Abstract
Anomalies of renal development comprise abnormalities in the amount of renal tissue (agenesis and hypoplasia); anomalies of renal position, form, and orientation; and renal dysplasia. There are previous reports of canine renal dysplasia in different breeds but none in the Beagle breed. This is the first report of renal dysplasia in this breed of dog. Morphologic descriptions of the range of microscopic features observed in four cases of renal dysplasia from preclinical studies in laboratory Beagle dogs are presented (including persistent primitive mesenchyme, persistence of metanephric ducts, asynchronous differentiation of nephrons, and atypical tubular epithelium), along with a basis for the classification of the lesion.
Introduction
Nonrodent species such as the Beagle dog play an important role in the drug development process, providing the best method for detection and limitation of predictable toxicity in the initial clinical studies in humans. They are used by pharmaceutical companies in preclinical toxicity testing before the potential drug is administered to humans (Peckman and Thomassen, 2007) because of their similarity with humans in biochemical responses and because of the similar pharmacological distribution and behavior of the target proteins, in addition to having comparable absorption, metabolic, pharmacokinetic, and distribution profiles for some chemicals (Lowenstine 2003).
We present here four cases of renal dysplasia found during preclinical studies in Beagle dogs over a period of 7 years. Three cases were in dogs treated with different compounds; the other case was in a dog with telemetry implants that had been used in safety pharmacology studies. No renal effects related to test article were found in any of these dogs, and the renal findings in each case were considered to represent a preexisting abnormality based on the short durations of the studies and the nature of the findings. There are previous reports of canine renal dysplasia in different breeds but none in the Beagle breed. To the authors' knowledge, this is the first report of renal dysplasia in this breed of dog.
Anomalies of renal development comprise abnormalities in the amount of renal tissue (agenesis and hypoplasia); anomalies of renal position, form, or orientation; and renal dysplasia. Minor congenital anomalies of the kidney such as cysts and polycystic kidneys are fairly common in dogs, but agenesis is infrequently reported in all species except when there is a familial incidence as in some dog breeds and in Large White pigs (Morita et al. 2005; Picut and Lewis 1987; Maxie and Newman 2007). However, the causative defect, pathogenesis, and mode of inheritance for most hereditary nephropathies are unknown. Although much less common than acquired renal disorders, congenital renal diseases are a cause of renal failure in dogs. Unilateral agenesis is compatible with life if the remaining kidney is normal, but concurrent contralateral renal dysplasia may ultimately lead to renal failure.
Materials and Methods
Records were collected retrospectively from 4 laboratory Beagle dogs of up to 5 years of age that were on preclinical toxicology studies and whose tissues were subjected to routine histopathologic evaluation as part of these safety evaluation studies.
The studies were planned in accordance with the local (Japan and UK) standards of animal care and ethics. For the UK, these are described in the Guidance on the Operations of the Animals (Scientific Procedures) Act 1986 issued by the UK Home Office. They were conducted so that any clinical expression of toxicity remained within a moderate severity limit as described in guidelines agreed with the UK Home Office inspector.
For light microscopic examination, the kidneys were sampled as part of the normal necropsy procedure and fixed in 10% buffered formalin. Paraffin embedded tissue was processed to slides and stained with H&E, with additional staining to demonstrate fat (Oil Red O stain in case 4) or mineralization (von Kossa stain, case 2). Additionally in case 4, samples of formalin-fixed renal cortex were trimmed into sections 1 × 7 mm and transferred to fixative containing 2.5% glutaraldehyde, 2% paraformaldehyde in 0.1M NaPO4 buffer for electron microscopic examination (case 4). These were postfixed with 1% osmium tetroxide in 0.1M phosphate buffer, dehydrated through graded alcohols, embedded in PolyBed 812 resin, and polymerized in a conventional oven at 60°C. Following polymerization, half-micron sections were cut using a Leica Ultracut UCT ultramicrotome, stained with Toluidine Blue, and evaluated by light microscopy. Areas representative of the lesions to be characterized were trimmed from selected blocks, and then 75 to 90 nm sections were cut, mounted on 200 mesh copper palladium grids, stained with uranyl acetate and lead citrate, and evaluated in the Hitachi 7100 transmission electron microscope at 75kV. Digital images were captured using a digital CCD camera system (Advanced Microscopy Techniques, Danvers, MA).
