Abstract
Tissue cross-reactivity (TCR) studies are screening assays recommended for antibody and antibody-like molecules that contain a complementarity-determining region (CDR), primarily to identify off-target binding and, secondarily, to identify sites of on-target binding that were not previously identified. At the present time, TCR studies involve the ex vivo immunohistochemical (IHC) staining of a panel of frozen tissues from humans and animals, are conducted prior to dosing humans, and results are filed with the initial IND/CTA to support first-in-human clinical trials. In some cases, a robust TCR assay cannot be developed, and in these cases the lack of a TCR assay should not prevent a program from moving forward. The TCR assay by itself has variable correlation with toxicity or efficacy. Therefore, any findings of interest should be further evaluated and interpreted in the context of the overall pharmacology and safety assessment data package. TCR studies are generally not recommended for surrogate molecules or for comparability assessments in the context of manufacturing/cell line changes. Overall, the design, implementation, and interpretation of TCR studies should follow a case-by-case approach.
Keywords
Introduction
Tissue cross-reactivity (TCR) studies are screening assays conducted with monoclonal antibodies and related antibody-like biopharmaceuticals primarily to identify off-target binding and, secondarily, to identify sites of on-target binding that were not previously identified. As presently utilized by the biopharmaceutical industry and regulatory agencies, TCR studies usually involve the ex vivo immunohistochemical (IHC) staining of a panel of frozen tissues from humans and animals. However, other methods of conducting TCR studies are possible. Data from ex vivo TCR studies are used to alert preclinical and clinical investigators toward potential target organs of toxicity based on the presence of staining in tissues. In addition, a comparison of the ex vivo patterns of staining between human and animal tissue panels may also be used to support the relevance of species evaluated in preclinical toxicity studies. TCR studies filed with the initial IND/CTA (investigational new drug application/clinical trial application) to support first-in-human (FIH) clinical trials have generally been performed in compliance with U.S. Food and Drug Administration (FDA) 21 Code of Federal Regulations (CFR) Part 58, European, and/or Japanese good laboratory practices (GLP) standards.
While ex vivo TCR studies have become routine in the development of antibody therapeutics, their value for the purpose of making safety assessment decisions by both industry and regulatory agencies has been questioned as experience with the assay has increased. This article provides both historical and contemporary perspectives regarding the TCR study as a component of the preclinical safety assessment of candidate antibody and antibody-like molecules. Drawn from a broad base of expertise, a detailed analysis of the technical aspects that affect implementation and interpretation of these studies is presented, and specific examples are provided. This article seeks to establish best practices for the conduct, interpretation, and use of TCR data in the discovery and development of antibodies and antibody-like products. These recommendations are intended to be specific enough to be useful and informative but flexible enough to allow for a case-by-case science-based approach to drug development, as well as to accommodate the potential impact of future innovations.
What Are TCR Studies?
TCR studies are screening assays designed to identify binding (i.e., reactivity) of the test article (i.e., antibody or antibody-like protein) in tissues (note—the term screening is used in this article to denote an assay whose data should be interpreted in the context of additional studies). Binding to the expected target is a desired outcome, and sometimes these assays will identify previously unknown sites of the target antigen. In some cases, TCR studies also identify binding to unexpected targets (i.e., cross-reactive epitopes), hence the term cross-reactivity. From a practical standpoint, it is usually not known based on TCR data alone whether binding at unexpected sites is related to cross-reactivity, to the presence of the target antigen in unexpected cells/tissues, to nonspecific binding, or to an assay artifact. As a result of these limitations, additional analyses are sometimes needed to further characterize the nature and/or specificity of the unexpected staining. While TCR studies are most frequently conducted using IHC, in which binding is detected by the presence of staining, other methods (such as tissue-based lysate western blots, flow cytometry with cultured cells/cell lines or peripheral blood samples) may also provide similar information. Increasingly, in silico modeling may be used to understand the potential for cross-reactivity and to predict possible antibody:epitope interactions. When using IHC, the standard study design applies the test antibody ex vivo to a panel of frozen tissues from humans and animal species considered or selected for toxicity evaluation to determine whether the same or related antigenic determinants are expressed on cellular or tissue elements other than the intended target and whether the tissue and cellular distribution of staining is similar in humans and animals. The patterns and distribution of staining (e.g., cell types and subcellular or extracellular localization) have been used to guide preclinical and clinical investigators toward potential target organs of toxicity. The intensity and frequency of staining are also evaluated, as increased intensity and frequency have been associated in some cases with greater in vivo toxicity. However, the true relevance of the staining pattern and intensity can only be determined retrospectively after toxicity and/or clinical data become available. TCR studies filed with the initial IND/CTA-supporting FIH clinical trials for essentially all antibodies and antibody-like products have generally been performed in a GLP-compliant manner.
Scientific, Technical, Regulatory, and Strategic Considerations: A Historical Perspective
An awareness of the history of TCR studies is important to understand how regulatory agencies and industry currently view these studies and to understand recommendations for the future. TCR studies have evolved over time, with technical, regulatory, and strategic considerations shaping the conduct, interpretation, and use of these studies.
Between the early 1980s, when development of mAb products began in earnest, and the present time, the technical feasibility of TCR studies has become increasingly complex and challenging. In the early 1980s, the first antibodies in development were mouse monoclonal antibodies with structures similar to endogenous molecules. The mouse origin and intact structure of these early products made detection of targets in human, nonhuman primate, and rat tissues relatively easy because a specific secondary anti-mouse antibody could be used to detect binding of the test article (i.e., the primary antibody) to the target or cross-reactive epitopes without interference by reactivity with endogenous human, non human primate, or rat IgG. As immunogenicity concerns related to the mouse origin of products became apparent and technology evolved, the antibody products became chimeric, then humanized, and finally fully human. While making the antibody products less prone to result in immunogenicity in humans, this also had the effect of making detection of the test article more difficult. This is because the anti-human secondary antibodies did not distinguish between the humanized or human antibody product used in relatively low concentrations in an IHC assay and the endogenous antibodies naturally present in much higher concentrations in human tissues. In addition to incremental humanization over time, many antibody-related products have emerged by modifying a naturally occurring structure by genetic engineering, for example, a receptor-Fc fusion protein. In these cases, the typically anti-human secondary antibodies available commercially might not recognize the new structures. Lastly, while the binding affinity of therapeutic antibodies has been increased to promote potential efficacy in the clinical setting, better affinity of the test article does not necessarily translate to better sensitivity as an IHC reagent. While low-affinity test articles sometimes are not good IHC reagents, medium or high affinity does not predict success in the assay, and in fact the increased binding affinity may broaden cross-reactive binding in IHC and other assays (Wu et al. 2007). For these reasons, TCR assays have become more difficult to develop.
In addition to changing technical considerations, regulatory recommendations and strategic considerations have also evolved, often together. Certainly the regulatory positions and use of TCR studies as described in guidance documents have been primary drivers in the strategic use of these studies by industry. The first guidance document addressing TCR studies from a regulatory standpoint was the “Points to Consider in the Manufacture and Testing of Monoclonal Antibody Products for Human Use” (points to consider [PTC] document), finalized by the FDA in 1983. This guidance was subsequently updated in 1987, 1994, and 1997.
The 1983 PTC document recommended that “an immunohistological survey of human vital organs, blood components and intended target cells or tissues should be carried out using both quick frozen and chemically fixed adult and, if available, fetal tissue samples.” In addition to immunohistochemical (IHC) methods of assessing TCR, the PTC document also indicated other methods could be used on blood or cultured cells, including microcytotoxicity, fluorescent antibody methods, and/or autoradiography. It was also recommended that tissues from unrelated humans should be tested, that staining should be quantified in some manner, and that several dilutions of the test article should be evaluated. If cross-reactivity was not identified in human tissues, it was not expected that animal tissues would be evaluated in TCR studies. However, in the event that cross-reactivity was identified, the 1983 PTC document recommended that attempts be made to identify an animal model or system with similar reactivity, and to justify whether toxicity testing needed to be conducted in nonhuman primates or could be conducted in other species. Ex vivo evaluations or in vivo studies could then be conducted in the selected animal species to determine any consequences of the cross-reactivity. Finally, in the event cross-reactivity was identified and there was no animal model, it was recommended that extensive histopathology evaluations and thorough distribution studies be conducted in humans, although the recommended methods were not described.
In 1987, the second version of the PTC document was released. Differences from the 1983 version related to TCR studies were generally minimal. The 1987 document no longer recommended that studies be done on chemically fixed tissues, and instead only quick-frozen tissues were suggested for testing. The use of colony-forming assays to detect cross-reactivity in blood or cultured cells was added. The 1987 document also recommended that a quantitative comparison of the binding between the expected target and the cross-reactive cells or tissues be conducted. The suggestion that cross-reactivity be evaluated in humans using extensive histopathology evaluations if there was no animal model was removed.
In 1994, the third version of the PTC document was released. The section related to TCR studies was rewritten. The document noted that TCR studies at that time were being conducted on human cells or tissues using immunocytochemical or IHC methods but that appropriate newer technologies should be employed as they become available. This version of the PTC document specifically indicated that TCR studies “should always be conducted prior to Phase 1” human studies. In addition, because a number of antibodies with modified structures were entering development, the document indicated the actual clinical material should be tested, and not a surrogate product. Increased concern was noted for pharmacologically active antibodies, or those antibodies that were conjugated to cytotoxins. For bispecific antibodies, each parent antibody as well as the bispecific product was to be tested. The number of unrelated human donors that should be tested was specified as at least three. The concept of considering antibody affinities and expected peak plasma concentrations in the selection of antibody concentrations for TCR studies was introduced, as was the use of inhibition with purified antigen to assess the specificity of potential cross-reactivity. Positive and negative controls were considered necessary in TCR studies for interpreting study results. Anti-transferrin receptor was noted to be a useful control to assess overall tissue and cell integrity, as transferrin is an abundant molecule on the surface of cells and its loss would suggest damage to antigens in the tissue, rendering that tissue unsuitable for TCR studies. Interestingly, the 1994 PTC document commented that if TCR studies on human tissues demonstrated no cross-reactivity, and there was no animal model of disease activity or animal that expressed the relevant antigen, then in vivo toxicity testing in animals could be limited to the General Safety Test for an unconjugated antibody. The 1994 PTC document also noted, in the section on pharmacokinetics and distribution, that mAbs should be tested whenever possible in an animal model in a species that shares a cross-reactive or identical target antigen with humans.
Lastly, and perhaps most significantly, the 1994 PTC document broadened the use of TCR studies to recommend the routine testing of animal tissues from different species to determine the most relevant animal for in vivo toxicity studies. This addition was driven in part by concerns about the appropriate use of animals for in vivo preclinical toxicity studies. As more antibodies and other biopharmaceuticals entered development in the late 1980s and early 1990s, it became apparent that many companies were simply defaulting to the use of nonhuman primates for preclinical toxicity studies without adequate scientific justification. In some circumstances, the target and biological activity might occur in a range of species beyond nonhuman primates, including rodents. Thus, in vivo toxicity studies might be more appropriately conducted in species other than monkeys. In some cases, the target and biological activity were not present in any animal species. This situation could arise if the target was only present in humans, or the target was not expressed in normal humans or animals (such as might occur if the target was a bacterial, viral, or fungal antigen; or if the target was only expressed in the disease state, including tumor-specific antigens). Thus, the recommendation that ex vivo TCR studies should be used in determining a relevant animal species for subsequent in vivo toxicity studies by comparison of the staining patterns across species was added to the 1994 PTC document. A relevant animal species, in the context of TCR studies, was one in which there was similar staining in human and animal tissues. This staining might be related to binding to the target, binding to a cross-reactive epitope (i.e., off-target binding), or a combination of target and off-target binding, and the ideal preclinical species would have staining localization and intensity that matched that observed in human tissues.
In 1997, the fourth and most current version of the PTC document was released. This version included minor changes from the 1994 version. For the first time, an “ideal” concentration of the test article was suggested as the lowest mAb concentration that produces maximum (plateau) binding to the target antigen, although testing more than one concentration was still recommended. The inhibition of staining with purified antigen, which had been previously recommended, was clarified as a possible method to distinguish nonspecific as well as Fc-mediated binding versus specific binding to the target epitope via the complementarity-determining region (CDR). Finally, the 1997 PTC document provided an update addressing those programs in which TCR studies in human tissues demonstrated no cross-reactivity and there was no animal model of disease activity or animal that expressed the relevant antigen. In these cases, it was suggested that in vivo toxicity testing in animals might not be necessary for an unconjugated antibody. This was in contrast to the 1994 document that suggested a General Safety Test might be needed.
ICH S6, also released in 1997, commented briefly on TCR studies, noting that they should be carried out by appropriate IHC procedures and should test a range of human tissues. Further technical details were not included in ICH S6. However, at a more strategic level, the importance of TCR studies in the selection of relevant species for toxicity testing was mentioned. According to ICH S6, “A relevant species is one in which the test material is pharmacologically active due to the expression of the receptor or an epitope (in the case of monoclonal antibodies).” Furthermore, ICH S6 stated that “a variety of techniques (e.g., immunochemical or functional tests) can be used to identify a relevant species.” In cases where no animal species expressed the desired epitope, ICH S6 suggested toxicity testing might still be warranted if unintentional tissue cross-reactivity similar to that seen in human tissues was demonstrated in the animal tissues. In 2007, the European Medicines Agency (EMEA) finalized the “Guideline on Strategies to Identify and Mitigate Risks for First-In-Human Clinical Trials with Investigational Medicinal Products,” which also indicated that TCR data may be considered in the selection of relevant animal species for toxicity testing. However, the same document indicated that other tests may also be considered in selecting a relevant species, including target expression, distribution and primary structure, pharmacodynamic endpoints such as binding and occupancy, functional consequences, cell signaling, and pharmacokinetics and metabolism.
To summarize regulatory and strategic considerations, there have been four versions of the PTC document, one document from ICH, and one document from the EMEA. These documents have clearly highlighted the relevance and importance of TCR data, while acknowledging that other methods could be developed and used in the future. In fact, data from ex vivo TCR studies could potentially be used to argue against the need to conduct any preclinical in vivo toxicity studies in certain circumstances.
Current Status
Antibody and antibody-based products have been developed for more than twenty-five years, and it has now been more than ten years since the 1997 PTC and ICH S6 were published. Over this period, the scope of TCR studies has increased, and in the past decade since the last publication of regulatory guidances, the use of these studies has expanded (Hall et al. 2008). In the absence of other data, broad TCR screens testing multiple species have been used by some as one approach to identify pharmacologically or toxicologically relevant preclinical species for toxicity testing. In addition, TCR studies have been conducted on novel protein structures that do not contain a CDR with questionable scientific rationale. At the same time, the technical feasibility of conducting TCR studies has in many cases become more challenging. The overall result is that concerns have been raised that regulatory agencies are requiring, and industry is conducting, ex vivo TCR studies that are generating data of questionable value that can then confound rational development strategies. In addition, various regulatory agencies have at times requested sponsors to conduct TCR studies for other purposes, such as comparability assessments related to manufacturing changes. As this use of TCR studies is not recommended or discussed in detail in current regulatory guidances, it is inconsistently applied.
