Abstract
The component model of human parenting has been extensively used to study parents’ interactions with their offspring and to examine variation across cultural contexts. The current study applies this model to nonhuman primates to investigate which forms of parenting humans share with other primates and how these interactions change over infants’ first year of life. We repeatedly observed 52 mother-infant pairs, including humans (N = 11), chimpanzees and bonobos (N = 21), and several species of small apes (N = 20), during different daily activities when infants were 1, 6, and 12 months of age. Humans differed from apes in their higher probability of face-to-face contact and the use of object stimulation. Moreover, parenting seemed to be characterized by more variability within humans than within and possibly between ape species. Overall, the component model of parenting appears to be an effective tool to study the functional systems of parenting behavior in a comparative developmental perspective, by allowing direct comparisons between human and non-human primate species across development.
Introduction
Parenting behavior is a central part of humans’ and other mammals’ behavior, as it is necessary for offspring’s survival after birth (Trevathan & McKenna, 1994; Trivers, 1972). In humans, parenting include behaviors like nurturing, warming, and safe-guarding (Keller, 2007), but also behaviors that facilitate the detection of infants’ negative and positive cues (Keller et al., 2005) and the regulation of their emotions (Holodynski & Friedlmeier, 2006). To categorize these different types of parent–child interactions, Keller (2007) has established the component model of parenting with six distinct parenting systems, which has been applied by different studies across several sociocultural contexts (e.g., Keller et al., 2009; Keller, Lohaus, et al., 2004). In the following, we will first introduce this component model and its different parenting systems, explain its link to other parenting frameworks, and then apply it to the study of parenting in primates other than humans. This systematic longitudinal and comparative approach will allow us to investigate the developmental patterns of parenting systems in several ape species during infants’ first year of life and to identify the human parenting behaviors that are shared with other primates and those that might be instead uniquely human. To other researchers, this work will provide an assessment of the component model of parenting as a new tool to study the functional systems of parenting behavior from a comparative perspective, allowing direct comparisons between human and nonhuman primate species.
Theories of Human Parenting
One of the most extensively studied questions in psychology is the effect of different forms of parenting behavior on children’s development. Theories of human parenting widely differ on a variety of crucial dimensions but mostly describe how the different ways in which parents relate to their offspring predict children’s development and/or modulate genetical and environmental influences on it (Holden, 2010). Attachment theory, for instance, views the affiliative relationship between mothers and children as an adaptive behavioral system that promotes offspring’s survival and competent functioning: by showing affection, helping to regulate children’s emotions, and satisfying their needs, parents support the development of secure attachments, they provide a secure base for offspring to explore the environment, foster social and cognitive competences and promote autonomy in their child (Ainsworth & Bowlby, 1991; Easterbrooks et al., 2013).
How affiliative relationships and attachment patterns are instantiated, however, can strongly differ across cultures. Ecological theories of human parenting situate the relationship between parents and children within the environment and highlight the influence of cultural context (e.g., cultural values, attitudes, customs, and norms) on this relationship (e.g., Bronfenbrenner & Morris, 2006; Whiting & Whiting, 1975). By observing others belonging to the cultural community, children, parents, and other caregivers develop expectations about their relationship, reproduce observed behavioral patterns in it (Rogoff, 2003), and develop routines that incorporate culture-specific values, emotions, and norms (Weisner, 2002). In this way, culture shapes parenting behavior, which in turn contributes to maintaining and transmitting cultural values and norms across generations (Bornstein & Lansford, 2010; Harkness et al., 2007).
Functional Approaches: The Component Model of Parenting
To study human parenting across cultures, researchers often differentiate between the form and function of parenting behaviors (Bornstein, 2012). Different parenting behaviors, indeed, can serve the same function across cultural contexts (Bornstein, 2012), for instance, to foster positive outcomes in school children (Leung et al., 1998) or to express love and affection (Cheah et al., 2015). These different parenting behaviors are clearly adaptive in some cultures, but not in others (Ogbu, 1993).
