Abstract
The controversies surrounding the heavily redacted contracts between the European Commission and Covid-19 vaccine producers have highlighted ‘transparency’ as a hotly debated concept in the pharmaceutical market. We combine research on transparency with literature on the organization of markets to investigate how such struggles over competing visions of transparency end up shaping markets and their politics. Focusing on the case of the European pharmaceutical market, we demonstrate how market transparency was implemented through devices that enacted specific visions of transparency and produced distinct market organizations over time: transparency for states (until about 1990), transparency for corporations (ca. 1990 to 2010) and transparency for state coalitions (since 2010). We discuss how the specific instrumentations and materializations of such visions of transparency play a crucial role in market politics. This debate also highlights why engaging in controversies over transparency has become increasingly important for those contesting the market status quo – in pharmaceutical markets and beyond.
Keywords
Introduction
Why aren’t markets more transparent? During the Covid-19 pandemic, this issue was a lively point of debate as activists and politicians questioned the opacity in which Covid-19 treatments and vaccines were ordered, priced and distributed (Centre for Global Development, 2021; Londeix & Martin, 2022). In May 2019, a few months before the virus started to spread, a resolution had already been passed at the World Health Assembly (WHA, 2019), which ordered member-states to ensure transparency in pharmaceutical markets. Making visible a push for transparency by state actors themselves, the resolution aimed to reduce the spaces of opacity in which high prices for medicines were said to proliferate (Shaw & Mestre-Ferrandiz, 2020). Yet, while states, corporations and civil society all agreed on the benefits of ‘more transparency’, its very definition was a contested one. Shortly after the WHA, a pharmaceutical spokesperson published a column suggesting an alternative vision of transparency, one that focused on making the decision-making processes of payers more transparent to corporations (Roedinger, 2019). How do such contestations around visions and definitions of transparency act to reorganize markets, and to whose benefit?
Once the exclusive concern of stock market regulators and neoclassical economists looking for markets emanating ‘correct’ informational signals, transparency has become a ubiquitous issue in public debate in markets ranging from pharmaceuticals, capital markets, international trade, raw materials and housing to food (BEUC, 2018; EPHA, 2021; Transparency International, 2016). This practical concern over market transparency as a governance ideal arises at a time when organization studies have started to illuminate how the notion of transparency is put to use by policymakers, civil society and corporations (Hansen & Weiskopf, 2021; Heimstädt & Dobusch, 2020; Reischauer & Ringel, 2023; Weiskopf, 2023). This research highlights the constitutive qualities of transparency, where ‘transparency as a form of ordering’ (Flyverbom, 2015, p. 168) shapes organizations and actor conduct.
We transpose this constitutive view of transparency into the context of markets, demonstrating how shifting meanings of transparency have concrete organizational consequences as they become translated into socio-material market arrangements. We highlight that visions of transparency are materialized through what Harvey, Reeves and Ruppert (2013) called transparency devices, and we investigate the role that these devices play in the organization of markets. Our study contributes to research that moves beyond normative views of market transparency (Roscoe, 2022; Roscoe & Willman, 2021) by analysing the struggles that occur around competing visions of transparency and, crucially, by tracing how these struggles shape markets.
Investigating such struggles, we claim, is vital to understand market politics. Focusing on the evolution of the European pharmaceutical market, we highlight how the strategic deployment of arrangements that promote selective visions of transparency is a key mechanism to reorganize markets and direct funding to specific actors. In this perspective, a lack of transparency is not the outcome of ill-designed markets – in the pharmaceutical market at least, our investigation shows how (shifting) definitions of transparency were carefully organized with political objectives in mind. Our paper highlights certain moments in time in which transparency assumed a ‘post-political’ role in markets. In post-politics, superficially consensus-based regulatory norms such as transparency are used to ‘hide fundamental differences in interests and power resources’ behind technical settlements (Garsten & Jacobsson, 2013, p. 422; Wilson & Swyngedouw, 2014). In our case, such settlements worked to conceal fundamental conflicts between corporations, states and patients. However, by focusing on transparency’s technical implementation through devices, we also expose the politics around this post-political concept, emphasizing how it has become not only a consensual key organizing principle but also a technocratic battlefield for promoting alternative market organizations. By highlighting these market politics, our discussion offers insights into how civil society and state activists currently reclaim transparency to challenge the market status quo, in pharmaceutical markets and beyond.
Theoretical Framing
Transparency and its contestations
The topic of transparency has presented a lively field of research in organization studies in the past decade, as studies have questioned a normative understanding of transparency in organizations (Fenster, 2015; Splitter, Dobusch, von Krogh, Whittington, & Walgenbach, 2023). Critical and, more recently, constitutive perspectives on transparency have sought to move beyond normativity to consider transparency ‘as a social process with constitutive ramifications’ (Albu & Flyverbom, 2019, p. 277). While the critical perspective has opened up important vistas into the ‘generative interactions between the visible and the “in-visible”’ (Quattrone, Ronzani, Jancsary, & Höllerer, 2021, p. 1199), it has remained broadly tethered to transparency as an (always-incomplete) governance ideal and opacity as its lesser, though persistently present, other. Recent calls for a constitutive view of transparency abandon this position to investigate ‘how people in and around organizations claim, contest and configure. . . these concepts, in which situations and with what consequences’ (Heimstädt & Dobusch, 2020, p. 3). The call is for transparency itself to become the focus of inquiry, as a ‘practice that is historically contingent and multiple, and thus negotiable and contested’ (Weiskopf, 2023, p. 326). Contestations proliferate particularly in situations of ‘ontological insecurity’ (Heimstädt & Dobusch, 2020), where actors have stakes in conflicting definitions of transparency. Such moments of contestation offer unique entry points for researchers studying how these definitions become generative of concrete organizational practices and arrangements.
Empirically, a constitutive perspective of transparency involves charting how actors organize their contexts according to decisions over ‘what should and should not be seen’ (Albu & Flyverbom, 2019, p. 278). Hansen and Weiskopf (2021) for instance study the Chinese social credit system, where different transparency matrices – law, discipline, security and care – are used to mobilize different mediating technologies. Hansen, Christensen and Flyverbom (2015) contemplate how so-called disclosure devices – due diligence, rankings and big data analysis – organize and channel organizational knowledge production and use. Deploying a practice-theoretical approach, Ringel (2019) and Reischauer and Ringel (2023) study enactments of transparency within the German Pirate Party and between the party and its audiences, respectively. In turn, Mehrpouya and Salles-Djelic (2019) take a historical tack to examine how shifting understandings of transparency relate to broader historical transformations, with the rise of neoliberal policies prompting a redefinition of transparency in macroeconomic governance.