As a result of these concerns, several initiatives were undertaken by industry. These included the conduct of an industry survey regarding TCR practices (which will be reported separately; Bussiere et al. n.d.) and the formation of this TCR working group. As a product of this working group, this article critically reexamines the TCR study and its role in supporting the clinical development of antibody and antibody-based products. This includes a discussion of the scientific and technical aspects of TCR studies, provides case examples, and makes suggestions and recommendations for the appropriate use of TCR studies based on the collective data obtained from more than twenty-five years of experience with hundreds of products. As was evident during the preparation of this article, the experience with TCR studies varied between individual scientists and companies, which may reflect the different test articles and targets evaluated by each person and company. Nevertheless, the suggestions and recommendations in this article represent a consensus of opinion based on the collective experience of the authors. Importantly, the broad spectrum of experiences highlights the overall need for a case-by-case approach to the application and interpretation of TCR studies in drug development.
Scientific and Technical Aspects of TCR Studies
Details of the technical methodology of conducting TCR studies using IHC are presented in the appendix. Information on the interaction between antibodies and their epitopes as they relate to TCR studies using IHC is presented in the sections below.
Science of Binding—Antibody CDR/Epitope Interactions
It is critical to understand the science behind antibody, target epitope, and potential off-target (cross-reactive) antigen interactions to interpret TCR data and to understand the relevance of TCR findings to the development program. The parts of the heavy and light chains of antibodies in closest contact with antigen are the CDRs; the CDRs interact with a specific region on the antigen called the epitope. Epitopes can be contiguous (i.e., linear, sequence-dependent), as in a consecutive sequence of adjacent amino acids, or can be discontiguous (i.e., conformational), with the epitope appearing only when the protein is folded into the correct secondary or tertiary structure. Each antibody heavy and light chain has three CDRs, which together make a single cleft to form the antibody-binding site. Antibodies bind their specific antigens using hydrogen bonds, hydrophobic interactions, Van der Waals interactions, and ionic bonds, but perhaps the most important is the physical structure that allows the hydrophobic portions of the CDRs to line up with the hydrophobic portions of the epitope. Binding may be dynamic, with both the CDRs and epitope adjusting configuration or deforming to provide better interactions; some small soluble antigens may become completely surrounded by the CDRs. In recent years, water associated with the epitope or CDRs has been recognized as an increasingly important aspect of affinity, as it serves to fill and cement gaps between CDRs and epitopes. Less is known about interactions with nonprotein epitopes, but similar interactions are likely for other types of molecules.
Interaction of the antibody and its epitope in tissue sections during a TCR assay may be different than in vivo. Several processes inherent in the tissue preparation for the TCR assays can alter the tissue target or tissue matrices or can create new chemical structures not found in vivo. Three such treatments include drying, fixation, and endogenous peroxidase inactivation. Simply drying tissue onto slides can drive water out of a target epitope and matrix and can impart rigidity to the tissue, thereby denaturing or immobilizing the epitope so it can no longer achieve a conformation suitable for CDR binding or restricting access of the antibody to the target (Emoto, Yamashita and Okada 2005a; Yamashita and Okada 2005b; Metz et al. 2004; Kakimoto et al. 2008). The effects of fixatives in tissue are discussed in detail below, but regardless of whether the fixatives are organic solvents that fix by desiccation or aldehydes that cross link, all fixation alters tissue chemistry. Similar to drying, fixation may alter protein folding or lock dynamic (mobile) epitopes into a rigid configuration that limits steric access to the epitope or fixes adjacent molecules over the target (Emoto, Yamashita and Okada 2005a; Yamashita and Okada 2005b; Metz et al. 2004, 2006). Solvents can wash out lipids in membranes; the removed lipids may include unattached protein islands containing cell surface receptors. The process of inactivating endogenous peroxides involves exposure of the tissue to oxidizing agents that can destroy labile epitopes. Other epitopes may be very short-lived and may degrade quickly before tissue collection or with time.
Not all tissue treatment has negative effects on every epitope; fixation seems to stabilize some epitopes and improve binding. Judicious selection of fixatives described below and good practices such as performing peroxidase inactivation after antibody binding can minimize the effects of some tissue treatments on epitope structure. Despite the best care, loss of binding to tissue related to epitope stability or processes inherent in the TCR assay is probably second only to low epitope numbers as the leading cause of failure of the TCR to detect a known tissue target. In the experience of the authors, creation of new structures may also occur with tissue processing, leading to the potential for artifactual binding.
The antigen exposure in a TCR assay does not mimic the exposure that would occur in vivo. Freezing, cutting, and fixation of the tissues disrupt cells and expose intracellular epitopes that are not normally accessible in vivo. Thus, the antibody has equal access to all tissues and cell components (membrane, cytosol, nucleus) on the tissue sections in TCR studies. This is not true in vivo, where access to the tissue may be governed by passive diffusion of the antibody into the tissue. In addition, unless there is receptor-mediated transport, cell membranes usually preclude entrance of the test article into cells. Also, there are blood-brain, blood-nerve, blood-eye, and blood-testis barriers characterized by specialized endothelium that reduce transfer of antibodies and related large molecules into these protected spaces. Therefore, some tissues have relatively little in vivo access by antibodies compared with other tissues. Likewise, antibodies have little chance of access to antigens within cells compared with cell membrane or transmembrane antigens (Hall et al. 2008).
For the purposes of this article, cross-reactivity is defined as the antibody CDR binding to an epitope different from the directed target (i.e., an off-target structure). It involves the same types of interactions of the CDR as described for specific binding. Off-target binding can occur due to sequential (compositional) elements or nonsequential (conformational) elements in the cross-reactive antigen. For example, sequence-dependent cross-reactivity was reported when therapeutic antibodies directed against prostate-specific membrane antigen, bound to neural enzyme NAALADase (glutamate carboxypeptidase II) (Berger, Carter, and Coyle 1995; Troyer, Beckett, and Wright 1995; Liu et al. 1997; Luthi-Carter et al. 1998; Chang et al. 1999). Later studies revealed close sequence homology between these two proteins as the likely cause for the cross-reactivity (Sacha et al. 2007). As another example, Ying et al. (1992) demonstrated monoclonal antibodies cross-reacted with partially homologous sequences in human C-reactive protein, human serum amyloid P component, and the horseshoe crab C-reactive protein homolog limulin.
In other cases, the relationship between the target epitope and the off-target epitope has not been described but is not thought to be related to shared sequences. Monoclonal antibodies directed against carcinoembryonic antigens that cross-react with macrophages and granulocytes are examples (Zoubir et al. 1990; Thompson, Grunert, and Zimmermann 1991). Certain aspects of the antibody/epitope pair may contribute to the potential for cross-reactivity. It is not unusual for antibodies that bind to strongly hydrophobic epitopes to have off-target binding to other hydrophobic epitopes, including those with dissimilar sequences compared with the target. Off-target binding appears to be less frequent when the CDR-antigen interaction involves strong salt bridges and electrostatic rather than hydrophobic interactions (Sinha et al. 2002). Some epitope targets might have a higher likelihood of cross-reactivity (“molecular mimicry”). Oldstone (1998) indicated that approximately 5% of eight hundred mouse monoclonal antibodies directed against a variety of human and mouse viral antigens demonstrated cross-reactivity with some normal mouse tissues.
While not considered cross-reactivity as defined by this article, unexpected binding can occur when a target epitope is distributed to tissues other than the expected target. For instance, cancer-associated epitopes may also be widely expressed in normal tissue. As an example, EpCAM (17–1A antigen) is overexpressed in uveal melanoma, head and neck, lung, breast, and ovarian cancer. In adult tissue, therapeutic and IHC diagnostic antibodies directed against EpCAM (17-1A antigen) show widespread cross-reactivity with the basolateral membranes of simple, pseudostratified, and transitional epithelium but not with normal stratified squamous epithelium or hepatocytes (reviewed in Balzar et al. 1999; Odashiro et al. 2006).
The potential for cross-reactivity is not limited to protein antigens. Monoclonal antibody and biochemical analyses of the core glycan structures of enteropathogenic Campylobacter jejuni have identified how a single antigenic determinant can act as a ganglioside mimic and elicit autoantibodies that associate with clustered human N-acetylneuramininic acid-containing gangliosides to precipitate the onset of neuropathy in Guillain-Barre or Fisher syndromes (Prendergast and Moran 2000; Houliston et al. 2007). Other antibody cross-reactivity might be related to shared glycosylation sites, as has been proposed for homology between platelet GpIIb/IIIa and bacuolovirus- but not vaccinia-virus-expressed human immunodeficiency virus gp160 (Bettaieb et al. 1992), or with GpIIa and Helicobacter pylori urease B (Bai et al. 2009). While specific binding of the antibody occurs via the CDR cleft, nonspecific interactions may involve other portions of the antibody outside of the CDRs (including antibody Fc binding to Fc receptors) and may also result in staining at sites that do not contain the target epitope.
In summary, because of limitations in the antibody/epitope pair and changes to the tissue during tissue preparation, IHC studies will not be feasible for some products and may provide challenges in interpreting data for other products. When an antibody epitope pair is conducive for developing a robust assay, the TCR study offers one of the broadest screens for target localization and cross-reactivity of any preclinical assay. When staining occurs, it is not usually known whether it is related to binding to the target in a previously unrecognized location, to a cross-reactive epitope, or in some cases to artifactual staining. In general, it is not necessary to differentiate between on- and off-target binding. However, in some cases, this information may be used in the overall understanding of the potential tissue target liability of the test article.
Suitability of the Test Article/Epitope Pair for IHC Staining/TCR Assays
TCR studies are based on IHC. To serve as a useful IHC reagent, an antibody (or antibody-like molecule) and its epitope need to have the appropriate physicochemical characteristics that result in good specificity and sensitivity in tissues ex vivo. Ideal test article/epitope pairs have epitopes that are generally linear, hydrophilic but insoluble, expressed at high levels, insensitive to fixation or desiccation or membrane rigidity constraints, and readily retrieved after fixation (Metz et al. 2004; Emoto, Yamashita and Okada 2005a; Yamashita and Okada 2005b; Sompuram et al. 2006; Blind et al. 2008; Pollard et al. 1987).
In contrast to the ideal circumstances, the epitopes selected as targets for many antibody and antibody-like test articles may lack these characteristics because they are conformational, expressed at low levels in normal tissues, dynamic and subject to rigidity constraints, hydrophobic, and/or have highly charged amino acid residues (Barlow, Edwards, and Thornton 1986; Costagliola, Panneels, et al. 2002; Costagliola, Franssen, et al. 2002; Costagliola et al. 2004; Huebner 2004; Sanders et al. 2004; Ruf et al. 2007). Similarly, candidate antibodies are selected based on desirable in vivo pharmacologic activity and not on optimal IHC characteristics or ability to function as IHC reagents. As a result, the methods used in TCR studies must be carefully selected and optimized on validated positive and negative control specimens that are known to express or not express the target antigen, respectively. Because the regulatory guidances stipulate that the test article should be the same as those planned for clinical use, there is no option for substituting the test article with another antibody with better binding characteristics, or for substituting a portion of the target that is more stable or conducive to IHC testing. Thus, the selection of the optimal method represents a delicate balance between preserving the highest signal with the least amount of background and nonspecific staining. In many cases, a robust sensitive, specific, and reproducible IHC assay can be developed for both linear and conformational epitopes (Hall et al. 2008). However, in some cases, the best TCR assay may have high background/nonspecific staining or may show no staining of target in tissue because of limitations imposed by the test article or its target. Given the variability in IHC performance, when a robust, reproducible, and sensitive TCR method cannot be developed despite the best documented efforts, the lack of an interpretable TCR assay should not prevent a program from moving forward.
Whether to Conduct a TCR Study
Whether to conduct TCR studies in a given program has become a topic of debate as more novel structures are being developed. The purpose of TCR studies is to identify potential binding targets, and IHC affords a broad assessment of potential targets in the context of tissue architecture. However, IHC assays are designed around using antibody-antigen binding as the basis of this technology platform. Therefore, it is recommended that TCR studies only be used for antibody and antibody-like test article molecules that contain a CDR, including antibody-conjugates. When the test article is a radioimmunoconjugate or chemical conjugate, the TCR assay should include the conjugate. In the case of a radioimmunoconjugate, a nonradioactive conjugate form of the test article can be used. Overall, it is not recommended to conduct TCR studies with test articles that do not contain a CDR. Furthermore, although IHC may be used to justify research use of a genetically engineered model, full tissue panel TCR studies in these animals are not recommended because protein distribution and expression in these models are influenced by the design of the transgene and the gene integration site(s), and they may not reflect the epitope as expressed in the human or the selected toxicology species (Matthaei 2007).
Other Uses—In Vivo Test Article Distribution Studies
Although distinct from the TCR studies discussed above, in vivo test article distribution or localization studies can also be performed in selected preclinical species and have become more common recently driven by improvements in IHC reagents that permit staining of human IgG (i.e., test article) in the presence of endogenous monkey, rabbit, rat, or mouse IgG (Rojko and Price-Schiavi 2008). However, these types of studies are usually conducted based on specific scientific need and not as a routine component of toxicity studies. They may provide value in determining whether the binding observed ex vivo actually occurs in vivo in preclinical species. These studies may be carried out using IHC on routinely collected paraffin tissues from toxicity studies and provide information regarding the cellular, subcellular, and extracellular localization of test article in vivo in normal and diseased tissues. The relationship between test article localization in vivo, tissue effects, and association with other findings can then be used to make integrated evaluations about the mechanism and relevance of any observed in vivo effects. However, specific localization of the test article following in vivo dosing by IHC has the same type of limitations that have been reported for radioimmunodetection (Moffat et al. 1999, Gratz et al. 1998, Cai et al. 2006); that is, the specific localization of bound drug cannot be determined unless it is much greater than the unbound drug in tissue and plasma. In addition, unlike radiolabeled studies, IHC is rarely quantitative. Therefore, determination of specific localization to a target versus distribution may be challenging, and these types of studies should only be conducted on a case-by-case basis to address specific issues.
Case Studies
To better understand the conduct, use, interpretation, and limitations of TCR studies, a variety of case studies are presented that highlight the collective experiences of industry with TCR studies. Cases were solicited from industry, and were voluntarily submitted by companies, or were available from the literature. All cases provided to the authors are included in the section below; there was no selection or exclusion of submitted cases. The cases are intended to provide specific examples of situations that have been encountered, including a few that do not reflect currently recommended approaches (i.e., TCR with a surrogate mAb, or with a fusion protein that lacks a CDR).
This set of cases is neither comprehensive in scope nor intended to represent a proportional distribution of outcomes but, rather, is designed to provide examples that cover many of the possible interpretive challenges arising from TCR studies and to show where additional information may ultimately be needed to understand the data more fully. When available, the results of additional studies, preclinical or clinical, have been included in the case descriptions. Because of confidentiality, most cases have a limited set of information available. However, in the collective experience of the authors, the information that is provided in these cases does highlight both the benefits and issues related to TCR studies.
Each case study is followed by a brief commentary on the impact of the TCR study. This commentary is based on the authors' retrospective analysis in light of the current recommendations of this article and in agreement with the contributors of the case studies. It should be recognized that these studies were conducted at varying times over the past two decades, and the knowledge and regulatory climates were not necessarily the same. As the cases collectively highlight, TCR data constitutes only one preclinical element contributing to the overall data available for the development program.
In cases 1 through 6, on-target binding in a previously unknown site of target expression or identification of cross-reactivity, that is, off-target binding, is demonstrated.
