Abstract
This brief communication describes a rare spontaneous background lesion in the lower urinary tract of two male laboratory beagles. Proliferative lesions comprising a constellation of histological features consistent with polypoid cystitis were observed in the bladder of two adolescent dogs from a routine preclinical toxicology study. Both animals were clinically asymptomatic and had only minor alterations in urinalysis parameters. While chronic polypoid cystitis is well-recognized in adult pet dogs, this is the first reported case in purpose-bred laboratory beagles. An awareness of this uncommon background finding is important for toxicological pathologists to distinguish it from potential test article-related findings.
Alongside monkeys and minipigs, beagle dogs are widely utilized as the non-rodent species in the preclinical safety evaluation of novel therapeutic agents. Canine age-related pathology and spontaneous neoplasms are well described in the literature, particularly for pet dogs. Conversely, normal anatomical variations, clinically asymptomatic lesions, and incidental findings are infrequently described as the numbers of necropsies routinely performed by clinician and veterinary pathologists, for several reasons, has been declining in recent years. 4 Literature describing common background findings in control beagles is, however, frequently updated by publications and textbooks.9,10 In this brief communication, we discuss two cases of polypoid lesions in the urinary bladder, in clinically asymptomatic laboratory beagles, which were histopathologically consistent with polypoid cystitis.
The affected dogs were from a regulatory toxicity study (4-week toxicity followed by a 4-week recovery period), which was approved by the local ethical committee (AWERB), and conducted at Labcorp, Harrogate, an AAALAC-accredited animal program, in accordance with the UK Home Office “Guidance on the Operation of the Animals (Scientific Procedures) Act 1986.” The dogs were sourced from a commercial supplier based in the United Kingdom and underwent routine clinical examination on arrival to the test facility, which revealed no clinical abnormalities. The dogs were both males and siblings from the same litter and were approximately 6 months old at the start of the study. One dog (Dog 1) was from the recovery control group and, therefore, did not receive the test article, while the other (Dog 2) was from the intermediate dose group and received the test article daily by oral capsule. No salient clinical observations were recorded for either dog during the study.
Hematology, coagulation, clinical chemistry, and urinalysis were performed for all animals once during the predose phase and once during week 4 of the study. Samples for urinalysis were collected via urinary catheter and, if urinalysis was not performed on the same day, the samples were refrigerated and stored, with urinalysis performed within 24 hours. For the affected dogs, hematology, coagulation, and clinical chemistry were unremarkable at both time points. Urinalysis revealed increased microscopic red blood cells for Dog 2 at predose (between 11 and 30 cells per field) and for both males (>30 cells per field) at week 4, compared to the rest of the males in all groups (for which values were either “not present” or comprised between 0 and 10 cells per field). Higher occult blood was also detected for Dog 1 at week 4 only; urine occult blood for Dog 2 was not evaluated at week 4 due to an insufficient sample. All other urinalysis parameters, including white blood cells, casts, crystals, and bacteria, were within normal limits for both dogs.
Dog 1 and Dog 2 were euthanized on study day 58 (recovery day 29) and study day 29, respectively, and a full necropsy was performed. Multiple black and firm raised areas (up to 5 mm) were noted on the mucosal surface of the urinary bladder for Dog 1, whereas a single dark red and soft raised area (up to 2 mm) was noted for Dog 2. No other macroscopic findings were evident for the urinary bladder of either dog or the urinary tract of the remainder of dogs on study. A full transverse section of the central region of the urinary bladder was sampled in addition to the grossly abnormal lesions and were fixed in 10% neutral-buffered formalin. Tissues were trimmed, routinely processed, and embedded in paraffin wax prior to sectioning (4-6 µm) and staining with hematoxylin and eosin (H&E) for routine histological examination. No test article–related histological findings were recorded for the urinary tract of any dog on study.
