Abstract
In this study, rasH2-Tg mice treated with N-methyl-N-nitrosurea (MNU) developed exuberant hematoproliferative changes in the spleen that included dysplasia and features of neoplasia. Hematoproliferative change was characterized as exuberant proliferation of hematopoietic cells within the spleen that distorted but did not displace normal splenic morphologic features. The hematopoietic cells were of mixed lineage, but one type, often erythroid, predominated. Cellular atypia was present in all mice with hematoproliferative change, and dysplasia was present in five of eight examined. Hematoproliferative neoplasia was characterized by similar cytologic features but also resulted in displacement/disruption of normal splenic architecture and increased numbers of unidentified blast cells. One case was differentiated toward myeloid proliferation, suggesting granulocytic leukemia. Affected mice had other neoplasms, such as lymphoma and anemia. These proliferative and dysplastic lesions of the spleen in rasH2-Tg mice treated with MNU require additional characterization to definitively differentiate them from the reactive hematopoiesis that can occur in response to inflammatory, neoplastic, or hematopoietic insults in mice.
Introduction
The rasH2 transgenic (Tg) mouse is an accepted alternative rodent model for carcinogenicity testing (MacDonald et al. 2004; Long et al. 2010). This hemizygous Tg mouse carries the human c-Ha-ras oncogene and the promoter/enhancer region for this gene on the genetic background of a BALB/cByJ x C57BL/6J F1 mouse. The transgene is stably expressed in all tissues. The rasH2-Tg mouse model has been extensively tested and shows similar predictivity to the conventional 2-year mouse bioassay for human carcinogens (Morton et al. 2002). The model will manifest a carcinogenic effect within 6 months when treated with various types of mutagenic agents and a limited number of nonmutagenic agents (Yamamoto, Mitsumori, et al. 1998; Yamamoto, Urano, et al. 1998).
Spontaneous tumors occur at a low incidence in the rasH2-Tg mice before 6 months of age. The spontaneous and induced tumor spectrum in the rasH2-Tg mouse is consistent with that of the background strain (Tamaoki 2001), and the tumors generally have morphologic features similar to those observed in B6C3F1 and ICR mice (Mitsumori et al. 1998). Differences in tumor spectra between the rasH2-Tg and non-Tg mice are related to the relative frequency of specific neoplasms and/or target organs.
A single intraperitoneal dose of N-methyl-N-nitrosurea (MNU) is generally accepted as a positive control agent for the rasH2 model (MacDonald et al. 2004; Long et al. 2010). A single dose of 75 mg/kg induces more neoplasms than five doses of 15 mg/kg each (Yamamoto, Mitsumori, et al. 1998; Yamamoto, Urano, et al. 1998). Administration of MNU induces more neoplasms in rasH2-Tg mice than in non-Tg mice of the same background strain, but the tumor types and sites are consistent in the rasH2-Tg and non-Tg mice (Takaoka et al. 2003). Commonly induced neoplasms are forestomach papillomas and carcinomas, lymphomas of the thymus, squamous cell papillomas and carcinomas of the skin, lung adenomas, and splenic hemangiosarcomas. Hemangioma and hemangiosarcoma are the most common neoplasms localized to the spleen, and malignant lymphomas are common tumors of the hematopoietic system (Usui et al. 2001). Granulocytic leukemia occurs at a low incidence in rasH2-Tg mice treated with a single dose of 75 mg/kg MNU (Yamamoto, Mitsumori, et al. 1996) and was diagnosed in two of eight female rasH2-Tg mice treated with 75 mg/kg MNU in one study (Mitsumori et al. 1998). The lesions involved the liver, spleen, and bone marrow. Neoplastic cells were described as large and blastic; segmented or lobed nuclei were rare.
Hyperplastic lesions reported in the spleen of rasH2-Tg mice treated with MNU have included lymphoid, erythroid, and endothelial hyperplasia (Takaoka et al. 2003). Nonproliferative lesions of the spleen were angiectasis, extramedullary hematopoiesis (EMH), fibrosis, and pigmentation. The incidence of a number of nonneoplastic lesions, including EMH and angiectasis in the spleen, was increased in rasH2-Tg mice compared with non-Tg mice treated with MNU. In a report on positive control studies, EMH was reported in 0 to 2 per group of 15 rasH2-Tg mice treated with 75 mg/kg MNU but was not reported in any untreated rasH2-Tg mice (Machida et al. 2008). Malignant lymphoma was reported in 12 to 14 in each group of 15 MNU-treated rasH2-Tg mice. There were no reports of myeloid neoplasia or dysplasia in spleen in these studies.
In this article, we report a high incidence of enlarged spleens associated with a spectrum of excessive hematopoietic proliferation with features of dysplasia and neoplasia in two studies in rasH2-Tg mice treated with MNU as positive controls. One study was conducted as a positive control study for the rasH2-Tg mouse model; the other was a positive control group in a study to examine the carcinogenic potential of a test compound. Splenic enlargement was attributed partially to infiltration of neoplastic lymphocytes in mice with lymphoma; however, involvement by malignant lymphoid cells was not extensive enough to explain the magnitude of increase in size of the spleen or the atypical histologic features of the hematopoietic cells in all affected animals. The current report is an initial characterization of this hematopoietic proliferation.