Case 1
The test article was a toxin-conjugated mAb specific for an antigen that is overexpressed on a variety of solid tumors. Toxicity studies were initially conducted in dogs, which were known to be a good model for the cardiotoxicity associated with the toxin. In the initial in vivo studies, effects were observed throughout the entire digestive tract (mouth to anus), and some dogs had acute gastrointestinal toxicity during or immediately following administration of the first dose. Similar findings were also observed following administration of the unconjugated antibody. IHC studies on colon biopsies demonstrated that those dogs that had high levels of binding of the test article to epithelial cells in the gastrointestinal tract developed significant GI toxicity, whereas low-expressing dogs did not have toxicity in vivo. IHC studies also showed that relatively high expression of the target in one area of the digestive tract was associated with higher expression in other areas, although the absolute expression was variable between different locations in the digestive tract. TCR studies in rats, dogs, cynomolgus monkeys, and humans were conducted that demonstrated that the staining of high-expressing dogs was similar to that observed in humans with staining of many gut epithelial cells, while staining in the cynomolgus monkey and rats was less intense and involved fewer epithelial cells (primarily limited to crypts). Phase I studies in patients showed toxicity of the upper GI that was considered to be related to binding of the antibody and was dose limiting.
Commentary
In this case, TCR studies identified the dog as the species with highest and most similar expression of the target relative to humans, and the location of the expression correlated with in vivo toxicity in the dog. The level of target expression was associated with the level of toxicity observed during in vivo studies. The dog also had the most similar target organ (intestine) expression compared with humans. In clinical trials, similar effects were observed. Thus, the TCR provided information on the correct preclinical species for toxicity studies and identified the target organ.
Case 2
The clinical candidate antibody was a toxin-conjugated mAb directed against an antigen expected to be restricted to tumor tissue in patients but was also known to be present in placenta. The test article used in the TCR studies was the unconjugated antibody. A TCR study evaluating human, rat, and cynomolgus monkey tissues was initially conducted and showed binding to some normal cell types/tissues in human, but not in rat or cynomolgus monkey tissues. Tissues from the dog, guinea pig, hamster, rabbit, CD-1 mouse, rhesus monkey, and common marmoset were then evaluated in TCR studies. The common marmoset was the only species with staining that was similar to the human. Based on this data, an in vivo toxicity study was conducted in common marmosets concurrent with other in vitro evaluations of the binding of the test article to the target across species. Toxicity was observed in the eye of the common marmoset, but not in cynomolgus monkeys, which had also been tested in an in vivo toxicity study. Although binding had been seen in several tissues in the TCR studies in humans and common marmosets, no binding was evident in the eye. The additional in vitro evaluations demonstrated that the test article bound to the target antigen from humans and common marmosets, but not cynomolgus monkeys. Further evaluations using laser capture microdissection demonstrated mRNA for the antigen in the affected substructure of the eye. The presence of the target was confirmed with IHC evaluations using a different antibody to the target protein that was a more robust IHC reagent. The antibody was not advanced into the clinic.
Commentary
Case 2 highlights several interesting points. The TCR study supported the common marmoset and not the cynomolgus monkey as an appropriate toxicity species based on the TCR binding pattern. During in vivo toxicity studies in common marmosets, ocular toxicity was observed. Interestingly, other tissues that did have staining in the TCR studies did not have in vivo effects. Additional studies later demonstrated the presence of the target in the target organ (the eye). Thus, TCR studies supported the correct pharmacologic species to use, which demonstrated a significant toxicity that precluded the program from entering clinical trials. However, the TCR study with the drug candidate itself did not identify the critical toxicity organ, while staining did occur in a few other tissues that did not demonstrate toxicity, highlighting some of the challenges that exist with interpreting TCR studies.
Case 3
The test article was directed against a human cytokine receptor (IL-6R) with similar but incompletely overlapping human and monkey TCR profiles (Kato et al. 2009). The test article had pharmacologic activity in the cynomolgus monkey, which was considered a relevant preclinical species. On-target staining that was observed in human, but not in the cynomolgus monkey, included hepatocytes, uterine epithelium, and glial cells in the brain and spinal cord. In the human clinical trials, an adverse effect on liver function (increased total cholesterol, HDL-C, and triglyceride levels as well as transient liver enzyme changes) was observed and attributed to a test article-related effect on hepatocytes. No alterations in liver function were observed in monkey toxicity studies. No adverse clinical effects were observed that could be attributed to the unexpected binding to glial cells or uterine epithelium.
Commentary
Case 3 highlights several points. Although the cynomolgus monkey was considered to represent a relevant pharmacologic species, they did not have the same on-target staining in TCR studies and did not have the same in vivo effects that occurred in humans that did demonstrate staining. However, in humans, in vivo effects were only observed in some tissues that were positive in human tissues, suggesting that other factors (genetic effects, reproductive cycling, tissue distribution [e.g., blood-brain barrier]) also affected potential for clinical toxicity (Kato et al. 2009). Thus, the TCR staining appeared to correlate with in vivo effects in some, but not all, tissues that were positive in humans. Once again, this highlights that TCR studies are screening assays, and the relevance of the staining needs to be addressed through other assays.
Case 4
The test article was a toxin-conjugated mAb against a surface antigen that is expressed predominantly in the prostate gland. This target also shared substantial amino acid sequence identity with an otherwise unrelated enzyme expressed in neural tissue. There were reports of this neural protein in human and rat brain tissue, as detected using polymerase chain reaction (PCR) and IHC approaches, respectively. As a further complication, the toxin was known to cause peripheral neuropathy in unconjugated form and when conjugated to mAbs directed against unrelated, nonprostate proteins. Thus, there were overall concerns about possible nervous system toxicity related to the mAb, or related to the toxin alone.
The unconjugated mAb was characterized for its ability to bind the prostate and neural proteins using flow cytometry. Prostate cancer cells expressing the prostate protein, and transfected cells expressing the neural protein were evaluated. The unconjugated mAb stained cells expressing the prostate protein but not the neural protein. The unconjugated mAb was also used as the test article for the TCR assays, and human, cynomolgus monkey, rat, and mouse tissues were assessed. No staining occurred in rat or mouse tissues, but similar staining was observed in human and monkey central nervous system (CNS) tissue. Prostate and epididymal staining was observed in humans, but only epididymal staining was observed in adult monkey tissue. The absence of staining in the monkey prostate was unexpected. Staining was additionally seen in a hemorrhagic, newly forming corpus luteum in the ovary of one human donor. While the ovarian staining was initially considered “nonspecific,” reports were subsequently located that described expression of the prostate protein on new vessels during nonprostate tumor angiogenesis (such as might occur in a corpus luteum). In addition, reports on expression of the neural protein in human ovary tissue were also found. Thus, it was thought that the ovarian staining might be related to possible prostate or neural protein expression.
To better understand the distribution of the prostate protein, reverse-transcription polymerase chain reaction (RT-PCR) was used to quantify the prostate protein mRNA in human and monkey tissues. Prostate protein mRNA was identified at high levels in human prostate, and at lower levels in liver, kidney, salivary glands, and CNS and peripheral nervous system (PNS) tissues. In tissues from monkeys, prostate protein mRNA was present in dorsal root ganglia, kidney, and in a sample containing prostate, seminal vesicle, and urethra. The method did not allow localization of the mRNA to the specific monkey urogenital tissue. Thus, based on mRNA, the prostate protein appeared to be distributed in several tissues in humans, including in tissues that were not positive in the TCR assay, and including human CNS and PNS tissues. In the monkey, mRNA was seen in fewer tissues, including limited nervous system tissues. In monkey urogenital tissue, it was not clear where the protein was localized based on mRNA expression. The difference between IHC and flow cytometric results for neural protein staining may reflect differences in antigen preservation and/or recognition (presentation).
In single-dose toxicity studies, the mAb-toxin conjugate induced toxicities in multiple nonneural tissues of both mice and monkeys, but there was no toxicity with the unconjugated mAb. In repeat-dose toxicity studies in mice and monkeys, the toxin-conjugated mAb induced toxicity in additional target organs, including peripheral nerves, the dorsal funiculus of spinal cord, and dorsal root ganglia and was associated with clinical signs of peripheral sensory neuropathy. There was no toxicity of the unconjugated mAb in repeat-dose studies. Of interest, at equimolar doses of toxin, the mAb-toxin conjugate was more toxic than the toxin alone but involved the same tissues as the toxin alone did. The data overall suggested the antibody was not causing toxicity (based on a lack of effects from the unconjugated antibody), and the antibody was not directing the toxin to certain tissues and causing increased toxicity (based on a lack of toxicity in some tissues that expressed the protein). The increased toxicity of the mAb-toxin conjugate was interpreted as being instead related to the much longer half-life of the toxin when conjugated to the mAb, relative to the much shorter half-life of the toxin alone. In clinical trials, peripheral neuropathy was a prominent finding.
Commentary
This case covers a complex program with a variety of assays to explore target distribution. The combination of TCR studies and PCR data identified the target antigen distribution in human and monkey tissues. This information supported the use of the monkey as a relevant species for in vivo toxicity testing. The lack of toxicity in some tissues that contained the target, coupled with the lack of toxicity of the unconjugated Ab, and the longer half-life of the toxin-conjugated mAb, led to the interpretation that the observed in vivo effects were related to the toxin, and not to targeting by the mAb. The case also demonstrates that TCR assays can be integrated with other methods that can be used to help understand the potential for human toxicity.
Case 5
The test article was directed against a human surface antigen present on leukocytes. TCR studies with human and monkey tissues indicated expected on-target staining of human and monkey leukocytes as well as unexpected staining which was later determined to be on-target staining in a previously unrecognized site, that is, perivascular microglial cells, in sections of brain from human and monkey. Human clinical trials revealed a higher incidence of headaches in subjects dosed with the test article. Monkey toxicity studies revealed perivascular brain inflammation with several different formulations of the test article. The sizes and locations of affected vessels in the toxicity study corresponded to the sizes and locations of perivascular staining in the monkey and human TCR studies. Although the blood-brain barrier protects certain parts of the brain, other parts (e.g., choroid plexus, cerebrospinal fluid, circumventricular organs, area postrema, Virchow-Robbins space, meninges, ependyma, and immediate subpial and subependymal white matter) are outside of it (Bernacki et al. 2008). In the case described, the primary vessels affected were in regions not protected by the blood-brain barrier.
Commentary
In case 5, TCR studies detected a previously unknown location of the target, that is, on target-binding. TCR studies confirmed binding in human and monkeys and correlated with the location of effects in monkeys.
Case 6
Hall and colleagues (2008) reviewed a case of a chimeric therapeutic antibody against colon carcinoma with off-target staining of endothelium in gray matter in human and monkey brains. Administration of this test article was associated with short-term memory loss and dizziness in three out of six Phase I clinical trial volunteers. The initial histopathology evaluation had indicated no lesions in the monkey brains; however, subsequent review demonstrated mononuclear leukocytes adherent to the lining the endothelium of cortical gray matter vessels, and the clinical trials were discontinued.
Commentary
A cross-reactive (off-target) epitope was identified in Case 6, and data supported the monkey as an appropriate toxicity species. The cross-reactivity was associated with effects in monkeys and subsequently in humans.
Cases 7 and 8 highlight examples where a robust (i.e., sensitive and specific) TCR assay could not be developed because staining could not be detected in positive control material. Case 9 also similarly describes an example where staining was not expected.
Case 7
The test article was a mAb specific for a cell-associated and circulating factor that is expressed on and by a number of cell types as detected by other methods. Both human and cynomolgus monkey TCR studies were conducted. The TCR assay did not show any binding with the test article under most circumstances tested. This included all normal tissue specimens from both species, diseased tissues expected to have the target antigen, and cell lines with confirmed overexpression of the target antigen. The only discriminating staining was to the target antigen bound to beads embedded in paraffin. The TCR assay was conducted using the beads as staining controls to comply with regulatory expectations. There were no cross-reactivity findings, and no additional regulatory concerns were identified.
Commentary
Case 7 shows an example where staining could not be demonstrated in tissues that were known to express the target. Because test articles are not selected based on their ability to serve as IHC reagents, such lack of binding will sometimes occur, as discussed in more detail in other sections of the article. In this case, the lack of staining of known positive tissues did not impact the development of the molecule.
Case 8
The test article was a mAb against a cell surface receptor predominantly expressed by neoplastic cells. Both human and cynomolgus monkey TCR methods development studies were conducted. The TCR assay showed an absence of definitive staining of a limited set of normal tissues or tumor tissues proposed as positive staining controls. This test article also failed to stain a cell line transfected with the target antigen. This same cell line was positive when stained with a commercially available diagnostic mAb against the target. Binding of the target antigen by the test article was confirmed by other in vitro assays. Unexpected and severe ocular toxicity, likely related to agonism of the target, was seen in a one-month in vivo toxicity study in cynomolgus monkeys. The original TCR assay had not included eye tissues, and follow-up studies did not demonstrate staining in the cynomolgus monkey eye.
Commentary
Case 8 demonstrated the issue of a lack of staining with the test article, even in tissues known to express the target. In this case, other assays were used to select the toxicity species, and the TCR study did not contribute to that selection. TCR staining was not predictive for the in vivo effects seen in the eye.
Case 9
The test article was a mAb against a soluble antigen, and therefore on-target tissue staining was not expected. As the molecule bound to human, cynomolgus monkey, mouse, and rat soluble antigen, TCR studies were conducted in multiple species. The TCR assay showed no specific staining in any tissues from any species. Both the cynomolgus monkey and mouse were used in toxicity studies.
Commentary
Case 9 shows an example where staining was not expected to occur in tissues, and none was detected, including no cross-reactivity to off-target epitopes. The selection of the appropriate species for toxicity testing was based on pharmacology data.
Cases 10 and 11 demonstrate programs where either staining was observed but did not correlate with any in vivo effects (case 10), or no staining was observed in a tissue that had in vivo toxicity (case 11).
Case 10
The test article was a humanized mAb to a receptor on T cells and macrophages. A TCR study evaluating human and cynomolgus monkey tissues was conducted. The assay showed widespread staining of vessels, especially in the renal glomeruli of both species. Initially, the vascular staining raised numerous questions internally related to the significance of the staining and the potential risk of moving forward with the program. However, there was no evidence of vascular toxicity in up to three–month dosing in toxicity study in monkeys. Thus, the TCR findings were not predictive of in vivo toxicity in this case.
Commentary
Case 10 highlights a common issue that occurs with TCR studies, that is, the presence of staining that is not associated with any in vivo effects. It also highlights that TCR studies are screening studies, and the relevance of any staining should be interpreted within the context of additional studies.
Case 11
The test article was a mAb against an anchored cell surface antigen. Results from TCR assays differed based on method used (fixed versus frozen tissues) and ranged from no specific staining (frozen tissues) to staining of numerous tissues (fixed tissues). Where staining was observed, it was similar between monkey and human. However, even under conditions that resulted in positive staining, the toxicity observed in the IND-enabling safety study was not predicted by the TCR studies.
Commentary
Case 11 highlights the variability that can occur with TCR studies, although the staining patterns between human and monkey were similar and confirmed the monkey was a relevant species for toxicity testing. Furthermore, although staining was observed in some tissues, toxicity was observed in a tissue that did not have staining. Thus, the TCR study did not predict the target organ, although it may have been useful in assisting in species selection.
Cases 12 and 13 describe experiences where data from a TCR assay led to several additional investigations.