One theoretical framework that has been used to study cross-cultural variation in human parenting is the component model of parenting, which assesses the functional link of specific parenting systems (i.e., sets of parenting behaviors that serve a similar function) to children’s motor, cognitive, emotional, and communicative development (Keller, 2007). The primary care system describes caring duties like nurturing or keeping the infant warm and safe. The body contact system includes every form of physical contact between parent and infant, while the body stimulation system is characterized by parents’ motor stimulation behavior of infants’ body or body parts. The face-to-face contact (FC) system comprises parents’ effort to move their face into the infant’s visual field, enabling the exchange of gaze contact and facial signals. The object stimulation system is defined as the use of an object to stimulate infant’s attention, while the narrative envelope system describes language as an interactive tool. According to Keller (2007), these systems are not mutually exclusive and can occur in various combinations, resulting in specific parenting styles. Common combinations include a frequent use of body contact and body stimulation practices (i.e., proximal parenting style) and a frequent use of FC and object stimulation (i.e., distal parenting style).
The occurrence and distribution of these systems vary across cultures, as caregivers’ actions are strongly influenced by the sociocultural context and corresponding cultural models of parenting (Bornstein, 2012; Bornstein & Lansford, 2010; Bronfenbrenner & Morris, 2006; Kärtner et al., 2010; Keller et al., 2018; Liebal et al., 2011; Rogoff, 2003; Whiting & Whiting, 1975). For example, urban-living mothers from Costa-Rica, Germany, and Greece often prefer a distal parenting style with frequent face-to-face interactions and object stimulations, while rural village-living mothers from the Cameroonian Nso and Indian Gujarati preferentially use a proximal parenting style with frequent use of body stimulation (Keller, Lohaus, et al., 2004; Lamm et al., 2014).
Applying the Component Model of Parenting to Other Primates
While the component model of parenting has been applied to study variability in human parenting behavior (e.g., Kärtner et al., 2010; Keller et al., 2009; Lamm et al., 2014), virtually nothing is known about whether this model is also suitable to study our closest relatives, the nonhuman apes. Humans (Homo) belong to the same taxonomic group (Hominoidea) as nonhuman apes (hereafter, apes), which includes great ape species like chimpanzees and bonobos (genus Pan), orangutans (Pongo) and gorillas (Gorilla), and small ape species like siamangs (Symphalangus), hoolock gibbons (Hoolock), dwarf gibbons (Hylobates), and crested gibbons (Nomascus) (Fuentes, 2018; Groves, 2005).
Like in humans, the survival of young apes strongly depends on maternal care, as mothers usually provide food, care, and protection to their offspring, forming strong, long-lasting bonds with them and facilitating their interaction with the socioecological environment (Lonsdorf, 2017; Maestripieri, 2018; van Noordwijk, 2012). In human and nonhuman primates, for instance, lactation increases offsprings’ survival by fostering their physical growth, but also their emotional, social, and cognitive development (Hrdy, 1999). However, there are clearly also important differences in parenting styles, both within and across species, which partially mirror the socioecological conditions in which primates live (Hrdy, 2009; Maestripieri, 1994).
