Abstract
Interpretation of chemical-induced effects on the female beagle mammary gland can be difficult owing to the wide variation of normal glandular morphology. In this retrospective study, morphological features of the gland in four (proestrus, estrus, diestrus, and anestrus) phases of the cycle are described. The gland was quiescent (inactive) in proestrus and estrus. In diestrus, with the rise of progesterone, four (I–IV) distinct morphological changes were evident. In phase I, there was exuberant stromal and ductal proliferation. In phase II, there was early lobular development with branching ducts and alveolar proliferation. In phase III, there was an abundance of glandular tissue with large lobules containing secretory material, whereas phase IV had features of early regression, increased interlobular connective tissue, and eosinophilic secretions in distended ducts and acini. In early anestrus, ducts were distended, with eosinophilic secretions with alveolar regression, whereas regression was complete in late anestrus. Glandular morphology was slightly variable in the mammary chain within the same dog. Progesterone receptor expression was prominent in estrus and early diestrus, and peak estrogen receptor expression was noted in diestrus II. Expression of proliferation marker ki-67 was highest in diestrus I, followed by diestrus II. There was excellent concordance between the estrous stage and the glandular morphology.
Keywords
Introduction
The association between the phase of the ovarian (estrous) cycle and the various developmental stages of the mammary gland serves as a common theme in most mammals, although morphological changes in the mammary gland are not uniform across all species. In rats, the mammary epithelium responds to cyclic changes in ovarian hormone levels, with slight changes in the glandular morphology (Schedin et al. 2000). Likewise, cycle-related morphological changes in the mammary glands of cynomolgus macaques are subtle, with slightly increased epithelial tissue in the luteal phase compared to the follicular phase of the cycle (Stute et al. 2004). In women, morphological changes in the breast in relation to the menstrual cycle had been a subject of debate, however, elegant studies performed in the 1980s (Longacre and Bartow 1986; Vogel et al. 1981) and more recently (Navarrete et al. 2005; Ramakrishnan et al. 2002) have provided conclusive evidence of mild cyclical changes in the normal breast lobules. In the human breast, lobular proliferative changes have been variously reported as peaking in the late follicular phase (Ramakrishnan et al. 2002; Vogel et al. 1981) or the luteal phase (Longacre and Bartow 1986; Navarrete et al. 2005), with the latter opinion supported by more quantitative methods.
In contrast to the short estrous cycle of rats or the monthly menstrual cycle of macaques, the canine estrous cycle is unique in that the luteal phase of the cycle is prolonged, as evidenced by long-standing persistence of functional corpora lutea, even in the absence of pregnancy (Concannon et al. 1989). In addition, unlike rats or macaques, the canine mammary gland does not require pregnancy to complete its total differentiation into lobuloalveolar secretory tissue during the luteal phase of the cycle. Because of the broad spectrum of essentially normal morphological (histological) changes in the gland (Nelson and Kelly 1974), interpretation of drug-induced changes in the canine mammary gland and associated reproductive tissues can be complex. Knowledge of the receptor-mediated, hormone-driven morphological changes occurring in the normal beagle mammary gland during the ovarian (estrous) cycle would enable the identification of chemical-induced alterations in the pituitary–ovarian–mammary gland axis. Histologic changes in the canine mammary gland were reported nearly a century ago (Marshall and Halnan 1917), and again in the 1930s (Turner and Gomez 1934) and 1970s (Nelson and Kelly 1974). However, most of the hormonal profiles associated with the estrous cycle, especially in the laboratory beagle, were published much later. Therefore, the purpose of this article is not to reiterate earlier studies but to illustrate, for the toxicologic pathologist, an integrated review of the morphological changes in the mammary gland and the associated changes in the hormonal profile in purpose-bred female laboratory beagles at an age (range 10–13 months) normally used in toxicology studies. The importance of understanding the normal morphological variation of the beagle mammary gland is vital for interpreting toxicity studies.
In a previous retrospective study, we reported the frequency of the different estrous stages and associated morphological features in the reproductive tissues of purpose-bred beagles (Chandra and Adler 2008). The data presented in this study are an extension of the previous study correlating morphological changes in the beagle mammary gland with the ovarian cycle. An adjunct part of this study was to characterize the immunohistochemical expression of hormone receptors (estrogen receptor (ER)-α and progesterone receptor [PR]) and a cell proliferation marker (Ki-67) in representative sections of the mammary gland in different stages of the estrous cycle. Interestingly, most publications on immunohistochemical expression of hormone receptors or proliferation markers in canine mammary tissue have generally used normal mammary tissue, most often from a single dog without due consideration to the stage of the estrous cycle (Geraldes et al. 2000; Martín de las Mulas et al. 2004; Millanta et al. 2005). In routine toxicity studies, a single section of skin with overlying nipple (usually from the inguinal region) is obtained for histological examination of the mammary gland as a representation of the entire gland. Because the concentration of estrogen and progesterone receptors is not uniform in all regions of the mammary gland, with posterior (fourth and fifth pair) glandular tissue having a higher concentration compared to the anterior glands (Donnay et al. 1995), we decided to evaluate any possible morphological differences among the different regions of the gland by examining the entire mammary chain in a few dogs.
Material and Methods
This retrospective study was conducted using data and archival histology slides from thirty-eight previously conducted (2002–2008) toxicity studies. Data from 102 control (vehicle/water treated) dogs from thirty-two studies were reviewed in the previous study (Chandra and Adler 2008) to identify the stage of the estrous cycle. In this study, tissues from an additional thirty-one dogs (for a total of 133) were reviewed from six additional toxicity studies. Each study usually had three dogs (or five if recovery was an end point) in the control group. The in-life portions of all studies were conducted at GlaxoSmithKline in RTP, North Carolina), USA, with identical housing and husbandry conditions. The dogs were housed individually, although commingling with dogs in the adjacent cages was permitted during some hours of the day. All studies were conducted in accordance with current guidelines for animal welfare (Guide for the Care and Use of Laboratory Animals, 1996; Animal Welfare Act, 1966, as amended in 1970, 1976, 1985, 9 CFR Parts 1-3). Procedures used in these studies were reviewed and approved by the internal Institutional Animal Care and Use Committee. All dogs (average age at necropsy was approximately 14.38 months [range 11 to 22.5 months]) used in these toxicity studies were supplied by Marshall Farms (NY).
At the end of the dosing period (two or four weeks duration), dogs were anesthetized and killed by exsanguination, and protocol-required tissues were weighed and subsequently fixed in neutral buffered formalin for histopathological evaluation. For histological evaluation of the mammary gland, a single piece of (inguinal) skin containing the nipple was routinely collected from all dogs in every study. The morphological changes in the mammary gland described in this study are based on identification of the specific estrous stage by histological examination of the ovaries, uterine horns, vagina, and cervix (Chandra and Adler 2008). Histologically, the estrous cycle of the laboratory beagle can be divided into four major phases: anestrus, proestrus, estrus, and diestrus (Chandra and Adler 2008). For this study, hematoxylin and eosin–stained sections of skin (nipple attached) containing the mammary gland from dogs in the four phases of the estrous cycle were examined to describe the morphological features.
All slides in this study were reviewed by a single pathologist (Dr. Chandra) to ensure consistency between dogs, and all tissues from a dog were reviewed simultaneously to ensure morphological changes in individual organs were in synchrony with the stage of the cycle. In addition, the images and histology slides were peer reviewed by the co-authors. The cycle-related glandular changes are gradual and contiguous, and there is no sharp distinction between the different phases of the cycle. Even within one phase, slight variations exist not only between dogs, but within the same dog depending on the anatomic location from which the sample was obtained (see below). This variation in the morphology was even more pronounced for dogs in diestrus. To describe the chronologic morphologic events that appear during the diestrous phase, the entire mammary chain from a few representative cases was selected and the morphological changes were studied in microscopic detail. Therefore, the average changes for individual phases are described here. Photomicrographs were obtained from studies conducted over a six-year period; therefore, staining differences are apparent in the images.