Case 12
The test article was an IgG4 mAb against a circulating cytokine. There was no tissue or cell line positive control tissue for this test article, so agarose beads coated with the cytokine were used as the positive staining control material. A TCR study evaluating human and cynomolgus monkey tissues was conducted. The assay showed unexpected cross-reactivity in brain tissue and optic nerve of human and monkey tissues. Subsequently, a cross-reactive protein was identified and was determined to be a brain matrix protein rather than a neuronal component. There were no effects in cynomolgus monkeys during in vivo toxicity studies. It was concluded that binding of the mAb to the CNS matrix protein, even if it occurred in vivo, would unlikely adversely affect CNS function, as the mAb was an IgG4 with a low potential for antibody-dependent cytotoxicity (ADCC).
However, regulatory agencies regarded the positive staining as a significant risk factor for CNS toxicity, even though no effects had been seen in existing in vivo toxicity studies in cynomolgus monkeys, and the test article exposure in most of the CNS is very low because of the blood-brain barrier under normal circumstances. Concern was expressed for patients that might experience inadvertent CNS exposure to the drug if a stroke or other disease resulted in abnormal bleeding or disruption of the blood-brain barrier. Therefore, extensive additional preclinical testing was required prior to initiating clinical trials, including studies evaluating intravenous, subcutaneous, and intrathecal administration followed by IHC test article distribution studies to assess in vivo cross–reactive binding. For both the intrathecal study and subsequent six- and twelve-month toxicity studies, cynomolgus monkeys were subjected to an extensive battery of in-life (including electrophysiology) and postmortem neurologic assessments (whole-body perfusion studies followed by detailed CNS tissue examination including sampling for electron microscopy and in vivo test article distribution studies). No morphologic or functional nervous system findings and no in vivo binding of the drug to cynomolgus monkey nervous tissues were ever demonstrated. There were also no effects in humans during the clinical trials that were eventually allowed to proceed.
Commentary
In case 12, the TCR study identified a cross-reactive epitope and supported an appropriate species in which to conduct in vivo toxicity testing. The in vivo toxicity studies suggested the binding was not associated with any effects. However, regulatory concerns led to significant additional studies and delays to the program. When the program was finally allowed to proceed into the clinic, there were no effects observed related to the off-target binding in the CNS, suggesting that TCR studies should not supersede well-designed and performed toxicity studies.
Case 13
The test article was a mAb against an anchored cell-surface antigen. Both human and cynomolgus monkey TCR studies were conducted. Specific staining with the test article was observed in numerous different tissues in both species. Staining was similar between species with the exception of unexpected binding in one human tissue that did not occur in the monkey tissue. This observation led the regulators to require additional clinical monitoring and exclusion criteria in Phase I study, based on the view that in vivo preclinical safety data were not likely to be representative because of the lack of staining in the tissue of concern in the monkey TCR study. No toxicity signal related to a theoretical concern driven by TCR study was detected in any clinical study.
Commentary
As was mentioned in case 12, case 13 highlights that TCR data can lead to additional evaluations and program delays, even when well-designed in vivo studies do not identify a risk. The staining observed led to regulatory concerns and the requirement for additional clinical monitoring and exclusion criteria in Phase I; however, no toxicities relevant to the staining were observed.
Cases 14 and 15 demonstrate the assay variability that is relatively common in TCR studies.
Case 14
The test article was a mAb against a tissue-diffusible ligand derived from a plasma protein. In a preliminary non-GLP TCR study, one tissue type stained in both the human and cynomolgus monkey. GLP TCR studies were then done in human and cynomolgus monkey tissues to confirm the relevance of the cynomolgus monkey as a species for toxicity studies. In this study, unexpected specific staining was observed in the cytoplasm and cell membranes of numerous different tissues in both species. This staining differed from the preliminary study results. The staining in the GLP study overlapped between human and monkey in only some tissues: in other tissues staining only occurred in human but not monkey tissues. Interestingly, the only tissue that was positive in the preliminary study was one of the few tissues that were negative in the pivotal study. However, the potential off-target tissues identified in either of the ex vivo TCR studies did not correlate with any in vivo effects as there were no microscopic findings in monkeys following a three-month repeat-dose toxicity study.
Commentary
Case 14 demonstrates the variability that can occur in TCR studies. Staining was not consistent between TCR studies. However, there was some overlap in the staining between human and cynomolgus monkey tissues. The species selection for toxicity studies was based primarily on amino acid sequence homology of the target in humans and monkeys. The TCR study did not contribute to the species selection. The program also highlights that ex vivo staining may not be associated with in vivo effects.
Case 15
The test articles were two humanized and one mouse mAb to a cytokine receptor. Despite inconsistent staining on positive control cells by the candidate antibody, TCR studies in human and chimpanzee were performed twice with the therapeutic candidate and with a humanized surrogate in an attempt to fulfill the regulatory requirement. The mouse antibody against the receptor was used as positive control in establishing receptor expression in the positive control cells and tested tissues, and when applied to human tissues, binding was seen on the membrane of stromal, nonhematopoietic, myeloid, dendritic cells and tumor cells. Although broader than anticipated, the binding of the mouse mAb in the tested species correlated with the expected receptor distribution. In contrast, both humanized mAbs turned out to be very poor IHC reagents with no consistent staining of positive control cells. Substantial time, effort, and resources were invested in trying to get the TCR study to work with the humanized test articles, ultimately with inconclusive results.
Commentary
Case 15 highlights again the variability related to IHC staining properties of different Abs, even when targeting a single antigen or epitope. This case also highlights that the use of a surrogate test article may not be useful in understanding the binding of the clinical candidate.
Cases 16 through 18 describe investigations using IHC or other alternative assays.
Case 16
The test article was a humanized (CDR-grafted) mAb against a surface antigen present on a specific subset of human lymphocytes. This particular surface protein facilitates the trafficking of these cells within a particular tissue. The mouse mAb had been shown to be efficacious in a nonhuman primate (marmoset) model of inflammation within this tissue. When the mouse mAb was bound to the lymphocyte receptor, the cells could not traffic properly and inflammation was resolved. The mouse mAb had no effector function and did not result in depletion of targeted lymphocytes.
The TCR study identified identical, highly specific lymphocyte staining patterns in human and cynomolgus monkey tissues. A TCR study with the mouse parent mAb also gave identical results. Therefore, the mouse mAb was used as a staining reagent in subsequent IHC studies to quantify occupancy (“blocking”) of the target receptor in monkey tissues collected after in vivo administration of the humanized mAb. When the humanized mAb was bound to the lymphocyte target in vivo, the mouse mAb could not bind to the tissues ex vivo. When the humanized mAb cleared from the target, the mouse mAb could bind again. This same principle was used to stain monkey lymphocytes with the mouse mAb to quantify the relative numbers of “free” and “bound” receptors on blood lymphocytes by flow cytometry. Furthermore, when monkeys dosed with the humanized mAb in repeat-dose toxicity studies developed primate anti-human-antibodies, which cleared the humanized mAb, this “blocking” effect was not observed, and it was possible to stain the target with the mouse mAb. No toxicity was observed with this mAb. When the humanized mAb was evaluated in clinical trials, it had no effector function, did not deplete lymphocytes, and yet resulted in marked improvement in inflammation within the target tissue. This improvement was associated with receptor blockade on lymphocytes. In humans that developed neutralizing human anti-human antibodies, free receptors became available and tissue inflammation returned.
Commentary
In case 16, the TCR study supported the species selection for toxicity studies. In addition, it demonstrates that information and methods learned from TCR assays can be adapted for use for nonclinical and clinical monitoring of the effects of treatment, such as receptor occupancy on circulating target cells and within the tissues in which the desired effect is intended to occur.
Case 17
The test article was directed against a cytokine/cytokine receptor. There were no off-target cross-reactivity findings. A short-term toxicity study revealed lung inflammation in a small number of test article-dosed animals. Follow-up IHC test article distribution studies using paraffin sections from the toxicity study examined the transport, uptake, and distribution of the test article to lung and selected other tissues following in vivo administration. These studies compared the staining patterns of test article in test article-dosed monkeys to the staining patterns of endogenous monkey IgG in test article-dosed and undosed monkeys. The test article staining patterns in the in vivo–exposed monkey lungs were consistent with physiologic uptake and distribution of test article as part of the endogenous IgG pool via Fc receptor (FcR)-mediated and other physiologic transport processes and did not indicate CDR-mediated binding to lung in vivo. Thus, consistent with the TCR findings, there was no distribution of test article in lung other than expected physiologic distribution. Lung inflammation was judged unrelated to test article binding, consistent with the absence of lung findings in subsequent longer-term toxicity studies and clinical trials.
Commentary
In case 17, in vivo IHC test article distribution studies were used to show that an in vivo finding in a short-term toxicity study was not likely related to the test article, and subsequent longer-term toxicity studies confirmed this. Thus, the in vivo distribution data helped allow the program to proceed.
Case 18
The test articles were affinity-matured anti-respiratory syncyticial virus (RSV) monoclonal antibodies with variably increased ability to neutralize RSV in vitro relative to the parental mAb. A subset of variant test articles demonstrated unexpected staining of lung and skin tissues in ex vivo TCR studies and also had altered pharmacokinetics, lower than expected lung bioavailability, and suboptimal efficacy in vivo as compared to the promising in vitro potency. However, a few test article variants had little or no ex vivo tissue binding, and these also had relatively improved pharmacokinetics, lung bioavailability and potency in vivo (Wu et al. 2007).
Commentary
In case 18, the ex vivo tissue binding pattern of test article variants correlated with in vivo pharmacokinetic and pharmacodynamic properties. In this example, affinity maturation of a therapeutic mAb against a viral target ultimately resulted in broad off-target binding that was reflected in both ex vivo and in vivo studies. Ultimately, these studies were used to select an appropriate clinical candidate.
Case 19 is an example of conducting a TCR study with a molecule that lacks a CDR.
Case 19
The test article was a humanized glycosylated fusion protein that was known to bind to T lymphocytes; it had an immunoglobulin Fc but did not have a CDR. A TCR study including tissues from humans and nonhuman primates was conducted to determine the presence of target and/or off-target binding. The assay did not show any binding. Regulatory agencies requested that the sponsor perform the assay a second time to prove that all efforts had been made to get the assay to work. Although the TCR assay did not demonstrate binding to the target tissues, a pharmacodynamic marker showed that the nonhuman primate model was relevant for this extremely potent drug, and therefore, in the end, the results of the TCR assay did not call into question the relevance of the preclinical model.
Commentary
Case 19 demonstrates a situation where a robust IHC assay could not be developed for this test article. Furthermore, when the first study did not demonstrate binding, regulatory requests were made to repeat the study. It is not surprising that mAb-like products that lack CDRs, as in this fusion-protein example, would not perform well in an IHC-based assay. This case is consistent with the authors' position that TCR studies are not recommended for molecules that do not contain a CDR.
In summary, the case studies outlined above and the experience of the authors indicate that the TCR assay by itself has variable correlation with toxicity or efficacy. The results highlight the fact that TCR studies are screening assays. In most of these case examples, toxicity would have been detected during in vivo toxicity studies. It should be particularly stressed that ex vivo TCR staining does not necessarily equate to in vivo toxicity. As is the case with almost all preclinical safety studies for biotherapeutics, applicability to providing meaningful information can only be determined on a case-by-case basis in conjunction with other data. Importantly, the presence of staining in TCR studies should not be overinterpreted, and conversely, the lack of the ability to develop a TCR study should not preclude the safe clinical development of candidate mAb therapeutics.
Discussion
Assessment for potential binding to known and unknown tissue targets is recommended for Ab and Ab-based biopharmaceuticals. A TCR study with a panel of human tissues is currently considered a standard component of the preclinical safety assessment package supporting initial clinical dosing for these molecules. TCR studies are recommended as screening assays (i.e., as assays whose data should be interpreted in the context of additional studies), which are designed to identify potential target organs. Importantly, as presented in the various case studies, the TCR assay by itself has variable correlation with toxicity or efficacy. Therefore, any findings of interest should be further evaluated and interpreted in the context of the overall pharmacology and safety assessment data package. Based on the current status of TCR studies, this article is recommending that TCR studies follow the algorithm outlined in Figure 1. This algorithm can be applied to many programs that are considering TCR studies, outlining appropriate decision points and suggesting when additional TCR studies are generally not necessary. The rationale supporting this algorithm is presented in the discussion below. However, as has been highlighted above, a case-by-case approach should be taken for each test article.
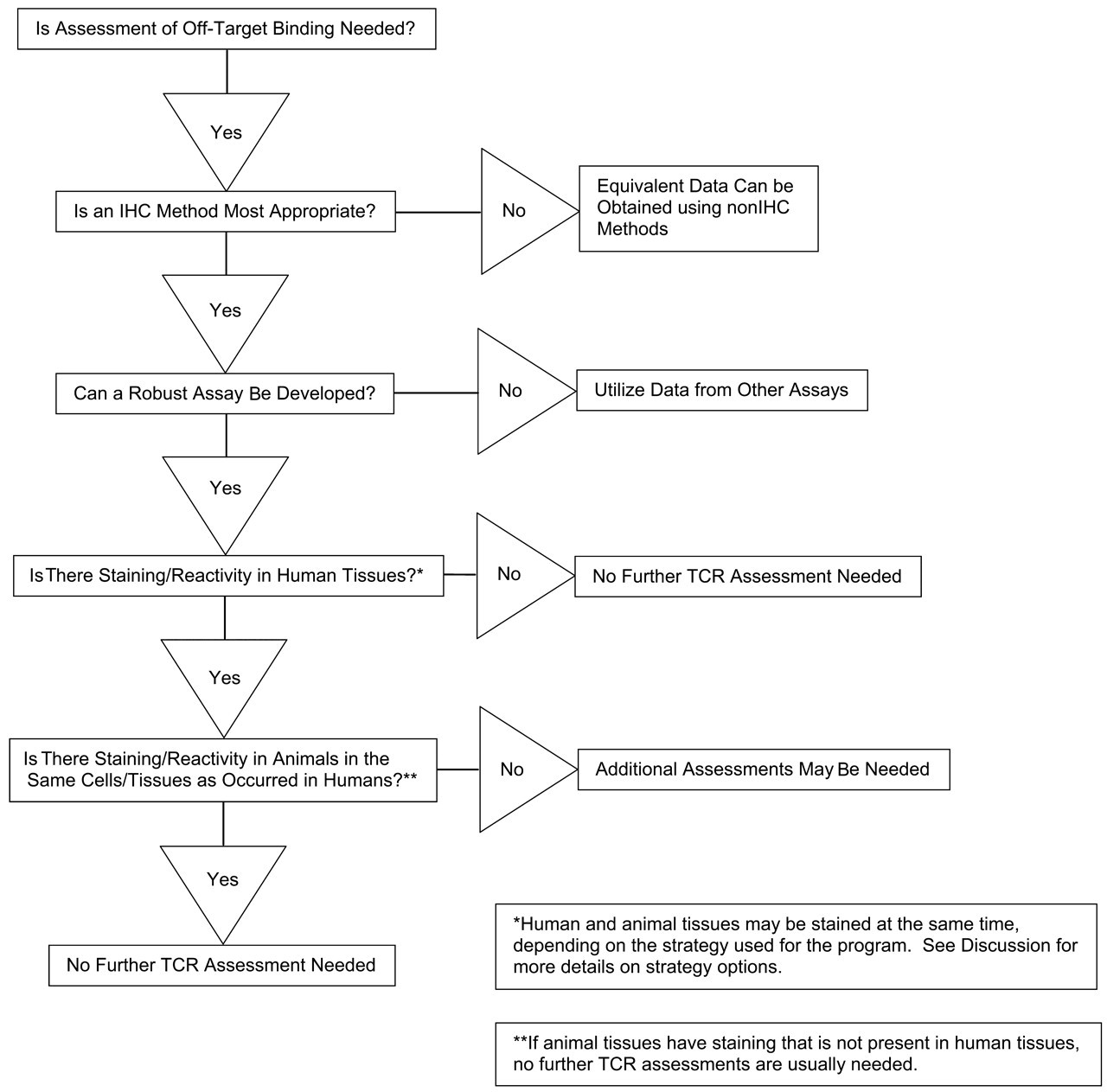
Recommended algorithm for tissue cross-reactivity (TCR) studies with antibody and antibody-based molecules.