Case 1
An 11-month-old female Beagle dog (Marshall Bioresources, Italy) from the high-dose group of a 2-week toxicity study died immediately after dosing on day 11. The cause of death was unrelated to the renal abnormalities. A full necropsy was conducted and a comprehensive set of tissues was examined microscopically. Although no clinical pathology samples were taken on day 11, there were no abnormalities in the clinical pathology parameters collected from this animal before the study started (day –1).
Case 2
This 9-month-old, 12 kg male Beagle dog (Marshall Bioresources, Italy) was in the high-dose group of a 7-day early in vivo toxicity study with a limited list of tissues to be sampled and examined. This group was euthanized on day 1 of dosing, as the animals experienced adverse clinical signs associated with exaggerated pharmacology during dose administration, and a full necropsy was conducted. There were no abnormalities in the clinical pathology parameters in the samples taken from this animal euthanized early.
Case 3
This 5-year-old, 16.5 kg male Beagle dog (Marshall Bioresources, Italy) was from a group of dogs used for telemetry studies: monitors implanted in the heart recorded heart rate and blood pressure data. No significant abnormalities were seen in any blood samples from this animal. Hematology, serum chemistry, and liver function tests were within normal limits.
Case 4
The 13-month-old female Beagle dog (Marshall Bioresources, Italy) presented here was from the high-dose group of a 7-day toxicity study. This individual and others in the same group were euthanized early on humane grounds unrelated to the renal anomalies discovered in this dog. A full necropsy was conducted with a limited list of tissues collected and examined. There were no clinical pathology samples taken from any animals euthanized early.
Results
Case 1
At necropsy, the right kidney was small and weighed approximately 5 grams (left kidney weighed approximately 15 grams). No other abnormalities were observed in the urogenital system. Microscopically, the cortex was partially thinned in the middle of the kidney and accompanied by a subcapsular segmental area consisting of many immature glomeruli and proliferative arterioles with slight interstitial fibrosis and lymphocytic infiltrates (Figure 1). These components of the lesion were present also at the edge of the cortex along the renal hilum. Remaining cortical tissue consisted of well-developed tubules and glomeruli and was of normal appearance except for some scattered immature glomeruli. The medulla consisted of persistent, loose, poorly differentiated stroma (persistent mesenchyme) that supported a paucity of collecting ducts. Isolated collecting ducts, which were dilated and lined by attenuated cuboidal to flattened epithelium, were occasionally found within the poorly differentiated stroma. The left kidney was microscopically normal.
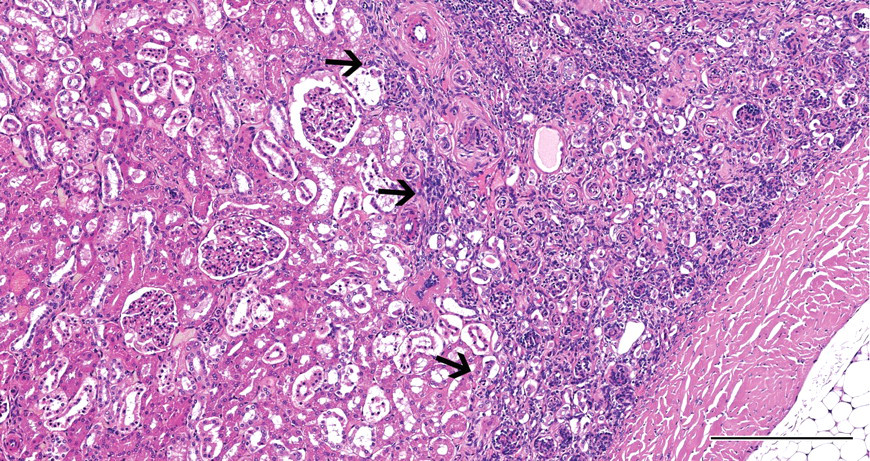
Case 1: Subcapsular area (arrows) with immature glomeruli and proliferative arterioles. H&E X40; bar = 250 μm.