Methods
Male and female CB6F1/Jic-TgN (RasH2+/−, heterozygous, Tg) and CB6F1/Jic-TgN (RasH2−/−, wild type, non-Tg) mice were obtained from Taconic Farms, Germantown, New York. Animals were randomized and assigned to groups based on weight. Male and female mice were group housed in suspended stainless-steel cages, acclimated to laboratory conditions for approximately 2 weeks, and then individually housed for the experimental period. Mice were offered food and water ad libitum. Environmental controls for the animal room were set to maintain 18°C to 26°C, a relative humidity of 30% to 70%, 10 air changes/hour, and a 12-hour light/12-hour dark cycle except when interrupted for study-related activities. Procedures were in compliance with the Animal Welfare Act, the Guide for the Care and Use of Laboratory Animals, and the Office of Laboratory Animal Welfare.
Study A was a positive control study in which rasH2-Tg and non-Tg mice, 20/sex/group, were treated with MNU or control vehicle. Study B was an alternative carcinogenicity study for a test compound in rasH2-Tg mice and included 25/sex/group. Groups from study B reported herein include the positive control (MNU-treated) group and the negative control group. Mice were treated with MNU (Isopac; Sigma-Aldrich, St. Louis, MO). The MNU was formulated at 7.5 mg/mL in acidified saline (physiological saline, 150 mM sodium chloride and 15 mM sodium citrate, adjusted to pH 4.5 with 1 N HCl). Mice received a single intraperitoneal injection of 75 mg MNU/kg body weight at 7 to 11 weeks of age. Control mice in study A received control vehicle (acidified saline) in a similar fashion. Control mice in study B received daily oral gavage doses of 10% acacia in water, the control vehicle used for the test article in the study.
A complete necropsy was conducted on all animals that died or were sacrificed at an unscheduled interval. When possible, samples were collected from animals sacrificed in a moribund condition for analysis of hematology parameters (described below). All surviving mice were sacrificed and submitted to a complete necropsy on days 184 to 187. Samples were collected from all surviving mice prior to scheduled sacrifice for analysis of hematology parameters (erythrocyte [red blood cell {RBC}] count, hemoglobin concentration, hematocrit, total white blood cell [WBC] count and differential, and platelet counts in both studies; reticulocyte counts in study A). Hematology parameters were determined on an Advia 120. RBC, platelet, and WBC counts were determined by flow cytometric light-scattering measurement, with the WBC count using peroxidase cytochemical staining. Hemoglobin concentration was determined by a modified cyanmethemoglobin method and hematocrit derived from the RBC count and RBC volume derived from the RBC volume histogram.
At necropsy, a complete set of tissues typical for a carcinogenicity study were preserved in 10% neutral-buffered formalin. Tissues included femur and sternum with bone marrow; spleen; mesenteric lymph node; thymus; lung; liver; kidney; heart; representative sections of gastrointestinal, reproductive, hormonal, and central nervous systems; and gross lesions. Tissues were embedded in paraffin, sectioned, and stained with hematoxylin and eosin (H&E). All tissues were examined. Bone marrow was evaluated on decalcified sections of femur and sternum. Bone marrow smears were not available.
Sections of spleen and liver from nine mice in study A and seven mice from study B were immunolabeled for immunophenotyping of hematopoietic cell types. Tissues were selected to represent the range of histologic appearances in the spleen from among those judged to have the best tissue preservation. Immunolabels were selected from among those previously described for analysis of hematopoietic neoplasms in mice (Kunder et al. 2007): CD45 (LCA): AbD Serotec (Raleigh, NC) MC1031GA rat monoclonal antimouse, pan-leukocyte marker; CD3: Dako (Carpinteria, CA) A0452 rabbit polyclonal antihuman, pan T-cell marker; CD45R (B220): BD Pharmingen (San Jose, CA) 550286 rat monoclonal antimouse, B cell marker; F4/80: eBiosciences (San Diego, CA) 14-4801 rat monoclonal antimouse, pan macrophage marker; Myeloperoxidase, granulocyte marker (MPO): Dako (Carpinteria, CA) A0398 rabbit polyclonal antihuman, granulocyte marker; and Ter-119 (Ly-76): BD Pharmingen (San Jose, CA) 550565 rat monoclonal antimouse, erythroid marker.
Immunolabeling was performed using a Dako autostainer and heat-induced antigen retrieval. Immunolabeled slides were examined in conjunction with examination of H&E-stained slides to relate cell type identification to cytologic features.
Results
Mortality was high in rasH2-Tg and non-Tg mice treated with MNU. In study A, mortality among rasH2-Tg and non-TG mice treated with MNU was 12/20 and 14/20 males and 12/20 and 15/20 females, respectively. In study B, mortality was 20/25 in males and 21/25 in females in rasH2-Tg mice treated with MNU. Most deaths occurred during the latter half of the studies and were associated with neoplasms, primarily lymphoma and neoplasms of the gastrointestinal tract, which were similar to common neoplasms reported previously in this model (Mitsumori et al. 1998; Takaoka et al. 2003). Other common MNU-induced neoplasms in the current studies included papillomas and squamous cell carcinomas of the skin and bronchiolar-alveolar adenomas and carcinomas of the lungs, and they were also similar to those reported previously. Neoplasms associated with MNU treatment at low incidence included renal transitional cell carcinoma, Harderian gland adenoma, subcutaneous sarcoma, and ureteral transitional cell papilloma.