We build on these studies’ cumulative insights into the organizational constitution of transparency, but our focus on markets also moves beyond the literature’s substantive concern to draw attention to transparency’s role in a broader post-political governance toolkit. Over the past decades, specific definitions of market transparency highly favourable to corporations – what Mehrpouya and Salles-Djelic (2019) call ‘neoliberal transparency’ – have been implemented, but their historical specificity is typically concealed behind technicalities. In post-politics, according to Wilson and Swyngedouw (2014, p. 6), ‘political contradictions are reduced to policy problems to be managed by experts and legitimated through participatory processes in which the scope of possible outcomes is narrowly defined in advance’. As Garsten and Jacobsson (2013, p. 424) highlight, post-political governance tools such as sustainability, choice, efficiency, or indeed transparency form part of a nexus of neoliberal governance tools in which they serve to transform political conflicts into economic settlements. Post-political governance seeks to establish jointly beneficial solutions and ‘win-win’ relationships but often occludes the hegemonic character of these agreements (Garsten & De Montoya, 2008). Leaning on Chantal Mouffe’s (2005) work, Saifer and Dacin (2022) for instance reflect on how data discourses depoliticize societal problems by turning them into narrow series of data points to be managed by technocratic experts. By restricting the voicing of legitimate or even thinkable alternatives, this channelling of politics into technologies serves to neutralize potential contestants such as civil society actors (Beveridge, Hüesker, & Naumann, 2014; Parés, 2011). Crucially, however, we propose that it is this devising that also enables the re-politicizing of the organizational orders that spawn from post-politics: to do so one needs to interrogate the specific organizational or technological configurations through which definitions of transparency are stabilized (Barry, 2002). Asking how transparency is ‘interpreted, practiced, inverted, or used to achieve particular ends’ (Garsten & De Montoya, 2008, p. 2), we suggest, thus involves moving back and forth between competing visions of transparency and the specific ‘organizational, technical, and political formations’ (Flyverbom, 2015, p. 173) they give rise to.
Markets, devices and their politics
We highlight how conflicts over visions and materializations of transparency play out in the context of one particular kind of organization: markets. Market transparency has been upheld as the North Star of market design both by defenders and detractors of neoclassical market orders to ensure informational efficiency (Best, 2005; see also Coslor, 2016; Roscoe & Willman, 2021). Governors of markets have had a longstanding interest in transparency both as a ‘disciplinary’ and ‘enabling’ tool – to restrict their own influence in market affairs and to simultaneously hold market actors accountable (Grossman, Luque, & Muniesa, 2008). Yet, far from being a natural corollary of a well-designed market, transparency has to be carefully worked into it – even in those markets said to be ‘liquid’ (Roscoe, 2022). From this viewpoint, opacity and transparency do not designate qualities of markets themselves, but different ‘transparencies’ become congealed in specific market configurations that have material consequences on the distribution and enactment of market power.
In tackling market transparency ‘as a set of devices, not as a principle’ (Grossman et al., 2008, p. 117), we build on previous studies inspired by actor-network theory (ANT) that have envisioned market power as embedded in the socio-material arrangements that structure market exchanges (Callon, 2017; Geiger & Gross, 2018; Chimenti & Geiger, 2023). More specifically, we consider studies that have focused on the blurry boundaries between markets and policy in ‘concerned markets’, that is, markets that are seen as a way of dealing with public interest problems (Geiger, Harrison, Kjellberg, & Mallard, 2014). In this literature, policymaking is understood as continuous market organization, where different conceptualizations of markets are mobilized to guide the work of market diagnosis, design and repair (Frankel, Ossandón, & Pallesen, 2019). Ossandón and Ureta (2019) for instance demonstrate how changing conceptualizations of market ideals such as ‘perfect competition’ or ‘choice’ create radically different organizations of health insurance and public transport markets in Chile. This literature chimes with the constitutive perspective on transparency by studying such concepts ‘at work’ in markets, that is, mobilized by an array of actors and instrumented through diverse socio-material arrangements.
The strong focus on devices in this literature is helpful in tracing the concrete organizational efforts that unfold from shifting definitions of transparency – an attention that is often missing from transparency studies and that, as we argued above, is vital in ‘repoliticizing’ the notion of transparency. We are particularly interested in the double role that transparency devices play in organizing the pharmaceutical market at a socio-material level all while being the object of political struggles. Market devices, according to Geiger and Gross (2018, p. 1360), ‘mediate the relationships between different market actors and coordinate their actions’. Devices articulate as well as shape what counts in a market, for instance by determining who participates in setting the terms of economic exchange (Neyland, Ehrenstein, & Milyaeva, 2019). But devices also tend to ‘misfire’ (Callon, 2010; Geiger & Gross, 2018), which can give rise to contestations and reorganizations. For our analytical purposes, we deviate from classical actor-network theoretical conceptions of full agentic symmetry of (human) market actors and devices in postulating that market actors and devices are likely to play differentiated agentic roles. We believe that this differentiated attention is needed in order to fully crystallize a market’s politics, or, with MacKenzie (2019, p. 1), to ‘bring together the focus on materiality of actor-network theory with an emphasis on [the] structural advantage’ enjoyed by certain market actors in constructing those devices. In building on Harvey et al. (2013), we define transparency devices as all the socio-technical, regulatory and organizational devices that result from and are entangled in struggles over definitions of transparency. We adopt a pragmatic understanding of these devices as those parts of a market’s organization that are explicitly embedded in the struggles around transparency by market actors – either because they are labelled as ‘transparency’ instruments by actors themselves or because they are being brought to bear in controversies around transparency.
We investigate how struggles around transparency devices reorganize markets in three aspects. First, we propose that they are likely to reorganize
In sum, our article combines the growing literature on transparency in organization studies with ANT-inspired research on market politics to address the following question:
Research Approach
In the pharmaceutical market, different visions of transparency are devised through legal, organizational and material interlinkages at national and supra-national levels. While our investigation is directed at the European level, we also chose one focal European Union country with a prominent domestic pharmaceutical industry (France) to examine the state level. We build our investigation on three sets of empirical materials – historical documents, observations and interviews. First, we rely on historical sources of public debates and regulations about pharmaceutical transparency in the EU and in France (Table 1). At the European level, we accessed the archives of the European Commission’s DG-Santé (formerly DG-Sanco) related to the 1989 Transparency Directive. We also accessed the reports and press releases of the two European pharmaceutical negotiation alliances, BeNeLuxA (since 2015) and the Valletta Declaration Group (since 2019). This material amounted to approximately 50 documents. To trace the implementation of these regulations at the state level, we consulted three historical sources. To capture the industry perspective, we studied the 280 issues of the French pharmaceutical lobby magazine
Archival sources used in this article.