Case 2
Macroscopically, the right kidney was small, lobulated, and firm with an irregularly pale, pitted, and thin cortex caused by wide radial bands of fibrous tissue extending from the capsular surface to the renal pelvis (Figure 2A). The left kidney appeared grossly normal.
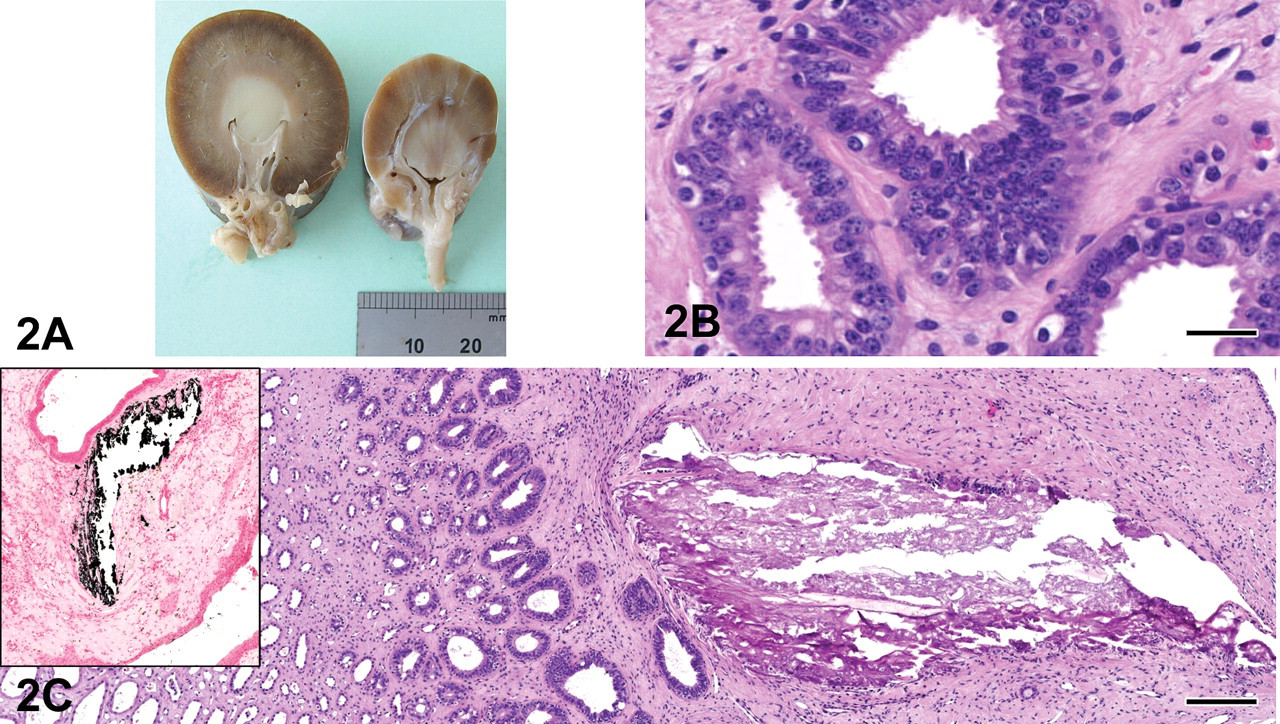
Case 2: 2A: Macroscopic view. Normal kidney is on the left, dysplastic kidney on the right. 2B: Persistent metanephric ducts. H&E X200; bar = 25 μm. 2C: Peripelvic area with mineralization. H&E X200; bar = 25 μm. Inset: same area stained with von Kossa technique to show mineralization (black). X200.