Hematology data in mice sampled at termination are summarized in Table 1 . rasH2-Tg and non-Tg mice treated with MNU had values for erythrocyte count, hemoglobin concentration, and hematocrit that were lower than those of the concurrent controls. MNU-treated mice sacrificed before study termination from which hematology parameters were obtained had erythron values similar to those in the animals sampled at termination. One male rasH2-Tg mouse treated with MNU in study B that survived to study termination had erythron values that were near the low end of the range of untreated controls (erythrocyte count of 9.23 million/μL); this individual had normal splenic histomorphology. Reticulocyte counts tended to be higher in MNU-treated mice at study termination (sampled in study A only), and the majority were greater than the range of the respective control group. WBC counts were higher in both rasH2-Tg and non-Tg MNU–treated mice compared with nontreated mice. The higher WBC counts were generally due to higher neutrophil counts. The neutrophil counts in MNU-treated mice were highly variable and were excessive (i.e., >100,000/µL) in 2 MNU-treated male rasH2-Tg mice in study B that were unscheduled deaths.
Hematologic Parameters in Control and MNU-Treated rasH2-Tg and Non-Tg Mice a Tested at Study Termination
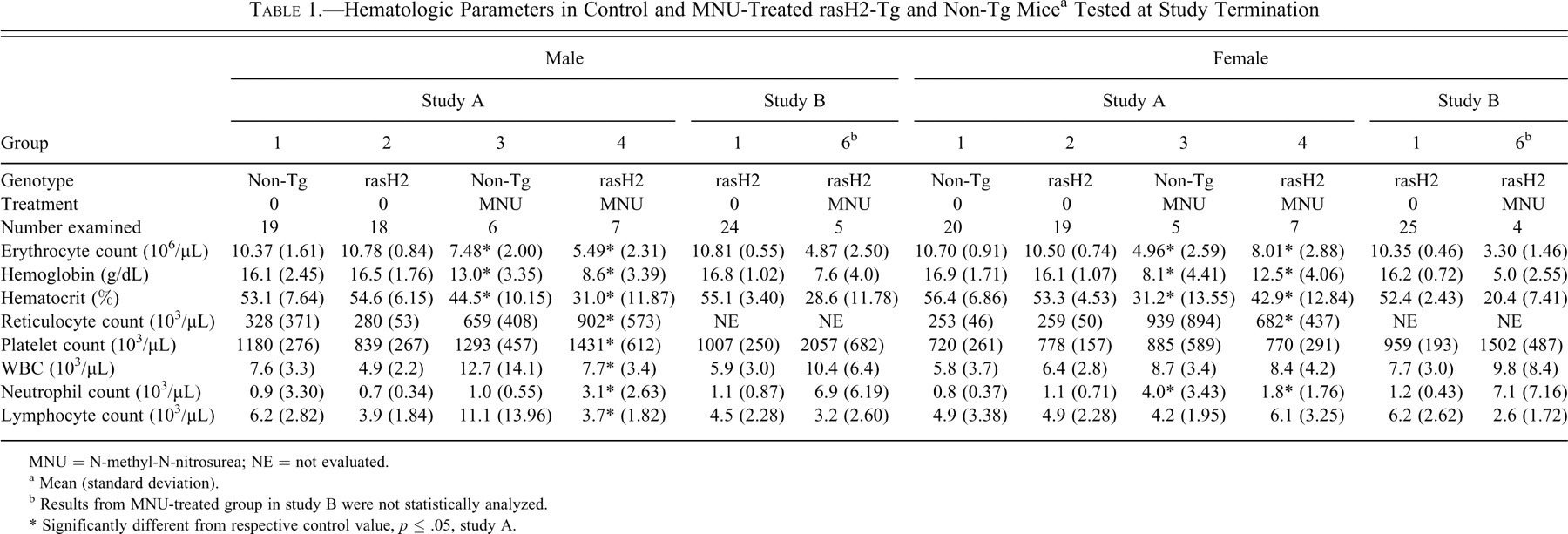
MNU = N-methyl-N-nitrosurea; NE = not evaluated.
a Mean (standard deviation).
b Results from MNU-treated group in study B were not statistically analyzed.
* Significantly different from respective control value, p ≤ .05, study A.
Splenic enlargement was observed in many MNU-treated mice, in both rasH2-Tg and non-Tg mice (Table 2 ). Splenic enlargement correlated with microscopic findings of increased amounts of hematopoietic tissue and/or lymphoma in MNU-treated mice, but not all animals that had histologic evidence of increased hematopoietic tissue had gross splenic enlargement.
