Abstract
Diuron (3-[3,4-dichlorophenyl]-1,1-dimethyl urea) is an herbicide with carcinogenic activity in rats and mice, which have developed respectively urothelial and mammary gland tumors in long-term studies. Accordingly, diuron has been categorized as a “likely human carcinogen” by the U.S. Environmental Protection Agency. Although the carcinogenesis-initiating activity of diuron has been reported in an early initiation-promotion mouse skin study, its genotoxic potential has been disputed. It is necessary to clarify the mode of action through which it has caused rodent neoplasia and verify its relevance to humans. Herein, two experiments were developed to verify the initiating and promoting potentials of diuron in a twenty-three- and a twenty-one-week–long mouse skin carcinogenesis protocol. In one, dimethylsulfoxide (DMSO) was the solvent for the herbicide; in the other, acetone was the alternative solvent in order to verify whether DMSO had inhibitory influence on a potential cutaneous carcinogenic activity. The adopted schedule for the tumor-promoting agent 12-O-tetradecanoylphorbol 13-acetate (TPA) resulted in skin ulcers, which demonstrates the need for careful selection of TPA dose levels and frequency of application in this model. In both studies, diuron did not exert any influence on the skin carcinogenesis process, in contrast with results already reported in the literature.
Keywords
Introduction
Diuron (3-[3,4-dichlorophenyl]-1,1-dimethyl urea) is a widely used herbicide for weed control in the cultivation of soy, cotton, sugar cane, citrus fruit, wheat, and coffee crops, as well as in non-crop areas such as airport runways, railroads, and oil-pipes. Occupational exposure is the main concern about this herbicide. Agricultural workers frequently neglect safety practices, including the use of personal protective equipment designed to avoid dermal exposure during pesticide mixing or application (Carpenter et al. 2002; Perry et al. 2002). Moreover, skin-protective equipment (SPE) may fail to cover the skin surface entirely, and transfer may occur when touching the contaminated exterior of the SPE. Additionally, SPE may enable permeation depending on the manipulated substance and/or the SPE material (Brouwer et al. 2005).
Diuron was recognized as carcinogenic to rodents orally exposed to 2,500 ppm through feed in two-year-long bioassays (Iyer 2002). In those studies, Wistar rats developed urothelial papillomas and carcinomas, and NMRI female mice developed mammary gland adenocarcinomas. Pursuant to these findings, the U.S. Environmental Protection Agency (EPA) has categorized this herbicide as a “likely human carcinogen” (US EPA 2004). Because diuron is permitted for agricultural use, it is necessary to clarify the mode of action (MOA) through which this product has caused rodent neoplasia and verify whether it is relevant to humans. In a recent study in this laboratory, it was demonstrated that diuron and/or its metabolites induce urinary bladder carcinogenesis in Wistar rats through direct chemical cytotoxicity followed by regenerative cell proliferation; the specific mechanism of action, however, has not yet been clarified (Rocha et al. 2009).
Despite having been evaluated in several in vitro and mammalian in vivo genetic toxicology assays, the genotoxic potential of diuron is disputable, depending on the experimental protocol, dose levels, and end points evaluated (Grutman et al. 1984; Iyer 2002; Liu 2001). A weak mutagenic activity of diuron was reported in a metabolically activated Ames test with Salmonella thyphimurium, but results were negative in a mouse bone marrow micronucleus assay (Seiler 1978). When tested in Swiss mice, diuron was positive in the bone marrow micronucleus assay (Agrawal et al. 1996) and in the germ cell dominant lethal assay (Agrawal and Mehrotra 1997). However, in the Mutatox test with a dark variant (M169) of the Photobacterium phosphoreum, the genotoxicity of diuron was only suspected (Canna-Michaelidou and Nicolaou 1996).
The biological significance of positive results obtained in genotoxicity cytogenetic assays has been questioned based on their low predictivity of rodent carcinogenicity (Witte et al. 2007). Additionally, it was suggested that the comet assay (single-cell gel assay) is the most preferable method to assess genotoxicity because it is relatively insensitive to cytotoxicity; hence, false-positive results are less frequent, even when high concentrations of the test compounds clearly affect cell viability (Witte et al. 2007). Previous studies from this laboratory reported an absence of diuron genotoxicity in the standard alkaline version of the comet assay with urothelial and peripheral blood cells (Nascimento et al. 2006). Besides, a modified version of the comet assay developed in vitro with CHO cells indicated that diuron also does not induce DNA cross-links (Rocha et al. 2009).
In an early skin initiation-promotion carcinogenesis study with female Swiss mice, the initiating activity of diuron (supplied by Bharat Pulverizing Mills, Bombay) was registered when applied in consecutive doses (250 mg/kg b.w., three times/week for three weeks) followed by applications of the tumor promoter 12-O-tetradecanoyl phorbol 13-acetate (TPA) for fifty-two weeks (Antony et al. 1989). This observation may suggest that diuron is a genotoxin, hence every initiating agent should be assumed to be genotoxic (Pitot and Dragan 2001). However, no initiating potential was verified by Antony et al. (1989) when diuron was applied at a single 250 mg/kg b.w. dose, which could indicate a possible threshold, inconsistent with its supposed initiating potential. In this case, a promoting influence on the mouse skin carcinogenesis process would be more likely, because in this condition a threshold level and a non-genotoxic MOA would be more suitable (Williams 1999). Antony et al. (1989) did not justify the diuron dose levels they evaluated, adding to the unsubstantial aspect of their study.