Key Points Regarding Design and Conduct of TCR Studies
TCR studies are complex ex vivo assays conducted for antibody and antibody-based molecules, naked or conjugated. Because the purpose of TCR studies is to identify the binding of the test article to tissues ex vivo, it is recommended that TCR studies be considered for antibody and antibody-like molecules that contain a CDR. Conversely, it is not recommended to conduct TCR studies with molecules that do not contain a CDR, as the assay methodology is not designed to be used in these circumstances. Overall, the decision on whether to conduct TCR studies for any given development program, and the thought process applied throughout the study conduct, should follow a case-by-case approach.
There are several methods that can be used to evaluate the potential for antibody and antibody-like molecules to bind to tissues, including TCR studies. Historically, IHC has been the most common method. In the future, additional methods such as in vivo distribution studies in relevant species, high-throughput protein binding screens, flow cytometry, in situ hybridization, or in silico epitope mapping and binding predictions may gain favor. However, for the purposes of this discussion, the primary method referenced will be IHC.
Ideally, development of a TCR assay requires “hands-on” experience and a fundamental understanding of the science and methods used; in IHC, there are many experimental variables, often exceeding the limits of practical testing during method development. Assay development usually begins as preliminary studies using methods that have been successful with similar test articles, followed by modifications designed to optimize the assay. Whenever possible, the preliminary studies should be conducted on a limited number of well-characterized positive and negative control tissues where the target is known to be present or absent. If it appears a TCR assay can be developed, the preliminary studies should attempt to establish the reagent and staining conditions required for a robust, specific, and sensitive assay that can detect the target antigen without significant background staining. Sometimes, the methods development process can also provide an understanding of the limitations of interpretation for the assay. However, it is clear that even with extensive method development, some antibody-epitope combinations do not have the qualities to allow development of a robust IHC assay regardless of the extent of optimization; that is, specificity and sensitivity cannot be achieved based on the positive and negative control materials. There are several possible reasons for this, including low target expression and altered antigen presentation ex vivo. If reasonable efforts that have been made to develop a usable TCR assay fail, drug development should not be impeded, as the TCR assay is only one of a number of evaluations that can assist in safety assessment.
Various strategies for timing of TCR studies may be used. Generally, it is suggested that TCR studies be conducted prior to FIH dosing to identify binding to known and unknown tissue targets. If conducted in this time frame, they can be used to confirm species selection for preclinical toxicity testing. Two commonly used strategies are presented, but other strategies are possible. One strategy is to conduct preliminary TCR studies early enough in the development program to be able to use the data to establish the ability to develop a robust definitive assay and support species selection for FIH-enabling, in vivo, GLP-compliant toxicity studies. These preliminary TCR studies may use a limited number of donors and tissues. Sequential staining of human and then animal tissues may be feasible because adequate time is available (see below for more discussion). In this strategy, a definitive TCR study, which is often conducted as a GLP study at the present time, is then performed concurrent with the in vivo toxicity studies using methods developed during the preliminary TCR study. This permits a rapid study start and helps prevent unexpected method development issues that can delay programs. A disadvantage with this approach is the potential need to reconcile inconsistent results, if any occur, between multiple studies.
Another timing strategy is to conduct only a definitive TCR study concurrently with the FIH-enabling, in vivo, GLP-compliant toxicity studies. While using fewer resources, this approach runs the risk that the methods development may be more difficult than anticipated and/or that the TCR data may show unexpected findings in either the human or animal tissues that may trigger a need for additional investigations. In either case, there may be delays in the program. A modification of this strategy is to conduct a definitive study earlier in the development program, provided test article of sufficient quality is available (see below).
Regardless of timing, for the definitive TCR study it is recommended that three unrelated donors be used for each human tissue when possible and two donors be used for testing other species (if other species are needed, as discussed below). The test article in the definitive TCR study should be comparable with that used in the FIH-enabling GLP toxicity studies and the initial clinical studies, but it is rarely the good manufacturing practices (GMP) clinical material. While most definitive TCR studies are currently conducted in a GLP-compliant manner, the authors do not believe this is necessary. If the data generated in preliminary or definitive TCR studies suggest that additional donors are needed for interpretation, they should be evaluated using relevant tissues to address specific questions. Other strategies, not presented here, can also be justified; ultimately, the decision on which timing strategy to use is made on a case-by-case basis.
Another factor to consider in the TCR study design is what species to test. In the past, some developers routinely conducted TCR studies over a broad range of species prior to initiation of any in vivo toxicity studies, and then used the results from these TCR studies to help justify the appropriate species for in vivo testing. This strategy had the potential for generating irrelevant data, along with using additional resources. With the technological advances of the past decade, a more common approach has been to use product characterization and pharmacology data to select relevant species to use in toxicity studies. These data may include results from cell-based functional assays, flow cytometry, surface plasmon resonance (Biacore®, Piscataway, NJ, USA) binding kinetics, KinExA binding affinity, high-throughput screening assays for protein binding, sequence homology, and in vivo pharmacology or efficacy models. Once pharmacologically relevant species are selected, TCR studies may be conducted in the species chosen for toxicity studies to document that the animal species also have similar “off-target” or “unexpected” findings as observed in human tissues. Factors to consider in comparing staining between species include the tissues involved, cell type, and cellular localization (e.g., membrane, cytoplasmic) of binding. It should be noted that it is not expected that staining in the animals will exactly match the human. If the pattern of binding in the pharmacologically relevant species covers similar or related cell types or tissues as observed in humans, then these species should be considered sufficient, and it is not recommended to evaluate additional animal species in TCR studies. However, if the staining patterns in human tissues are not adequately covered by the animal species initially selected for testing, then the TCR data in combination with other types of information may lead to reconsideration of the species selected for toxicity testing. Extensive TCR evaluations across multiple species are not usually recommended.
Another point to consider in the design of TCR studies is whether to start testing only in human tissues or to test both human and animal tissues (from species planned for use in in vivo toxicity studies). Because the primary purpose of the TCR study is to detect cross-reactive epitopes in human tissues, initially only human tissues need to be stained. This strategy is especially useful if the target antigen is not expected to be present in normal tissues (which might occur for test articles against targets only present in diseased tissue). However, practical experience has suggested that for certain targets that are not expected to be present in normal tissues, staining (either cross-reactivity or binding to the target epitope that is unexpectedly present) can occur with some regularity. This can occur with test articles to infectious agents (Oldstone 1998) and with tumor antigens, which are often also present on some normal cells. If there are no positive human tissues (i.e., no on- or off-target staining), then additional TCR studies are usually not necessary in animals. If there is staining in human tissues, then at a minimum those positive tissues should be tested in the animal species planned for use in toxicity studies to determine if similar staining patterns are present. If no comparable staining occurs in similar cells or tissue types in the planned animal species for toxicity studies, the need for additional investigations may be considered on a case-by-case basis using TCR data in combination with other information. One disadvantage to the sequential approach is that, if additional studies are needed, they have the potential to delay the program, and for this reason, animal and human TCR studies are often performed in parallel.
In addition to the ex vivo TCR studies, it may be possible to attempt test article localization using IHC following in vivo administration. These studies are conducted to address specific issues, such as investigating whether test article location correlates with in vivo effects and therefore should not be conducted on a routine basis.
Overall, the decision on whether to conduct TCR studies for any given development program, and the design and implementation of such studies, should follow a case-by-case approach, recognizing that although IHC remains the predominant method used at the present time, there may be other ways to conduct this assessment in the future.
Key Points Regarding the Conceptual Goals, Interpretation, and Use of TCR Studies
Conceptually, TCR studies are screening assays that evaluate the binding of the test article to tissues, and in this regard they are somewhat analogous to receptor screens that are typically conducted for small molecules. TCR studies do not provide stand-alone data for use in safety assessment. They are designed to identify signals of concern that may require follow-up. Thus, if staining is observed, some additional evaluations, such as in vivo toxicity testing, are usually needed to appropriately interpret the TCR findings.
The primary goal of TCR studies is to screen for potential off-target, cross-reactive epitopes across a wide range of tissues. In this regard, TCR studies have the ability to evaluate a large number of tissues for binding to unknown epitopes in a relatively rapid manner, and there is currently no other assay available for this purpose. A secondary goal of TCR studies is to confirm known sites of target expression, as well as to identify previously unknown sites of target antigen expression (which may initially be considered to represent cross-reactivity to a different epitope). Regardless of whether the binding is off-target or on-target, from a pragmatic standpoint TCR studies can identify possible target organs of toxicity related to this binding, provide comparative information regarding the distribution of the target epitope or cross-reactive epitopes between different species, and suggest additional evaluations or monitoring that may be useful in vivo in animals and humans following administration of the test article.
Although TCR studies may provide useful information to support clinical development, it is also well recognized that they can present significant challenges related to a high frequency of unexpected negative or positive staining. Specifically, TCR studies often do not demonstrate staining in some or all of the “expected” positive tissues or, conversely, they do demonstrate staining in many tissues that are expected to be negative. When staining is not observed in tissues that are known by other methods to contain the target or a cross-reactive epitope, or in tissues that have effects in vivo, there are several possible explanations. These include the following: (1) the epitope binding the test article in other assays may not exist in the ex vivo TCR setting, possibly related to fixation or tissue handling; (2) the epitope may exist, but at expression levels too low for detection in TCR studies; or (3) the in vivo effects are not related directly to binding of the test article. From a practical standpoint, the reason for unexpected negative staining is usually not determined.
In contrast, and usually more problematic, is the positive staining of some or many tissues that are expected to be negative, that is, those that are not known to have the target epitope and do not demonstrate any in vivo effects. As demonstrated by the Case Reports above and the recent industry survey (Bussiere et al. n.d.), the positive staining of some or many tissues that do not correlate with any known epitope or any in vivo effects are common. There are several possible explanations for this: (1) the epitope that is binding the test article may be an artifact of the ex vivo condition and may not exist in vivo, (2) the epitope may exist in vivo but not be accessible to the test article (e.g., staining of intracellular structures that are not accessible in vivo), (3) binding to the epitope in vivo may not cause any biological effect, or (4) nonspecific interactions may involve other portions of the molecule outside of the CDRs (including the antibody Fc binding to Fc receptors). Because positive staining in TCR studies is a relatively common occurrence, the significance of positive staining in TCR studies that do not demonstrate effects in the in vivo studies should not be overinterpreted and generally should be of limited or no concern.
One likely reason for the unexpected positive or negative staining that frequently occurs in TCR studies is because Ab and Ab-like products and their target epitopes are not selected for their ability to produce a robust TCR assay but, rather, for their ability to cause an intended biological effect. Thus, test articles are often not good IHC reagents, despite significant efforts at developing a robust TCR assay. Because of the significant challenges, the interpretation of TCR assays should be made with the understanding that they are ex vivo screening assays that may or may not be representative of in vivo conditions. For example, as discussed in the Science of Binding section, above, producing high-quality sections suitable for histology requires drying and usually fixation of the tissue, which alters the biochemical structures in the tissue and may destroy a target epitope or create new epitopes.
The lack of staining of expected positive tissues, coupled with the presence of staining in tissues that do not have any associated in vivo consequences, has resulted in significant controversy regarding the appropriate use and value of TCR in development of antibody and antibody-like test articles, including whether TCR studies should be conducted at all. However, despite the clear technical and interpretive challenges, TCR studies on normal tissues from humans and animals have, on occasion, identified targets that alert the developer to toxicities that are evident in subsequent toxicity studies, and TCR studies have on occasion helped guide species selection for toxicity studies. Examples are provided in the case studies above and in Hall et al. (2008). Furthermore, in a few programs (<10% based on the authors' collective experience and the industry survey [Bussiere et al. n.d.]), data from TCR studies have been the only preclinical information correlating with toxicities that were subsequently observed in humans; that is, the human toxicities were not predicted by the standard preclinical in vivo toxicity studies. For these reasons, the authors support the continued conduct of TCR studies or their equivalent for determining cross-reactive, off-target binding as part of the preclinical safety assessment of antibody and antibody-like test articles that contain a CDR, as long as a reasonable method can be developed and the data are interpreted appropriately. TCR findings should be confirmed using other assays (for example, determining whether in vivo effects are observed), and all findings should be considered in the context of the overall preclinical safety data package.
When staining is observed in human and animal tissues in TCR studies, a comparison of the TCR data between species can support the relevance of the selected species. For example, when there is unexpected binding to human tissues, but similar binding is observed in the toxicity species, there may be greater comfort that any potential effect resulting from this binding, adverse or otherwise, may be revealed in the in vivo preclinical safety studies. Although infrequent, there have been some cases where a relationship between staining frequency/intensity and toxicity has been demonstrated. However, if human tissues did not show any staining, then TCR studies in animals may not be necessary.
Other Key Points
In vivo toxicity studies in relevant species remain the primary basis for assessment of toxicity for antibody and antibody-based test articles, and data from these studies should supersede in vitro or ex vivo data. Tumor antigens are often found on normal cells at some level and, depending on the sensitivity of the TCR assay, can be detected. The extrapolation of safe starting doses and target organs are best informed by in vivo toxicity studies in relevant species, and not based on TCR data. It is recognized that the slides from the TCR study are evaluated relatively early in the development process when there may be limited in vivo data, yet the report may be consulted throughout clinical development in the context of emerging findings from subsequent clinical or toxicity studies. Interpretation of the future clinical or toxicity studies and program decisions may be made based in part on the content of the TCR report. Therefore, very clear documentation of specific tissue binding of the test article, in the context of assay performance, is needed to accurately reflect the appearance of the slides and to enable informed decision making that will be useful at that time the study is conducted and in the future as the program proceeds. TCR studies are generally not recommended for surrogate molecules. The TCR assay is designed to identify off-target binding of the actual clinical candidate in tissues from humans and animal species used in toxicity studies, and information about the binding of a surrogate is not relevant to this purpose. The relevance of surrogate molecules to the intended clinical material is determined by in vitro binding and functional assays. TCR studies are not recommended for comparability assessments in the context of manufacturing/cell line changes. If necessary, preclinical comparability is based upon physicochemical characterization, pharmacokinetic, and/or bridging toxicity studies as needed. If results of the TCR study demonstrate no staining in human tissues and there is no relevant animal model to assess toxicity, for example, in the case of a specific bacterial, viral, fungal, or tumor antigens, then only limited in vivo toxicity studies may be needed.