Microscopically, single or multiple pedunculated, polypoid growths, composed of a loose connective tissue stroma covered by transitional epithelium, were evident in the urinary bladder of both dogs (Figure 1A). The transitional epithelium was diffusely hyperplastic and formed invaginations or epithelial down-growths within the polyp stroma (Figure 1B). Rarely, Brunn’s nests (solid nests of transitional cells located within the lamina propria and detached from the overlying urothelium) were also noted (Figure 1C). Transitional epithelial cells had mostly indistinct borders, with abundant eosinophilic cytoplasm, often containing intracytoplasmic vacuoles or cavitations containing eosinophilic material, which have previously been referred to as intraepithelial lumina. 5 Mild anisocytosis, frequent mitotic figures, and small areas of hemorrhage were evident multifocally within the epithelium (Figure 1C). The stroma was composed of moderately edematous, loose connective tissue, infiltrated by lymphocytes, macrophages, and occasional plasma cells; neutrophils were also rarely observed (Figure 1D). Within the stroma, abundant newly formed blood vessels were also evident, and at the luminal extremity of the polypoid growths, hemorrhage, with associated hemosiderophages, was prominent. Small, multifocal areas of hemorrhage were noted in the urinary bladder mucosa outwith the polypoid lesions for Dog 1, whereas no microscopic abnormalities were noted for the remainder of the urinary bladder for Dog 2. Mucoid degeneration and cystitis glandularis (glandular metaplasia) have been previously reported in association with chronic polypoid cystitis; however, these were not observed in the current cases. 6 A variant of chronic polypoid cystitis in which an eosinophilic infiltrate predominates has been also previously reported in dogs but was also not evident in these cases. 11
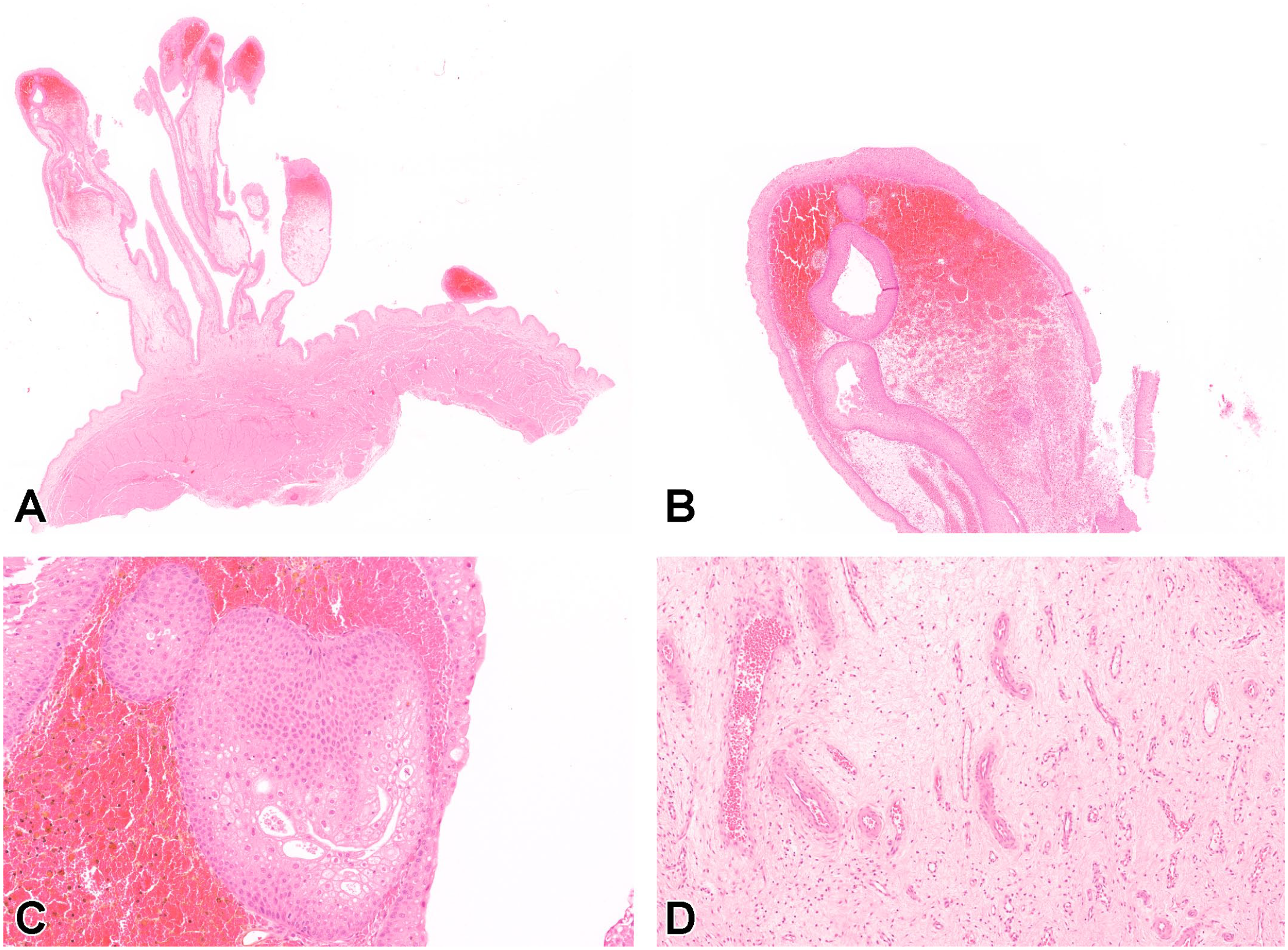
Polypoid cystitis, beagle dog, urinary bladder (hematoxylin and eosin). (A) Multiple, variably sized, polypoid growths, projecting from the urinary bladder wall and extending into the lumen. (B) Higher magnification of the luminal extremity of a polyp. The surface is lined by an hyperplastic transitional epithelium and epithelial downgrowths create invaginations into the stroma. (C) Features of mild anisocytosis, intraepithelial lumina, and mitotic figures are visible within Brunn’s nests and the surface transitional epithelium. Surrounding stroma contains hemorrhage and associated hemosiderophages. (D) Numerous newly formed blood vessels within the edematous connectival stroma of a polyp.