Herein, the initiating potential of diuron was evaluated in a mouse skin carcinogenesis protocol aimed to reproduce the Antony et al. (1989) experimental design. Besides, the promoting potential was also evaluated using basically the same model. Two experiments were developed; in the first one, dimethylsulfoxide (DMSO) was the solvent for diuron, as used by Antony et al. (1989). Dimethylsulfoxide has been reported to reduce tumor responses in the two-stage skin carcinogenesis assay. In DMBA-initiated Sencar mice, the promoting activity of TPA diluted in DMSO was decreased by up to 99% when compared with TPA diluted in acetone (Slaga and Fischer 1983). In Tg.AC mice, the use of DMSO as the TPA solvent reduced the skin tumor response by up to 66%, whereas administration of TPA in acetone caused nearly maximal tumor responses (Stoll et al. 2001). The reason for this decreased promoting response, however, was not explained in either study. Hypothetically, DMSO rapidly penetrates the skin barrier, carrying noxious molecules like carcinogens, in such a way that the contact with epithelial cells is too short to cause damage (Volden et al. 1983). Considering the possibility that DMSO might have masked the diuron carcinogenic potential in the first experiment, which showed negative results for carcinogenesis, acetone was used as an alternative solvent in the second study.
The present study aimed to evaluate the carcinogenic potential of diuron in a female Swiss mouse skin carcinogenesis assay. The potential role of DMSO or acetone—the herbicide solvents—was also investigated in this process. Regardless of the solvent used, diuron did not exert any carcinogenesis-initiating or -promoting activities in mouse skin in the present study.
Materials and Methods
Animals and Chemicals
These experiments were approved by the Committee for Ethics in Animal Experimentation of the UNESP Medical School, São Paulo, Brazil (protocols 2006-567 and 2008-672). Four-week-old female Swiss mice, obtained from the Multidisciplinary Center for Biological Investigation (CEMIB, UNICAMP, Campinas, SP, Brazil), were kept in polypropylene cages (five animals/cage) with metallic grill covers, and maintained in a room at 22°C ± 2°C, 55% ± 10% humidity, and a twelve-hour light/dark cycle. Animals were provided with commercial pelletized diet (Nuvital, Nuvilab, Curitiba, PR, Brazil) and tap water ad libitum.
After a two-week acclimatization period, mice were randomized to the experimental groups according to body weights. The dorsal skin was shaved using electric clippers one day before the first treatment and weekly thereafter, except for the untreated control.
All animals were examined daily for clinical signs. Incidence, number, and size of gross papillary tumors and ulcers were registered. Body weight and food and water consumption were measured weekly. Mice were euthanized by carbon dioxide inhalation at the end of the experiments or when bearing severe cutaneous ulcers. At necropsy, patchy skin samples were collected, including both normal and ulcerated areas. Skin patches with lesions (ulcers and tumors) were cut randomly for histological analyses. Liver, spleen, kidneys, and lymph nodes were also collected and weighed. All tissues were fixed in 10% buffered formalin, followed by routine histological processing.
Treatments
Positive control groups were initiated with 9,10-dimethyl-1,2-benzantracene (DMBA, Sigma-Aldrich Co., St. Louis, MO, USA), and promoted with 12-O-tetradecanoylphorbol 13-acetate (TPA, Sigma-Aldrich). Diuron (CAS No. 30-54-1) was purchased from Sigma Chemical Co.
The chemical substances were alternatively or successively applied using a micropipette to a shaved dorsal area of approximately 9 cm2, at the following concentrations: DMBA, 52 µg/100 µL acetone; TPA, 3.5 µg/100 µL acetone; diuron, 250 mg/kg b.w. dissolved in 100 µL DMSO or in 300 µL acetone; acetone, 100 µL and 300 µL (vehicle controls). DMBA was applied once, and TPA was applied three times per week until the end of each experiment. Diuron was applied three times per week for three weeks when tested as an initiator, and three times per week until the end of each experiment when tested as a promoter. Acetone was applied once when tested as an initiator and three times per week until the end of the experiment when tested as a promoter.
Experiment 1 consisted of six groups with twenty animals each (Figure 1): Group 1 (G1), untreated control; Group 2 (G2), DMBA followed by TPA; Group 3 (G3), 100 µL acetone followed by TPA; Group 4 (G4), diuron/DMSO followed by 100 µL acetone; Group 5 (G5), diuron/DMSO followed by TPA; Group 6 (G6), DMBA followed by diuron/DMSO. There was a three-day interval between initiation and promotion treatments in groups G4 and G5, and a one-week interval in groups G2, G3, and G6.

Experimental design.
Experiment 2 consisted of nine groups with fifteen animals each (Figure 2): Group 1 (G1), untreated control; Group 2 (G2), DMBA followed by 300 µL acetone; Group 3 (G3), DMBA followed by diuron/acetone; Group 4 (G4), DMBA followed by diuron/DMSO; Group 5 (G5), 100 µL acetone followed by diuron/acetone; Group 6 (G6), 100 µL acetone followed by diuron/DMSO; Group 7 (G7), DMBA followed by TPA; Group 8 (G8), diuron/acetone followed by TPA; Group 9 (G9), diuron/DMSO followed by TPA. There was a one-week interval between initiation and promotion treatment in all groups.

Experimental design.