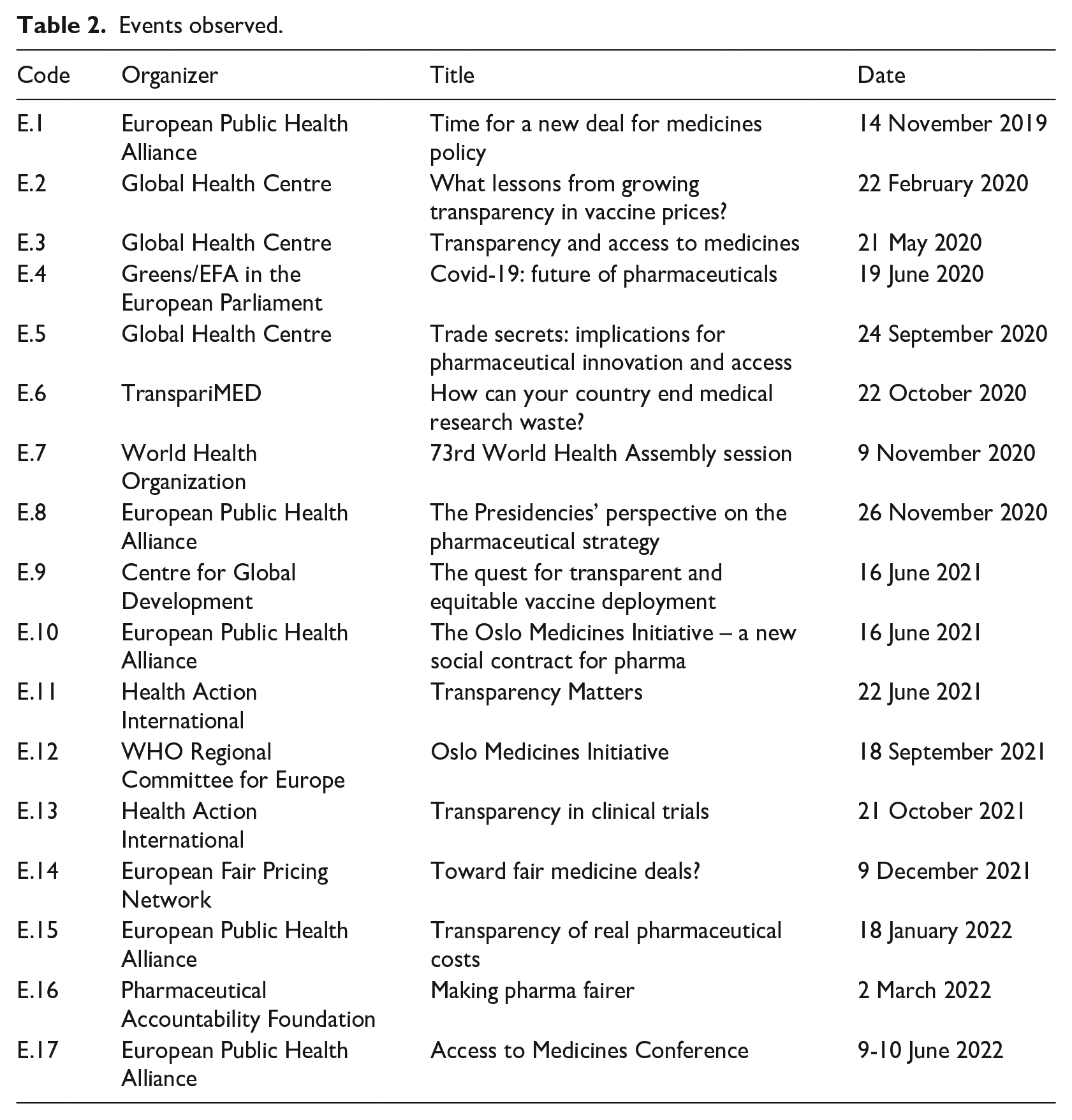
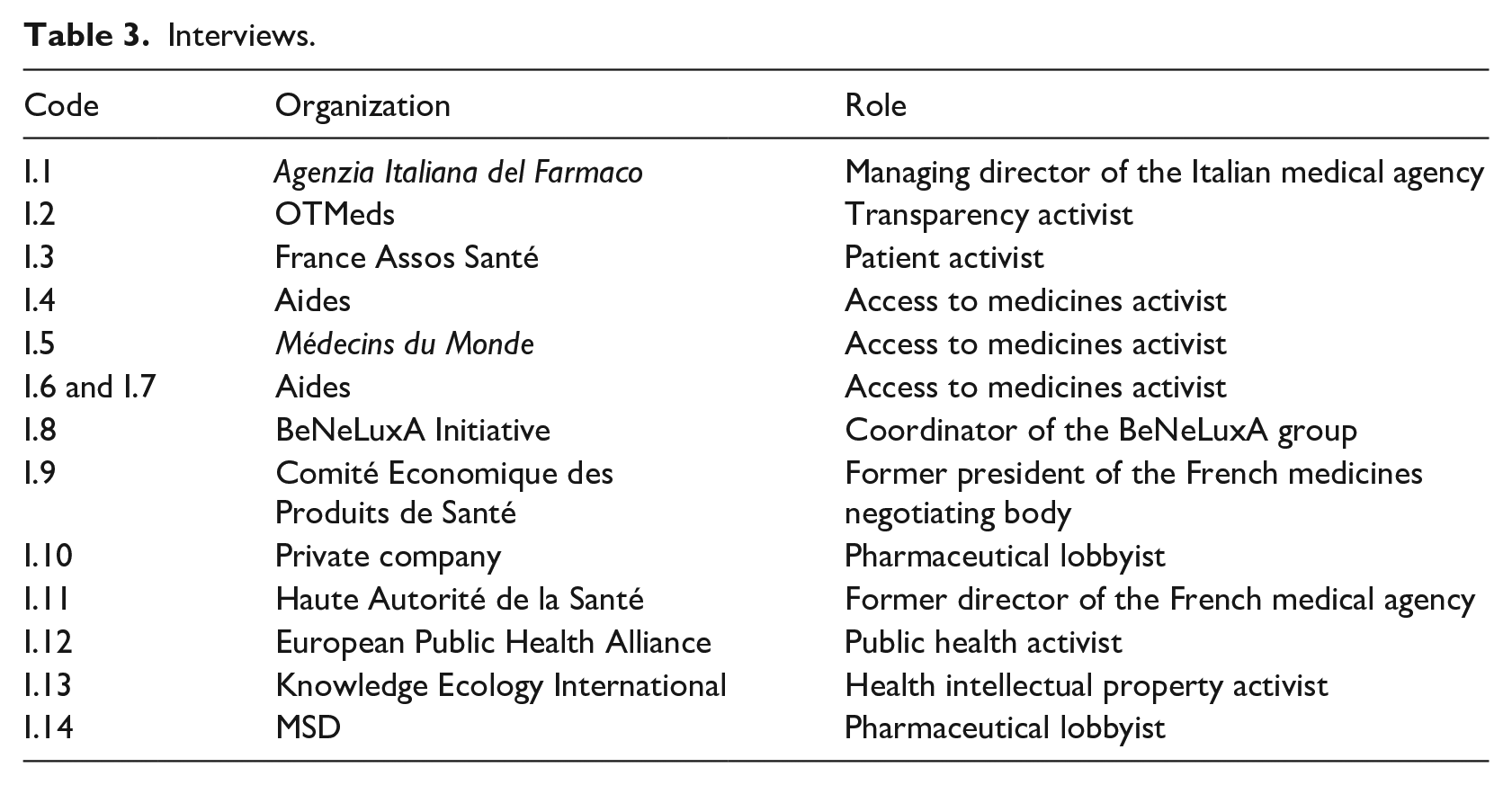
To complement our historical with a contemporary perspective, we carried out participant observation at 17 EU-level conferences and seminars from 2019 to 2022, gathering pharmaceutical regulators, industry representatives, and access to medicines activists (Table 2). These events helped us understand alternative and emergent definitions of pharmaceutical transparency and how these may be devised. We finally conducted 13 interviews with former pharmaceutical lobbyists, French and European civil servants, and access to medicines activists (Table 3). They proved useful to contextualize changes in the past and better comprehend present dynamics.