Immunohistochemical stains were done on formalin-fixed, paraffin embedded sections of mammary glands obtained following euthanasia of fourteen stock dogs (control dogs that are not part of any toxicity study). Briefly, two sections of the mammary gland from each stage of the estrous cycle (except proestrus) were immunohistochemically stained for the presence of ER-α, PR, and Ki-67 (cell proliferation marker). Sections of mammary gland were labeled with antibodies specific for ER-α (rabbit anti-human; Invitrogen, Carlsbad, CA, at 1:200 dilution), PR (rabbit anti-human; Beckman Coulter, Fullerton, CA, at 1:25 dilution), and Ki67 (rabbit anti-human; Neomarkers, Fremont, CA, at 1:400 dilution). Ki-67– and ER-α–labeled slides were developed using 3,3′-diaminobenzidine (DAB) (Ventana Medical, Tucson, AZ) as the chromogen. Progesterone receptor–labeled slides were developed using Fast Red (Leica Microsystems, Bannockburn, IL). Sections were counterstained with hematoxylin II (Ventana Medical) (to label the nuclei). The immunoreactivity was scored semiquantitatively as low (+), moderate (++), or strong (+++), incorporating both the intensity of the (chromogen) staining and overall proportion of labeled epithelial cells in the mammary tissue. Nuclear staining was considered specific, and staining of secretory material in ducts or acini (Figure 5) was considered nonspecific.
The beagle mammary chain is generally represented by five pairs of nipples corresponding to cranial and caudal thoracic; cranial and caudal abdominal; and inguinal glands (Miller 1964). To assess any possible variation among the thoracic, abdominal, and inguinal regions, the entire mammary chain was histologically examined from fourteen dogs (stock dogs and not part of any toxicity study) representing all stages of the estrous cycle, except dogs in proestrus. For this procedure, a single section of the nipple (skin and underlying dermis) was trimmed from all grossly visible nipples for microscopic evaluation. The number of nipples varied between the dogs, and a section of the skin was obtained only when a nipple was apparent.
Results
Morphological changes in the mammary gland parallel the cyclic changes in the ovarian (estrous) cycle. A schematic representation of the closely intertwined relationship between the ovarian (estrous) cycle and the morphological changes in the canine mammary gland are depicted in Figure 1. The mammary gland is quiescent (inactive) in the late anestrus, the proestrus, and the estrous phase of the cycle. Proliferation, differentiation, and regression of the mammary gland occur during the Diestrus phase of the cycle. In this study, based on the four distinct histological changes that are evident in the mammary gland during the Diestrus phase of the cycle, we have divided this phase of the estrous cycle in to four phases (I–IV) to describe the changes in the mammary gland.
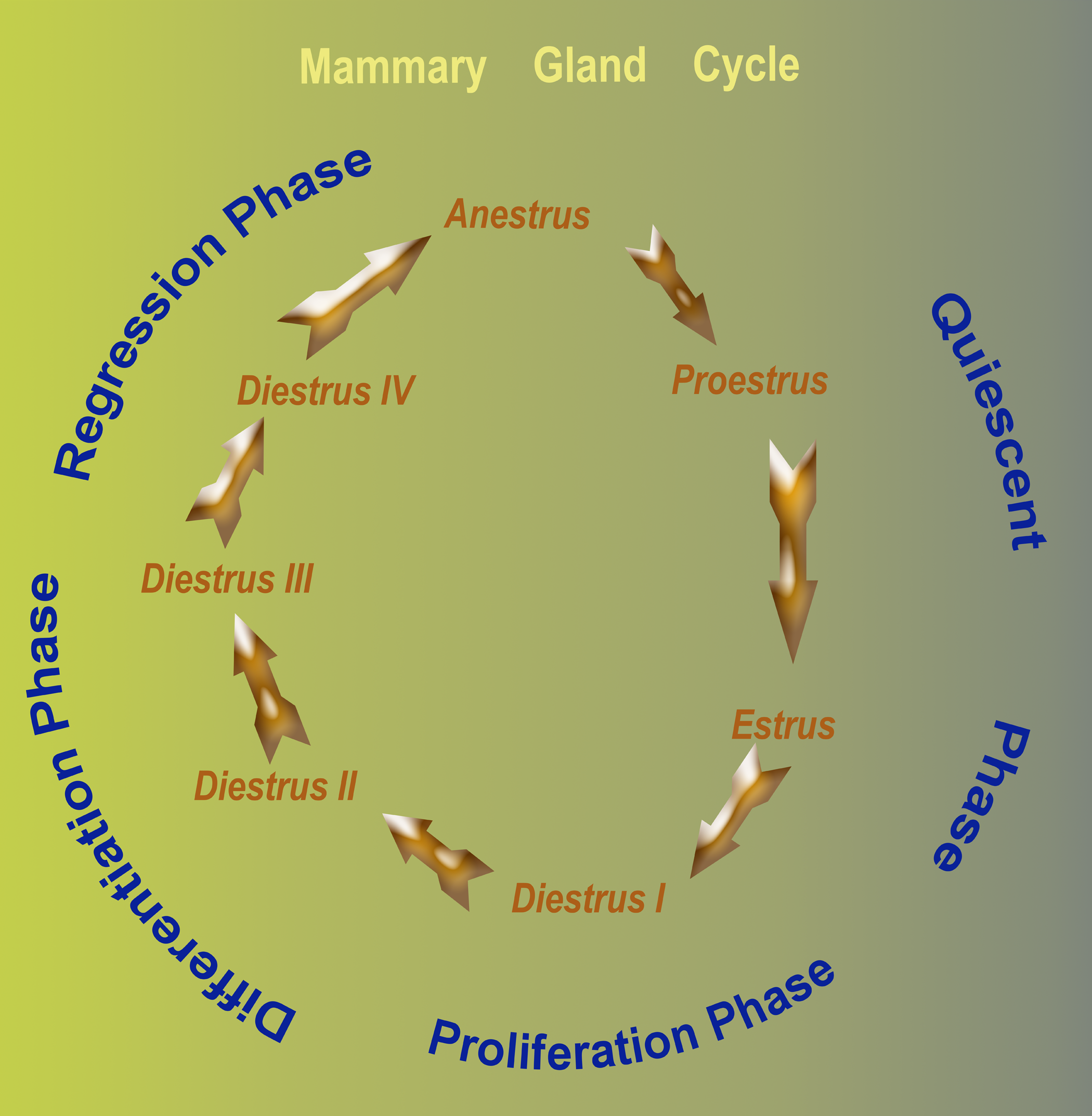
Schematic representation of the histologic changes in the mammary glands of sexually mature laboratory Beagles. The mammary gland in non-pregnant Beagles undergoes cyclical changes that parallel the ovarian (estrous) cycle. The gland is quiescent in anestrus, proestrus and estrus phase of the cycle. Proliferation, differentiation and regression of the mammary gland occur during the diestrus phase of the cycle. Based on the four distinct histological changes in diestrus, a proposed four (I - IV) phase classification this stage is illustrated.