Incidence of Hematoproliferative and Selected Other Lesions in the Spleen of Control and MNU-Treated rasH2-Tg and Non-Tg Mice
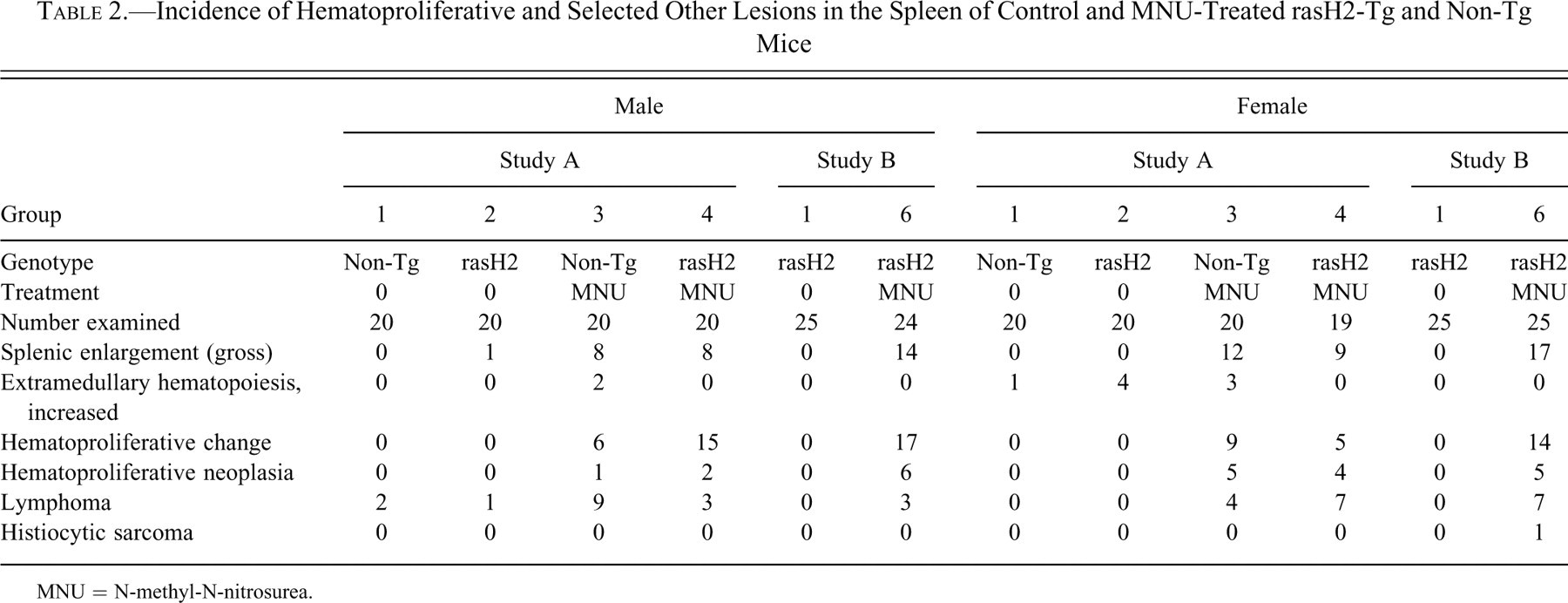
MNU = N-methyl-N-nitrosurea.
The following terms were used within the studies to diagnose increased hematopoietic tissue in the spleen: increased EMH, myeloproliferative change, and myeloproliferative neoplasia. In this publication, these changes are identified as increased EMH, hematoproliferative change, and hematoproliferative neoplasia, respectively. Increased EMH in the spleen described hematopoietic tissue that was minimally to slightly increased over that in controls, maintained a normal maturation sequence, and did not substantively alter the overall morphologic relationships in the spleen. Hematoproliferative change was defined as exuberant proliferation of hematopoietic cells within the spleen that distorted overall splenic morphologic relationships, resulting in considerable expansion of the red pulp. It caused limited compression but not displacement/disruption of splenic architecture (e.g., lymphoid structures). The change sometimes occurred in conjunction with other proliferative lesions (e.g., lymphoma or splenic hemangiosarcoma). By examination of H&E-stained sections, the hematopoietic cells most often appeared to be of mixed lineage (erythroid, myeloid, and megakaryocytic), but in some cases, one lineage predominated. A feature of hematoproliferative change was dysplasia of one or more hematopoietic lines. Dysplastic changes included multinucleated cells, abnormal nuclear to cytoplasmic ratio, disorderly left shift, tinctorial changes in cytoplasm, and maturation arrest. Hematoproliferative change occurred in rasH2-Tg and non-Tg mice treated with MNU (Table 2) but did not occur in any controls. Hematoproliferative neoplasia was characterized by an exuberant proliferation of hematopoietic cells that caused displacement/disruption of normal splenic architecture. Similar to hematoproliferative change, the hematopoietic cells appeared dysplastic and of mixed lineage on H&E-stained slides, but one type sometimes predominated. Large blastic undefined leukocytes (on H&E-stained slides) were sometimes present. A similar-appearing discrete cell population was sometimes present in other organs, most often the liver, lung, and thymus. Hematoproliferative change occurred in rasH2-Tg and non-Tg mice treated with MNU (Table 2) but did not occur in any controls.