Events observed.
Interviews.
We gathered all data (archival copies and notes, observation notes, interview transcripts) in a dedicated database, which we used for thematic and processual analysis. We started by mapping all key events and actors involved in transparency debates along a chronological timeline at national and EU levels. In doing so we followed ‘transparency’ back in time from the 2019 WHA Resolution, which we took as a crucial contemporary moment that highlighted it as a major concern for state actors. We used transparency as a native construct, charting the actors, regulations, policy deliberations and regulations that explicitly referred to concerns around transparency or opacity and tracing any shifts back to earlier debates, regulatory drafts and public consultations. In a second step, we analysed regulatory and policy shifts that more broadly affected how transparency was organized in our focal market, and we drew on data across our sources to establish the histories and chronologies of these shifts (Langley, 1999). Doing so ‘backward’ in time helped us to denaturalize market processes that are now taken for granted, such as the value-based approach to pricing innovative medications. This allowed us to identify three phases in the construction of transparency in the pharmaceutical market. We compared and contrasted the multiple data sources we had gathered to drill into the main dynamics we saw occurring in each temporal bracket, as well as ruptures and continuities across brackets. These efforts at grounded processual theorizing (Holton, 2017) allowed us to grasp how market politics was related to the definitions and instrumentations of transparency and how these constellations changed over time; they resulted in our analytical framework in Table 4.
Transparency visions and transparency devices.
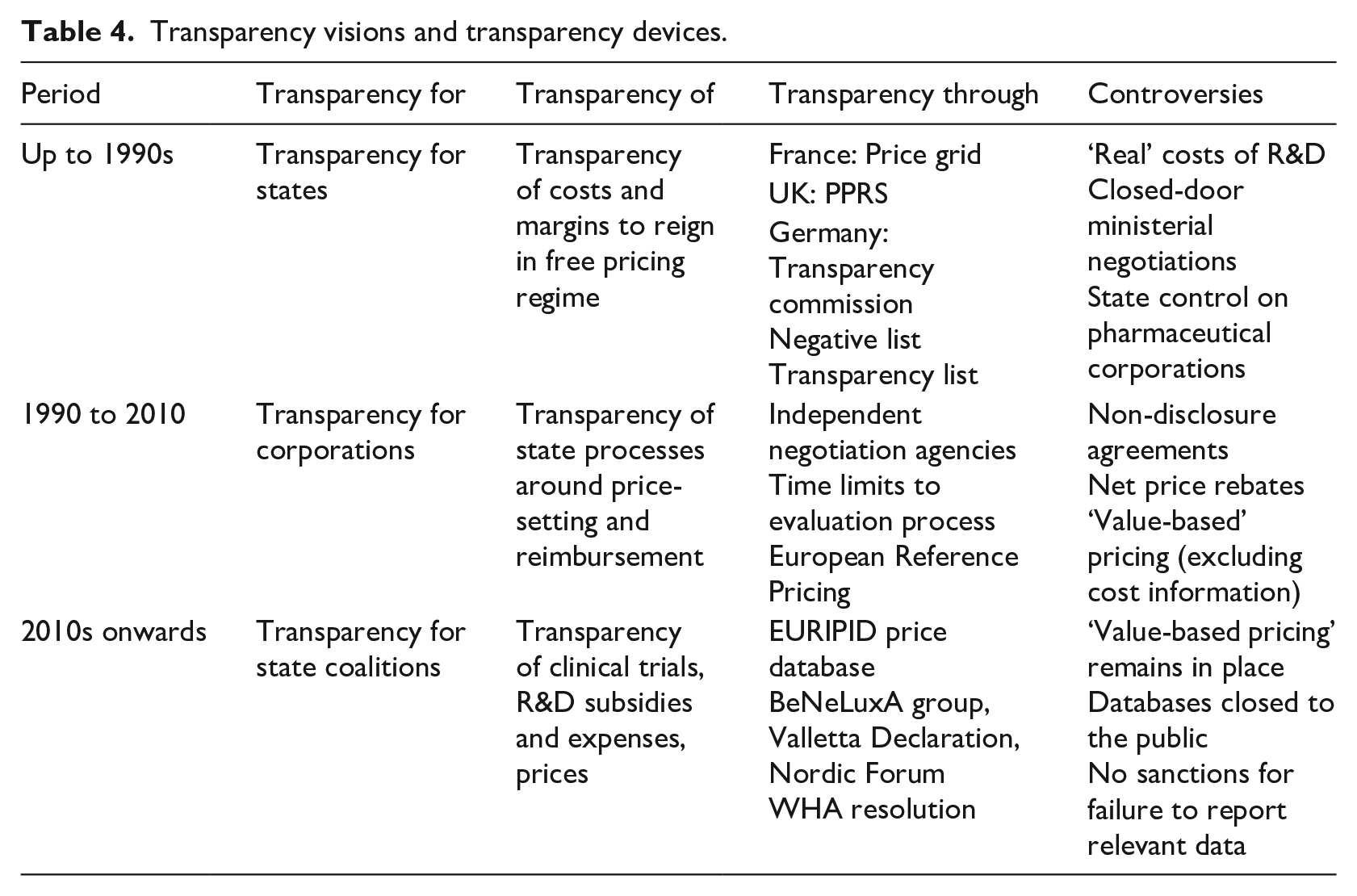
To note, while the temporal brackets presented below emerged analytically as distinct governance periods from our data and broadly coincide with previous research insights (Mehrpouya & Salles-Djelic, 2019), we also observed certain discontinuities within and continuities between brackets. This is to be expected in a multi-level analysis where organizational arrangements shift at different speeds and where local contingencies have differential stabilizing and precipitating forces. The brackets below should thus be read as ‘idiographic maps of the territory’, reflexive of the map maker as much as of the territory itself (Van Maanen, 1979, p. 520). In providing corroborating evidence for the dynamics traced in our analysis, we use the shorthand ‘A’ (plus year and number) for quoted archival sources listed in Table 1; ‘E’ designates observational data gathered from the events listed in Table 2; and the letter ‘I’ signals interview data (Table 3).