In nonhuman primates, one framework that has often been used to capture this variation typically assesses parenting behavior along two independent dimensions: protection and rejection (e.g., Fairbanks, 1996; Maestripieri, 2018). Protective mothers, for instance, are more likely to spend time in social interactions with their offspring but also more likely to restrain them; in contrast, rejective mothers more frequently avoid proximity with their offspring, also recurring to aggression to interrupt physical contact (e.g., Bardi & Huffman, 2002, 2006; Fairbanks, 1996; Maestripieri, 1994, 1999, 2018; Sekizawa & Kutsukake, 2019). Some primates may also vary along these dimensions depending on the structure of their social groups. Across macaque species, for instance, maternal protection is usually more common in despotic species like rhesus (Macaca mulatta) and pig-tailed macaques (M. nemestrina), where mothers need to mitigate the higher risk of severe aggression by other group members; in more tolerant species like stump-tailed macaques (M. arctoides), in contrast, mothers are less protective toward their offspring (Berman, 1980; Maestripieri, 1994; Thierry, 2000). These studies have provided crucial information about the factors that likely explain variation in parenting styles, but also about the possible effects that specific maternal behaviors might have on the offspring’s social development (e.g., Amici et al., 2019; Bard, 1994; Fairbanks, 1996; Lonsdorf, 2017; Maestripieri, 2018; Maestripieri et al., 2006; Tomonaga et al., 2004). However, the component model of parenting (Keller, 2007) may provide a valid complimentary approach to studying the functional systems of parenting in nonhuman primates, for several reasons. First, it provides a clear theoretical framework to directly assess the functional link between specific parenting systems and the offspring’s motor, cognitive, emotional, and communicative development. Second, it focuses on a different set of behaviors that occur during interactions between mothers and offspring (see below), providing complimentary information as compared to other approaches focusing on protection–rejection measures. Third, the component model of parenting has been repeatedly used to study humans and also across cultures, and it therefore allows direct comparisons between human and nonhuman primate species—a comparative approach that is not possible with other theoretical frameworks.
Regarding the parenting system of body contact, chimpanzees and bonobos seem to differ from other apes, since infants need their mothers’ constant support in the first months of their life (Bard, 2002; Hopkins & de Waal, 1995). For example, chimpanzee mothers and their infants almost constantly maintain body contact over the infants’ first months of life, until they can cling to their mothers’ body on their own (Bard, 1994), while newborn orangutans and gorillas can already cling to their mothers without support (Fossey, 1979; Galdikas, 1982). Humans differ from other apes as infants are not able to cling independently on their mothers’ hairless body, which provides no hold (Hayashi & Matsuzawa, 2017). In contrast, touch is an important form of body contact across humans and nonhuman primates, which can occur during grooming, infant carrying, and comforting, and plays an essential role in developing strong bonds between mothers and offspring (Hertenstein et al., 2006).
Regarding the parenting system of body stimulation, Bard (1994) observed “physical exercises” of chimpanzee mothers to stimulate their infants’ motor development by supporting their crawling and walking attempts. Because body contact is almost always maintained during the first months of an infant’s life, mothers often perform such exercises on their infant’s body (Bard, 2002). Very little is known about other apes, and existing studies largely focus on mothers’ encouragement to stimulate infants’ early locomotion (Maestripieri et al., 2002). In humans, the significance of body stimulation varies across cultures, being considered crucial for children’s healthy development in some communities (Keller, 2007), whereas in others, it is assumed to follow a rather predetermined pattern of development and is thus not specifically enforced (Keller et al., 2009).
Regarding the parenting system of FC, such interactions have been described for rhesus macaques (Macaca mulatta) and chimpanzees (Bard et al., 2005; Dettmer et al., 2016; Ehardt & Blount, 1984). In chimpanzees, this is often referred to as “mutual gaze,” and chimpanzee mothers gaze relatively frequently toward their offspring, often directly in their face (Amici et al., 2023; Bard, 1994; Tomonaga et al., 2004), and especially when physical contact is interrupted (Okamoto-Barth et al., 2007). With infants’ increasing age, mutual gazes increase in chimpanzees (Amici et al., 2023), possibly because infants spend less time in body contact with their mothers and therefore might engage more frequently in this distal form of interaction. Interestingly, differences in the frequency of FC were also found between chimpanzee groups (Bard et al., 2005; Okamoto-Barth et al., 2007), although they were explained by “the interchangeability of visual and tactile modalities” (Bard et al., 2005, p. 621) and not as systematic differences between groups.
Regarding the parenting system of object stimulation, human mothers frequently include objects in interactions with their infants to attract and direct their attention, although this occurs more frequently in urban-living families with a Western cultural background than in rural-living families with a non-Western background (Keller, 2007). In apes, comparable stimulations that trigger infants’ interest in objects have not yet been described (Bard & Vauclair, 1984; Maestripieri et al., 2002). Developmental studies with chimpanzees showed that infants start to manipulate objects from 3 to 5 months onward (Tomonaga et al., 2004), but infants and their mothers rarely looked at each other while manipulating objects, nor did they engage in showing or giving these objects to each other. Thus, unlike humans (Carpenter & Liebal, 2011), chimpanzees do not seem to engage in such triadic interactions (Tomonaga et al., 2004).