Conclusions
Assessment for potential binding to known and unknown tissue targets is recommended for Ab and Ab-based biopharmaceuticals. A TCR study with a panel of human tissues is currently considered a standard component of the preclinical safety assessment package supporting initial clinical dosing for these molecules. TCR studies are ex vivo assays conducted for antibodies and related antibody-like products that contain a CDR to characterize the binding to antigenic determinants using complex IHC techniques. Over the past two decades the use of TCR studies has expanded and limitations have been identified. When technically feasible, TCR studies can identify potential target, off-target, or unexpected interactions in human and animal tissues.
Data from TCR studies should not be overinterpreted. TCR studies should be viewed as screening assays designed to identify potential target organs and may also be used to confirm the selection of species for toxicity studies. Any findings of interest should be further evaluated and interpreted in the context of the overall pharmacology and safety assessment data package to determine the risk of binding in human and animal tissues and the relevance of any binding to humans. The presence of ex vivo binding does not necessarily mean there will be any associated functional activity or toxicity. Because the studies are complex, scientists designing and interpreting TCR studies should have an excellent understanding of the methods used as well as the limitations of the assay in order to optimize the use of the data generated.
It may not be feasible to develop a TCR assay for some products, and there may be challenges in interpreting data in other cases. If the test article is not effective for IHC staining, the inability to develop a TCR assay should not prevent a development program from moving forward, because assays other than the TCR study may provide additional or better information about on target binding, off-target binding, and confirmation of species for use toxicity studies.
TCR studies are not recommended for assessing comparability of the test article as a result of manufacturing changes over the course of a development program or for determining tissue binding of surrogate mAbs. Overall, the requirement for TCR studies and their design and implementation should follow a case-by-case approach, recognizing that there is no single way to approach the TCR assay. The relevance and value of the TCR study should continually be assessed as experience in animals and humans accumulates.
Tissue Collection and Fixation
The initial U.S. Food and Drug Administration (FDA) points to consider (PTC) document in 1983 recommended TCR studies on both frozen and routine formalin-fixed paraffin-embedded (FFPE) tissues (U.S. FDA 1983; updated 1987, 1994, 1997). However, antigens can be damaged or rendered nondetectable by routine chemical fixation, paraffin embedding, oxidation during sectioning, or storage as paraffin blocks (Pollard et al. 1987; Blind et al. 2008). This fact, coupled with the general belief in the early 1980s that frozen tissues were the “gold standard” for immunohistochemistry (IHC) (Dabbs 2008), led the PTC documents beginning in 1987 to specify the use of only frozen tissues in TCR studies. It is currently known that frozen tissues are not optimal for all antibody targets (Nap et al. 1996; Shi et al. 2008). Regardless, frozen tissues remain the recommended and most commonly used material for TCR studies. Frozen tissue sections may have poor morphology compared with FFPE tissue sections (Dabbs 2008), and this may hamper precise localization of antibody binding in some cases. The morphology of frozen tissue specimens is highly affected by tissue collection procedures, storage conditions, and time in storage. Therefore, tissues for TCR studies should be frozen in optimal cutting temperature (OCT) compound as quickly as possible (termed quick frozen or snap frozen) following removal from the donor. After snap freezing, tissues are placed on dry ice until being transferred to more permanent storage. Freezing tissues directly on dry ice or by placing in a freezer (–20°C or –80°C) is generally not recommended as this may allow formation of large ice crystals, which can impair morphology and make the interpretation of staining more challenging or impossible. Ice crystal artifacts can also occur during long-term storage, particularly in tissue blocks that have been repeatedly warmed to cryostat temperature for sectioning then returned to storage, so tissue banks should be routinely monitored for the presence of ice crystal artifacts and damaged tissues should be culled as needed.
Frozen tissues should be stored at –80°C in sealed containers to minimize “freezer burn” damage to the tissues caused by dehydration due to sublimation of water from the tissues into the surrounding air. A cryostat is used to prepare 4 to 6 µm cryosections that are mounted on slides and allowed to dry, placed in isotonic buffer, or immediately fixed in an appropriate fixative. Dried, unfixed slides may be stored at 4°C for a few days or at –80°C for longer time periods; prolonged storage should generally be avoided as antigen or cryosection deterioration can occur even at –80°C. In rare instances, such as with some phosphorylated antigens, antigen deterioration can occur within hours after preparing the slides. In these cases, slides should be stained immediately following cryotomy. An example of rapid antigen deterioration would be IHC staining of p63, a protein antigen expressed by breast myoepithelial cells and stem cells, which can deteriorate even in stored paraffin sections (Burford, Adams, and Hameed 2009). Another example would be soluble P-selectin, which is unstable for even a short time at 4°C in ELISA (Hartweg et al. 2007). In the experience of some of the authors, unstable antigens complicate IHC assessment of TCR frozen sections less than 5% of the time (J. Rojko and C. Chan, unpublished data ).
Although fixation can damage or destroy antigens, or otherwise alter the immunostaining of the fixed tissues, some form of fixation also helps preserve tissue morphology and in some cases may immobilize soluble antigens. Thus, except in rare cases, a brief period of fixation is usually applied to frozen tissue sections after cryotomy, but before the test antibody is applied, to provide better morphology. No single fixative is best for all target antigens; thus, different fixatives such as acetone, methanol, neutral-buffered formalin, or paraformaldehyde should be evaluated. Each fixative has a different mechanism of action, and the effect of these fixatives on target (expected) and off-target (unexpected) antigen preservation should be considered during methods development. For example, aldehyde fixatives (formalin, paraformaldehyde) can denature epitopes by cross-linking N-terminal amino acids or modifying arginine, cysteine, histidine, tyrosine, and/or lysine side chains in proteins via using methylol groups, Schiff-bases, or methylene bridges (Metz et al. 2004, 2006).
As mentioned previously, drying or fixation has adverse effects on preservation of some epitopes and beneficial effects on others. When fixation or drying has an adverse effect on target antigen preservation in frozen tissue sections, TCR studies can be conducted on unfixed tissues maintained in isotonic buffer, or the fixative can be applied after the test article, and the sections are then washed prior to application of the remaining detection reagents. Other corrective strategies can include the use of aqueous mounting medium to eliminate lipid or protein loss, or structural alterations that might occur with the organic solvents used in preparation for typical permanent slide mounting media. In rare cases, heat-induced antigen retrieval strategies might be considered; these may restore antigenicity in unfixed frozen sections, consistent with the idea that some antigens are obscured when frozen tissue is cut and dried onto a slide (Kakimoto et al. 2008). For other epitopes, retrieval efforts in frozen or FFPE sections may permanently destroy the immunoreactivity (Kakimoto et al. 2008).
Although not currently recommended by regulatory guidances, for some target antigens, the evaluation of sections of FFPE tissues might be superior to the evaluation of frozen tissue sections, particularly if the target antigens are better preserved in FFPE tissues than in frozen tissues. This has been demonstrated for some cytoplasmic or nuclear antigens, while some cell surface antigens are equivalently or better demonstrated in frozen tissue sections (Nap et al. 1996; Shi et al. 2008). One concern regarding the use of FFPE sections is that they are subject to staining variability, with three major factors being time in the fixative prior to transfer to alcohols and organic solvents, temperature of the embedding process, and oxidation during storage and resectioning. Some target antigens in FFPE tissues may require the use of antigen retrieval methods that may affect tissue quality and the preservation of cross-reactive (off-target) antigens. Thus, the use of FFPE tissues in TCR studies is not considered routine and should be justified on a case-by-case basis.
Human Tissues
Regulatory documents state that normal tissues from at least three unrelated human donors should be evaluated in a TCR study to screen for individual variation (1997 PTC), and thus it is recommended that TCR studies include a diverse tissue pool with different donors, sexes, ethnicities, and ages based on scientific rationale and tissue availability. Appendix I of the 1997 PTC document provides a list of the normal human tissues to be used in cross-reactivity testing, as does Annex 1 of the recently adopted European Medicines Agency (EMEA) Guideline on Development, Production, Characterization and Specifications for Monoclonal Antibodies and Related Products (2008). A complete tissue list combining these documents is found in Table A1 .
Suggested list of normal human tissues used in tissue cross-reactivity testing.

a Suggested in Appendix I of U.S. Department of Health and Human Services, Food and Drug Administration, Center for Biologics Evaluation and Research, “Points to Consider in the Manufacture and Testing of Monoclonal Antibody Products for Human Use,” February 28, 1997 (www.fda.gov).
b Suggested in Annex I of European Medicines Agency (EMEA) Committee for Medicinal Products for Human Use Document CHMP/BWP/157653/2007, “Guideline on Development, Production, Characterization and Specifications for Monoclonal Antibodies and Related Products,” adopted by CHMP on December 18, 2008, effective date July 1, 2009 (www.emea.europa.eu).
c Evaluated in all tissues where this cell type is distributed.
In the United States, the most common sources of human tissues are tissue banks that specialize in collecting human tissues for research. Scientists conducting human TCR studies should work with sourcing tissue banks to ensure the best collection procedures. Clinical information provided with standard specimens often includes age and gender and may also include race, clinical history, and/or cause of death (autopsy specimens). Surgical biopsy samples are a preferred tissue source, if frozen within a relatively short time after removal, but are not always readily available for normal tissues (and are essentially unobtainable for some vital tissue organs, such as heart and central nervous system tissues). Additionally, because surgical tissues are often removed in conjunction with a disease process, biopsy tissue may not be normal even when designated as “normal adjacent tissue.” Therefore, many to most human samples used in TCR studies come from adult (eighteen years and older) human autopsy donors and may be less than optimally preserved. The time to collection can impact some antigens as some degrade in minutes or hours. However many epitopes are not affected by autolysis during the first twenty-four hours postmortem (Maleszewski et al. 2007), and autopsy tissue may adequately display off target antigens. Specialty tissues (e.g., pediatric, fetal, diseased, or defined ethnic or racial origin) may be difficult to obtain and are generally not used in a standard TCR study unless there is a specific scientific rationale for their inclusion (e.g., when the therapeutic indication is for pediatric patients or reproductive toxicity studies are not feasible [e.g., target expressed only in humans and chimpanzees] and fetal exposure is possible based on the clinical indication). Regardless of the source, it is recommended that tissues used in TCR studies be fully consented and de-identified or anonymized so that tracking back to individual donors is not possible.
Animal Tissues
As noted previously, the 1997 PTC document states that for biological products, preclinical testing should be accomplished in a relevant species and that “a comparison of in vitro cross-reactivity in tissues from different species is important in determining the most relevant animal for subsequent toxicology testing.” In current practice, much of the pharmaceutical industry determines the most relevant species using methods other than TCR (such as in vitro cell based functional assays, receptor binding assays, etc.) but often conducts TCR testing on tissues from animal species that are being used or considered for use in preclinical toxicology studies, in addition to human tissues.
Animal tissues are usually collected specifically for research purposes shortly after death and can be renewed periodically to minimize the use of old tissues, which may have a greater incidence of antigen deterioration and morphologic damage due to ice crystal damage. Therefore, animal tissues are often better preserved and of higher quality than human tissues collected for TCR studies. Even so, it is important that groups collecting animal tissues for TCR studies make an effort to ensure the best collection and storage procedures possible, as better-quality tissues allow for better evaluation of potential cross-reactivity.
There has been debate as to the appropriate number of donors for animal TCR studies. It is now recommended that for relevant animal species, at least two donors be used. In the case of highly inbred strains of rodents and rabbits, the animals are genetically very similar and would be expected to have minimal staining variability when it comes to cross-reactivity, so the evaluation of tissues from two donors would be appropriate. In the case of nonhuman primates and other species, at least two unrelated donors should be used whenever possible for each tissue, though additional donors can be included for selected tissues when considered necessary to clarify whether there is individual animal heterogeneity in epitope expression or to study unusual findings in human or animals in vivo where heterogeneity of response is suspected.
Tissue Microarrays
The use of tissue microarrays has increased in a number of research settings, and tissue microarrays that contain frozen or FFPE tissue core samples of all the required tissue types (PTC document and EMEA 2008) are commercially available. Tissue microarrays may be useful for a preliminary evaluation of cross-reactivity, but they have significant disadvantages minimizing their usefulness in TCR studies. The tissue cores are small (usually ≤ 2 mm each) and often do not contain the variety of tissue elements present in larger samples. As an example, even if separate 2 mm cores are taken from renal cortex and medulla, they may not contain all critical tissue elements, including medullary tubules, stroma, papilla, proximal and distal tubules, and glomeruli. In addition, individual cores may vary in depth, and some may be exhausted earlier than others or may come off the slide during staining, leading to loss of those tissue elements for evaluation and hampering overall interpretation. Therefore, if used, tissue microarrays need to be carefully evaluated to determine if they include the relevant tissue components in all test and control sections. In most cases, whole tissue sections are more reliable. Tissue arrays may be more useful in special situations where a single tissue needs to be rapidly evaluated from a large panel of donors or for preliminary screening studies.
Controls
Positive and Negative Target Controls (Control Materials)
A minimum criterion for an acceptable IHC assay is that the test article needs to stain the positive (target) control material but not irrelevant (negative, nontarget) control material, and the negative control antibody does not stain either the positive or negative control materials. The selection of appropriate positive and negative control materials (tissues, cells, purified protein) is often the first crucial step in a successful TCR evaluation. When possible, an ideal positive control is a normal tissue with well-documented expression of the target antigen. However, there are situations where choosing a normal tissue as the positive control sample for the study is not possible. For example, the expression in normal tissues might be below the level of sensitivity of the TCR assay, or the location of the target in normal tissues may not be known. In cases where the target is a soluble antigen, it might still be detectable within cells (i.e., prior to secretion), on the cell surface, in blood or lymphatic vessels, or attached to extracellular matrix; or alternatively it might rinse out of the tissue during the staining or may be terminally clipped in the plasma or not exist in cells.
In cases where a normal tissue control cannot be found, alternative positive control tissues that have a higher level of target antigen expression than normal tissues may be useful. Examples include neoplastic tissues, disease tissues, or tumor xenografts. When positive and negative tissues can be identified, these provide the best control materials, as the control tissues contain multiple cellular and noncellular tissue elements that are frozen, cryosectioned, dried, and fixed similarly to the human and/or animal tissues used as the TCR test system.
Other options for positive and negative (target) control materials are a cell line or purified target proteins. This could include cell lines that either express the target antigen naturally or have been transfected to express the target antigen. The level of expression in transfected cell lines may be above or below normal target tissue levels and can result in the development of methods that are not optimized for staining in normal tissues. In addition, cell lines do not contain multiple cell types, which can make target-specific binding easier to interpret but also can result in a failure to identify staining issues that might occur in test tissues (such as background or nonspecific staining). These issues potentially can affect the results and interpretation of the study. For this reason, if a TCR method has been developed and tested on positive and negative control cell lines, it is recommended that attempts be made to transfer the method to positive and negative tissue controls at the earliest opportunity and, if necessary, modify the method to work optimally in tissues.
Purified target and control antigens may provide adequate control materials especially for some types of antigens, such as soluble targets not immobilized by fixation (i.e., some cytokines), when the target is expressed at levels below the sensitivity of normal IHC assays in normal or diseased tissues or when a tissue or cell-based control cannot be developed. Purified antigen is often the best solution when the test article is directed against an agent or substance not normally found in tissue (i.e., Drug Enforcement Agency (DEA)–controlled drug substances or infectious/toxic agents). The purified target antigen (positive control material) can be covalently linked to a support medium such as agarose or polystyrene beads (similar to an affinity column) and the beads embedded and treated similarly to tissue. Alternatively, the purified antigens can be spotted directly onto ultraviolet (UV)–activatable, plastic resin-coated slides (UV activation causes the resin to polymerize, adhering the control material to the slide). It should be noted that with the purified antigen spot method, the positive and negative controls are not treated the same as a tissue, and this may affect the staining and assay development. In addition, without a firm covalent binding, some antigens spotted onto slides rinse off during staining.