Findings: Struggles Over Transparency Devices Shape Markets
The European pharmaceutical market is organized around two main sets of actors: state bodies (as buyers, evaluators and regulators) and pharmaceutical corporations. Most European countries have developed universal healthcare systems where prescription items are wholly or partly reimbursed by national or regional public insurance bodies, called ‘payers’, with the proportion of pharmaceutical expenses covered by public health insurance reaching more than 70% in most European countries (Chauveau, 1999; Vogler, Haasis, Dedet, Lam, & Pedersen, 2018). Public bodies in the pharmaceutical market fall into two sets of organizations: negotiating bodies and evaluation agencies. Since the 1990s, most European countries have created independent bodies tasked with negotiating on behalf of the health insurance system. They negotiate list prices (the official ‘sticker’ price of a medicine) and net prices (the mostly secret ‘real’ price after rebates) according to the efficiency of the medicine, which is assessed by another independent state body. In the UK for instance, the negotiating body is the National Health Service (NHS) and the evaluation body is the National Institute for Care and Health Excellence (NICE). This market configuration, however, is contested. Since the late 1990s, the real prices of new pharmaceutical drugs in the EU have increased dramatically, following a pattern that the European Commission (2019, p. 5) itself has deemed to be ‘unsustainable’. The heads of European pharmaceutical agencies involved in reimbursing medicines have expressed concern regarding their weakness in negotiations over pharmaceutical contracts (EPHA, 2020). National payers assert that they are not able to negotiate on an equal footing with large pharmaceutical companies acting as if they were in an oligopolistic market (Larsen, 2019).
In this context, calls for transparency as an instrument to limit pharmaceutical firms’ market power have multiplied. However, such calls are not exactly new. In fact, the pharmaceutical market is a striking example of a market in which transparency has been a constant organizing principle. For decades, its main administrative bodies and regulators have been carrying unequivocal names, such as the ‘transparency committees’ and ‘transparency lists’ created in the 1970s and 1980s, the 1989 European ‘transparency directive’, or most recently the 2019 World Health Assembly ‘transparency resolution’. Yet, while being continuously reorganized in the name of transparency, European pharmaceutical markets were transformed in very different ways through time. The following three sections will trace how transparency has been mobilized by different actors, embedded into diverse devices, and resulted in distinct market organizations over time (see Table 4 for an overview).
Period I – Mobilizing transparency for state payers (until 1990)
Until around 1990, European pharmaceutical markets were nationally distinct ones. Each country had its own regulatory model to manage access to pharmaceuticals, organized according to a vision of transparency in which states control the margins of pharmaceutical manufacturers and ensure affordability for public payers. But this situation came to be heavily criticized by pharmaceutical companies and European policymakers, who denounced the spaces of opacity this vision was arguably creating and its detrimental consequences for the industry. Sketching out three main European markets of the time – France, the UK and Germany – we describe the transparency devices in place during this period and how they were contested.
The domination of European pharmaceutical markets by state payers
In period I, European pharmaceutical markets were organized around a vision of transparency that gave a central position to state regulators. Crucial to this market configuration were transparency devices that enabled public authorities to gain relevant information on the costs and margins of pharmaceutical production.
In France, since 1968, controls on prices were enacted through an official ‘price grid’ that allocated a price to each medicine (Nouguez & Benoît, 2017, p. 405). The prices of the grid were determined by the state to give pharmaceutical manufacturers an average margin for the medicine, compared to their peers, based on the calculation of the ‘real costs’ of the pharmaceutical (though these were known to be exaggerated by industry, see I.9 and I.10). Although initially following rigid formulas based on accounting ratios, this system was progressively relaxed to leave room for negotiations between state authorities and pharmaceutical companies (Chauveau, 1999; Nouguez & Benoît, 2017). From the 1980s onwards, the prices of the grid were increasingly determined through negotiations across industrial and health ministries. As one French health ministry advisor of the time recalls (I.10):
For some drugs where pricing discussions were complicated, civil servants from these three ministries [industry, health, and finance] had to agree, they were gathered in an ad-hoc committee on prices. . . [. . .] In most cases, we would end up in the office of the Prime Minister’s health advisor, who was arbitrating between us.
In the UK, there was no ‘price grid’, but pharmaceutical companies were constrained by sectoral regulations that limited their margins and levels of profit (Gross, Ratner, Perez, & Glavin, 1994). Pharmaceuticals were submitted to a ‘pharmaceutical price regulation scheme’ (PPRS), through which the return on capital invested was capped at between 17 and 21 percent (Gross et al., 1994). Each company set prices for new medicines freely but had to ensure that their return on capital did not exceed the maximum level, as excess profits had to be paid back to the NHS (Mrazek, 2002).
In the federalized German market, dominated by powerful domestic firms, pharmaceuticals were traditionally priced according to a free pricing system (Gross et al., 1994). Successive reforms sought to establish a system that would allow comparing costs between medicines, though these were often inconsequential to price negotiations. In 1977, a ‘Transparency Commission’ was tasked with comparing prices and therapeutic efficacy of the most frequently prescribed medications (Anon., 1977). In the 1980s, exploding state health insurance budgets and continued lack of transparency around medication costs became widely debated issues. Regulatory reforms in 1989 introduced two national transparency devices: a list of therapeutically equivalent drug classes, establishing a fixed price for each class (which led to dramatic price decreases, see Paetow, 1989); and a so-called ‘negative list’ of cost-ineffective medications, based on the previously published ‘transparency list’ (Glaeske, 1989).
These national transparency devices had deep consequences for the organization of pharmaceutical markets. Regarding the
Contesting transparency for state payers
This mobilization of transparency came under attack by pharmaceutical lobbies who argued that the cost-based or fixed-margin systems in place were too opaque and the transparency lists implementing price caps for each drug class too restrictive. The German ‘transparency list’ was dubbed by one industry representative as a ‘torturing device’ as its price ceilings were arguably arbitrary (Glaeske, 1989). In France, manufacturers were unhappy with the case-by-case evaluation of medicines behind the closed doors of ministries, which they argued did not allow them to make strategic R&D investment decisions (I.9 and I.10). The head of the UK’s pharmaceutical lobby too called for member-states ‘to abandon their broad set of price and profit control instruments’ (A.3 1992, p. 13). These calls made it clear that manufacturers were not simply asking for ‘less’ transparency – what they had in mind was a very different kind of transparency, one that would give corporations deeper insights into states’ decision-making processes.
The devising of transparency through lists and accountancy instruments aimed to help control costs for states was also questioned by European policymakers. Cost-based evaluations had them worry about whether manufacturers were artificially inflating costs (Chauveau, 1999). They also feared that it may lead to maintaining costly production lines that would make manufacturers unable to compete in the European single market (A.1 1991; A.2 1992; A.3 1992). In 1992, the head of the pharmaceutical products unit in the European Commission asserted that ‘the Commission [was] in favor of deregulation, at least when it [came] to direct price control’ (A.3 1992, p. 13). He thereby directly attacked the cost-based devices that organized transparency for state payers in the market; more indirectly, he also confronted the well-known practice of supporting national pharmaceutical manufacturers through an often highly political price determination process.