Regarding the parenting system of narrative envelope, nonhuman primates lack this system as they have no language. Therefore, we excluded this system from a cross-species comparison. We also excluded the parenting system of primary care because both humans and apes provide their offspring with food, shelter, and hygiene. Therefore, we did not expect any general differences between humans and apes.
Taken together, existing studies suggest some forms of human parenting are shared with other apes, although face-to-face interactions and object stimulation may be more pronounced in humans. However, our knowledge about parenting behaviors in apes is still limited, because direct comparisons are scant, have usually included no species other than humans and chimpanzees, and/or have investigated infants only at a specific age, neglecting possible changes over the course of development. However, systematic cross-species comparisons with a longitudinal approach are crucial, as pace and pattern of infants’ development and their corresponding parental systems might vary across species because of primate different life histories (Rosati et al., 2014).
Aims of the Current Study
The present study aims to address current limitations in comparative and developmental research, by applying the component model of parenting to other primate species, to investigate the extent to which the parental systems of body contact, body stimulation, object stimulation, and FC are observed not only in humans, but also in chimpanzees, bonobos, and small ape species. We used a longitudinal approach and observed infants at the age of 1, 6, and 12 months to investigate longitudinal pathways of mother-infant interactions during infants’ first year of life. Since paternal care is rare in primates other than humans (Lappan, 2008; Santos et al., 1997), we only focused on mothers to enable comparisons across species. Unlike most existing studies with humans (e.g., Keller et al., 2003; Keller, 2007; Keller, Yovsi, et al., 2004; Lamm et al., 2014), we covered different daily activities and not only interactions in playful contexts. By directly comparing humans, great apes, and several small ape species, we provided a broader perspective on the possible evolutionary origins of different parenting systems. We expected that (1) ape mothers would spend more time in body contact with their infants across all ages, as compared to human mothers, while (2) they would spend less time in FC and (3) object stimulation. However, we had no specific prediction with regard to body stimulation, as little is known about this parenting system in primate species other than humans. We also expected longitudinal changes in the first year of life for some of the parenting systems, with (4) body contact decreasing with infants’ increasing age across all species and (5) FC gradually increasing through age, especially in human mother-infant pairs.
Methods
Recruitment
In humans, research was conducted according to the ethical standards of the Deutsche Gesellschaf für Psychologie (German Psychological Association) and in line with the ethical guidelines of the research institution (Freie Universität Berlin), as approved by the graduate school of the cluster “Languages of Emotion” and the department of Comparative Psychology, under supervision of Prof. Dr. Katja Liebal, in March 2021. The recruitment of women took place at the Freie Universität Berlin via public advertisement and the participant pool of the Excellence Cluster “Languages of Emotion,” either in their third trimester or when they recently gave birth (less than 1 month before the start of data collection). Women who met the criteria (full-term delivery and the lack of signs of pre- or post-natal depression) were informed about the aims and procedure of the study. Before the start of data collection, mothers gave their written consent to participate in this study.
In apes, we contacted zoological gardens and animal parks across Europe to give their permission to conduct this observational study and to inform us if new infants were born. Permits to conduct research on apes were obtained separately from each institution between June 2011 and October 2012. All zoos fulfilled the terms of the WAZA Code of Ethics and Animal Welfare (World Association of Zoos and Aquariums [WAZA], 2003), the Guidelines for the Treatment of Animals in Behavioral Research and Teaching of the Association for the Study of Animal Behavior (ASAB, 2006) and the EAZA Minimum Standards for the Accommodation and Care of Animals in Zoos and Aquaria (European Association of Zoos and Aquaria [EAZA], 2014).