Experiments 1 and 2 were planned to last until week 25. However, a humane decision was taken to anticipate termination at weeks 23 and 21, respectively, owing to the high number of animals with severe skin ulcers.
Statistical Analysis
The Fisher exact test was applied for comparison of skin lesion incidence between experimental groups and for comparison between ulcer-bearing and “non-ulcer”–bearing animals in relation to the incidence of carcinomas per group. The Mann-Whitney test for independent samples was used to compare the variation of the number of papillary tumors per mouse between ulcer-bearing and “non-ulcer”–bearing animals of the positive control group in Experiment 2. Results were considered significant when p < .05.
Results
From the first to the last week of experiment, mean body weights increased from 25.32 ± 0.20 g to 39.32 ± 1.82 g in the first study, and from 26.63 ± 0.25 g to 38.85 ± 1.60 g in the second study (data not shown). Taking into account the lowest and highest mean body weights, diuron doses ranged from 6.30 mg/application to 9.80 mg/application, representing 250 mg/kg b.w. Food and water consumption varied between groups in both studies (data not shown), but apparently they did not significantly affect body weight gain and viability, since all animals grew during the study.
Table 1 shows the incidence of macroscopic and histological lesions verified in Experiment 1 (Figure 3 ). Only the positive control group (G2, DMBA+TPA) yielded a significantly increased incidence (100%) of gross papillary tumors, which started to appear at week 7; the other two groups also treated with TPA presented few animals with tumors, irrespective of previous exposure to acetone (G3) or diuron/DMSO (G5). When compared with the controls, animals treated with diuron either at the initiation (G4, G5) or promotion steps (G6) did not develop significant incidence of papillary tumors. From week 14 on, animals treated with TPA (G2, G3, and G5) developed skin ulcers in significantly higher numbers when compared with the other groups. Histologically, epidermal hyperplasia was verified in all treated groups, but only the groups exposed to TPA (G2, G3, and G5) showed significantly higher incidence of accentuated hyperplasia, characterized by more than four cell layers (acanthosis) and hypergranulosis. Most of the skin papillary tumors were papillomas, some of them with dysplastic epithelial foci; few of them were keratoacanthomas. Well-differentiated squamous cell carcinomas were found within ulcerated lesions; the incidence was significantly higher in the groups treated with TPA.
Experiment 1: Incidences of cutaneous lesions at the end of week 23 in a female Swiss mouse two-step skin carcinogenesis assay using DMSO as the solvent for diuron.
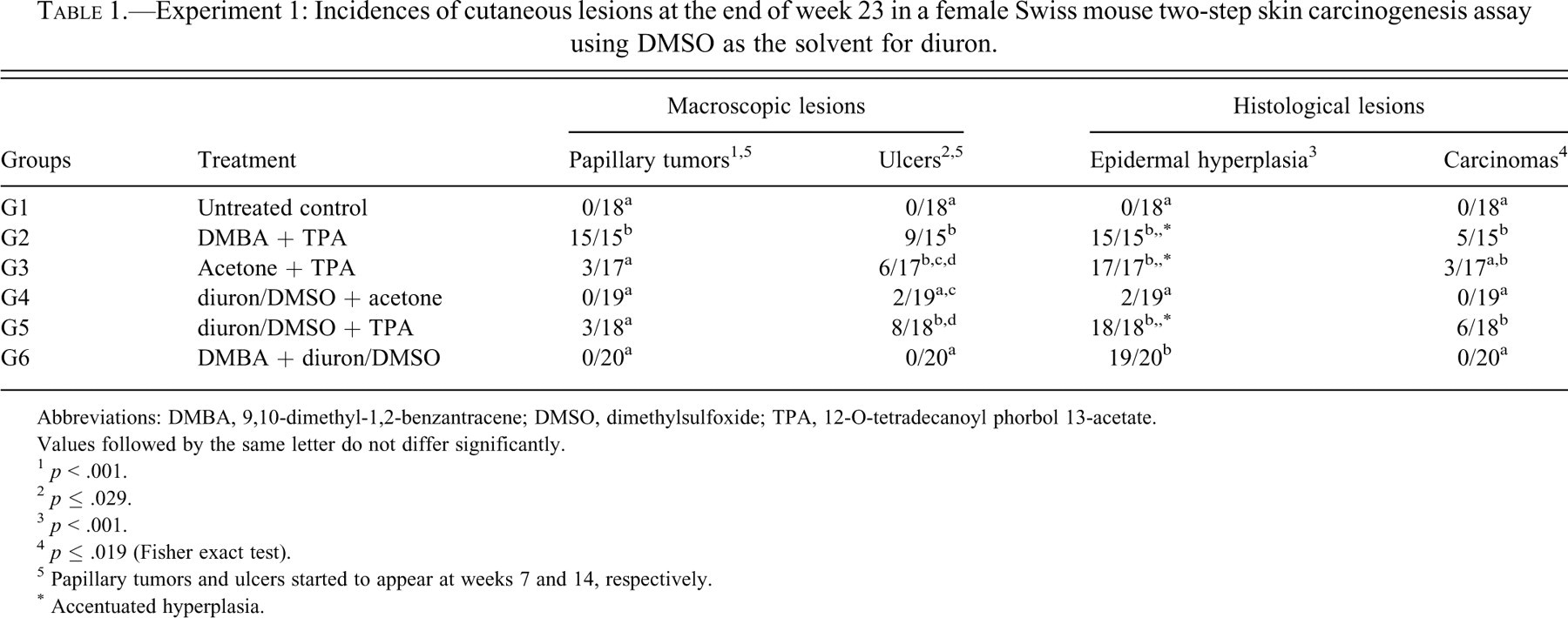
Abbreviations: DMBA, 9,10-dimethyl-1,2-benzantracene; DMSO, dimethylsulfoxide; TPA, 12-O-tetradecanoyl phorbol 13-acetate.