Microscopically, in the right kidney there were multifocal, fairly demarcated, linear bands of scarring in the cortex that extended to the capsule, surrounding, compressing, and partially replacing tubules, glomeruli, and vessels and extending variably into the medullary interstitium. Associated with these bands were abundant narrow, hyperplastic (proliferative) arterioles, primitive mesenchyme, and interstitial fibrosis (scarring). In addition, there were small (primitive) to mildly ectatic tubules lined by variably attenuated epithelial cells, small immature (foetal) glomeruli with peripheral nuclei, and inapparent capillaries and larger glomeruli that exhibited enlarged glomerular tufts with hyperplasia of mesangial cells, segmental to global sclerosis, and/or cystic dilatation of Bowman’s spaces. Within the medulla, there were scattered, irregular-sized collecting ducts lined by pseudostratified cuboidal to columnar epithelium (persistent metanephric ducts; Figure 2B) that were surrounded by primitive loose mesenchyme. Irregular areas of mineralization as indicated by H&E and von Kossa stains were observed in the corticomedullary and peripelvic areas (Figure 2C and inset). Multifocally in the interstitium, there were small infiltrates of plasma cells, lymphocytes, and few macrophages. Scattered tubules that contained pale eosinophilic, globular to homogeneous (proteinaceous) substance and exfoliated cells were observed (hyaline and renal tubular epithelial casts). The adjacent cortical tissue was more normally developed; however, it contained occasional fetal glomeruli.
No extrarenal lesions were observed macroscopically or microscopically. The left kidney was histologically normal.
Case 3
The right kidney was pale, smaller than the left kidney, moderately firm, and had an irregular surface.
The histological examination showed that approximately 30% of renal parenchyma was replaced by multifocal cortical and medullary interstitial fibrosis, hyperplastic (proliferative) small arteries and arterioles, along with immature mesenchyme (Figure 3). Primitive ducts and cysts lined by up to 3 cellular layer thick atypical columnar epithelium and surrounded by concentric layers of mesenchyme (with occasional mitotic figures) were present in these areas. Some of these ducts contained finely granular to homogeneous pale eosinophilic substance. Also, there were scattered clusters of lymphocytes, plasma cells, and few macrophages. Cystic glomerular atrophy and mature glomerular tufts were observed in the adjacent cortex. A few glomeruli exhibited markedly dilated Bowman’s spaces; others were sclerotic and distributed within the adjacent cortex. Some tubules were filled with eosinophilic granular debris and exfoliated epithelial cells. The left kidney appeared histologically normal.
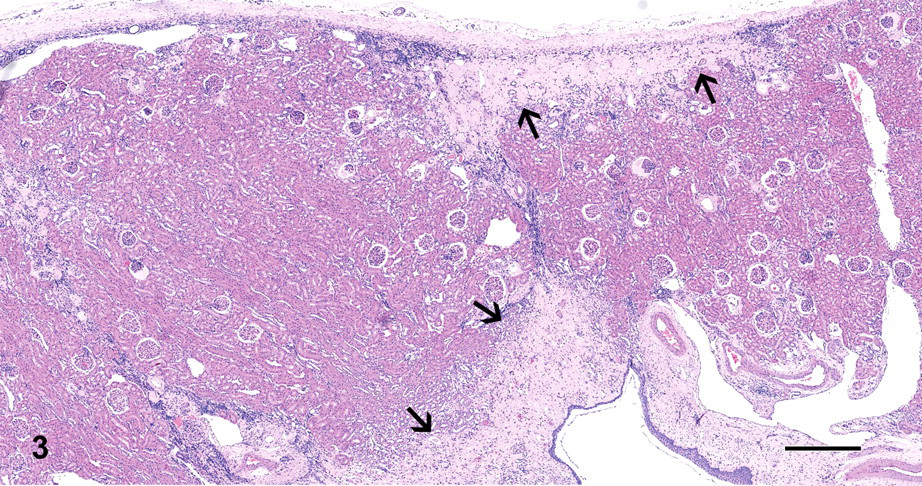
Case 3: Renal parenchyma was replaced by multifocal cortical and medullary areas composed of interstitial fibrosis, hyperplastic (proliferative) small arteries and arterioles, along with immature mesenchyme (arrows). H&E X40; bar = 500 μm.
Case 4
At necropsy, the right kidney of this female dog was missing, and the left kidney had an abnormal shape.