Subjects
The study comprised a total of 52 mother-infant pairs, including 11 human pairs (Homo sapiens), 11 chimpanzee pairs (Pan troglodytes), 10 bonobo pairs (Pan paniscus), and 20 hylobatid pairs of seven different species (3 pairs of pileated gibbons, Hylobates pileatus; 5 pairs of white-handed gibbons, Hylobates lar; 1 pair of silvery gibbons, Hylobates moloch; 5 pairs of siamangs, Symphalangus syndactylus; 2 pairs of Northern white-cheeked crested gibbons, Nomascus leucogenys; 1 pair of Southern white-cheeked crested gibbons, Nomascus siki; 3 pairs of Yellow-cheeked crested gibbons, Nomascus gabriellae). For a complete list of study subjects and their characteristics, please see Table S1 in Supplementary Materials. All infants were healthy and typically developing at the time of the first contact and were raised by their biological mothers.
All human mother-infant pairs lived in an urban setting (n Berlin = 10, n Leipzig = 1). The number of primiparous and multiparous mothers was balanced (n first child = 6, n later child = 5). On average, mothers were 30.6 years old (range = 25–38 years, SD = 4.01 years) at the time of their infant’s birth (either first- or later-born). The sample included six boys and five girls (Supplemental Table S1). Ape mothers’ age at infants’ birth ranged between an average of 18 years for small apes and 23 years for bonobos and chimpanzees. Similar numbers of male and female infants could be recruited for the genera Pan and Hylobates, while for Nomascus and Symphalangus, only male infants were available (Supplemental Table S1). The majority of mothers were multiparous. The number of mother-infant pairs included in the study was constrained by the limited availability of observable ape pairs during the study period. The number of human pairs was accordingly constrained, as for modeling purposes, it was more convenient to have a similar number of observations across species (see below).
Procedures
Data collection was conducted by MEL and three research assistants. We visited each mother-infant pair repeatedly when the infants were 1, 6, and 12 months (±8 days) old (Supplemental Table S1). We conducted spot observations (Rogoff, 1978), a modified version of focal animal sampling, to record naturalistic interactions (Altmann, 1974). We used a digital video camera (Panasonic, HDC-HS30) to film apes from the visitor areas during the zoos’ regular opening hours and humans at their homes at different times of the day to cover varying activities of their normal daily routine (e.g., feeding, cleaning, playful interactions, resting, and sleeping).
We included a total of 197 hr of video footage in the study (Supplemental Table S1), including 130 hr of film footage collected by MEL and 67 hr of additional footage from previous developmental studies with chimpanzee and bonobo mother-infant pairs (Schneider et al., 2012) and gibbon mother-infant pairs (Waller et al., 2012). Some of these videos were used in other studies to address different research questions (Amici et al., 2023, 2024). For some infants, it was not possible to collect data at all ages because of illness (one human infant) and death of infants (one chimpanzee and two gibbon infants). For the additional video footage, total observation time varied across dyads, ranging from 20 to 120 min per age stage (Supplemental Table S1), because of different methods of data collection. These differences in the length of observations were controlled for in the statistical analysis.
Coding
To enable comparisons across studies, we applied the same coding instructions from previous studies using the component model of parenting in humans (e.g., Keller et al., 2009). We divided each video clip into 10 s-intervals, in which one or more of the following four parenting systems were coded: (a) body contact, whenever the mother remained in physical contact with the infant for at least 5 s within a 10 s-interval, with any body part (e.g., fingers, arms, torso, head, or face); (b) body stimulation, consisting of repeatedly touching or moving the infant’s body (e.g., moving infant’s limbs repeatedly back and forth or up and down, stroking, tickling, grooming, or washing, rocking, tossing, supporting the infant’s own crawling or walking attempts), whenever it occurred at least once within a 10 s-interval; (c) FC, consisting of mother and infant directing their faces toward each other to enable or support an exchange of communicative signals (e.g., eye contact, vocalizations, and facial expressions) whenever the mother engaged in it at least 5 s within a 10 s-interval; (d) object stimulation, describing the mother’s effort to stimulate the infant’s attention with an object (e.g., toys or sticks), whenever it occurred at least once within a 10 s-interval.