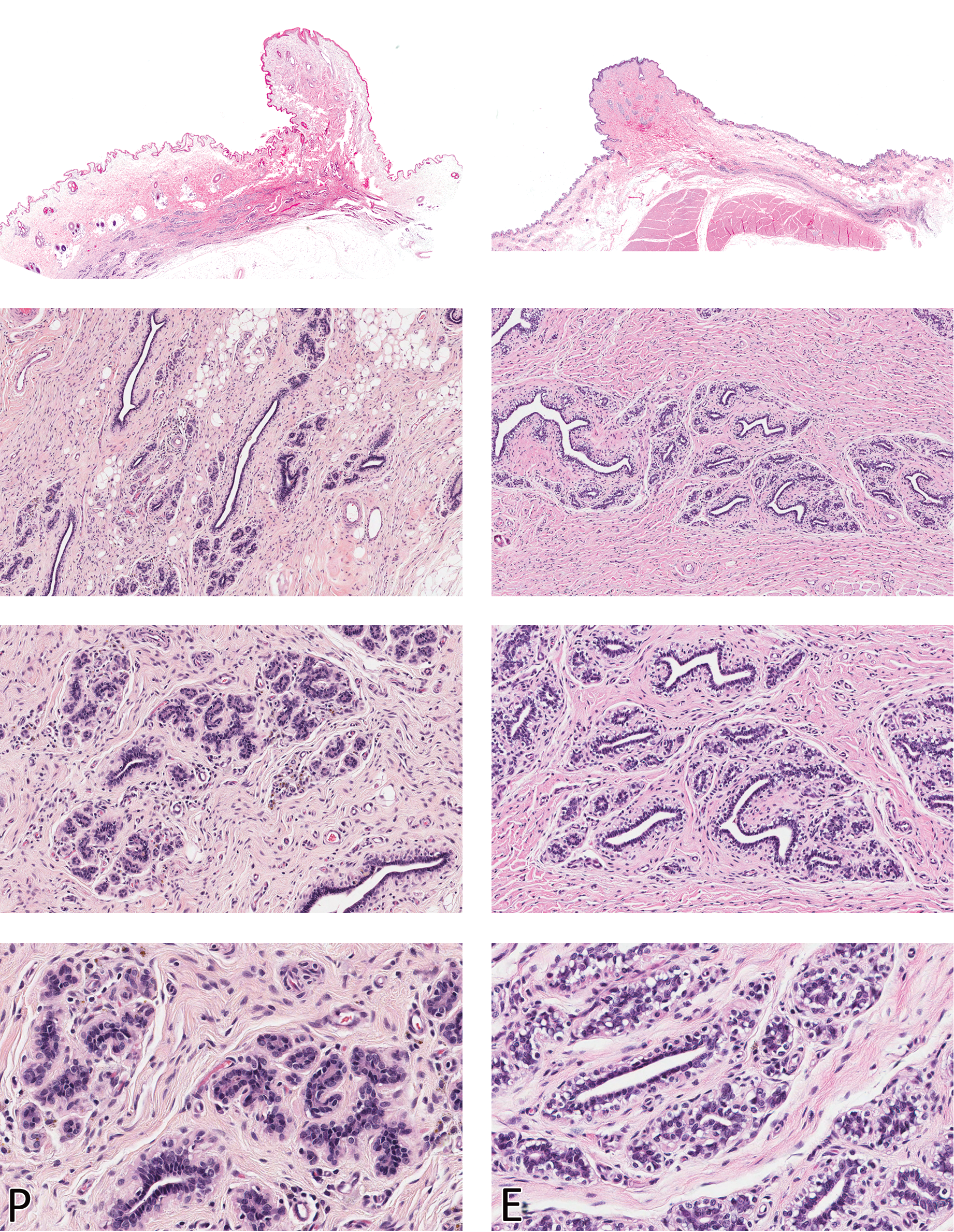
Morphologic characteristics of the Beagle mammary gland in proestrus (P) and estrus (E) are shown in the left and right columns, respectively. The gland is quiescent (inactive) in these two phases and glandular tissue is barely visible in the dermis (top panels). Compare with Figure 3. H&E. Magnification from top to bottom - Sub-gross, 100X, 200X, and 400X
Growth and differentiation of the mammary gland is dependent on ovarian and pituitary hormones. A profile of key hormones involved in the mammary gland morphology and their levels in different phases of the canine estrous cycle are listed in Table 1. Because the mammary gland is often viewed as an endocrine organ secreting growth hormone (GH) (Peaker 1995; Selman et al. 1994), resulting in systemic effects, data on GH levels are also included. This is not an all-inclusive list, and other hormones such as luteinizing hormone (LH), follicle stimulating hormone (FSH), and insulin-like growth factor (IGF) are also important. It is beyond the scope of this article to elaborate on the specific roles. Understanding the relationship between the ovarian cycle and the impact on the subsequent morphology of the mammary gland is important, because dogs can be euthanized during any one of those stages.
Profile of key hormones involved in mammary gland morphology and their levels in different phases of the estrous cycle.

b Olson et al. (1982).
e de Gier et al. (2006).
Mammary Gland Morphology
The number of dogs examined and the stage of the estrous cycle are indicated in Table 2. The average age of dogs at necropsy was approximately 14.38 months (range 11 to 22.5 months).
Frequency of the different phases of the estrous cycle in control beagles (N = 133).

Mammary Gland in Immature Dogs
In immature dogs, before the onset of the first estrous cycle, glandular tissue was barely discernible in the dermis (Chandra and Adler 2008). Examination of the nipple and underlying dermis for evidence of glandular tissue is a surrogate marker for assessing sexual maturity in beagles.
Mammary Gland in Proestrus
With the start of the estrous cycle, the mammary gland in proestrus was essentially quiescent and mostly composed of inactive ducts. Hemosiderin pigment and occasional apoptosis were observed, but not mitoses. The entire complex of skin and gland was relatively thin compared to that in diestrus, owing to the compact stroma and inactive glands (Figure 2).
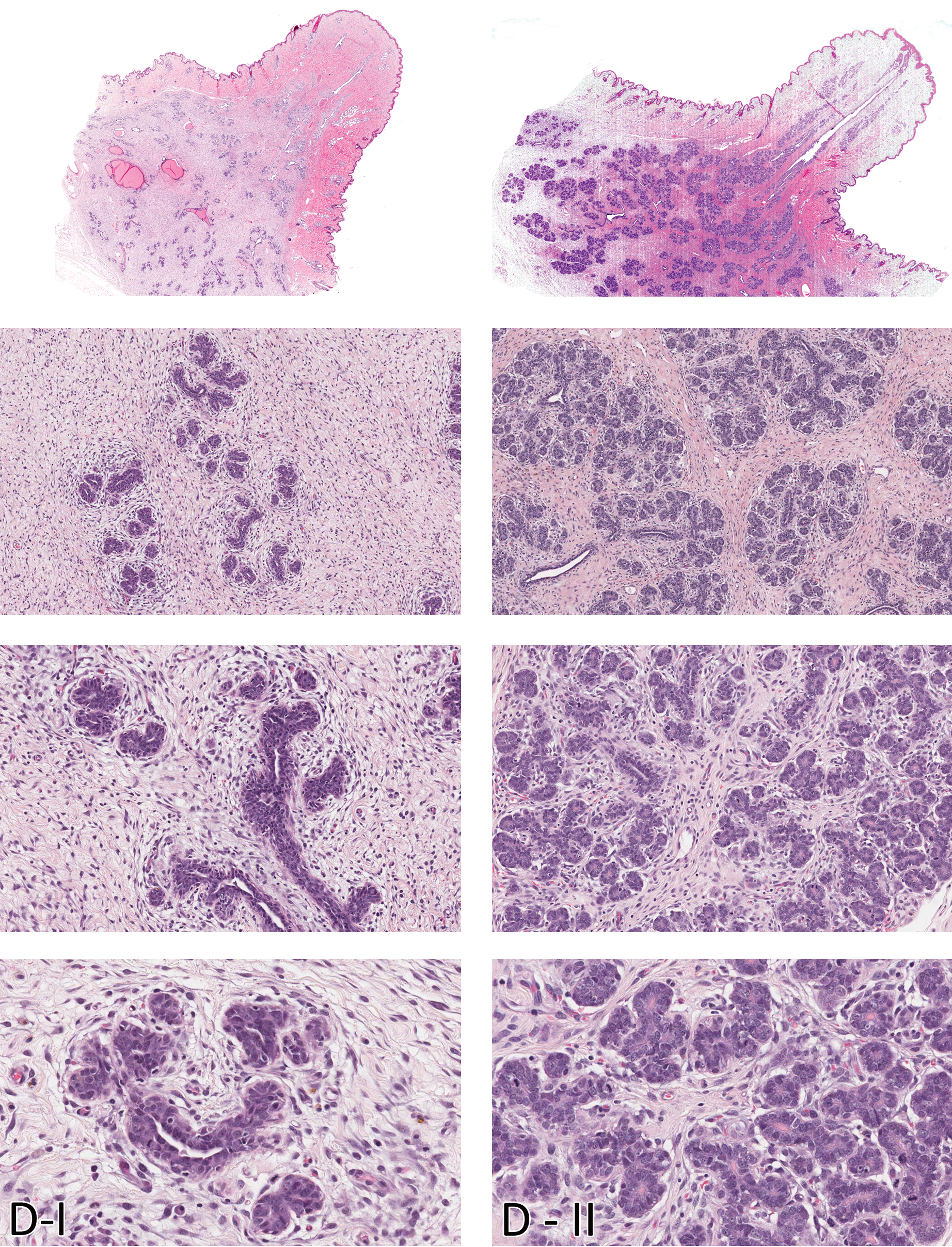
Morphologic characteristics of the Beagle mammary gland in diestrus I (D- I) and diestrus II (D - II). Mitotic figures are conspicuous in the epithelial cells in diestrus I and II. Compare with Figure 3B. H&E. Magnification from top to bottom - Sub-gross, 100X, 200X, and 400X.