In summary, until 1990, European pharmaceutical markets were organized through a vision of transparency that revolved around ensuring budgetary predictability and affordability for state payers. Several sets of transparency devices (the ‘price grid’ in France, the PPRS in the UK, or the German ‘transparency list’) implemented this form of transparency, but they still allowed state actors to build political considerations such as domestic investments into the pricing system. This devising of transparency was accused of creating opacity around states’ price determination practices, leaving too much scope for political considerations beyond cost control to enter decision-making processes.
Period II – Mobilizing transparency for pharmaceutical corporations (ca. 1990 to 2010)
From around 1990 to 2010, a new vision of transparency was mobilized, which we call ‘transparency for corporations’, with the construction of devices that deeply transformed European pharmaceutical markets. With the purpose of leaving behind the allegedly ‘politicized’ market procedures of the 1980s, this redefinition aimed primarily at ensuring the competitiveness and innovativeness of European pharmaceutical firms. Quickly enough, though, the devices that implemented this redefinition of transparency became the subject of intense struggles between smaller EU states and the Commission.
Mobilizing a corporate-centric vision of transparency
Early on, period II saw industry representatives and EU policymakers work together in redefining transparency around corporate interests. The EFPIA, the pharmaceutical lobby in Brussels, had lobbied for a shift of power from national state payers to corporations, which would support the emergence of a unified European market for pharmaceuticals (I.10; I.14). The lobby was backed by successive European Commissioners for Industry, who advocated for limiting the power of member-states (A.4 1993; A.7 1997). This alliance culminated with the organization of the ‘G10’ in March 2001, gathering EU Health and Industry Commissioners with ministers from member-states and representatives of the pharmaceutical industry (A.8 2002). The G10 aimed to ‘enhance competitiveness in the pharmaceutical industry’, elaborating policy recommendations that invoked ‘greater transparency’ (A.10 2002, p. 13). This transparency had a radically distinct meaning from the transparency mobilized in period I, as it focused on transparency of payer processes: ‘the G10 believes that much could be done to improve the transparency of national decision-making. . . Greater transparency would allow industry and other regulators a clearer understanding of the criteria used and lead to greater consistency of decisions’ (A.10 2002, p. 13). The push was supported by many patient organizations hoping that it would level out national differences in access to healthcare (Neroth, 2005).
This new vision of transparency was translated into successive transparency devices, which triggered major shifts in the market’s organization. This occurred most notably through the ‘Transparency Directive’ of 1989 (Directive 89/105 EEC), which sought ‘to ensure that any measures taken by EU countries to set the prices of and to reimburse medicinal products are transparent’ (European Commission, 2009). Following the Single Act of 1986, this directive aimed at creating a European-wide market for pharmaceuticals, replacing the previous system of state aids to national manufacturers with a transnational market. Here, transparency was equated to the principle of ‘fairness in pricing’, to be guaranteed to all European manufacturers (Directive 89/105 EEC, article 8). The directive enacted three rules. First, it set explicit time limits for national drug negotiations (with a maximum of 180 days between application and reimbursement decision); second, it compelled states to base their pricing decisions on ‘objective and verifiable’ criteria and explain disagreements with the price proposed by the pharmaceutical manufacturers (allowing the latter to take legal action); third, it decreed that unilateral administrative measures, such as price decreases, must be as fully justified as the initial price setting (again allowing for legal action).
The devising of these rules deeply affected the organization of European pharmaceutical markets. First, they resulted in the adoption of a new system for
Second, these new transparency regulations reconfigured the
Third, the new transparency devices strongly affected
State payers promoting an alternative vision of transparency
This transparency vision created significant issues for state payers. Despite the new ‘objective’ evaluation mechanisms, they complained that they had lost visibility on the likely costs and value-added of new medicines, making the control of national insurance budgets difficult. Though Germany had been a strong proponent of these changes, toward the end of this period its health minister Philipp Roesler highlighted the need to put ‘average drug prices [back] under control’ (A.12 2010), while his advisor Jens Spahn advocated for ‘breaking the price monopoly of the pharma industry’ (A.13 2010).
State payers accused this new system of making prices themselves opaque. Under the unified European market, the negotiation of prices for new medicines became progressively framed by the outcome of price negotiation in comparable countries – a process known as ‘external reference pricing’. In this system, the price and medical value-added of a given medicine are indexed on a basket of prices published by comparable EU countries (Rémuzat et al., 2015). However, it emerged that this transparency device actually allowed companies to ‘hide’ prices from state buyers. Even if national negotiating bodies publish the list price of the pharmaceutical, net reimbursed prices include rebates negotiated under non-disclosure agreements (OECD, 2008). Defending a decision to reimburse the high-priced pharmaceutical Orkambi, the Irish health authority for instance voiced the fear that if it ‘disclosed the details of any confidential negotiations . . . other pharmaceutical companies would refuse to negotiate with it’ (A.15 np). The president of the main French payer explains how these rebates were used by pharmaceutical companies to create further opacity (in Barbier & Daudigny, 2016, p. 49; our translation):
[Pharmaceutical industry representatives] defend high list prices (. . .), even if it means accepting rebates on the net price. Rebates are covered by trade secret regulations and are not known by market actors. The consequence is that it becomes difficult to do international comparisons and that there is no more price transparency.
Conflicts over this corporate-centric vision of transparency emerged most clearly when the European Commission attempted to strengthen its ‘Transparency Directive’ in the late 2000s. State payers rejected the draft proposal as ‘solely driven by pharmaceutical competitiveness, to the detriment of public health’. They promoted a vision of transparency that would reassert ‘the authority of member states. . . on their reimbursement list, and also on pricing’ (A.14 2012, p. 1-3). States successfully blocked the proposal, illustrating how contested the prevailing vision of transparency for corporations had become.
In summary, from the early 1990s pharmaceutical representatives and EU policymakers mobilized a new kind of transparency to converge all national markets within the European single market under the banner of ‘fair pricing’ to ensure European pharma corporations’ competitiveness and innovativeness. This vision materialized through a set of rules and devices that culminated in a value-based and reference-driven pricing system for new medicines. The new transparency devices led to a single European pharmaceutical market that was favourable to manufacturers but soon turned out to be unsustainable for many European states.
Period III – Mobilizing transparency for state coalitions (2010 onwards)
Since the early 2010s, transparency has yet again been redefined in the pharmaceutical market, this time in the name of health system sustainability and access. This latest struggle saw national payers become state activists, forming coalitions with other payers and civil society groups to fight for increased public scrutiny over the costs of medicines. While embracing transparency as the main organizing principle for this market, they started to promote a different version of it, focused on sustaining broad access to medicines. New transparency devices were introduced to transform the organization of the European pharmaceutical market, enabling states to pull together their resources. This new vision of transparency in the interest of state coalitions, however, has come to be criticized in its turn at the end of the period, with civil society arguing for a need to go beyond transparency for state coalitions.