Inter-rater reliability was established for a randomly selected subset of video footage (5% of the total sample), which was coded by a research assistant naïve to the hypotheses of this study. We calculated reliability using Cohen’s kappa, reaching substantial to high agreements (body contact = 0.94, body stimulation = 0.93, FC = 0.75; object stimulation = 0.85; Landis & Koch, 1977).
Analyses
First, we calculated the probabilities of occurrences of the four parenting systems within each of the 10 s-intervals (Table 1). We conducted analyses at the genus level, comparing humans (Homo), chimpanzees and bonobos (Pan), siamangs (Symphalangus), lar gibbons (Hylobates) and crested gibbons (Nomascus). Second, we ran generalized linear regression models using the package lme4 (version 1.1-10; Bates et al., 2010) for R (version 3.5.2, R Core Team, 2018). Given that each parenting system could be present or absent in the 10 s-intervals, we conducted four sets of models with a logit link function and a binomial structure, including body contact, body stimulation, FC, and object stimulation as response in each set of models. For each set of models, we used the same sequential steps of analysis: an intercept-only model with dyad as random effect, allowing variation in the outcome over dyads (Model 0); a model also including genus as test predictor (Model 1); a model further including age as test predictor (Model 2); a model also including a random slope for age, allowing for age-related changes to vary between dyads (Model 3); and a model further including the interaction of genus and age as test predictor, to investigate inter-specific differences in developmental changes for each parenting system (Model 4).