The selection of the appropriate negative control material is equally important to the proper design of TCR studies, as it is critical that the test article demonstrates specificity and does not bind to the negative control material. The negative control material ideally should be similar in format to the positive control material but should not express or contain the target antigen or any off-target antigens to which the test antibody might cross-react. For tissue-based controls, the negative control material is typically a tissue or a tissue element that does not express the target antigen and to which the test antibody does not bind. In some cases, both positive and negative control materials can be found in a single tissue; that is, one can use positive and negative elements from a single tissue. This strategy is a realistic approach for target antigens with widespread expression. For cell lines, the best negative control material may be a cell line of similar or identical origin as that of the positive control cell line that does not express the target. In the case of transfected cells, the negative control cell line is often the nontransfected or mock-transfected cells of the same type as the positive control cells. If a purified target antigen is used as the positive control material, then the negative control material should be an irrelevant antigen that is nonreactive with the test antibody.
Isotype Control Antibody (Negative Control Antibody, Control Article)
In the TCR study, it is important to be able to distinguish (as best as possible) specific binding mediated through interaction of the complementarity-determining region (CDR) from non-CDR-mediated binding such as binding via an Fc receptor. In addition, some tissue components (for example keratinizing epithelium, colloid, and collagen) appear to be associated with nonspecific antibody binding through unknown mechanisms. While nonspecific interactions can be minimized with blocking and carrier protein (strategies discussed later in the text), an irrelevant negative (isotype-matched) control antibody is used to facilitate interpretation of which interactions of the test article are likely to be specific and which are not. This is particularly important as increasing antibody affinity by antibody maturation strategies can lead to increased nonspecific staining (Wu et al. 2007). In the case of whole antibodies, a human IgG of the same isotype as the test article (IgG1, IgG2, etc.) should be used as the negative control antibody. There are isotype (negative) control antibodies for most of the common human immunoglobulin isotypes (IgG1, IgG2, IgG4, IgM, etc.) commercially available, although they often have minor structural differences from the test article beyond having a different CDR. When the test article is a mutated antibody or an antibody-like molecule, the optimal negative control should be as structurally identical to the test article as possible. However, structurally similar negative control proteins are often unavailable from in-house or commercial sources, and in these cases the most scientifically rationale negative control protein should be selected. Regardless of the source and structural similarity of the negative control with the test article, the minor structural differences between the negative control and test article, beyond having a different CDR, may translate into a different nonspecific tissue interactions from the test article and should be considered during methods development and slide evaluation (see below). If the test article is labeled, a similar label should be on the negative control antibody.
Selection of the Form(s) of the Test Article (Test Antibody) and the IHC Method Used in TCR
No one method is optimal for all the various types of antibody and antibody-like test articles, nor is there a way to predict the best method for any given test article. In many cases, multiple methods are evaluated to determine the optimal method for each individual antibody test article.
At the present time, most candidate antibodies are either humanized or fully human antibodies. In a typical TCR study, test article staining concentrations range from 0.5-50 µg/mL (see below), while endogenous IgG is present intravascularly and extravascularly in human tissues at much higher concentrations (typically about 10 mg/mL in serum; Roitt 1994; Perkins 1999). Thus, detecting the binding of a human mAb test article in human tissues in the presence of such high levels of endogenous human antibodies can be challenging. Excessive background staining usually precludes interpretation of staining if a conventional (standard indirect immunoperoxidase) approach were used (i.e., first incubating the tissue with the human mAb test article and then detecting binding using a labeled anti-human antibody detection reagent). Most TCR studies use one of three basic methods to circumvent this problem: Direct immunoperoxidase with biotin (horseradish peroxidase [HRP] or other)–labeled human antibody test articles Indirect precomplexing immunoperoxidase with unlabeled human antibody test articles Indirect sequential immunoperoxidase with fluorescein isothiocyanate (FITC)- or Alexa Fluor®–labeled test articles
Direct immunoperoxidase methods rely on detection of a label or detection enzyme covalently linked to the test article (primary antibody), while indirect methods use a secondary and sometimes tertiary (rarely quaternary) antibody to detect the test article (A1 and A2, courtesy of Shari Price-Schiavi, PhD, DABT). Secondary antibodies bind to the test article, and tertiary antibodies bind to the secondary antibodies. The secondary or tertiary antibodies may be linked either to biotin or a reporter enzyme and are used to increase assay sensitivity. Other methods employing anti-idiotypic secondary antibodies or anti-HuIgG4 secondary antibodies are employed in some instances (e.g., when the test article is a HuIgG4 isotype).
Schematic representation of direct avidin-biotin complex (ABC) immunohistochemical staining. For tissue cross-reactivity (TCR) assays, the direct ABC procedure is useful for mAb test articles that retain specificity and affinity when conjugated to a biotin label. The biotinylated mAb test article binds to its target (or off-target) antigen in tissue sections. Next, the horseradish peroxidase (HRP)–conjugated ABC complex is applied, followed by a substrate chromogen (typically diaminobenzidine [DAB]). The strong affinity of avidin for biotin and the numbers of biotin-avidin-HRP molecules in the ABC complex can provide up to a 4- to 8-fold amplification compared to HRP-conjugated mAb test articles. HRP cleaves the DAB substrate, allowing the deposition of precipitated brown stain to mark the site of the binding of the mAb to its target (or off-target) antigen. Slides are counterstained with hematoxylin, which detects nuclei, and interpreted by a pathologist.
Schematic representation of indirect precomplexing immunohistochemical staining. For tissue cross-reactivity (TCR) assays, the indirect precomplexing procedure is useful for unlabeled test articles. The unlabeled primary test article is precomplexed with the labeled (e.g., biotinylated) anti-human IgG secondary reagent. Then, excess human gamma globulin is added to “mop up” any unprecomplexed labeled secondary antibody, and the precomplex is applied to the cryosections. When the secondary reagent is biointylated, the staining reaction is detected using a horseradish peroxidase (HRP)–conjugated avidin-biotin complex (ABC) complex as described in A1. The use of a secondary antibody can provide up to an additional 3- to 10-fold amplification of staining compared to the direct ABC procedure in A1. This allows detection of lower-display antigens (fewer antigens per tissue or fewer binding sites per cell) compared to the direct ABC procedure.
The decision on which method(s) to attempt first should be based on a combination of experience with similar test articles and experience of the laboratory. The goal is to have a sensitive, specific, reproducible TCR assay, regardless of the method developed.
Direct Immunoperoxidase with Biotin (HRP or Other)–Labeled Human Antibody Test Articles
As applied to TCR studies, direct IHC methods use the antibody candidate (test article) covalently linked to a detection enzyme (e.g., HRP, alkaline phosphatase) or some type of marker (e.g., biotin) as the primary antibody. Direct methods eliminate the need for an anti-human IgG secondary antibody, and thus have technical advantages for use in human tissues. Enzyme-labeled primary antibodies are detected with the appropriate chromogen substrate (e.g., diaminobenzidine [DAB] for HRP, nitroblue tetrazolium chloride [NBT] for alkaline phosphatase). Biotin-labeled primary antibodies are typically detected using avidin (or streptavidin) linked to a detection enzyme followed by the appropriate chromogen substrate (A1); the biotin-avidin loop provides amplification of signal compared with HRP-labeling alone. However, as no secondary antibody is used, there is no additional amplification and assay sensitivity is less than with other (indirect immunoperoxidase) methods. This may be of particular concern when the epitope is expressed in low concentrations or at low density and detection in tissue requires a high level of amplification. More sensitive staining procedures (e.g., using fluorescein- or Alexa Fluor®–labeled test articles described later in the text) may provide greater amplification and can reveal staining of low-display epitopes (Hall et al. 2008).
The labeled antibody test article is not identical to the unlabeled test article (the actual drug candidate) and therefore may not behave identically. Labeling of the primary antibody can lead to no effects on staining, increased specific or cross-reactive staining (Panyutich et al. 1993), or a loss of affinity and staining (Siena et al. 1991). Labeling of an antibody can also differentially alter binding to the target or off-target epitope. The target antigen-binding characteristics of the labeled antibody should be verified by comparing the binding of the labeled and unlabeled antibodies to the target epitope using techniques such as surface plasmon resonance (Biacore®, Piscataway, NJ, USA), flow cytometry, ELISA, or KinEXA assay. Under ideal circumstances, the binding of the antibody to the target would be the same as the unlabeled antibody within the reproducibility of the assay. However, from experience, labeling of the test article may alter binding affinity, which may or may not translate to altered TCR staining patterns for the target and off-target epitopes. The effect of labeling can have variable effects on the antibody as shown in the following examples. (1) An unlabeled antibody test article showed good reactivity with the human and cynomolgus monkey target epitope, while the fluoresceinated test article had equivalent reactivity with the human target but reduced reactivity with the monkey target using flow cytometry. (2) Biotinylation of a test article increased its cross-reactivity with off-target cytoplasmic structures. (3) Labeling of a test article led to only a 5-10% change in binding affinity but led to staining of new targets that were not detected by the unlabeled antibody. (4) Conversely, other test articles can be less affected; in one case, even though binding affinity was reduced by 90%, the cross-reactivity staining patterns were not affected. These examples demonstrate that the effect of labeling on target binding and staining in the TCR study is not predictable and binding characteristics should be documented for each labeled test article.
Methods other than measuring binding affinity can also be used to show equivalency. It may be possible to run a head-to-head comparison of binding to a limited panel of nonhuman primate tissue (where the test antibody can be differentiated from the endogenous monkey IgG) and document whether labeling caused any meaningful differences in binding. In the final analysis, even if labeling affects affinity, it is important to note that direct labeling may represent the best method for conducting the TCR study, and from a practical standpoint, the ability of the labeled test article to appropriately stain the positive and negative control material is the best way to determine whether the labeled material is acceptable for use.
Indirect Precomplexing Immunoperoxidase with Unlabeled Human Antibody Test Articles and Anti-Human IgG Secondary Antibodies
As almost all antibody candidates currently under development are humanized or fully human antibodies or antibody fragments, they all contain human elements constituting a significant portion of the antibody. Standard (sequential) indirect IHC staining using a biotin-labeled anti-human IgG secondary antibody can result in excessive background staining of endogenous human IgG present in the tissue sections. To overcome this difficulty, the unlabeled test article (candidate antibody) and a labeled anti-IgG secondary antibody are noncovalently precomplexed together, and then applied to tissue specimens (A2). It is important to note that the precomplexing method uses the actual candidate antibody, theoretically eliminating concerns that a labeled antibody might bind differently in the TCR study.
Care must be taken to properly adjust the ratios of the primary antibody, secondary antibody, and blocking immunoglobulin in the precomplex mixture so that the optimal binding to the target antigen is achieved. Partially bound or unbound secondary antibody can cross-react with endogenous IgG and generally results in higher background staining compared with using directly labeled antibodies. Additionally, since different secondary antibodies (particularly from different host species) have different affinities for test articles, it may be important to evaluate the use of different secondary antibodies from multiple host species in the precomplexing procedure.
It should be noted that modifications (including mutations) are sometimes intentionally introduced in candidate antibodies to alter their function, such as to reduce antibody-dependent cell-mediated cytotoxicity (ADCC) and complement binding. In some cases, these modifications may alter the ability of secondary antibodies to recognize the primary antibody test article.
In summary, the precomplexing method is technically more challenging than some other methods. In the experience of the authors, the precomplexing method is successful in detecting positive control material in only about 33-50% of cases because of high background staining and/or a lack of staining of the target. Failure to develop a successful assay may be related to the formation of complexes that are too large for effective binding or whose CDR is blocked from binding to the target epitope. Other difficulties can arise when modifications in the antibody test article hamper binding by the secondary antibody. Precomplexing strategies have also been addressed by Hall et al. (2008).
Indirect Sequential Immunoperoxidase with FITC- or Alexa Fluor®-Labeled Human Antibody Test Articles and Antilabel Secondary Antibodies
An alternative to precomplexing is to conjugate the primary human test article to FITC or Alexa Fluor®, and then use a secondary antibody that detects the FITC or Alexa Fluor® label. The addition of the secondary antibody confers increased sensitivity compared to direct immunoperoxidase methods with biotin- or HRP-labeled test articles. Modifications in the test article antibody framework (e.g., modifications in the Fc region designate to reduce ADCC or complement binding) generally do not affect the ability to label such molecules. However, as described above, the labeled test article may have altered binding affinity and/or TCR staining patterns compared to the unlabeled test article, and efforts should be made to verify that the labeling of the test article does not adversely affect binding.
Anti-idiotypic Antibody Methods
If anti-idiotypic antibodies (antibodies specifically generated to detect the hypervariable region of the test article) are available, they may be useful as a secondary antibody to detect test article binding to the tissues, particularly if the anti-idiotypic antibody is directed against a portion of the hypervariable region that does not overlap with the binding site. Binding of the secondary anti-idiotype antibody to the primary antibody (test article) is detected with a biotin- or HRP-labeled tertiary antibody directed against the immunoglobulin of the host species of the anti-idiotype antibody (generally mouse or rabbit). Anti-idiotype antibodies are useful to detect binding of human antibody test articles to human and nonhuman primate tissues but are not useful to detect binding to tissues from the species in which the anti-idiotype antibody was generated because the tertiary antibody will bind to endogenous antibody in this case.
Anti-human IgG4 Secondary Antibody Methods
Antibody test articles with human IgG4 as their isotype backbone are finding more widespread use. Human IgG4 has the lowest expression among the four IgG subclasses and makes up approximately 4% of circulating IgG; therefore, normal tissues have only low concentrations of endogenous human IgG4 present. In many cases, it is possible to use a biotinylated anti-human IgG4 secondary antibody in a standard indirect immunoperoxidase assay to detect binding of the unlabeled IgG4 test antibody on tissues with low background IgG4 concentrations.
Other Antibody-like Molecules
A number of novel antibody-like molecules are currently in various stages of development, and more anticipated in the future. As noted above, TCR studies are recommended for molecules that contain a CDR. For antibody-like molecules with a CDR that have unique structural properties, creative alterations to standard TCR methods may be necessary to develop a usable assay.
Blocking Strategies to Reduce Background and/or Nonspecific Staining
Ideally, the methods development studies will explore a number of blocking reagents to identify a method that reduces background and/or nonspecific staining as much as possible. Various proteins (serum, albumin, and/or casein) are applied before the application of the primary antibody to reduce nonspecific attachment or adherence. If the applied protein (protein block) contains immunoglobulins, these will bind to tissue Fc receptors and inhibit nonspecific (non-CDR-mediated) binding of the test article or negative control antibody. Other applied proteins can saturate many sites in tissues that bind proteins, nonspecifically reducing the likelihood that the test article, negative control antibody, or detection reagents will nonspecifically adhere to tissue components. While these proteins may also coat or bind to the target antigen, they generally have little impact on binding of the test article for the target antigen because the high-affinity binding of the test article should displace any low-affinity binding of the protein block.