Characterization of Splenic Changes
Normal splenic morphology, with small amounts of EMH, was characterized in two rasH2-Tg mice treated with control vehicle. These mice were sacrificed at week 27. The hematopoietic tissue included a normal distribution of erythroid, myeloid, and megakaryocytic cells. Circulating numbers of erythrocytes, reticulocytes, and leukocytes in these two mice were within the range of the rest of the concurrent control group. These mice had no neoplasms. Hematopoietic tissue in the bone marrow was normal in these two mice (by routine light microscopic evaluation of H&E-stained sections). Normal morphologic features were well defined in the spleen. Small nests of hematopoietic cells in the red pulp had poorly defined borders (Fig. 1A
). Results of immunohistochemical labels were as follows: CD45 (pan-leukocyte marker): Most cells in the lymphoid tissues were labeled, as well as individual cells throughout the red pulp. CD45R (B-cell marker): Moderate number of cells in mantle zones were labeled, as well as small aggregates of lymphoid cells (follicles) adjacent to periarteriolar lymphoid sheaths (PALS; Fig. 1B). CD3 (T-cell marker): Most cells in PALS were labeled, as well as a few scattered individual cells in red pulp (Fig. 1C). F4/80 (macrophage marker): Moderate numbers of cells in the red pulp were labeled (Fig 1D). Labeled cell processes were primarily in sinuses. MPO (granulocyte marker): Individual cells scattered throughout the red pulp were labeled; many were within areas that appeared to be myeloid hematopoietic based on H&E-stained sections (Fig 1E). Ter-119 (erythroid marker): Erythrocytes were labeled throughout the red pulp and scattered in lymphoid areas. Variable numbers of cells were labeled within areas that appeared to be erythroid hematopoietic based on H&E-stained sections (Fig 1F).
Mildly increased EMH in the spleen was characterized in two rasH2-Tg mice treated with control vehicle. These mice were sacrificed at week 27. Neoplasms in these two mice were limited to Harderian gland adenomas. Hematopoietic cells in the red pulp were more numerous than in the two controls described with normal morphology. The nests of hematopoietic cells had poorly defined borders, but normal morphologic features of the spleen were otherwise well defined. The hematopoietic tissue included a normal distribution of erythroid, myeloid, and megakaryocytic cells, with a left shift (a greater proportion of immature cells) and a normal maturation sequence. Circulating numbers of erythrocytes, reticulocytes, and leukocytes in these two mice were within the range of the rest of the concurrent control group, and hematopoietic tissue in the bone marrow was histologically normal. Immunohistochemical labels in the spleen were similar to those in the two normal controls.
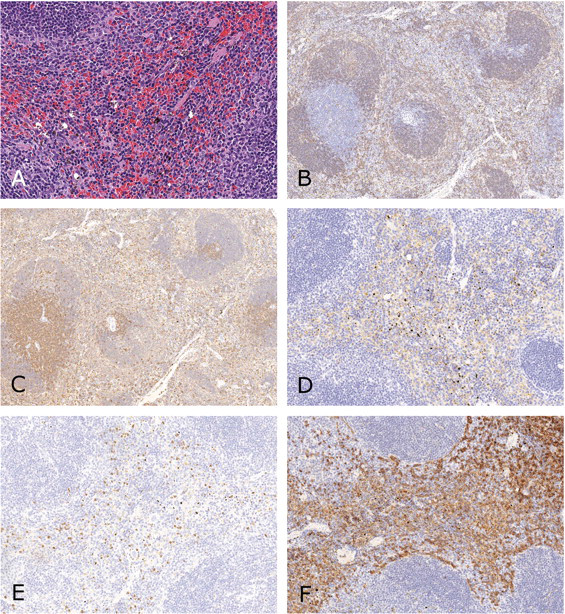
Spleen of rasH2-Tg mice with normal hematopoietic tissue. (A) Small nests of hematopoietic tissue in red pulp, hematoxylin and eosin (300×). (B) Moderate number of B lymphocytes in mantle zones and follicles, CD45R immunolabel (100×). (C) T lymphocytes are numerous in periarteriolar lymphoid sheaths and are scattered in red pulp, CD3 immunolabel (100×). (D) Moderate numbers of macrophages/histiocytes are in the red pulp, F4/80 immunolabel (200×). (E) Individual neutrophil precursors are scattered in the red pulp, myeloperoxidase, granulocyte marker, immunolabel (200×). (F) Erythrocytes are present throughout the red pulp, and erythrocyte precursors are present in areas of hematopoiesis, Ter-119 immunolabel (200×).