Probability of Each Parenting System Being Observed for Each Age (in Months) and Genus (Homo: N = 11, Pan: N = 21, Symphalangus: N = 5, Hylobates: N = 9, Nomascus: N = 6).
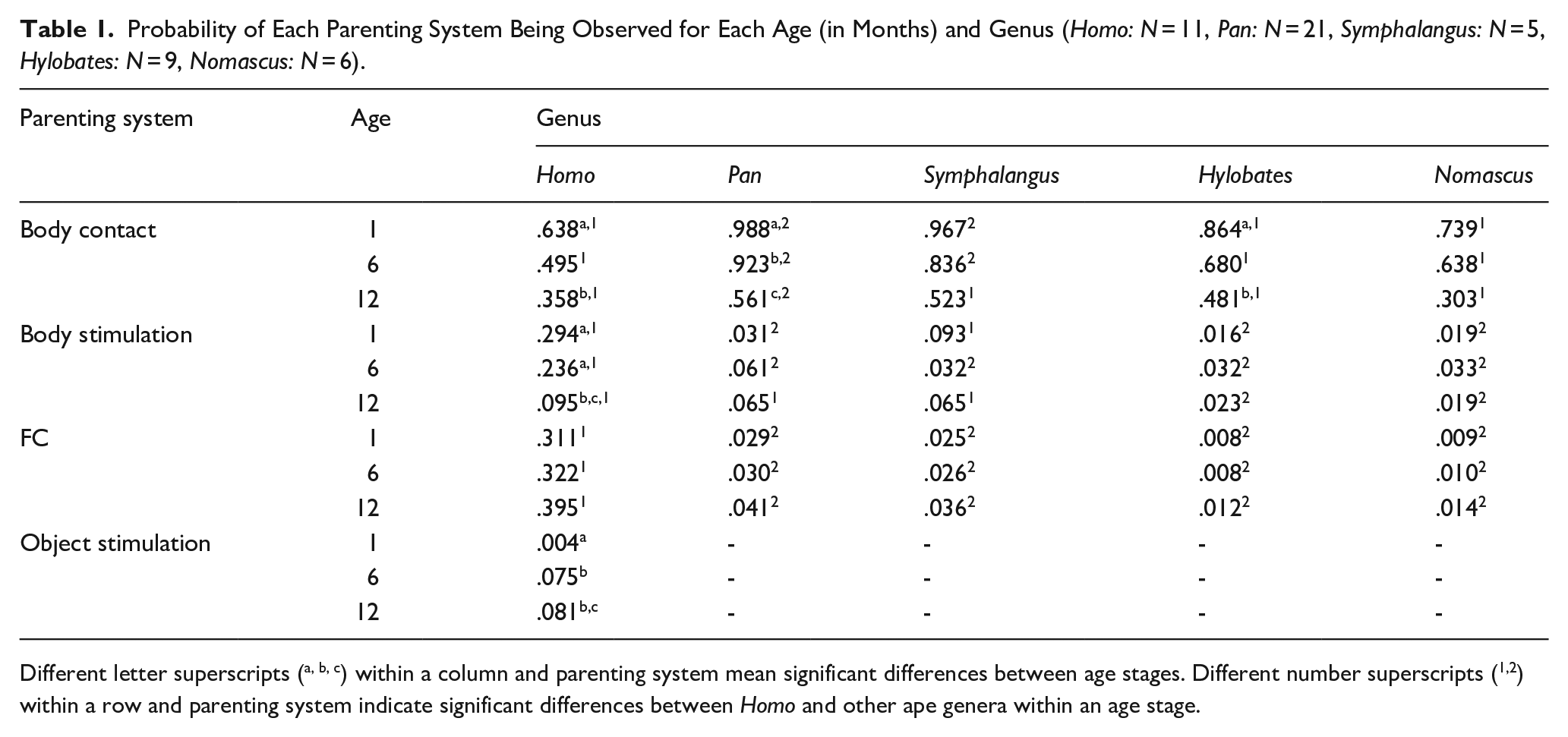
Different letter superscripts (a, b, c) within a column and parenting system mean significant differences between age stages. Different number superscripts (1,2) within a row and parenting system indicate significant differences between Homo and other ape genera within an age stage.
Within each set of models, we used a likelihood ratio test (LRT) to compare subsequent models and explore the necessity of more complex models and pseudo-R2 in the R-package MuMIn (version 1.43.15, Bartón, 2018), following Nakagawa and colleagues (2017). Both the marginal and the conditional pseudo-R2 are reported, with the former providing information on the fixed effects in the model and the latter also on the random effects. If LRTs revealed the best fit for the model, including the interaction between genus and age (Model 4), we conducted contrasts with the multcomp package (1.4-12, Hothorn et al., 2008) to detect developmental changes within each genus.
Results
Table 1 summarizes the probabilities of occurrence of each parenting style, depending on genus and age, and Table 2 the results of model comparisons. Significant contrasts are presented below, whereas non-significant ones are included in Tables S2 to S5 as Supplementary Material.
Sequential Comparison of GLMER Models Using Likelihood Ratio Tests for the Following Body Systems: Body Contact, Body Stimulation, Face-to-Face Contact, and Object Stimulation, Based on the Data Collected in Homo (N = 11), Pan (N = 21), Symphalangus (N = 5), Hylobates (N = 9), and Nomascus (N = 6).