Light Microscopy
Multifocal nodular to elongated areas up to 500 μm in diameter or length with moderate tubular dilatation were present in the renal cortex and the medulla of the left kidney. Generally, the atypical tubular epithelial cells of dilated tubules were attenuated, cuboidal with pale eosinophilic cytoplasm except in foci of tubular regeneration with nuclear crowding and pale basophilic cytoplasm. There was multifocal vacuolation of the tubular epithelium that was Oil-Red-O (ORO) positive. In addition, there were multiple minimal lymphohistiocytic and neutrophilic inflammatory foci in the interstitium, and some tubules contained hyaline casts or cellular debris. A 900-μm–wide band of fibrosis extended radially from the medullary area (Figure 4) to the capsule, which was focally depressed and moderately thickened. This fibrous area (persistent mesenchyme) comprised a few immature glomeruli; small numbers of dilated, cystic glomeruli with a compressed glomerular tuft and pale eosinophilic fluid in the Bowman’s space; a few dilated tubules with flattened epithelium (atrophic tubules); and rare persistent metanephric ducts lined by basophilic pseudostratified epithelium sometimes surrounded by concentric layers of fibrous tissue. Also present were numerous small-diameter blood vessels (proliferative arterioles) and several foci of lymphocytes, macrophages, and few plasma cells. Overall, approximately 40% of the glomeruli in this kidney were abnormal. Their appearance varied from immature, shrunken glomeruli, some of which had a dilated Bowman’s space filled with eosinophilic proteinaceous fluid, to those that were moderately dilated with accumulation of enlarged foamy, presumably mesangial cells (Figure 5A) that were ORO positive (Figure 5B), obscuring up to 90% of the glomerular tufts. The Bowman’s capsule of dilated glomeruli was diffusely thickened up to 3 times normal thickness, and there was crowding of parietal nuclei.
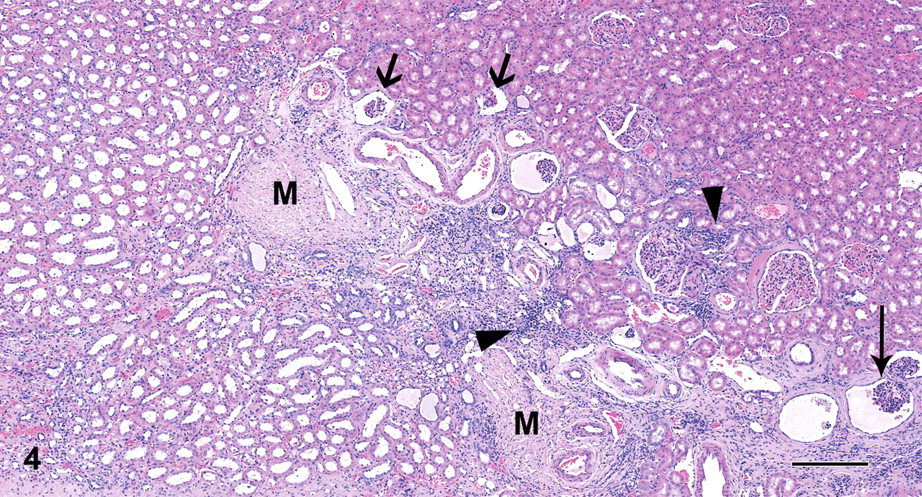
Case 4: Fibrotic band comprising immature (thick arrows) and/or dilated glomeruli (thin arrows), inflammation (arrow heads), and persistent mesenchyme (M). H&E X40; bar = 250 μm.