Mammary Gland in Estrus
Likewise, the morphology of the mammary tissue was nearly identical in the next stage of the cycle (estrus - Figure 2). In some dogs, very slight stromal/periductal edema was evident, although this was not a consistent feature in all dogs. Although early proliferative changes have been described in late proestrus and in estrus (Nelson and Kelly 1974), this feature was not evident in the current population of dogs. The entire mammary chain from a dog in estrus was also examined (see below) to elucidate any possible variation in the morphology of the gland based on the different anatomic location. Microscopic evaluation of the entire mammary chain revealed inactive (quiescent) glandular tissue irrespective of the location, and early proliferative changes were not evident.
Mammary Gland in Diestrus
The most striking morphologic changes in the mammary gland were noted during the diestrus stage of the cycle. The varied morphology of the gland in diestrus is illustrated in Figure 4A and 4B. Diestrus in beagles is usually about seventy-five days in duration (Schaefers-Okkens 1996), and unlike other domestic animals, a functional corpus luteum (CL) with a lifespan identical to that in a pregnant dog is present in the ovary (Hoffmann et al. 2004). To describe the morphologic events that appear chronologically during the different phases of the Diestrus phase, the entire mammary chain from a few representative cases was selected and the morphological changes studied in microscopic detail. Because even within one phase of the cycle variations exist in the grade to which the morphological changes appear, the average changes are described here. The cycle-related changes gradually appear and disappear, and no sharp distinction between the different subphases can be made. There are no established morphological criteria for further classifying the diestrus stage; however, based on the four distinct histological features (often with slight overlapping changes), we have classified this stage into diestrus I, II, III, and IV subphases.
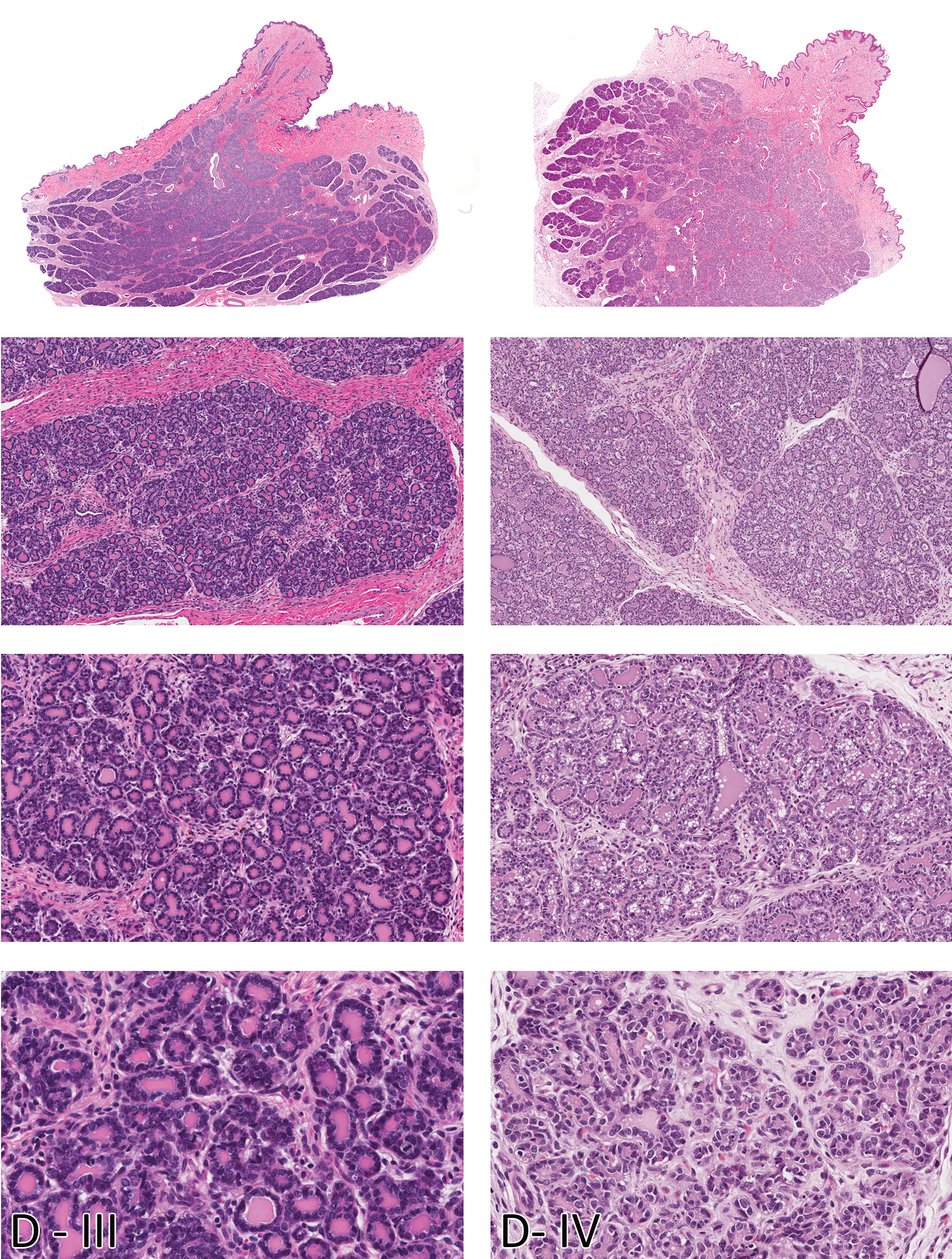
Morphologic characteristics of the Beagle mammary gland in diestrus diestrus III (D - III) and diestrus IV (D - IV). Note the progressive increase in glandular tissue and lobuloalveolar differentiation compared to diestrus I and II. H&E. Magnification from top to bottom - Sub-gross, 100X, 200X, and 400X.