Hematoproliferative change in the spleen was characterized in eight rasH2-Tg mice treated with MNU. Six of these mice were sacrificed at scheduled termination, week 27. The other two were sacrificed at week 19 and week 22. Each of the mice with hematoproliferative change had one or more neoplasms, as listed above. Three of the mice had lymphoma involving the spleen. Spleens ranged grossly from normal size to moderately enlarged. The hematoproliferative change was characterized by extensive hematopoietic elements that were left shifted, with blast cells predominating. The maturation sequence was mildly disrupted in five of the eight mice. Hematopoietic cell lineages were normally distributed in about half of the mice; the rest had a higher relative population of erythroid cells (Fig. 2A ). Erythrocytes and erythroid precursors, cells labeling for TER-119, were similar to those in controls but with increased numbers of erythroid precursors labeled (Fig. 2B). The relative number of myeloid hematopoietic cells, characterized by cytologic features and labeling for MPO, were normal to relatively reduced in most mice. In one mouse, increased numbers of cells in apparent areas of myeloid hematopoiesis were labeled for MPO (Fig. 2C). Lymphoid structures were normal or only slightly effaced. Expansion of cells that labeled for CD-3 occurred in the three mice with T-cell lymphoma (Fig. 2D), but margins were poorly defined. Lymphoid structures were normal or reduced in size in the mice with hematoproliferative change but without T-cell lymphoma (Fig. 2E). The number of macrophages/histiocytic cells, cells labeling for F4/80, was generally similar to controls, but there appeared to be increased size and/or number of cells labeled in red pulp/sinuses in three of eight mice. Low to moderate numbers of hematopoietic cells had atypia, with abnormal features such as prominent angular nucleoli. Dysplasia, present in five of the mice, was characterized by disruption of the maturation sequence of any of the three lines (erthyroid, myeloid, or megakaryocytic). Dysplasia was often readily apparent in megakaryocytic cells, with fragmented nuclei attached by filamentous strands (Fig. 2F). Dysplasia was more recognizable in the erythroid line than in the myeloid line. Disruption of maturation in the erythroid line was evident as discontinuity in nuclear and cytoplasmic development stages (i.e., cells with similar nuclear development stages having different cytoplasmic development stages; Fig. 2F).
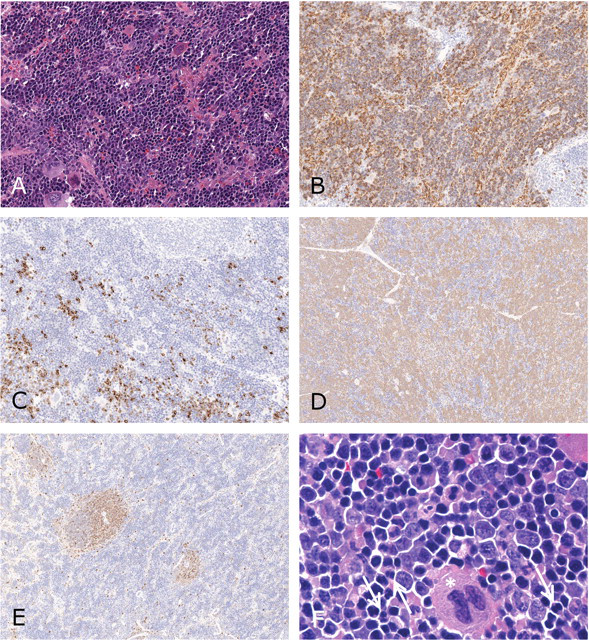
Spleen of rasH2-Tg mice with hematoproliferative change. (A) Exuberant hematopoietic tissue with all lineages and predominance of erythroid precursors, hematoxylin and eosin (H&E; 300×). (B) Cytoplasm of many hematopoietic cells labels as erythrocytes, Ter-119 immunolabel (200×). (C) Hematopoietic cells labeling for myeloperoxidase, granulocyte marker (MPO) increased in some areas and decreased in others, MPO immunolabel (200×). (D) T lymphocytes are numerous and diffusely present in splenic tissue of mouse with lymphoma, CD3 immunolabel (100×). (E) Reduced numbers of T lymphocytes in mouse with hematoproliferative change without lymphoma, CD3 immunolabel (100×). (F) Predominance of erythroid hematopoiesis with binucleated metarubricytes (arrows), and discrepancies in nuclear/cytoplasmic development. A megakaryocyte has nuclear fragmentation (asterisk), H&E (1,000×).
Hematoproliferative neoplasia in the spleen was characterized in four rasH2-Tg mice treated with MNU. These mice were sacrificed because of moribundity or died during the study. All had multiple neoplasms. Other than the hematoproliferative neoplasia, neoplasms involving the spleen were T-cell lymphoma and hemagiosarcoma, each affecting one mouse. Two mice had foci of sinusoidal ectasia, thrombosis, hemorrhage, and/or necrosis in the spleen. Spleens were grossly enlarged in all four mice (0.6 × 2.8 to 1.0 × 3.3 cm). In the three mice with hematoproliferative neoplasia but without lymphoma, lymphocytic structures were obliterated by the hematopoietic cells. Only remnants of the PALS or follicles were evident on immunohistologic-labeled sections (Fig. 3A , 3B). Dysplasia affected all three major hematopoietic cell types, was easily recognizable in megakaryocytic cells, and occurred to a greater degree in erythroid cells in two mice. Cells that were cytologically consistent with erythroid precursors but were blastic and/or dysplastic labeled less frequently for TER-119 than normal erythroid hematopoietic cells. One mouse had an apparent predominance of immature myeloid cells; most of the splenic parenchyma was replaced by immature granulocytic cells with predominantly immature and abnormal forms (Fig. 3C). Smaller areas of splenic parenchyma in this mouse had erythrocytic and megakaryocytic proliferation. The erythrocytic maturation was generally normal; many megakaryocytes had fragmented nuclei (Fig. 3D). Labeling for MPO was generally increased in this mouse (Fig. 3E), consistent with the morphologic appearance of myeloid cells. In this mouse, macrophages/histiocytes (cells labeling for F4/80) were similar in relative number or slightly increased over controls but often appeared to be increased in size, with more prominent cell processes, in splenic sinuses (Fig. 3F). Hematology parameters were measured in three of the four mice with hematoproliferative neoplasia. Erythrocyte counts were decreased in all three. Neutrophil counts were increased in two mice, and lymphocyte counts increased in one. Bone marrow was hypocellular in the mouse with lymphoma.