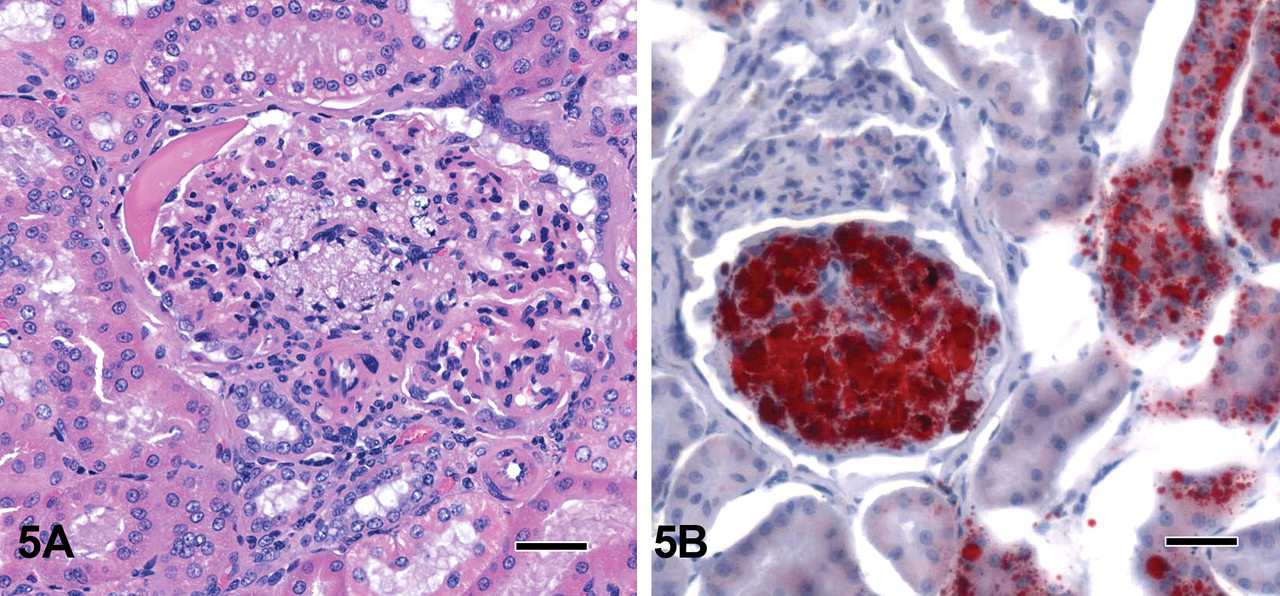
Case 4: 5A: Dilated tubules with cytoplasmic vacuolation and abnormal, moderately dilated glomeruli with accumulation of enlarged foamy, presumably mesangial cells. H&E X400; bar = 25 μm. 5B: Oil-Red-O (ORO) staining of section in 5A: shows positive fat staining cells in the tubular epithelium and in the glomerulus. ORO X400; bar = 25 μm.
Electron Microscopy
Electron microscopy examination confirmed the degeneration of tubular epithelial cells with occasional intratubular debris, rarefaction and swelling of mitochondria, and loss of the brush border. Cytoplasmic blebs containing amorphous cytoplasmic debris were present at the cellular surface (Figure 6A). In some tubular epithelial cells, there were occasional 150 to 400 nm accumulations of lamellated material centered around electron dense cores, consistent with lipid globules (Figure 6B). This correlated with the lipidosis observed in ORO stained sections. Immature glomeruli showed intra- and extra-cytoplasmic accumulations of granular and fibrillar material and a thick Bowman’s membrane up to 3 times normal (Figure 6C). Foamy mesangial cells contained numerous vacuoles of varying sizes and heterogeneous globules, correlating with the ORO positive glomerular cells and suggestive of lipid deposition (Figure 6D).
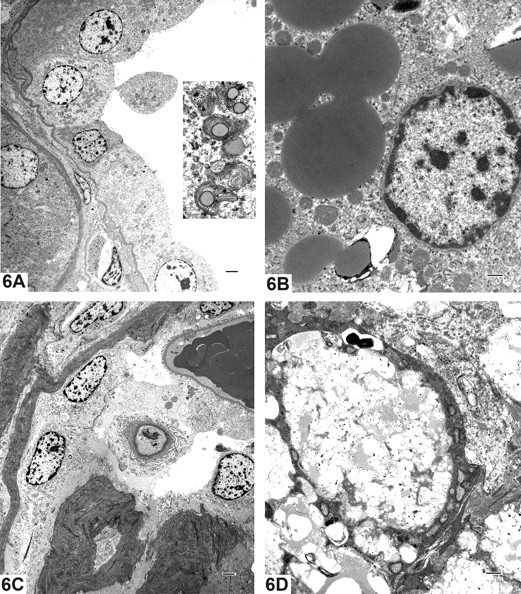
Case 4: Electron microscopy photomicrographs. 6A: Degeneration of tubular epithelial cells with occasional intratubular debris (Inset; X12,000), rarefaction and swelling of mitochondria, and loss of the brush border; X1,000. 6B: In some tubular epithelial cells, there were occasional 150 to 400 nm accumulations of lamellated material centered around electron dense cores, consistent with lipid globules; X5,000. 6C: Immature glomeruli showed intra- and extra-cytoplasmic accumulations of granular and fibrillar material and a thick Bowman’s membrane up to 3 times normal; X1,000. Fig. 6D: Foamy mesangial cells contained numerous vacuoles of varying sizes and heteregeneous globules, correlating with the ORO positive glomerular cells and suggestive of lipid deposition; X5,000.