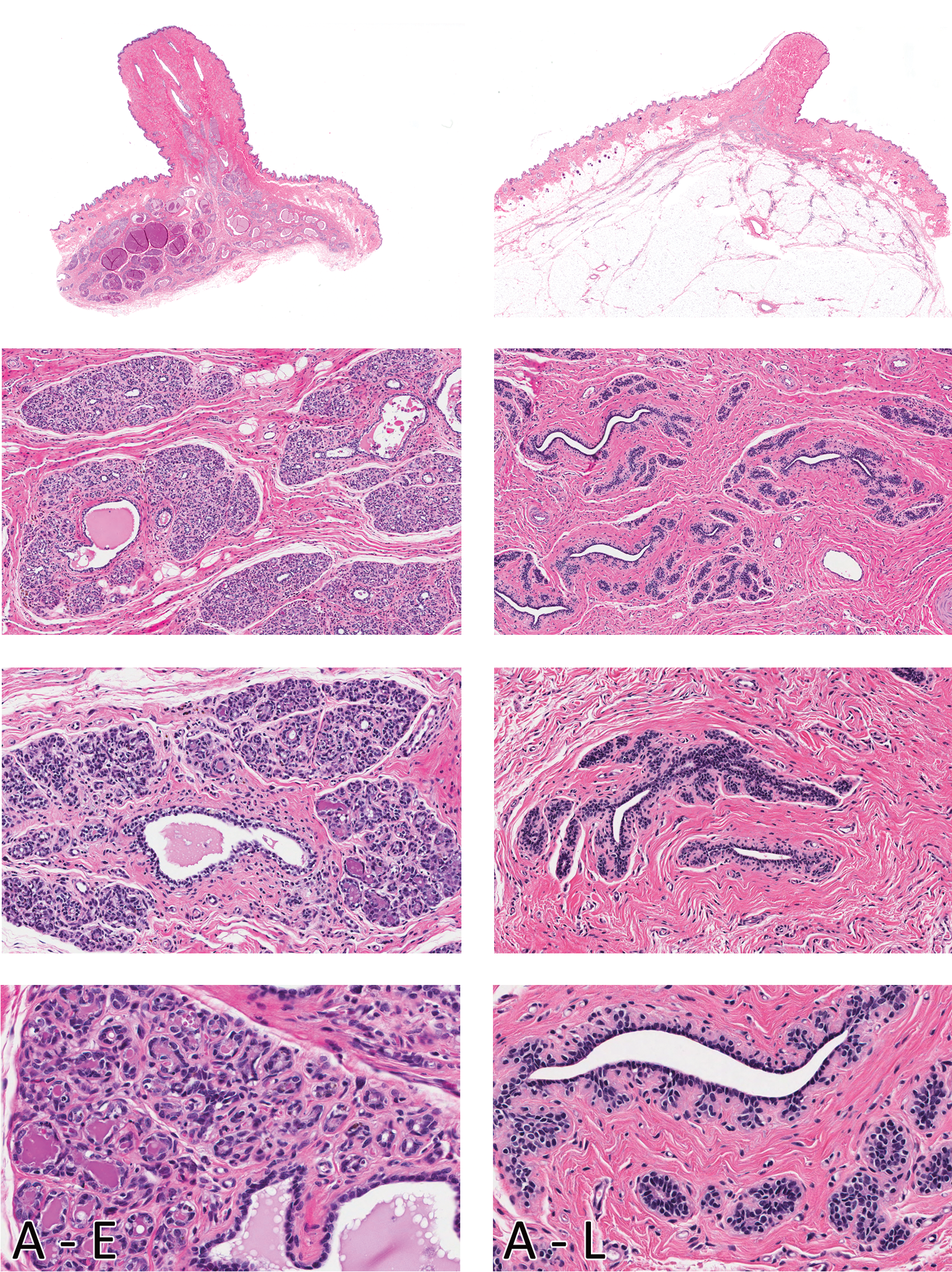
Morphologic characteristics of the female Beagle mammary gland in early (A -E) and late anestrus (A - L) are shown in the left and right columns, respectively. Note the ducts distended with secretions in early anestrus and involuting glandular lobules with increased interlobular connective tissue. Glandular morphology in late anestrus is nearly identical to that observed in Proestrus. Compare with Figure 2. H&E. Magnification from top to bottom - Sub-gross, 100X, 200X, and 400X.
During phase I, exuberant stromal proliferation was observed, predominantly composed of plump fibroblasts arranged in a loose, swirling pattern around basophilic ductular structures expanding the entire dermis. The stromal background matrix has a myxomatous appearance, with foci of extravasated erythrocytes and minimal scattered infiltrates of lymphocytes. Relative to the stroma, there was less glandular tissue, and it was composed primarily of plump basophilic, stratified epithelial cells forming immature ducts and structures resembling terminal end buds (TEB). Mitoses of epithelial cells were frequently observed. Morphological changes in the mammary glands of pubertal dogs in their first cycle have some similarities to those noted in diestrus I. Mammary glands of dogs that have not cycled before have multiple, somewhat discrete foci of undifferentiated, branching ducts surrounded by faint swirls of basophilic myxomatous stroma. However, there are two key distinguishing features. Mammary glands of dogs in diestrus I have distended ducts containing eosinophilic secretions (from the previous cycle), a feature lacking in pubertal dogs, whereas pubertal dogs have multiple clusters of dermal adipocytes entrapped in the newly expanding stroma amidst the island of newly forming ducts. In phase II, the stroma is less cellular (mature), with relatively equal proportions of stroma and glandular tissue. Early lobular development was evident, with branching ducts and alveoli. Alveoli in phase II in some lobules had scant eosinophilic proteinaceous secretions (milk). Mitotic figures were decreased compared to the previous phase and were mainly in the acinar structures. Phase III was characterized by abundance of glandular tissue, with large lobules occupying the dermis. Lobules were composed of closely apposed acini containing abundant eosinophilic secretions and ducts distended with secretions. Rare mitotic figures as well as apoptotic cells were observed in the acinar epithelium. Morphology of the mammary gland in phase IV of the diestrus stage was quite variable within the same dog and among the different dogs that were in this phase. In some dogs, a dual morphology was evident. Within the same histological sections, some glandular lobules had morphology similar to that noted in the previous phase, whereas other lobules were decreased in size with distended ducts and acini containing eosinophilic secretions. There was early lobular atrophy characterized by decreased lobular size (compared with phase III) and increased connective tissue within and between lobules. Acinar epithelial cells had a rarefied appearance, and apoptosis was prominent. Secretions were present in distended ducts and alveoli.
Mammary Gland in Anestrus
The long duration of anestrus (up to 10 months) reflects the slight variation in morphology of the gland in this stage of the cycle (Figure 4). In the early part of this stage, with regressing CL, the ducts were still distended with secretions, and acinar regression was not complete. Apoptosis of acinar epithelium was frequently observed. In late anestrus, with completely regressed CL, the lobular architecture of the glandular tissue was barely evident. There was abundant mature connective tissue, and collapsed ducts were present. From a practical perspective, the glandular morphology observed in late anestrus was generally indistinguishable to that seen in proestrus and estrus.
Immunohistochemical Expression of ER-α, PR, and ki-67
Nuclear expression of ER-α was observed in estrus and all four phases of diestrus in both the acinar and ductal epithelial cells, with strong (+++) expression noted in diestrus II (Figure 5). Estrogen receptor expression was moderate (++) in diestrus III, whereas low (+) expression was noted in estrus and in diestrus I and IV. Estrogen receptor expression was also noted in a few scattered ductal epithelial cells in early anestrus, but no ER expression was evident in late anestrus. Nuclear expression of PR was evident in all stages (mammary gland in proestrus was not stained) of the estrous cycle in the acinar and, to a lesser extent, in the ductal epithelium (Figure 6). Progesterone receptor expression (number of labeled cells) was quite variable within the same section and between the two sections from an individual dog. Subjectively, PR expression was moderate to strong in estrus (+++), diestrus I (+++), and diestrus II (++) compared with anestrus (+), diestrus III (+), and Diestrus IV (+). Nuclear expression of ki-67 matched the increased numbers of mitotic figures that were observed on H&E–stained sections (Figure 7), with strong (+++) epithelial expression detected in diestrus I, followed by dietrus II, with generally similar moderate (++) expression patterns between diestrus III and IV. Within the diestrous phase, expression was detected in both epithelial and stromal fibroblasts (only in diestrus I). Rare, individual acinar and/or ductal epithelial cells had ki-67 labeling in the anestrous and estrus stages of the cycle.