Coalescing to redefine transparency
By the early 2010s, faced with escalating pharmaceutical prices that had reached six-figure sums per patient (Bourgeron & Geiger, 2022b), state payers and civil society representatives started to work together to mobilize a new vision of pharmaceutical transparency. They started to pinpoint how the spaces of opacity created in the value-based system of the previous decades were increasingly threatening universal access to medicines. A new definition of transparency centring around healthcare system sustainability was identified as a crucial lever, as this French healthcare activist describes (I.4):
The great thing is that there was a very consensual aspect to transparency, it’s very hard to be against transparency. . . So, we put a lot of energy into transparency because we thought we would manage to obtain victories. . . transparency appeared to be a better battle-horse than other topics.
Enacting this definition of transparency became a central focus for civil society activism, with a number of non-governmental organizations (NGOs) created specifically to advocate for it. Advocacy events directed at mobilizing a new vision of transparency quickly multiplied (see Table 2), but NGOs were only one part of this movement. State activism became central, with the Italian and the Norwegian medical agencies playing a pivotal role in leading contestations against previous definitions of transparency (I.1; I.12; E.15; E.17). These actors sought to redefine transparency ‘as a whole package that needs to apply to the entire value chain. . . to the benefit of all stakeholders’, according to the former head of the Italian agency Aifa (E.15). Or, as stated in the Oslo Medicines Initiative: ‘transparency could be used to build trust between stakeholders, thereby enhancing negotiations and supporting access’ (Larsen, 2022, p. 33).
This mobilization of transparency around safeguarding access to medicines culminated with the Italian medical agency’s proposal for a transparency resolution at the 2019 World Health Assembly, bringing struggles around transparency from a European to a global level. The WHA Resolution redefined transparency in several respects. First, it demanded greater price sharing between countries, to ‘enhance the publicly available information on the prices applied in different. . . countries’ (World Health Assembly, 2019). Second, it demanded transparency on patents, allowing payers to better identify the real R&D efforts behind specific drugs. Third, it asked for transparency on funding, encouraging states to identify the share of pharmaceutical development that was paid for by public sources.
This broadened vision of transparency led to the creation of several new transparency devices aimed at fostering information sharing between European member-states. From 2014, the European Commission developed the European Integrated Price Information Database (EURIPID), through which partner countries can share price information for reimbursed drugs. EURIPID also became a platform for countries seeking to rebalance the negotiating position of states and implement the WHA transparency resolution after 2019. Additional databases were built for sharing information on patents and clinical trials, though their use has remained uneven (E.3; E.13; E.17; Bruckner, 2018).
Small state payers also entered into coalitions to share price information and negotiate prices together. The BeNeLuxA group, gathering Belgium, Netherlands, Luxemburg, Austria and Ireland, was founded in 2015 with the explicit goal to ‘improve transparency on pricing between the collaborating countries’ (BeNeLuxA, 2018, 4; I.8). The Valletta Declaration Group was created two years later around similar objectives, with an original group of six countries (Cyprus, Greece, Italy, Malta, Portugal and Spain), later joined by four others. The Visegrad Group was set up in 2017 by Czechia, Hungary, Poland and Slovakia. Finally, the Nordic Pharmaceuticals Forum was established in 2015 and gathered Iceland, Norway, Sweden and Denmark. These state coalitions explicitly challenged the contractual device of price non-disclosure, with the Greek Minister of Health declaring in a Valletta Group meeting that ‘it [was] time for states to send a message that the era of opaque and arbitrary price setting. . . [had] gone for good’ (Fletcher, 2019).
The WHA resolution resulted in the implementation of additional regulatory devices in several European countries. A 2019 Italian decree for instance compelled pharmaceutical companies to divulge the real prices of drugs in other countries. If the price requested significantly diverged from the real price in those countries, they had to communicate to the regulator the R&D expenses incurred and the public funding it benefitted from (I.1; E.3; E.17). A French clause on transparency of public funding, passed with the 2020 healthcare bill, also compelled manufacturers to communicate to the state negotiator the amount of public funding they had received in the development of a drug (OTMeds, 2021).
Together, these databases, buyer pools and information-sharing devices reorganized the pharmaceutical market in three main ways. First, regarding
Pushing transparency further: transparency for democratic control
The vision of transparency for health system sustainability and access through inter-state collaboration and information sharing largely excluded communicating information to the public. While it was seen as a marked improvement over previous mobilizations of transparency by some activists (I.12) and policymakers (I.1), others remained sceptical as to the leverage these interstate initiatives really had (E.14):
Voluntary collaboration can be a first step to increased transparency: it allows countries to exchange information on prices. But the tools do not lead to transparency automatically. We think it should be a specific requirement for new pharmaceutical initiatives to be [fully] transparent. . . (access to medicines activist)
Here, the activist made an implicit distinction between transparency for coalitions of European states and creating visibility into pharmaceutical costs and prices for the broader public. At stake in these latest contestations is a further redefinition of transparency to reshape this market’s politics, moving toward ‘building a democratic control’ over public expenditures (Barré-Sinoussi et al., 2019). This current situation begs the question of how the aftermath of Covid-19 will reshape the pharmaceutical market. Will we see the devising of another vision of transparency – transparency for democratic control – that could finally reorganize this market toward being more answerable to public concerns?
Discussion
This article asked: how do struggles around transparency reorganize markets? In addressing this question, it follows recent calls to deploy a constitutive perspective of transparency to study how actors claim, configure and contest the concept in different contexts (Heimstädt & Dobusch, 2020). We add to this literature in three crucial respects. First, we combine research on transparency with the literature on market devices to highlight the central role played by the transparency devices through which competing visions of transparency are enacted. We contend that transparency devices play a triple role in organizing markets, as they shape the evaluation of market objects, the topology of market actors, and the resulting prices. Second, drawing this device focus together with an investigation of the shifting political economies in which transparency mobilizations have been situated, we demonstrate how the enactment of different transparencies produces profound shifts in market power, including attempts to depoliticize transparency altogether and place it in a ‘post-political’ toolbox (this was particularly the case in our period II). Third and most important, leveraging insights from political economy for organization studies scholars, we investigate the struggles around transparency and its devices, showing how such struggles are instrumental to those seeking to challenge prevailing market orders and to reverse efforts to depoliticize market transparency. We discuss these contributions in turn.
Devising transparency
Building on a ‘market politics’ perspective that is sensitive to the struggles around concrete market organizations, we were able to show how the successive shifts in visions of transparency were mediated through a range of devices that together reorganized the European pharmaceutical market in various and often contentious ways. Rather than studying ‘governance by transparency’ in its normative sense (Fung, Graham, & Weil, 2007, p. 171), we traced how transparency was instrumented through these device assemblages and how this influenced market power and profit flows. While previous organization studies leveraged a Foucauldian perspective to study the governmental effects of transparency quests, these tended to revolve around the production and management of visibilities as a form of disciplinary control (Flyverbom, 2015). In broadening this perspective to the organization of markets, we find similar links between visions of transparency, their instrumentations, and the power of the agencies that promoted them, for instance when transparency devices in our period I enabled regulators to intervene in markets instead of using direct state aids.