Discussion
Renal dysplasia and hypoplasia are very common kidney defects in dogs and have been reported in many dog breeds, including Alaskan Malamutes, Bedlington Terriers, Chow Chows, Cocker Spaniels, Doberman Pinschers, Keeshonden, Lhasa Apsos, Miniature Schnauzers, Norwegian Elkhounds, Samoyeds, Shih Tzus, Soft-Coated Wheaten Terriers, and Standard Poodles (Lees 1996). It can be either hereditary or result from neonatal infections such as feline panleukopenia virus, bovine viral diarrhea virus, or canine herpes virus infections. The genetic origins of the dogs discussed here were investigated, and they were found to be unrelated.
Renal dysplasia is defined as the disorganized development of renal parenchyma leading in some cases to a macroscopic appearance of small, misshapen, fibrosed kidney(s). Another major differential diagnosis for a similar macroscopic appearance would be end-stage kidney, which occurs in older dogs and in a different clinical setting. Microscopically, renal dysplasia can demonstrate a range of morphologic changes and includes features that do not fit the development stage of the animal (Picut and Lewis 1987; Maxie and Newman 2007). Picut and Lewis (1987) characterize five primary morphologic aspects of canine renal dysplasia, the presence of at least one of which is required for a diagnosis of dysplasia. These include asynchronous differentiation of nephrons; persistent mesenchyme; persistent metanephric ducts lined by tall, pseudostratified, columnar epithelium; atypical tubular epithelium; and dysontogenic metaplasia. Although increased numbers of immature glomeruli can be observed in areas of renal dysplasia, they can also be observed occasionally in small numbers scattered in the cortex of normal dogs. Secondary degenerative and/or inflammatory changes may be present in wedges extending from the pelvis to the cortex and are thought to represent degenerative changes due to obstruction or infarction (Picut and Lewis 1987; Maxie and Newman 2007). The presence of lipid in the renal tubular epithelium has been ascribed to anoxic or toxic changes, similar to that which occurs in the liver (Jones, Hunt, and King 1997).
Based on the description above, the unilateral small kidney in case 1 was ascribed to renal dysplasia on the basis of microscopic evidence of areas of persistent mesenchyme containing reduced numbers of tubules, some of which showed atypical epithelium, along with immature glomeruli and proliferative arterioles. Similarly, renal anomalies present in cases 2, 3, and 4 were attributed to renal dysplasia and included areas of persistent, undifferentiated, loose mesenchyme; proliferative arterioles; immature glomeruli; atypical tubular epithelium; and primitive/metanephric ducts lined by cuboidal or columnar epithelium. In case 4, there was also unilateral renal agenesis.
Glomerular lipidosis is infrequently observed as a spontaneous finding in a few glomeruli in toxicity studies using Beagle dogs and has been described before (Agut et al. 2002; Taney et al. 2003; Greaves 2007; Jones, Hunt, and King 1997). In the example presented here (case 4), approximately 40% of glomeruli showed various degrees of lipidosis. This finding was considered to represent part of the degenerative and inflammatory process secondary to the dysplasia.
Cases of familial renal agenesis have been reported in Beagle dogs (Maxie and Newman 2007), and cases of unilateral agenesis were described in dogs including Beagle dogs (Morita et al. 2005; Agut et al. 2002; Taney et al. 2003; Robbins 1965). Renal dysplasia has been reported in some dog breeds (Morita et al. 2005; Picut and Lewis 1987) but, to the best of our knowledge, not in Beagle dogs. Because of the diversity of changes that may result from renal dysplasia, such renal changes presenting in Beagle dogs in toxicity studies can sometimes be difficult to differentiate from possible effects of test compounds when there is only a small number of animals in each individual study. Furthermore, published data on this topic in the Beagle dog were difficult to obtain. It is hoped therefore that this report will help by providing examples of the range of renal developmental background findings in Beagle dogs, along with a framework for defining the diagnosis of renal dysplasia.