Chronic cystitis classically occurs in three variants: diffuse, follicular, and polypoid. Although polypoid masses are typical of polypoid cystitis and may occur in any domestic species, they are more commonly reported in dogs. 11 No reports of polypoid cystitis have previously been described for laboratory monkeys, minipigs, or rodents. Polypoid cystitis in dogs appears macroscopically as multiple nodular to polypoid masses, pedunculated, or broad-based, generally located in the cranioventral urinary bladder wall (apex). In contrast, urothelial cell carcinomas (previously called transitional cell carcinomas) typically arise in the trigone, which is located caudodorsally between the ureteral and the urethral openings,6,11 and topographical location may be a useful discriminating feature between polypoid cystitis and neoplastic lesions. As the region of urinary bladder from which the gross abnormalities were sampled was not recorded, we could not confirm whether the current cases occurred in the urinary bladder apex.
No breed predilection has been reported for chronic polypoid cystitis in dogs and information regarding sex predisposition is conflicting. Some authors report that polypoid cystitis predominantly occurs in male dogs, 6 whereas other publications make no mention of any sex predisposition.3,11 In a recent paper describing 17 cases of chronic polypoid cystitis, a strong sex predilection for females was noted (88% of cases) 5 ; however, the authors also reported that males were less likely to exhibit clinical signs of chronic polypoid cystitis and the incidence of disease in males may be higher than previously identified as cases could frequently remain undiagnosed.
Chronic polypoid cystitis in domestic animals is thought to occur in response to chronic irritation of the urinary bladder, most likely caused by the presence of uroliths and/or recurrent urinary tract infections. 11 The most common clinical manifestation of chronic polypoid cystitis is intermittent hematuria, which likely occurs when the fragile structure of the polyps breaks down. The shape of the polyps is also thought to favor urine retention and bacterial adhesion to the mucosa, instigating a vicious cycle of infection-proliferation-inflammation.3,11 While canine polypoid cystitis typically occurs in adult dogs, Ruiz et al 8 reported the occurrence of the disease in a 14-week-old springer spaniel puppy that presented due to recurrent hematuria. In that previous case, bacterial urine culture was negative, however, urinalysis intermittently revealed the presence of struvite crystals. Histopathology also identified the presence of refractile crystalline material within the vicinity of the polypoid inflammatory lesions and the authors hypothesized that the initiating cause was chronic irritation secondary to crystalluria. In the present study, the two young beagle dogs diagnosed with polypoid cystitis were siblings and from the same litter; hence, genetic factors and/or breed predisposition may be implicated in the pathogenesis of polypoid cystitis in some instances. Interestingly, no evidence of bacteria or crystals was identified in the urine of either of these asymptomatic dogs, which might indicate that urinary tract infection and urolithiasis could be the consequence, rather than predisposing factors, in the onset of polypoid cystitis.
In humans, chronic polypoid cystitis occurs as a hyperplastic response to mechanical abrasion of posterior wall of the bladder secondary to the placement of indwelling urinary catheters. Polypoid cystitis is present in up to 80% of patients and most lesions are reported to resolve within 6 months after catheter removal.1,2 However, in rare cases, patients have been diagnosed with polypoid cystitis without any history of previous catheterization or urinary problems. 7 In this study, urinary catheterization was used to collect urine on two separate occasions. As an increased number of red blood cells was detected microscopically for both dogs at the second urinalysis, it could be hypothesized that the initial catheterization resulted in mechanical trauma to the bladder wall, which subsequently lead to the development of induced polypoid cystitis. However, this is considered unlikely in isolation given the relative frequency with which urinary catheterization occurs in dogs, either as a medical or laboratory procedure, and the lack of any previous association with polypoid cystitis.
While environmental stressors may contribute to a multifactorial pathogenesis of polypoid cystitis, since the administration of the test article in our study was via oral capsule, we do not believe stress played a major role in these current cases.
To our knowledge, polypoid cystitis has not been previously observed either as an outcome of xenobiotic treatment or as a spontaneous finding in preclinical toxicity studies and, furthermore, it is not described in the published International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) guide for the dog. 10 Therefore, if found solely in the test article-treated groups, this unusual finding might erroneously be interpreted as a test article-related effect.
In conclusion, polypoid cystitis is a rare, previously unreported, spontaneous background finding in the laboratory beagle, an awareness of which is important for toxicological pathologists to distinguish it from potential test article-related findings, particularly if such a lesion were to arise in a high-dose animal. Further studies are required to investigate the likely multifactorial pathogenesis of this condition in pet and laboratory-bred dogs and to identify predisposing factors for the onset of the disease.
Footnotes
Acknowledgements
The authors would like to acknowledge the Labcorp Harrogate pathology and histology team for their support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