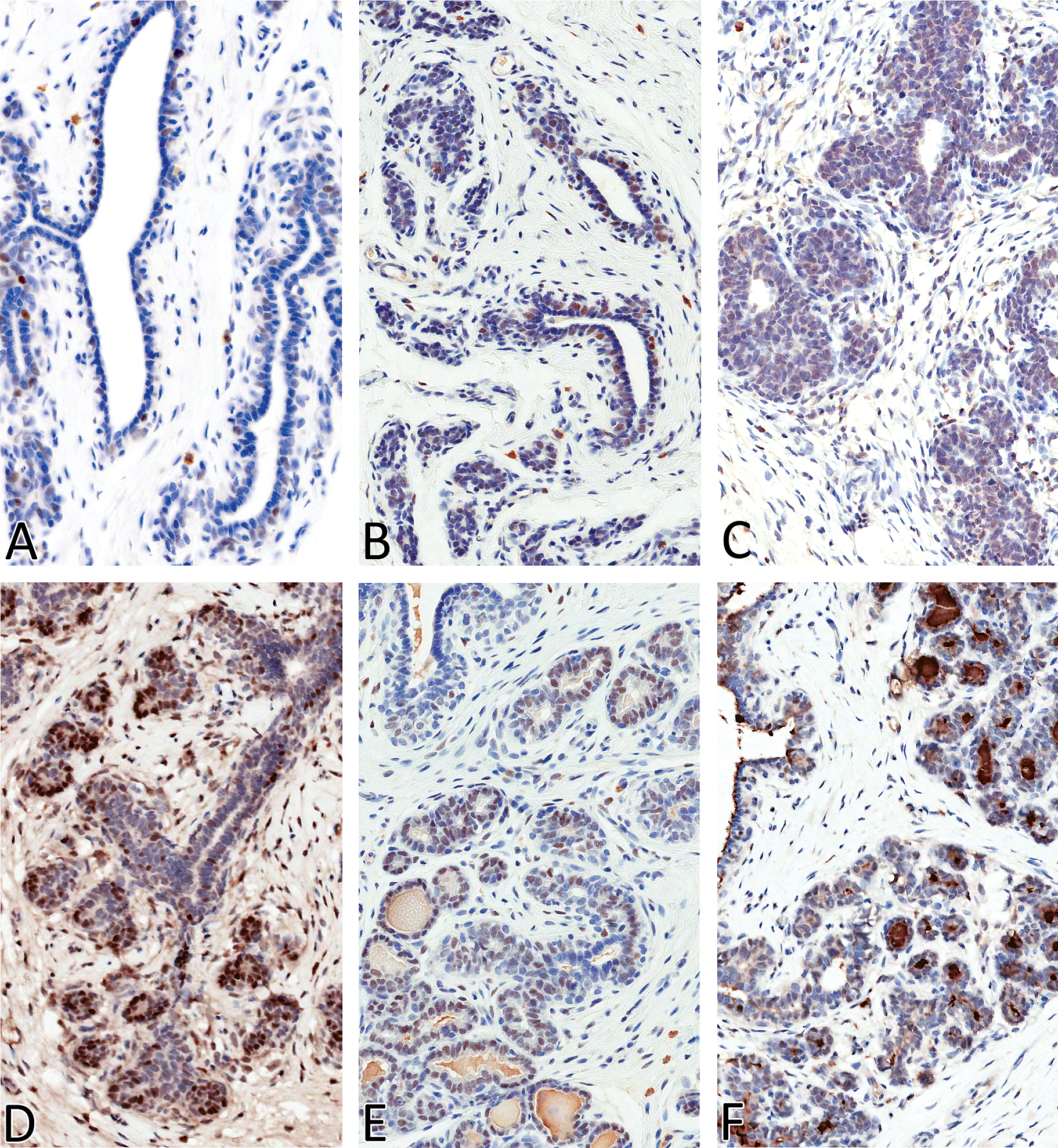
Estrogen receptor (ERα) expression in mammary gland of Beagles in anestrus (A), estrus (B), diestrus I (C), diestrus II (D), diestrus III (E), and diestrus IV (F). Magnification 400X.
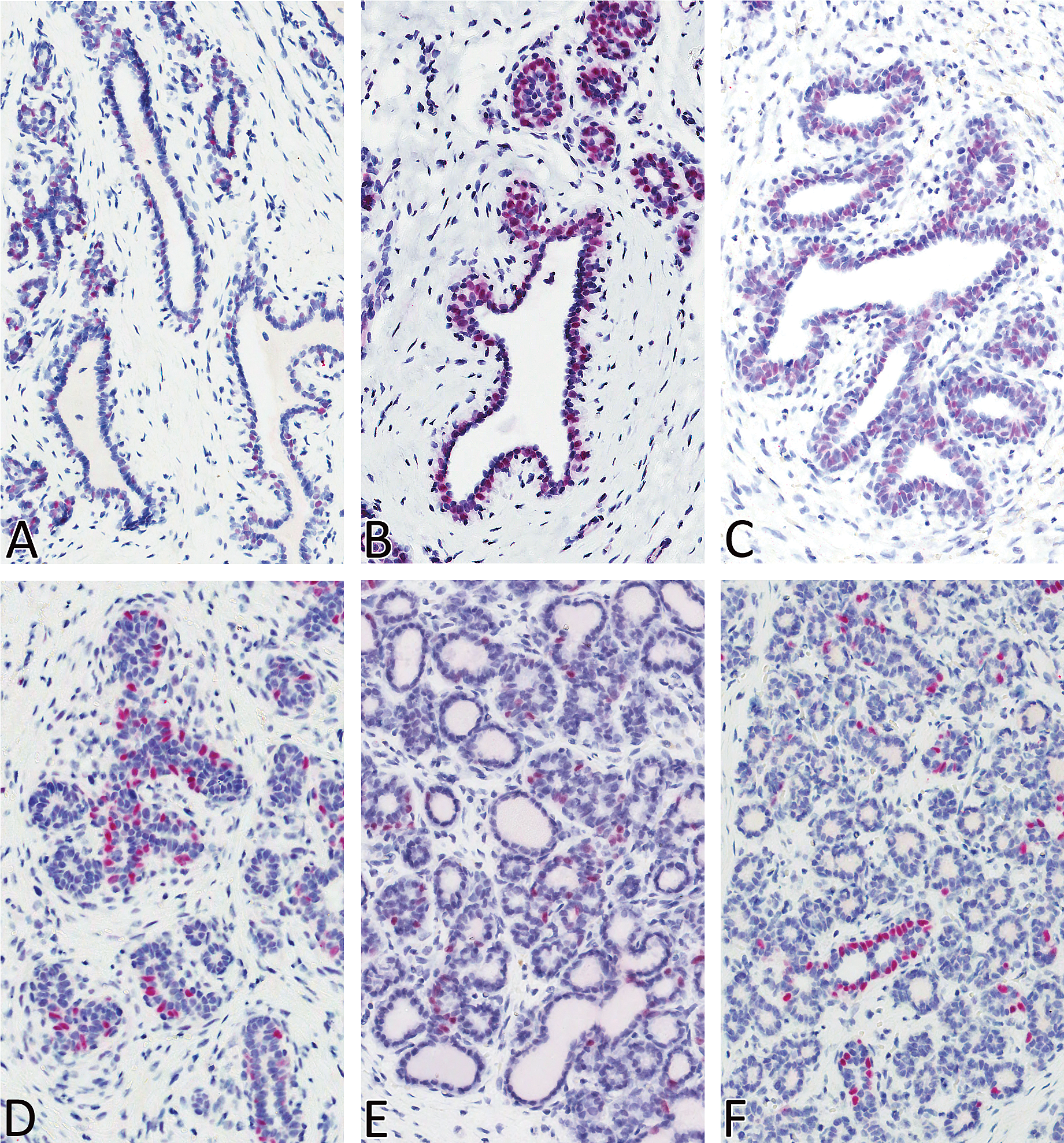
Progesterone receptor (PR) expression in mammary gland of beagles in anestrus (A), estrus (B), diestrus I (C), diestrus II (D), diestrus III (E), and diestrus IV (F). Magnification 400×.