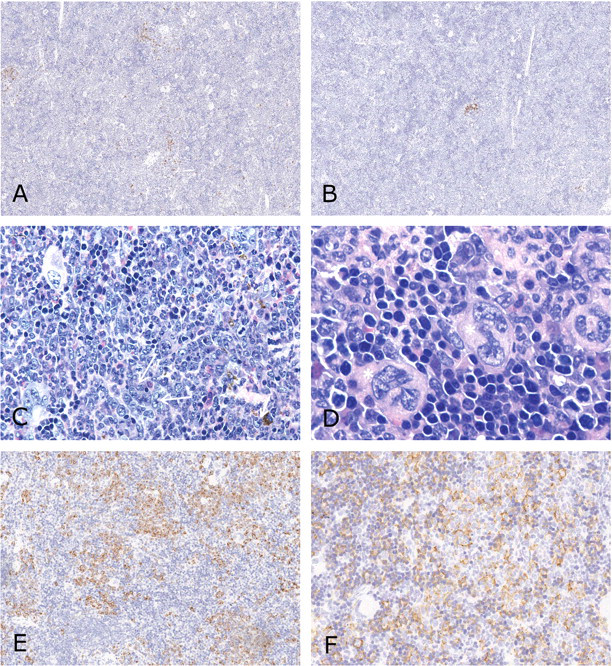
Spleen of rasH2-Tg mice with hematoproliferative neoplasia. (A) Greatly reduced numbers of T-lymphocytes, CD3 immunolabel (100×). (B) Greatly reduced numbers of B-lymphocytes, CD45R immunolabel (100×). (C) Spleen with predominance of blastic and immature granulocytic cells. A number of abnormal band forms are present (arrows), hematoxylin and eosin (H&E; 500×). (D) Area of megakaryocytic and erythrocytic hematopoiesis in same spleen as previous figure. Megakaryocytic nuclei are fragmented (asterisks). Erythrocytic proliferation generally shows normal development, H&E (1,000×). (E) Predominance of neutrophil precursors shown by labeling for myeloperoxidase, granulocyte marker, immunolabel (200×). (F) Increased size and/or number of macrophages/histiocytes in sinuses, F4/80 immunolabel (350×).
Mice with hematoproliferative neoplasia had increased cellularity within the hepatic sinuses and often in aggregates around vessels and portal areas of the liver. These cells generally labeled for CD45. In the mouse with lymphoma, most of these cells labeled for CD3, but there were a number of neutrophil aggregates that labeled for MPO. In the mouse with a predominance of myeloid precursors in the spleen, few cells in the hepatic sinuses labeled for MPO. Many of the cells labeled for CD3, and Kupffer cells (labeling for F4/80), appeared increased in size and number. Kupffer cells were also increased in size and number in the other two mice with hematoproliferative neoplasia. Variable numbers of cells labeled for CD3 and MPO.
Discussion
In this report, we describe proliferative changes in hematopoietic tissue in the spleen that have features suggesting that the proliferative process is atypical compared with the reactive hematopoietic process that commonly occurs in mice. The primary features suggesting this include dysplasia and displacement/disruption of normal splenic architecture. Increased EMH and erythroid hyperplasia in the spleen have been described previously in the rasH2 mouse treated with MNU (Machida et al. 2008; Takaoka et al. 2003), but it has not been reported as atypical. Lymphoma is a common neoplasm in this model and often involves the spleen (Takaoka et al. 2003; Usui et al. 2001). Granulocytic leukemia has been reported at a low incidence in rasH2-Tg mice treated with MNU (Mitsumori et al. 1998; Yamamoto, Mitsumori, et al. 1996).
The most likely reason this entity has not been reported previously is that excessive hematopoiesis in the mouse spleen is generally recognized as a reactive process. The mouse spleen has extensive potential for reactive hematopoiesis in response to inflammatory, neoplastic, or hematopoietic insults, and this has been described as being so extensive as to mimic neoplasia (Ward 1990). The spleen may enlarge with increased hematopoiesis that may involve erythropoiesis, myelopoiesis, and/or megakaryocyte hyperplasia with histologic features of splenic sinuses filled with hemopoietic elements and atrophic white pulp. A distinguishing feature of granulocytic leukemia is invasiveness and/or replacement of normal tissue of the spleen (Long et al. 1986), but myeloid hyperplasia may appear similar to a neoplastic process when the white pulp is depleted and the spleen is filled with immature and mature myeloid elements (Ward 1990).