Values followed by the same letter do not differ significantly.
1 p < .001.
2 p ≤ .029.
3 p < .001.
4 p ≤ .019 (Fisher exact test).
5 Papillary tumors and ulcers started to appear at weeks 7 and 14, respectively.
* Accentuated hyperplasia.
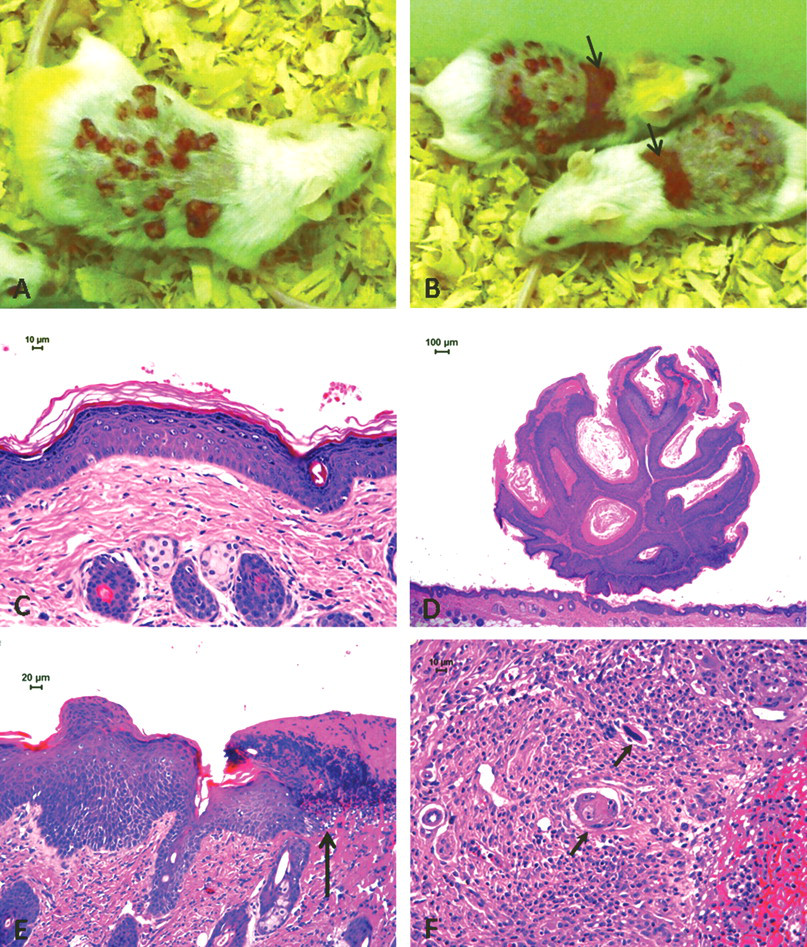
TPA-treated animals. Macroscopic lesions: (A, B) papillomas and ulcers (arrows). Histological lesions: (C) accentuated epidermal hyperplasia; (D) papilloma; (E) acanthosis and paraceratosis at the ulcer border (arrow at the limit); (F) squamous cell carcinoma infiltrating granulation tissue at the ulcer bottom (arrows).
Table 2 shows the incidence of cutaneous lesions in Experiment 2 (Figure 3). Again, macroscopic papillary tumors were observed only in the TPA-treated groups (G7, G8, and G9), but only the positive control group (G7, DMBA+TPA) yielded a significantly increased incidence (100%) of tumors, which started to appear at week 7. Starting from week 13, ulcers occurred in animals exposed to TPA during the promotion step, irrespective of previous initiating treatment with DMBA (G7) or with diuron diluted in acetone (G8) or in DMSO (G9). Histologically, lesions were similar to those found in Experiment 1. Accentuated acanthosis occurred in all animals treated with TPA. Squamous cell carcinomas occurred at the same incidence in the positive control group (G7) and in the groups initiated with diuron diluted either in acetone or DMSO and promoted with TPA (G8 and G9). Figure 3 presents macroscopic and histological lesions from animals treated with TPA.
Experiment 2: Incidence of cutaneous lesions at the end of week 21 in a female Swiss mouse two-step skin carcinogenesis assay using acetone or DMSO as the solvent of diuron.