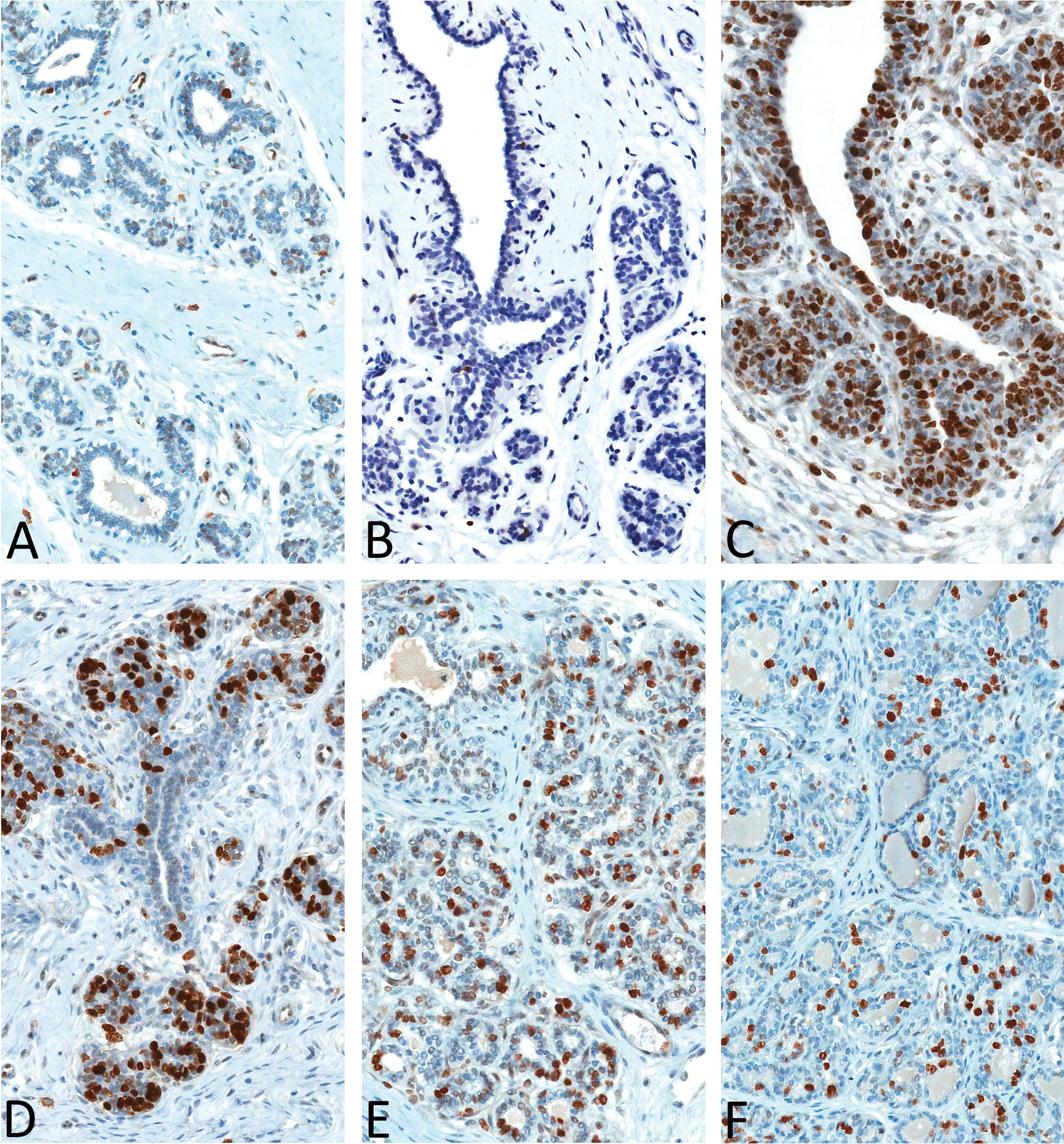
Ki-67 expression in mammary gland of beagles in anestrus (A), estrus (B), diestrus I (C), diestrus II (D), diestrus III (E), and diestrus IV (F). Magnification 400×.
Sections of the Entire Mammary Chain
The beagle mammary chain is generally represented by five pairs of nipples corresponding to cranial and caudal thoracic; cranial and caudal abdominal; and inguinal glands (Miller 1964). Histologic examination revealed differences, with regard not only to the quantity (volume) of mammary tissue (epithelial component) but also to the morphology. There were no apparent differences between the amount of mammary tissue or the morphology when dogs were in late anestrus or in estrus. Dogs in early anestrus and diestrus had slight differences between the posterior three pairs (inguinal and abdominal) compared with the anterior two pairs (thoracic). For example, dogs in early anestrus had quiescent (inactive) mammary tissue consisting mostly of ducts in the posterior gland, whereas the anterior glands still had remnants of regressing (involuting) alveolar tissue with dilated ducts. Likewise, glandular tissue was fairly abundant in the posterior three pairs, compared to the anterior two pairs when dogs were in the Diestrus stage of the cycle (Figure 8).
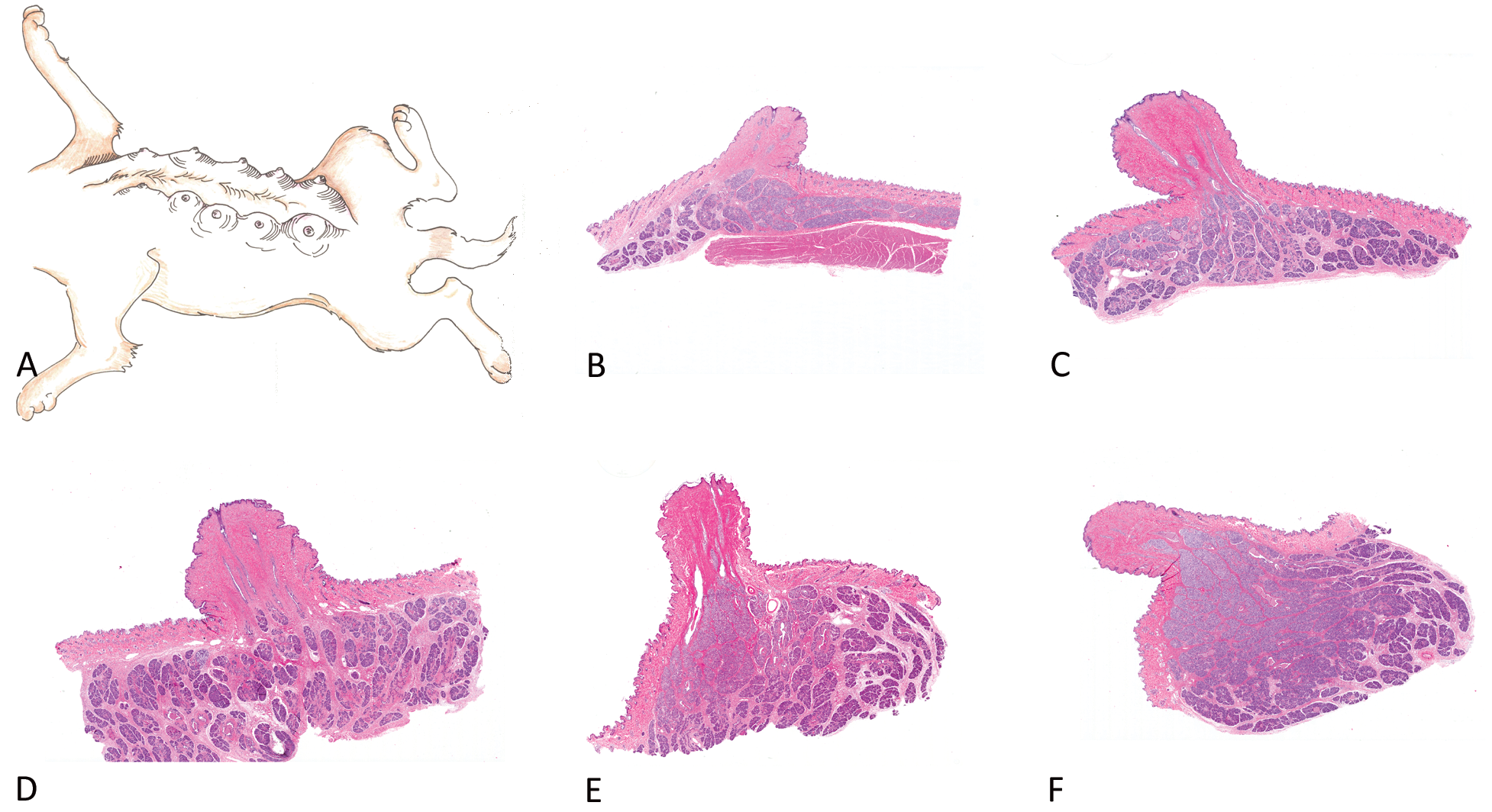
Mammary chain representing a dog in diestrus to show the variability in glandular tissue within an individual dog. A topographic representation of the mammary glands in beagles is depicted in (A). Adapted and modified from Miller (1964). Location and number of nipples are variable in individual beagles. Photomicrographs show a section of the nipple and underlying mammary glandular tissue from the cranial thoracic (B), caudal thoracic (C), cranial abdominal (D), caudal abdominal (E) and inguinal glands (F). Note the abundance of glandular tissue in the inguinal gland compared to the cranial thoracic gland in the same dog.