However, our account also painted a more complicated picture, where actors’ own capacities to act, instrumented into the devices, also came to be questioned through them. The transparency devices we analysed shaped agencies – with ‘states’ and ‘the pharmaceutical industry’ themselves transforming into different actors depending on the transparency devices these actors deployed. But the actor topologies that were enacted through the devices were never settled for long, with struggles giving rise to further reshufflings. This was the case for instance with the European Reference Price system, a system favourable to pharmaceutical firms whose increasing complexity became a focal rallying cry for state activists contesting the market’s organization. Thus, our study points toward a more complex engagement of agencies in devising transparency than previous studies may have suggested, and it opens fruitful avenues in studying the various ‘misfires’ that transparency devices may entail.
Varieties of transparency, varieties of markets
Our pragmatic approach allowed us to ground our definition of transparency devices in the practices of market actors themselves, tracing over time all those devices that actors mobilized in controversies around market transparency. As a result, the devices we encountered in our study substantially broaden the list of those previously studied, such as Hansen et al.’s (2015) three ‘disclosure devices’ (due diligence, rankings and big data analysis) or Hansen and Weiskopf’s (2021) ‘mediating technologies’ (leagues, lists, social media and written testimonies). Our transparency devices included evaluation criteria, state committees, databases, coalition agreements, price grids, medication lists, accountancy rules and others. This array points to the multitude of organizational tools that can be deployed as transparency devices and, in different configurations, make specific political concerns knowable and governable (Flyverbom, 2015). We were able to highlight how actors themselves generate and fight over distinct visions and versions of transparency through this array of tools. Transparency, then, becomes a battlefield over technologies, where devices are mobilized and work together to change market power.
It is the ‘working together’ that we wish to stress: our transparency devices did not act on their own, but only in the context of specific socio-material arrangements, as and when mobilized by market actors. Together with Hansen and Weiskopf (2021) and Mehrpouya and Salles-Djelic (2019), by being sensitive to this multiplicity our account demonstrates the extent to which the heterogeneity of the ‘transparency’ concept leads to a wide variety of social organizations. In our case, varieties of transparency existed not only in the objects of transparency (
The power of transparency struggles
Finally, our study has significant implications for researchers interested in how the boundaries and overlaps between political and economic spheres are organized. While organizational scholars have made good inroads into studying the constitutive features of transparency – or how discourses and practices of transparency help build organizations – there is a dearth of work highlighting how the concept’s economic and moral normativities are deployed and concealed. Our case encourages researchers to more fundamentally question the ‘discursive and moral triumph’ (Birchall, 2011, p. 66) of transparency as a pervasive organizational concept and an undisputed economic and social ‘good’ – in other words, to question the politics of the concept itself. Our study shows that what is organized in the name of transparency over time in fact represents highly selective politics – in our case, market politics. In his classic piece on the ‘antipolitical economy’, Barry (2002) argues that politics properly speaking can be fundamentally anti-political if contestations are shut off through technical arrangements, while conversely the politics of such organizational technologies is often vastly underestimated. This view chimes well with the analysis that we put forth in this paper, given how disputed the concept of transparency has been in the pharmaceutical market over the last decades and given the significance of what is at stake there.
Taking a step back with its historical approach, our article helps better situate the specific organizational effects that concepts such as transparency have had in the rise of neoliberal market reforms. Similar to ‘efficiency’, ‘accountability’, or ‘competition’, transparency has held a central role among those post-political principles mobilized to organize economic life in the neoliberal society. While market transparency as a way of controlling firms and ensuring fair prices emerged as early as the 1970s, our article shows our period II (‘transparency for corporations’) from the 1990s as a crucial moment in history in which a specific vision of transparency, directly related to the implementation of New Public Management policies, was heralded as a universal one. Here, our work highlights dynamics similar to those detected by Mehrpouya and Salles-Djelic (2019), with the rise from the 1980s of what they call ‘neoliberal’ transparency. More crucially, however, we show how this moment was not the end of history for market transparency. The neoliberal, post-political definition of transparency has become increasingly contentious in recent years, with policymakers, pharmaceutical lobbyists and civil society activists arguing with strongly opposing views of the same principle. And this battlefield has extended well beyond pharma: during the recent wave of inflation, activists, trade unionists and policymakers called for ‘transparency’ over excessive profit-making by commodity firms, energy suppliers and property investors (AFP, 2022; Jones & Bruce, 2022). In our case, an unlikely coalition of NGOs and smaller European states has started to re-politicize the transparency principle by establishing struggles over its practical mobilizations in national health systems. We can draw parallels from these recent contestations to other post-political principles, for instance ‘efficiency’. Once the monopoly of neoliberal policymakers, ‘efficiency’ has recently been reclaimed by critical activists to promote measures such as the nationalization of corporations (Mazzucato, Li, & Darzi, 2020) and the curbing of lobbying, which is deemed ‘inefficient’ (Transparency International, 2016). While it may be too early to tell whether we have entered the era of ‘post-post-politics’, our case clearly shows how contemporary contestations over such post-political concepts as transparency can re-enliven political debate. But, as our case demonstrated, such re-enlivening only becomes possible when civil society, state activists and researchers expose the socio-material arrangements behind these post-political concepts.
Concluding Thoughts
This paper is concerned with how struggles over transparency reshape the European pharmaceutical market. As Grossman et al. (2008, p. 108) quipped: ‘the devil of transparency is mostly in the details’ – and so, by extension, is its politics. Transparency has been a strategic concern in the organization of the European pharmaceutical market: a tool of marketization, corporate advancement and regulatory intervention. Highlighting the struggles around transparency in one public interest market is crucial not only for comprehending the concrete organizational effects of concepts that may often seem beyond contestation, but also in considering broader issues of how to transform market orders. As Wilson and Swyngedouw (2014) observe, the post-political age has brought to the fore how consensual principles such as transparency work to disempower critical voices and movements. But our article suggests that it has also opened new battlefields for state and civil society activists trying to disturb consensus politics. In our case, struggles over transparency have had a deep influence on the organization of the pharmaceutical market, leading to expanding corporate power and spiralling drug prices until the 2010s, but currently working in the opposite direction. Overall, as we have illustrated, the pharmaceutical market’s failures are unlikely to be resolved by normative and overly general calls for more ‘transparency’, but only through a deep engagement with the specific, multiple and shifting orderings this concept entails.
Footnotes
Acknowledgements
We would like to sincerely thank our Senior Editor, Professor Martin Kornberger, for his expert guidance throughout the publication process. The three anonymous reviewers he chose for this paper were extremely generous with their expertise and time and contributed greatly to this paper’s evolution – thank you! We thank Liliana Doganova and Étienne Nouguez who provided detailed comments on a previous version of this paper as well as the participants at research seminars in Sciences Po Paris and Sciences Po Grenoble in November 2022 for sharing their insights with us. Finally, we wish to thank all our research participants for taking time to share their views with us. All remaining errors are our own.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This project has received funding from the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation programme (grant agreement No 771217). Théo Bourgeron’s research has also been funded by the Leverhulme Trust during the review process of this article through an Early Career Fellowship (reference number ECF-2021-607).