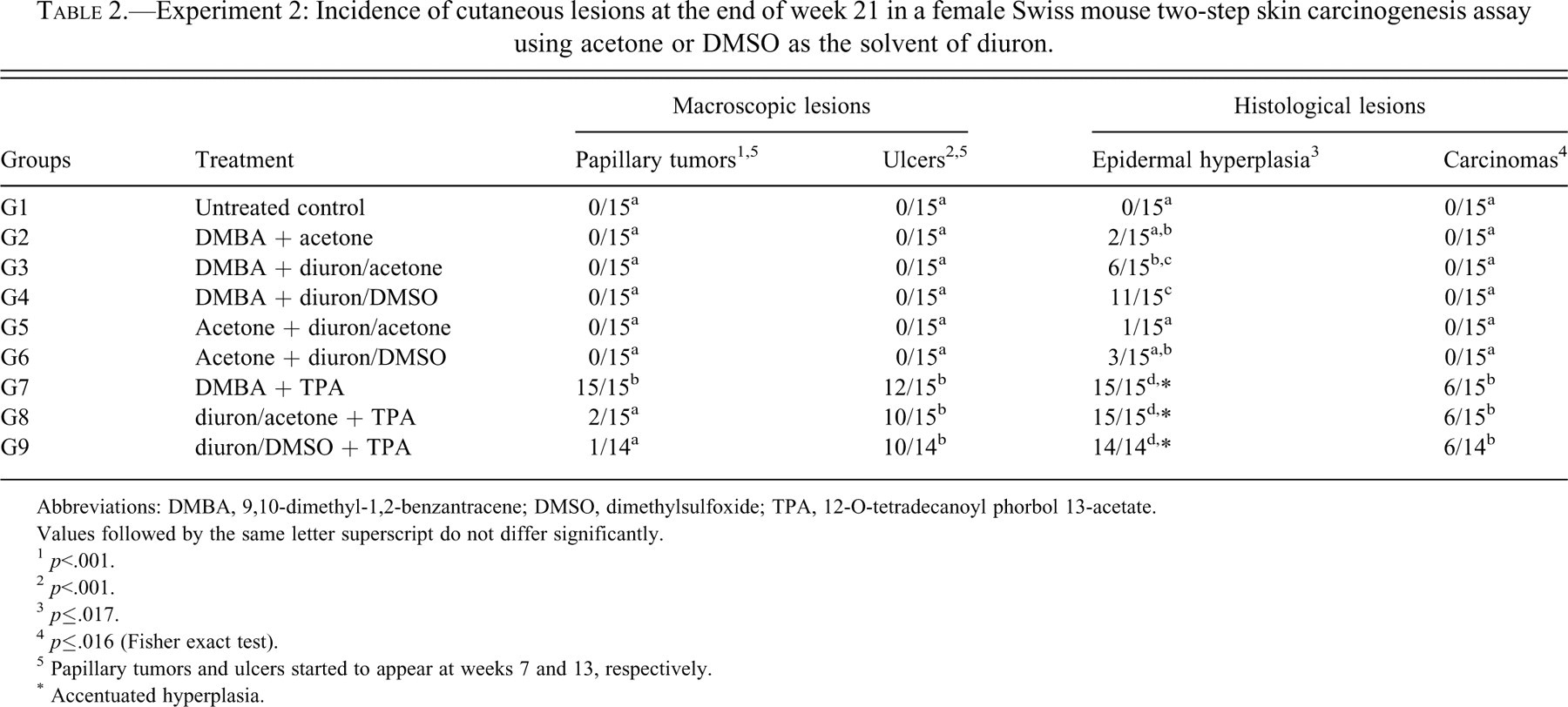
Abbreviations: DMBA, 9,10-dimethyl-1,2-benzantracene; DMSO, dimethylsulfoxide; TPA, 12-O-tetradecanoyl phorbol 13-acetate.
Values followed by the same letter superscript do not differ significantly.
1 p<.001.
2 p<.001.
3 p≤.017.
4 p≤.016 (Fisher exact test).
5 Papillary tumors and ulcers started to appear at weeks 7 and 13, respectively.
* Accentuated hyperplasia.
In Experiment 2, gross papillary tumor counts per mouse were recorded at weeks 9, 12, and 16. Because in diuron-initiated, TPA-promoted animals (G8 and G9) these tumors occurred in few animals, which developed no more than one papillary tumor each, tumor counting was restricted to the positive control group (G7, Table 3 ). In general, at week 16, ulcer-bearing animals presented fewer tumors when compared with “non-ulcer”–bearing animals. The association between the number of gross papillary tumors and ulcer development was statistically evaluated considering tumor multiplicity in ulcer-bearing and in “non-ulcer”–bearing animals (Table 4 ). The number of papillary tumors in ulcer-bearing animals decreased by 34.6% between weeks 9 and 16 and by 43.7% between weeks 12 and 16; in “non-ulcer”–bearing animals, the number of tumors increased by 102.6% between weeks 9 and 16, and by 14.2% between weeks 12 and 16. These observations indicate that skin ulcer development compromised the persistence of papillary tumors, whereas in “non-ulcer”–bearing mice, the multiplicity of these tumors increased continuously throughout the study.
Female Swiss mouse two-step skin carcinogenesis bioassay. Experiment 2: Number of papillary tumors per mouse and ulcer occurrence at week 16 in the positive control group (G7, DMBA + TPA).