Blocking the endogenous counterparts of detection enzymes such as HRP or alkaline phosphatase, or detection markers such as biotin, greatly reduces background staining. Endogenous myeloperoxidase (mast cell, eosinophil and neutrophil granules) or thyroperoxidase (thyroid follicular epithelium) reacts with the DAB chromogen unless blocked by hydrogen peroxide or other peroxidase blocking solutions. Endogenous biotin blocks are needed to prevent detection of endogenous biotin in cryosections, but are not required in FFPE as biotin activity is lost during paraffin embedding.
Assay Control
An assay control is often included for each tissue in the TCR study to provide information regarding background staining attributable either to the tissue (e.g., endogenous/exogenous pigments) or to the staining method (e.g., residual endogenous peroxidase). In the assay control slides, the primary antibody (test article or negative control antibody) is omitted from the staining reaction.
Tissue Acceptability/Staining Control
Human test tissues need to be confirmed as acceptable for each TCR study by staining with a tissue acceptability antibody directed against a ubiquitous antigen. This is important as the time between death and collection of human (but not usually animal) tissues may result in antigen or tissue degradation. The tissue acceptability/tissue staining control provides evidence that the TCR test system tissues have been appropriately preserved to allow for IHC staining but does not guarantee that the individual target epitope recognized by the test article was also properly preserved. However, generally when the tissue acceptability control antibody does not stain a tissue, neither will the test article.
Antibodies against CD31 (PECAM-1), transferrin receptor (CD71) or β2-microglobulin may be used as tissue acceptability control antibodies. In practice, a slide of each test human tissue is prepared and stained with the selected tissue acceptability control antibody. If the tissue does not stain with the tissue acceptability control antibody, it is not considered acceptable for further testing.
Concentration Selection
While regulatory documents such as ICH S6 (1997) and the 2008 EMEA guidance do not define the test article concentrations to evaluate, the 1997 PTC suggests “several concentrations” should be evaluated in TCR studies and that affinities as well as expected peak plasma concentrations should be considered when choosing concentrations for TCR studies. Although not stated in the PTC document, tissue concentrations may also be factored in if known. The 1997 PTC also states that one concentration to test is defined as the “ideal” concentration, which is the lowest concentration that produces maximum binding to the target antigen. The ideal concentration should be identified during methods development by evaluating a range of test article concentrations. From experience, this ideal concentration has historically been in the range of 2-10 µg/mL. Depending on avidity conditions, this concentration range may be inappropriately high for antibodies with affinity in the picomolar or femtomolar range, and each test article must be evaluated on a case-by-case basis.
The PTC document does not further define what other concentration(s) should be tested. However, a paradigm has evolved for most definitive TCR studies supporting FIH dosing in which at least one concentration that is higher than the defined ideal concentration is tested to help ensure that potential targets with weaker binding affinities are detected, provided this concentration does not result in excess nonspecific or background staining. Frequently, the highest feasible concentration is about 5 to 20 times the ideal concentration, which historically has resulted in a high concentration in the range of 20-50 µg/mL. However, these general guidelines may not be applicable in all cases, depending on avidity conditions and with antibodies engineered for higher affinities. Some test articles have a very narrow working range of concentrations, with only a 2- or 3-fold difference between the ideal concentration and the highest feasible concentration, and thus, for these test articles, the range is smaller. For other test articles that work in the nanogram, picogram, or femtogram/mL concentrations, the working range may be greater and more flexible, allowing the high concentration to be 5 to 20 times the optimal concentration. When the target antigen is not found in tissue or is below limits of detection, a typical approach would be to use either the highest feasible concentration or 20 to 50 µg/mL and choose a lower concentration for the second test condition. While the testing of two concentrations (i.e., the ideal concentration and the highest feasible concentration) in the definitive TCR study that supports FIH dosing is the most common paradigm currently in use, additional concentrations can also be tested if considered necessary. In addition, in the experience of the authors, the highest test article concentration tested in TCR studies generally should not exceed 50 µg/mL to preclude the risk of nonspecific background staining.
It should be noted that the highest feasible concentration is usually lower than the predicted highest serum and/or tissue concentration of the test article, but concentrations of the test article above the highest feasible concentration often result in excessive nonspecific or background staining, which precludes evaluation of higher concentrations. Thus, in most cases, attempting to use test article concentrations in TCR studies that are similar to human serum or tissue concentrations is not feasible.
Study Design, Conduct, and Validation
Once an acceptable TCR method has been determined in preliminary studies, a panel of tissues from humans and/or animals can be evaluated. A typical study design following regulatory recommendations would evaluate a complete set of human tissues (see Table A1) from three unrelated donors per tissue. For animal studies, two unrelated donors per tissue are usually adequate. Subsequent studies may also be conducted, usually using a limited number of tissues and donors to address specific issues, and usually under non-GLP (good laboratory practices) conditions.
Serial sections of each tissue are made and stained with the various control and test antibodies using at least two different staining concentrations. Inclusion of an assay control (omission of primary antibody test article) allows determination of the level of background staining. The use of serial tissue sections allows careful comparison between the same areas of the tissue to assist in interpretation of staining. Replicate human sections also are stained with the tissue acceptability antibody to ensure that the tissue is acceptable for IHC staining. Method validation is based on the performance of the test article and negative control antibody with the positive and negative control materials throughout the multiple staining runs required to conduct the TCR. A typical human TCR study with two staining concentrations generates about 700 to 750 IHC slides; usually multiple staining runs are required. Variability between staining runs should be documented in the study report if it has the potential to affect study interpretation. With a few test articles, the best TCR study that can be developed is not robust and may have staining run failures because of limitations of the target or the test article. Documenting these limitations in an individual TCR study can influence how much weight is placed on TCR results from that study.
Review of the TCR Study
The evaluation of slides from TCR studies is generally conducted by a pathologist familiar with IHC. The study pathologist is responsible for ensuring the tissues and overall study are adequate for assessment and can be adequately interpreted. This is done based on a number of evaluations described in more detail below, including assessment of tissue adequacy and acceptability, followed by determination of background versus nonspecific versus test article-related staining. An overall interpretation of the study results and possible relevance to in vivo can then be prepared. A pathology peer review is usually conducted for TCR studies that support initial clinical trials.
Assessment of Tissue Adequacy and Acceptability
The pathologist evaluates each tissue for adequacy by confirming the specimen is appropriately fixed; is of sufficient size for evaluation; contains representative tissue elements; and is free of excessive sectioning or freezing artifacts such as folds, cracks, or ice crystal damage. Tissue acceptability is confirmed by positive staining in the slides stained with the tissue acceptability control antibody (usually only human) and indicates the additional serial sections from that tissue stained with the negative control and test article are suitable for evaluation. Tissues should be rejected for evaluation if there is no staining by the tissue acceptability antibody, if representative tissue elements are lacking, or if sectioning or freezing artifacts preclude interpretation of test article staining. Rejected tissues should be replaced by alternate tissues when available.
Background Staining versus Nonspecific Staining
Background and nonspecific staining are common in TCR studies, reduce assay sensitivity (signal:noise differentiation), and can hamper assessment of both target and off-target binding of the test article. Although background and nonspecific staining are often used interchangeably, for the purposes of this article, they are defined as representing separate events. Background staining refers to staining intrinsic to the tissues (e.g., endogenous or exogenous pigments) or to the IHC method used (e.g., incompletely blocked endogenous peroxidase, secondary antibody binding to residual endogenous IgG in precomplexing methods). Some tissues/cells inherently tend to have higher background staining, especially kidney tubular epithelial cells and hepatocytes, related to high levels of endogenous pigments (e.g., lipofuscin or hemosiderin) or transport of endogenous IgG. Other tissue elements (e.g., peripheral blood eosinophils, mast cells, thyroid follicular epithelial cells) have higher background staining due to high levels of endogenous peroxidase.
In contrast, nonspecific staining refers to excessive, often indistinct, staining attributed to the primary antibody (test article or negative control antibody) mediated by nonspecific (non-CDR-mediated) attachment (or adherence) of the antibody protein molecule to various cellular or extracellular tissue elements. This can occur through binding to Fc receptors or through nonspecific hydrophobic or electrostatic interactions with the tissues. In addition, some antibodies have higher intrinsic nonspecific reactivity than others. If the antibody is labeled, using high concentrations of normal human IgG can sometimes decrease the nonspecific interaction with tissue but still retain specific binding. Excessive primary or secondary antibody concentrations can increase the likelihood of nonspecific staining, which often limits the maximal feasible concentration selected as the higher staining concentration in TCR assays.
Regardless of the cause, the presence of excessive background or nonspecific staining can make it impossible to determine if the observed staining represents true binding of the test article to the target or a cross-reactive epitope, or is an artifact of the assay. Ideally, the methods development studies will explore a number of blocking reagents to identify a method that reduces background staining as much as possible, and the various controls can help differentiate nonspecific from real staining.
Test Article Staining
The study pathologist relies on careful evaluation of the staining observed in the test article-stained slides compared with the isotype control (negative control antibody) and/or assay control slides to attempt to distinguish specific (CDR-mediated, test article-related) staining from nonspecific and/or background staining. The study pathologist determines the cell type or tissue elements stained, the subcellular (e.g., cytoplasmic, membrane, or nuclear) or extracellular location of staining, the intensity of staining, and the frequency and/or density of staining for each stained tissue component. Identifying the subcellular or extracellular location of staining is as important as identifying intensity and frequency. As reviewed by Hall et al. (2008), membrane staining (whether target or off-target) may be of greater concern because test articles are generally considered to be able to access these sites in vivo. In contrast, there may be less concern for staining of cytoplasmic or nuclear structures because they are unlikely to be in contact with the test article in vivo. However, some membrane targets are synthesized in the cytoplasm or in cytoplasmic organelles (e.g., Golgi), and the detection of ample target in a cytoplasmic structure such as granule may be a more sensitive indicator of cells containing target than detection of a narrow band of receptors at the cell surface or membrane that results in weak or no membrane staining. Other morphologic criteria including the quality (e.g., crisp versus indistinct staining) or precise histologic location (e.g., perivascular versus subepithelial) of staining are also used.
Staining is usually characterized using a scoring system that assesses staining intensity coupled with a description and/or a score of the distribution or percentage of tissue or cells affected, although different scoring systems can be used. For example, one commonly used system uses a 5-point scale for intensity (0 [negative] = no stained cells; 1+ = minimal/light stain; 2+ = mild/light-medium stain; 3+ = moderate stain; 4+ = marked/intense/dark stain) and for distribution (frequency, density) (0 [negative] = no stained cells; 1+ = rare/occasional cells/types of cells or <25%; 2+ =small number or 25-50%; 3+ = medium number or 50-75%; 4+ = frequent/large number of cells/types of cells or >75%). Scoring of extracellular tissue elements (e.g., collagen fibrils, intravascular proteinaceous material) is done similarly except that the frequency scores are based on the approximate percentage of that tissue element stained. In addition, the pathologist provides comments on background staining. Alternatively, adapting intensity and distribution scoring systems, such as those widely used in human pathology for HER2 or EGFR, may have some advantages in that they have been standardized by the medical field and may help translate the staining into intensities familiar to a medical reviewer.
Because human TCR studies include 32 to 37 different tissues from 3 donors, a thorough pathology interpretation can also provide information regarding the biology of the target epitope in vivo. Test article staining patterns can reflect uptake of the epitope by a cell type (e.g., receptor-mediated endocytosis of bound ligand, uptake of shed cancer antigens by macrophages) and/or secretion into the external milieu well as the more typically considered cell types/subcellular locations of target epitope synthesis/expression.
Strategies to Determine Specificity of Binding
In the event staining believed to be related to the test article is observed, studies may be conducted to further evaluate the staining. Blocking studies using the target antigen or unlabeled antibody to prevent binding of the primary antibody can be conducted (Hall et al. 2008) and can be used to differentiate off-target binding (e.g., CDR-related binding to unexpected or cross-reactive antigens) from nonspecific interaction(s) of the test article (e.g., FcR-mediated or nonspecific binding).
Blocking studies are generally performed in two different ways. Inhibition assays mix various concentrations of purified antigen with the test article and apply the mixture to tissues to evaluate whether specific binding can be inhibited. Antibody competition assays are conducted to evaluate whether IHC staining by labeled test articles is specific or nonspecific. The unlabeled test article is incubated with the tissue prior to application of the test article, or may be included in the labeled test article diluent. If addition of the unlabeled test article does not compete away the IHC staining, the staining is considered nonspecific. If addition of the labeled test article does compete away the IHC staining, the staining is considered to be test article-related and potentially CDR-mediated, although FcR or other nonspecific binding cannot be excluded. In a different variation, a different cross-competing antibody is used in place of unlabeled test article. It is a good practice to include the unlabeled test article or competing antibody in the solution containing labeled test article. The outcome of these types of studies is usually a reduction in staining by the cross-competing antibody rather than a complete blocking of staining.
Several cautions should be made regarding interpretation of data from blocking studies. TCR blocking studies generally can only be conducted across a narrow range of staining concentrations. Blocking with a soluble antigen works only if the test article has a similar or higher binding affinity for the soluble target compared with the target affixed in tissues. For some test articles with a high affinity for tissue or solid substrate targets, no amount of soluble antigen will block, possibly leading to an erroneous interpretation that the CDR is not involved in the staining. In addition, the concentration of the antigen needs to saturate all the CDR regions of the antibody molecules, or immune complexes may form. The free CDRs in the immune complexes could then bind to the target and give an amplified signal, also possibly leading to an erroneous interpretation. If a cross-competing antibody is used as a blocking agent, it should have relatively high binding affinity and avidity to help reduce displacement by the test article. Some displacement of the blocking antibody by the test article always occurs, and this can possibly lead to positive staining. This needs to be considered when interpreting the study to avoid erroneous interpretations.
Antigen or antibody blocking studies are conducted on selected tissues on an as-needed basis, either during methods development or as a follow-up to the definitive TCR study.
Footnotes
Acknowledgments
The authors thank the following individuals for their submission of case reports used in this article or assistance in the preparation of the article: Karissa Adkins, Joel Cornacoff, Noel Dybdal, James Fikes, Frank Geoly, Antoinette Hayes, Danuta Herzyk, Christopher Horvath, Pat Losco, Shari Price-Schiavi, and Garvin Warner.
Abbreviations
Appendix
Current Considerations for Technical Success in the Conduct of Tissue Cross-reactivity (TCR) Studies
When it is appropriate to conduct TCR studies, there are certain best practices that can be followed. While it is beyond the scope of this article to discuss all possible scenarios, the general principles presented should be applicable to most situations.
Methods development is conducted to identify the best IHC method for the human (and/or animal) TCR study by considering factors important to each test article-target epitope pair being evaluated. The following factors should be considered: Optimization of tissue antigen retention/availability in test tissues (test system) Collection of/access to “normal” human and/or animal tissues from appropriately diverse tissue banks Selection of most appropriate sectioning, drying, and fixation conditions Confirmation of tissue adequacy and acceptability by morphology and acceptability staining controls Selection of most appropriate form of test article for IHC Selection of positive and negative control materials (assay validation) Selection of isotype (negative) control antibody Selection of immunohistochemical (IHC) (staining) method Selection of staining concentration(s) Inclusion of other specificity or staining controls Tissue acceptability control Assay control
The procedures deemed optimal are then applied to the definitive staining runs in the subsequent TCR study.