This exuberant proliferation of hematopoietic elements in the spleen of rasH2-Tg mice treated with MNU has some similarities to a myelodysplastic or a myeloproliferative disorder. Myelodysplastic syndromes in humans present as a heterogeneous group of lesions that occur as idiopathic conditions as well as after therapy with alkylating agents and/or radiation (Cazzola and Malcovati 2005). A hallmark of myelodysplasia is cytopenia, often presenting initially as anemia. Other cytopenias (neutropenia, thrombocytopenia) may appear early or late, and abnormalities of neutrophils and platelets may be evident on blood smears. These changes are reflective of ineffective hematopoiesis (Malcovati and Cazzola 2008). Conditions with features overlapping myelodysplasia and myeloproliferative disorders are identified in the World Health Organization (WHO) classification of myeloproliferative neoplasms, and this category includes a diagnosis of myelodysplastic/myeloproliferative disease, unclassificable (Vardiman et al. 2002). The WHO classification of these conditions relies on morphologic, cytochemical, and immunophenotypic features of neoplastic cells, with a heavy reliance on the percentage of blast cells in blood and bone marrow. Cytologic variability is a feature of many of the conditions. Some of the myeloid neoplasms are also defined by specific abnormalities of genetic pathways that affect myeloid differentiation.
The current studies have a number of limitations preventing complete characterization of the hematopoietic proliferation. Hematology evaluations were conducted in only some of the affected mice, and then only at a single time point. Many of the mice with the more advanced hematoproliferative disorder had anemia. Reticulocyte counts were not measured in all mice, and reticulocyte counts were elevated in only some of the affected mice in which they were measured. While a potential source of anemia (e.g., intestinal neoplasm) was present in some mice, this could not be confirmed as the cause of anemia. Blood smears were not available for evaluation for abnormal cells, and the evaluation of bone marrow was limited to routine examination of H&E-stained sections of decalcified bone. These factors, coupled with the lack of an apparent source of blood loss, confounded interpretation of whether hematopoiesis were inefficient. The immunohistochemical evaluation was limited to a subset of the mice, with a selection to those with better tissue preservation.
The mice were not tested for genetic alterations that could possibly be related to differentiation of hematopoietic cells. Genetic alterations in addition to the expression of the ras oncogene in these mice were likely necessary for the expression of the hematologic abnormalities since these abnormalities have not been identified previously in the rasH2-Tg mice not treated with MNU. Additional genetic abnormalities would not be unexpected, considering the mutagenic/clastogenic properties of MNU. There is also a possibility that the hematoproliferative processes described herein are a response to abnormal cytokine production associated with the occurrence of other neoplasms. All of the rasH2-Tg mice treated with MNU had at least one other neoplasm, so there was no possible comparison to MNU-treated mice without other neoplasms.
In the mouse, granulocytic and erythroid leukemia are described as originating in the spleen (Fredrickson 1990; Frith et al. 2001; Long et al. 1986); therefore, detection of involvement of the spleen only would not rule out a diagnosis of hematoproliferative neoplasia. Characteristics of granulocytic leukemia in mice include early involvement of the spleen, possibly late involvement of bone marrow, dominance of one early stage of development, and invasiveness and/or replacement of normal splenic lymphoid and hematopoietic tissues (Long et al. 1986; Seki and Inoue 1990). Cells appearing in the blood in granulocytic leukemia may be either predominantly juvenile or mature type, but leukemic cells generally have variable nuclear size and shape and often indentation of the nucleus surrounding the Golgi apparatus (Seki and Inoue 1990). Based on these criteria, one of the cases described as hematoproliferative neoplasia (with blastic myeloid cells) in the current study may have been an early granulocytic leukemia, with overt involvement limited to the spleen. There was increased cellularity in hepatic sinuses in this mouse, but most immunohistochemical labeling indicated that most of these cells appeared to be T lymphocytes and Kupffer cells.
While many of the mice in our study appeared to have predominantly an erythroid component in the proliferating hematopoietic cells, on examination of H&E-stained sections, we did not identify this by immunohistochemistry. This may be because the erythroid immunolabel we used, TER-119, is reported to label the later stages of the murine erythroid lineage (i.e., early proerythroblast to mature erythrocyte) but not early cells and not erythroid leukemia cells (Kina et al. 2000). Erythroid leukemia in the mouse spleen can be difficult to distinguish from severe EMH, if the EMH is severe enough to replace the red pulp with immature erythroid cells that compress and/or displace the white pulp (Fredrickson 1990; Frith et al. 2001). One differentiating feature of severe EMH from erythroid leukemia is that a stimulus (e.g., inflammation, blood loss) is generally present for EMH. Use of this criterion for diagnosis imposes a functional assessment on a morphologic diagnosis and would not rule out the occurrence of independent events.
Additional characterization of this spectrum of hematoproliferative changes in rasH2 mice is necessary to differentiate it from the reactive hematopoiesis in the spleen that can occur in mice in response to inflammatory, neoplastic, or hematopoietic insults. This characterization includes critical evaluation of blood, spleen, and bone marrow smears. While these evaluations might further the characterization of the splenic changes, they are not necessary to substantiate the positive carcinogenic response in the rasH2-Tg mouse treated with MNU.
Footnotes
Acknowledgment
The authors would like to acknowledge Dr. A. E. Shultze for his assistance in reviewing histologic slides, consulting on descriptions, and reviewing the article.