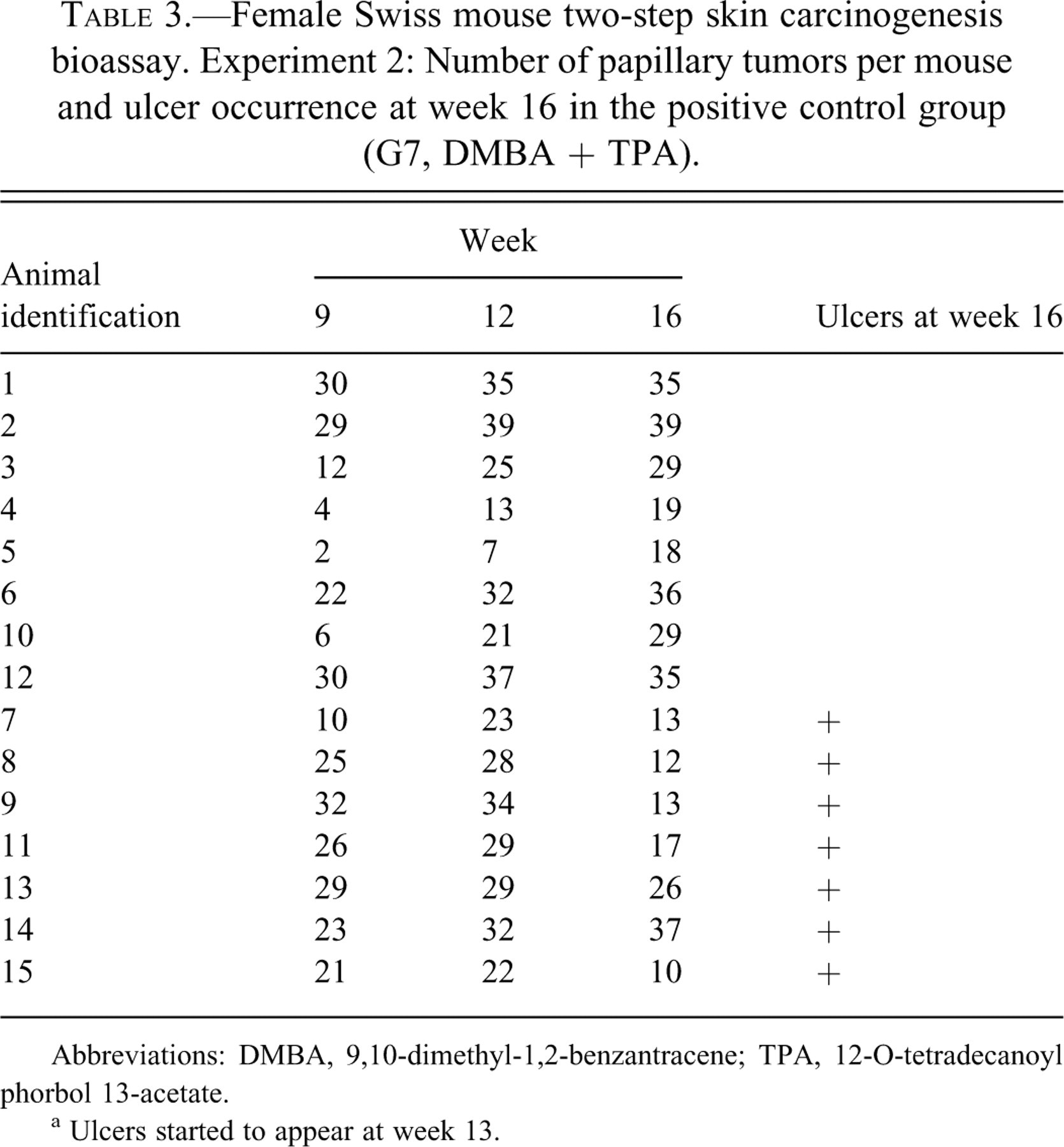
Abbreviations: DMBA, 9,10-dimethyl-1,2-benzantracene; TPA, 12-O-tetradecanoyl phorbol 13-acetate.
a Ulcers started to appear at week 13.
Female Swiss mouse two-step skin carcinogenesis bioassay. Experiment 2, positive control group (G7, DMBA + TPA): Association between ulcer development and number of gross skin papillary tumors per mouse from week 9 to week 16. *

* Data are expressed in median and interquartile range.
a Ulcers started to appear at week 13.
b Mann-Whitney test for independent samples.
Tables 5 and 6 present the comparison between ulcer-bearing and “non-ulcer”–bearing animals in relation to the incidence of histologically detected carcinomas in both experiments. In Experiment 1, carcinomas occurred at incidences of 56%, 50%, and 75% only in the animals that developed ulcers during TPA treatment (G2, G3, and G5, respectively). Association between ulcers and carcinomas was statistically significant in those groups, pointing to the critical role of TPA in establishing ulcerations and carcinomas. In Experiment 2, carcinomas occurred in ulcer-bearing animals at incidences of 42%, 60%, and 60% in groups G7, G8, and G9, all of them treated with TPA during the promotion step. One “non-ulcer” animal in the positive control group (G7, DMBA + TPA) developed a papillary carcinoma. The association between ulcers and carcinomas was significant for group G8 (p = .044), but not for G7 and G9.
Female Swiss mouse two-step skin carcinogenesis bioassay. Experiment 1: Association between ulcer development and incidence (%) of carcinomas per group.
