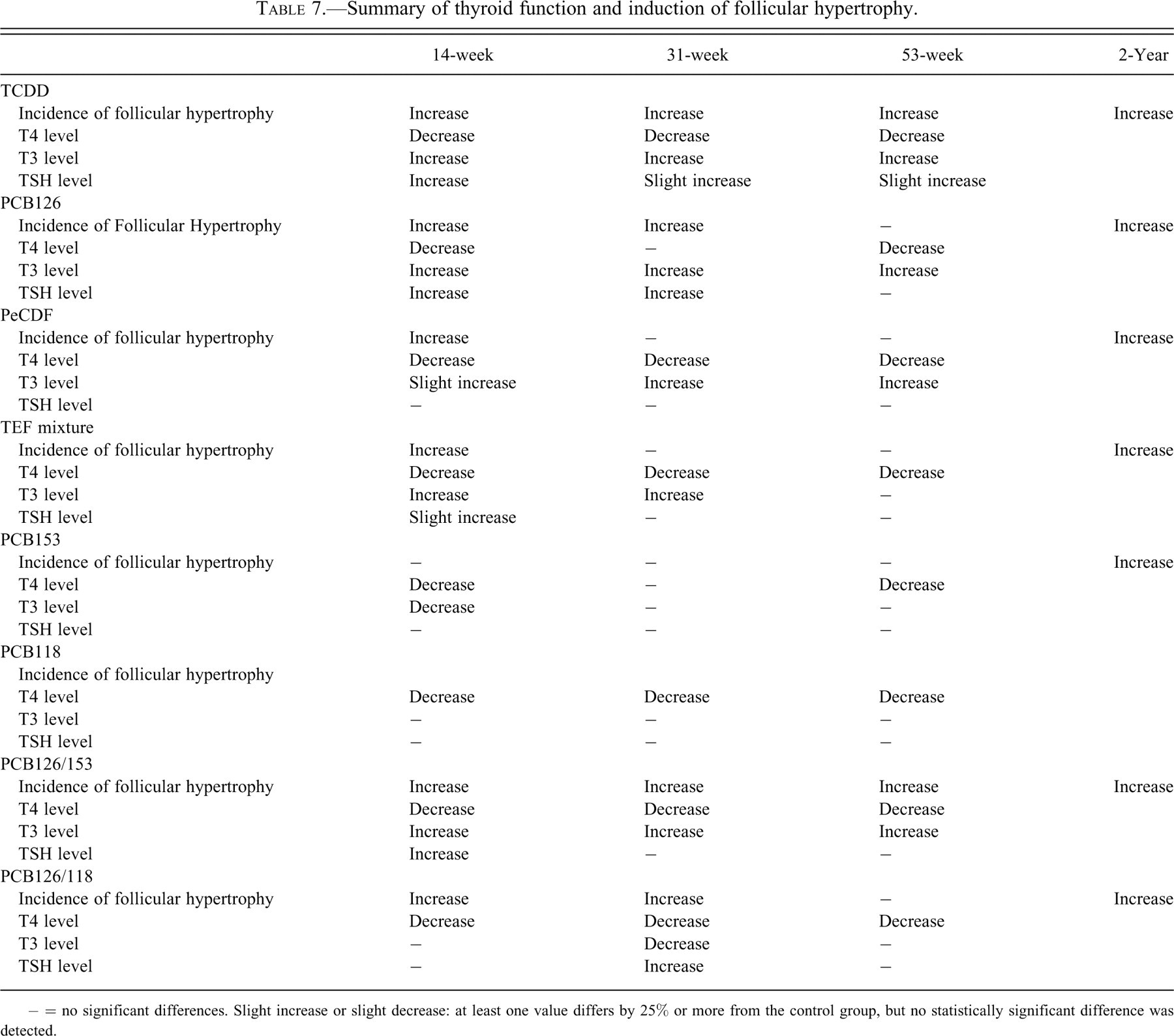
− = no significant differences. Slight increase or slight decrease: at least one value differs by 25% or more from the control group, but no statistically significant difference was detected.
The induction of follicular cell hypertrophy by the DLCs in the current studies is consistent with this mechanism and previously observed TCDD-induced increased incidences of follicular cell hypertrophy (Nishimura et al. 2002, 2003; Sewall et al. 1995). Although altered thyroid hormones and increased incidences of follicular cell hypertrophy were consistent with the proposed mechanism for TCDD-induced thyroid neoplasia, there were no follicular cell adenomas or carcinomas observed in any of the studies of the dioxin-like congeners or any of the mixtures. It is possible, therefore, that the lack of follicular cell neoplasia in this study reflects a lack of a sustained long-term increase on TSH sufficient to promote neoplasia.
Administration of Aroclors 1242, 1254, and 1260 (complex mixtures of PCBs) induce slight, but not dose-dependent, increases in the incidence of follicular cell adenomas in male SD rats, but not females (Mayes et al. 1998). These results differ from the previous NTP study of TCDD in Osborne-Mendel rats (NTP 1982) in that the nonneoplastic hypertrophy in the current studies did not progress to follicular cell adenomas. The differences between these studies might result from some differing protocol parameters such as vehicle, dosing schedule, diet, and the biological behavior of rat strain used (Walker et al. 2006).
The relative resistance to the development of thyroid cancer in humans with elevated serum TSH levels is in marked contrast to the response of the thyroid gland to chronic TSH stimulation in rodents. Effects of TCDD and DLCs on the thyroid were reported, such as alterations in TSH, T3, or/and T4 in various exposed populations, including veterans of the Vietnam War, children, and infants (Arisawa et al. 2005; Pavuk et al. 2003; Zober et al. 1994, 1997/1998). With respect to thyroid diseases by TCDD, an increased prevalence of combined thyroid disorders (goiter, thyrotoxicosis, hypothyroidism, and thyroid adenoma) was found in German workers. An increase in thyroid tumors in humans was observed in a large cohort of workers exposed to chlorophenoxy herbicides, including those contaminated with TCDD (Saracci et al. 1991). A possible association between exposure to dioxin and thyroid tumors has been suggested by epidemiological research for the industrial accident in Seveso (Pesatori et al. 1993, 2003). In general, the human thyroid is much less sensitive to this pathogenic phenomenon than rodents (Capen 1992, 1999; Stevens 1997). The differences in plasma half-life of thyroid hormones and binding to transport proteins between rats and humans may be one factor in the greater sensitivity of the rat thyroid in response to chronic TSH stimulation.
It can be concluded from the data that the DLCs evaluated in these studies transiently increased the levels of TSH accompanied with decreased levels of T4 and hypertrophy of the thyroid follicular epithelium. Interestingly, the continued exposure to each of the compounds did not lead to a progression from follicular cell hypertrophy to hyperplasia. These data suggest that, generally, even prolonged exposure did not severely affect the pituitary-thyroid axis related to thyroid carcinogenesis.
Footnotes
Acknowledgments
The authors thank all involved in the design and conduct of these NTP studies, with special appreciation expressed to Drs. John Bucher, Rick Hailey, Angelique Braen, and Milton Hejtmancik and Ms. Denise Orzech. We gratefully acknowledge Dr. Bhanu Singh, Dr. Rick Irwin, and Ms. JoAnne Johnson for critical review of the article. The authors declare that they have no competing financial interests.