Discussion
There is fairly abundant literature on the normal morphology of the mammary gland in laboratory animals used in toxicity studies, especially rodents (Lucas et al. 2007; Richert et al. 2000; Schedin et al. 2000) and nonhuman primates (Cline 2007; Stute et al. 2004; Wood et al. 2007). Current literature on the canine mammary gland is centered primarily on neoplasia in pet dogs, with limited references on normal glandular morphology of laboratory beagles (Nelson and Kelley 1974). In a previous retrospective study, we characterized the histological features of the ovary, cervix, and uterus, which are rather specific for each stage of the estrous cycle (Chandra and Adler 2008). This study is an extension of the previous report, characterizing the morphological features of the mammary gland with the stage of the estrous cycle. Although proliferative lesions of the mammary gland are of primary concern, careful evaluation of the beagle mammary gland is important in preclinical safety studies to avoid erroneous interpretation of normal morphology and secondly, to detect effects of chemicals that mimic sex steroids on the mammary gland or the female reproductive tract.
The canine mammary gland is unique among laboratory animals owing to wide variation in normal histological appearance that is dependent on the stage of the estrous cycle. From a morphological perspective, four stages (quiescent, proliferation, differentiation, and regression) of the mammary gland can be identified in young (average age of 14.38 months), nulliparous female laboratory beagles.
The gland is essentially quiescent in late anestrus, proestrus, and early estrus. Although, ductal and/or stromal proliferation has been reported in proestrus and estrus (Nelson and Kelly 1974), it was not evident in the population of dogs reviewed in this study. In our study, the earliest evidence of glandular and/or stromal proliferation was evident only in the early diestrous phase (diestrus phase I), similar to that reported by Rehm et al. (2007), although these authors refer to this stage as the metestrous phase. The difference in findings might be owing to differing definitions of the successive stages of the canine estrous cycle. The determination of the beginning and end of estrus can be especially difficult, because this period is rather brief. Hormonal data may indicate estrus, whereas histological and behavioral aspects may indicate early metestrus (Vermeirsch et al. 1999). Morphologically, the reproductive tissues (ovary, cervix, vagina, and uterus) of dogs in metestrus have features of dogs in diestrus, and therefore dogs in this stage were included under the diestrus category. Unlike nonhuman primates (Stute et al. 2004), women (Ramakrishnan et al. 2002; Buist et al. 2006), or rats (Strange et al. 2007), wherein there are no major differences in the amount of mammary epithelial tissue between the follicular and luteal phases, beagles have relatively scant glandular tissue in the follicular phase (proestrus and estrus) compared to the luteal phase (diestrus). The follicular and luteal phases have been used in a broader sense, since the dog differs from other species in several reproductive aspects.
The proliferative phase of the mammary gland in beagles begins in diestrus, with the CL as the unique source of circulating progesterone. In dogs, the CL has equivalent functionality in pregnant as well as nonpregnant animals (Concannon et al. 1989; Onclin and Verstegen 1997). Progesterone plays a key role in the proliferation of the mammary gland, and the rise in endogenous progesterone during the luteal phase of the cycle results in proliferation of the gland (Mol et al. 1999). Because the length of the luteal phase in dogs is extended and generally lasts about seventy-five days (Schaefers-Okkens 1996), this phase has been divided into early and late phases (Rehm et al. 2007) or into early, middle, and late phases (Nelson and Kelly 1974). In this study, based on the distinct morphological features, a four-phase (diestrus phases I–IV) classification scheme was employed. There are no established morphological criteria for further classifying the diestrus stage, however this stage has been subdivided into four phases based on levels of progesterone and prolactin (Kooistra et al. 2000; Kooistra and Okkens 2001; Kooistra and Okkens 2002). Phase I, phase II, phase III, and phase IV are 19 (±2), 38(±2), 57 (±2), and 78 (±2) days after the estimated day of ovulation. In this retrospective study, serum hormones were not measured, and estrous status (day of ovulation or previous cycle) prior to the animals being euthanized was unknown. Although hormone levels and day of ovulation were unknown, from a pure pragmatic perspective to the toxicologic pathologist, the four different morphological changes may be roughly correlated with the four luteal phases based on levels of progesterone and prolactin (Kooistra, et al. 2000; Kooistra and Okkens 2001; Kooistra and Okkens 2002). Beyond the morphological changes, it is important to view the canine mammary gland in diestrus as an endocrine organ (Peaker 1995; Selman et al. 1994). The mammary gland of the dog secretes GH in response to endogenous (and exogenous) progestins in sufficient amounts to cause systemic effects (Eigenmann et al. 1983).
The regression phase of the gland essentially begins in late diestrus (phase IV) and continues through the extended anestrus phase of the cycle. The end result (late anestrus) is inactive mammary tissue consisting mostly of ducts and occasional remnants of alveoli. The morphological appearance of the mammary gland in anestrus was variable because of the protracted (two to ten months) length of this stage in dogs (Bouchard et al. 1991; Concannon 1993). A sharp distinction between the late stages of diestrus (diestrous phase IV) and early anestrus was not evident.
The ovarian steroid hormones estrogen and progesterone play an important role in the development of the mammary gland, and the serum concentrations of these hormones fluctuate during the estrous cycle. However, published reports on immunohistochemical expression of hormone receptors in the mammary tissue are generally performed on normal mammary tissue without due consideration given to the stage of the estrous cycle (Martín de las Mulas et al. 2004; Geraldes et al. 2000; Millanta et al. 2005). In this study, expression of ER-α was detected in all stages of the mammary gland cycle, with strong expression in phase II of diestrus (mammary gland tissue from proestrus was not evaluated owing to lack of tissue from this stage of the cycle). Immunohistochemical expression of ER observed in this study is similar to the biochemical assays for ER in mammary tissue (Donnay et al. 1995), in which ER concentration was higher in the mid-luteal phase of the estrous cycle, although ER was detected in all stages of the cycle (Donnay et al. 1995). In cynomolgus monkeys, ER expression remains unchanged across the cycle (Stute et al. 2004). Biochemically, PR concentrations in the mammary gland were higher in the mid-luteal and lower in the early luteal phase (Donnay et al. 1995). Although subjective assessment of PR expression by immunohistochemistry revealed a similar pattern, the low numbers of animals in our study, and the lack of proestrous samples were limitations of our study. In addition to the methodology (biochemical vs. subjective immunohistochemical data), comparison of the studies may be inappropriate since the age, breed (purpose bred vs. pet dogs), reproductive status (virgin vs. parous), and environmental conditions were significantly different.
Histological evaluation of the entire mammary chain revealed the variability of the glandular appearance within the same dog depending on the location of the glandular tissue. Generally, the last three pairs (cranial abdominal, caudal abdominal, and inguinal) had similar morphologic features compared with the anterior pair (cranial and caudal thoracic). Based on review of the entire mammary chain, we recommend sampling of the inguinal gland for routine evaluation of the mammary gland.
In conclusion, the mammary glands of the female laboratory beagle undergo cyclic morphological changes that parallel the ovarian cycle. From a morphological perspective, four stages (quiescent, proliferation, differentiation, and regression) of the mammary gland can be identified in young, nulliparous laboratory beagles. There was a good concordance between the ovarian cycle and morphological changes in the gland. However, histological assessment of the mammary glands in beagles should always be in conjunction with evaluation of the associated reproductive tract.
Footnotes
Acknowledgments
The authors would like to thank Ms. Joanna Barton, Ms. Lisa Gates, Ms. Denise Frailey (for the elegant drawing of the beagle mammary gland), and Mr. Kerry Crabb for the generous help throughout this project. We would like to acknowledge Mr. David Krull and Dr. Richard A. Peterson for their help with the immunohistochemical stains.