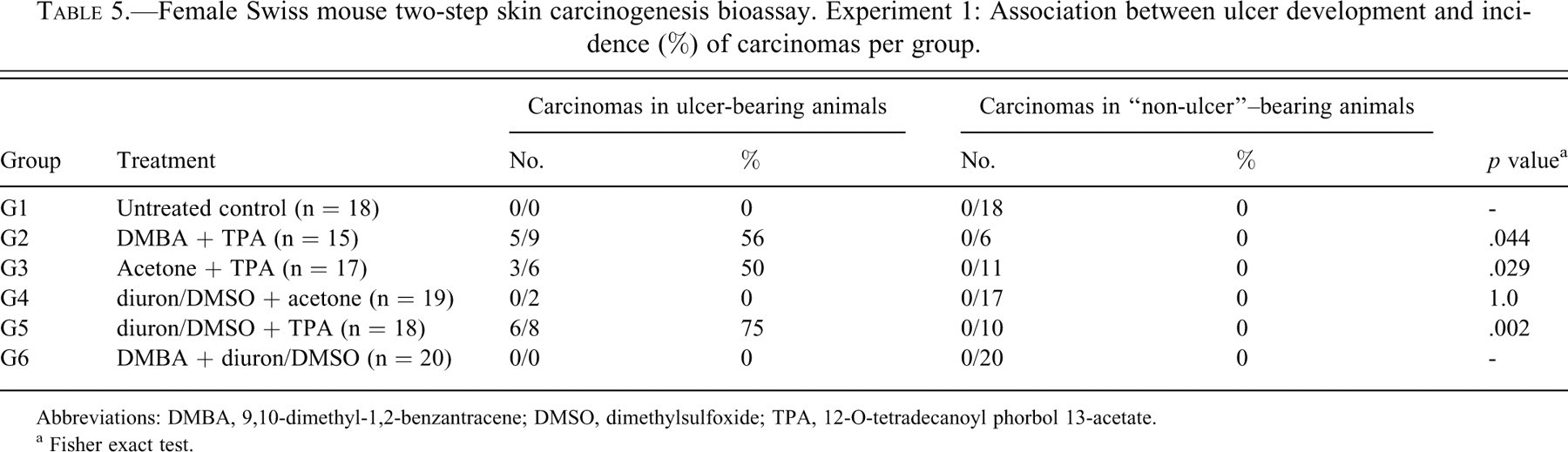
Abbreviations: DMBA, 9,10-dimethyl-1,2-benzantracene; DMSO, dimethylsulfoxide; TPA, 12-O-tetradecanoyl phorbol 13-acetate.
a Fisher exact test.
Female Swiss mouse two-step skin carcinogenesis bioassay. Experiment 2: Association between ulcer development and incidence (%) of carcinomas per group.
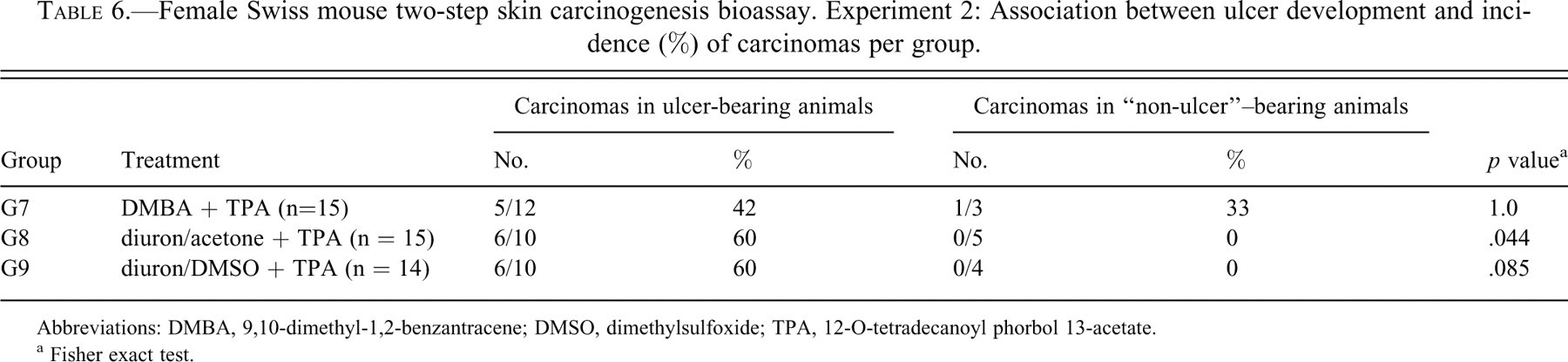
Abbreviations: DMBA, 9,10-dimethyl-1,2-benzantracene; DMSO, dimethylsulfoxide; TPA, 12-O-tetradecanoyl phorbol 13-acetate.
a Fisher exact test.
Discussion
In the present studies, gross skin papillary tumors were registered in significantly higher incidence, as expected, in the positive control group, treated sequentially with DMBA and TPA. In remarkably lower incidences, papillary tumors were also observed in other groups treated with TPA in the promotion step, irrespective of the initiating treatment with diuron/DMSO, diuron/acetone, or acetone only. When diuron was administered during the promotion step, despite being diluted in DMSO or acetone, it did not induce papillary tumors or other relevant skin lesions, irrespective of previous treatment with DMBA or acetone. If diuron had a genotoxic/initiating potential, it would have induced skin tumors after being successively applied during the extended period that characterizes the promotion step, since initiating agents are often complete carcinogens (Iversen 1994).
Therefore, under the experimental conditions adopted, diuron did not exert carcinogenesis-initiating or -promoting activities in female Swiss mouse skin, and varying the herbicide solvent did not influence the results. The present data strongly diverge from previous findings that indicated tumor initiatory activity by diuron in a mouse skin assay (Antony et al. 1989). In addition, they suggest that diuron is not a genotoxic agent, which is in line with information from this laboratory that reported non-genotoxicity of the herbicide under two different comet assay protocols (Nascimento et al. 2006; Rocha et al. 2009).
A striking observation in these studies was the development of severe skin ulcers and histologically detected carcinomas in the groups exposed to TPA. This non-genotoxic compound has been reported to be a weak but complete carcinogen when applied repeatedly in the hairless Oslo mouse skin, in which even a positive dose-response has been demonstrated (Iversen 1985; Iversen 1994). In the Iversen studies, 6.17 µg/100 µL of TPA diluted in acetone applied twice a week to non-initiated mouse skin produced 9% incidence of tumor-bearing animals after fifty-five weeks. The NTP has designed a protocol in which 5 µg of TPA applied once a week for fifty-one weeks was sufficient to induce tumors in 13% of non-initiated female Swiss mice (NTP 1996). In a recent review of the multistage skin chemical carcinogenesis protocol, it was suggested that TPA should be applied only once or twice a week, depending on the dose and mouse strain used (Abel et al. 2009).
In the early Antony et al. (1989) study, which reported initiating potential of diuron when applied in multiple doses, TPA 5 µg was applied three times a week, for fifty-two weeks. Thus, it is possible that the high frequency of TPA applications and the dose levels adopted by Antony et al. (1989) were sufficient to induce tumors, even in the absence of any initiating procedure, leading to the 73% tumor incidence verified in that group. This possibility, however, does not explain the absence of tumors verified by those authors in the group initiated with a single dose of diuron and promoted with the same TPA schedule (Antony et al. 1989), and this controversy remains to be elucidated. The TPA dose level used in our studies was slightly lower (3.5 µg/100 µL) than those suggested by others (NTP 1996; Abel et al. 2009), but it was applied three times a week, the same method as used by Antony et al. (1989) and in dissonance with the recommendations of Abel et al. (2009). It is intriguing that Antony et al. (1989) did not report on the histological evaluation of lesions and did not refer to ulcer or carcinoma developments in their study. The occurrence of these lesions in the current study is discussed below.
The present experiments were planned to last twenty-five weeks. This duration was based on the verification of conspicuous development of papillary tumors at this time point in the Antony et al. (1989) study and on other reports using the mouse skin carcinogenesis protocol, which did have the same or shorter lengths (Chan et al. 2004; Chaudhary et al. 2009; Ridd et al. 2006). However, owing to the severe ulcers developed in the TPA-treated animals, experiments were terminated at weeks 23 and 21 for humane reasons. Highly reproducible papilloma burden is expected after twenty weeks of promotion, and progression to squamous cell carcinoma usually occurs within twenty to fifty weeks of treatment with tumor-promoting agent (Abel et al. 2009). Therefore, it can be assumed that the experiments' duration was sufficient to detect potential diuron-induced skin papillary tumors.
High incidence of skin ulcers in groups treated successively with TPA is a relatively common finding owing to the irritating properties of this chemical, especially in the skin of the Swiss mouse (Klein-Szanto et al. 1986; NTP 1996). In this study, it was demonstrated that several papillary tumors were eradicated by the process of ulceration, as the number of tumors per mouse decreased concurrently with ulcer development.
Similarly to the ulcer incidence, the significantly higher incidence of histologically detected carcinomas in the TPA-treated groups is probably a consequence of TPA cytotoxicity, regardless of the initiating treatment. Besides TPA being a weak skin carcinogen (Iversen 1985; Iversen 1994), it is possible that the repetitive cell injury it provoked led to epithelial disruption and ulceration, inducing the surviving epithelium at the ulcer borders to regenerate and eventually progress to malignancy. This possibility is supported by the strong association registered between carcinomas and ulcers. Although a significant positive association was not verified in all groups from Experiment 2, there was a clear biological relationship between ulcers and carcinomas, and it is very likely that the absence of statistical significance of this association may be a result of the size of these groups. Additionally, there are no significant differences between the incidence of carcinomas in the positive control group and in the other TPA-treated animals in both Experiment 1 and Experiment 2, suggesting that TPA treatment itself, and not the initiating treatment, was critical for the development of epithelial malignancies. These observations reinforce the recommendations that experimental skin treatment by TPA should follow careful selection of doses and frequency of applications (Abel et al. 2009) in order to avoid incidental events that may hinder the interpretation of results owing to the cytotoxicity of this compound.
At relatively high dietary concentration (2,500 ppm), diuron is carcinogenic to the rat urinary bladder mucosa. The mode of action the herbicide plays on that target tissue is probably through cytotoxicity followed by sustained urothelial regeneration (Rocha et al. 2009). In mouse skin, diuron was associated with significantly higher incidences of epidermal hyperplasia–an indirect indication of enhanced cell proliferation–only when associated to DMBA or TPA; otherwise, when applied with either DMSO or acetone, the incidences of epithelial hyperplasia were not relevant. Therefore, contrary to what is observed in the rat urinary mucosa, diuron seems not to induce epidermal cell proliferation. Overall, the present data strongly suggest that diuron acts neither as an initiator nor as a promoter of carcinogenesis in the female Swiss mouse skin assay. Taking into account the limits of extrapolation between rodent and human skin biological events (Enzmann et al. 1998), it can be assumed that potential cutaneous occupational exposure to this herbicide probably does not represent a risk for human skin cancer development.
Footnotes
Acknowledgments
This work was supported by the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP; Grants # 06/60506-1, 06/04630-5 and 08/01809-0). Paulo Roberto Cardoso, Maria Luiza Falaguera, Paulo Cesar Georgete, and Cristina Aparecida Alquati Dorico (TOXICAM Laboratory) provided helpful technical assistance.