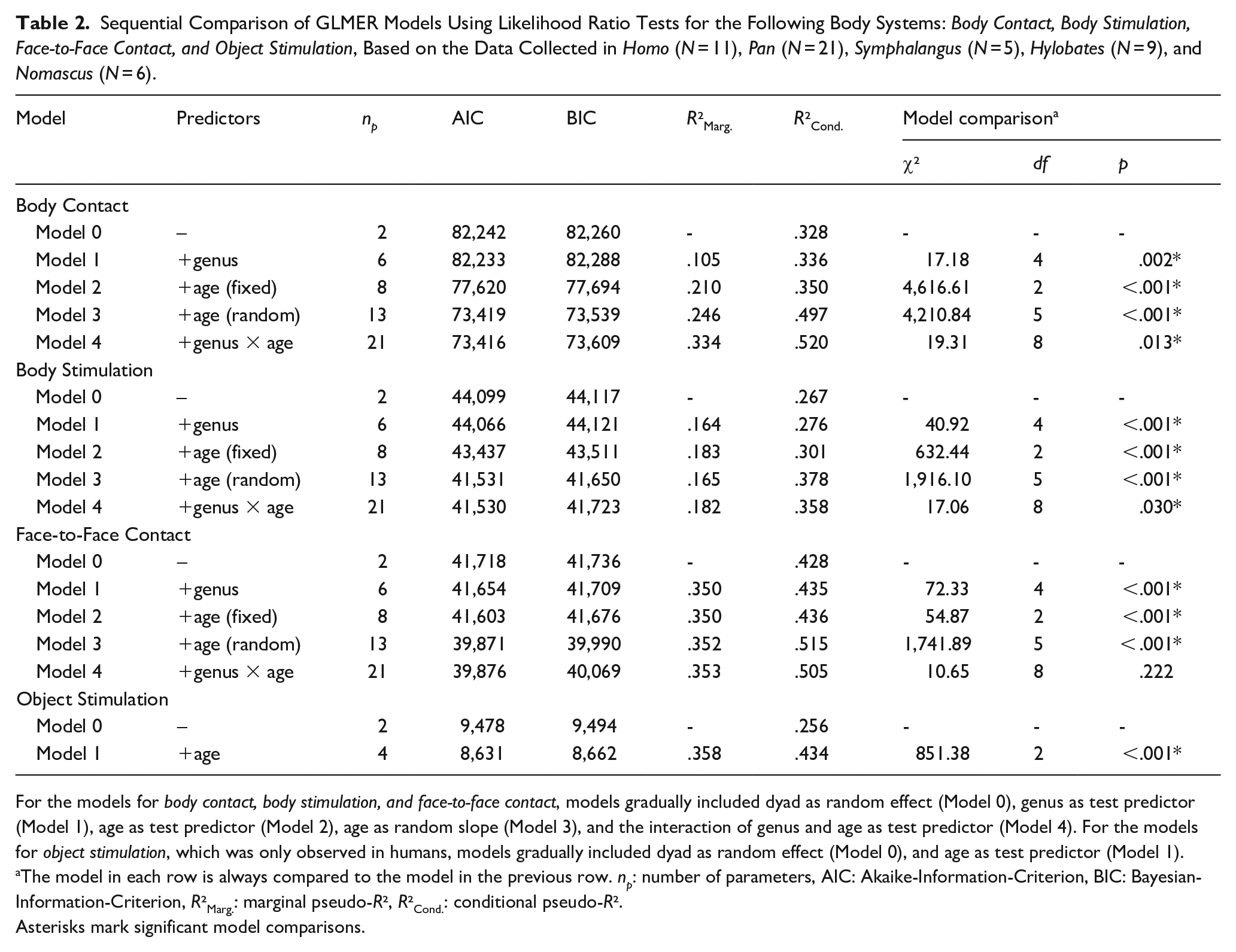
For the models for body contact, body stimulation, and face-to-face contact, models gradually included dyad as random effect (Model 0), genus as test predictor (Model 1), age as test predictor (Model 2), age as random slope (Model 3), and the interaction of genus and age as test predictor (Model 4). For the models for object stimulation, which was only observed in humans, models gradually included dyad as random effect (Model 0), and age as test predictor (Model 1). aThe model in each row is always compared to the model in the previous row. np: number of parameters, AIC: Akaike-Information-Criterion, BIC: Bayesian-Information-Criterion, R2Marg.: marginal pseudo-R2, R2Cond.: conditional pseudo-R2.
Asterisks mark significant model comparisons.
The body contact system was the most frequent parenting system across genera and ages (Figure 1). LRTs revealed the best fit for Model 4, including the interaction between genus and age. The probability of body contact tended to decrease with increasing age across all genera, reaching significance in Pan (p < .001 for all age comparisons), Homo and Hylobates (only for the comparison between months 1 and 12; p = .018 and p = .007, respectively), but not in Symphalangus and Nomascus (Supplemental Table S2). Moreover, mother-infant pairs differed in their slopes of decrease.
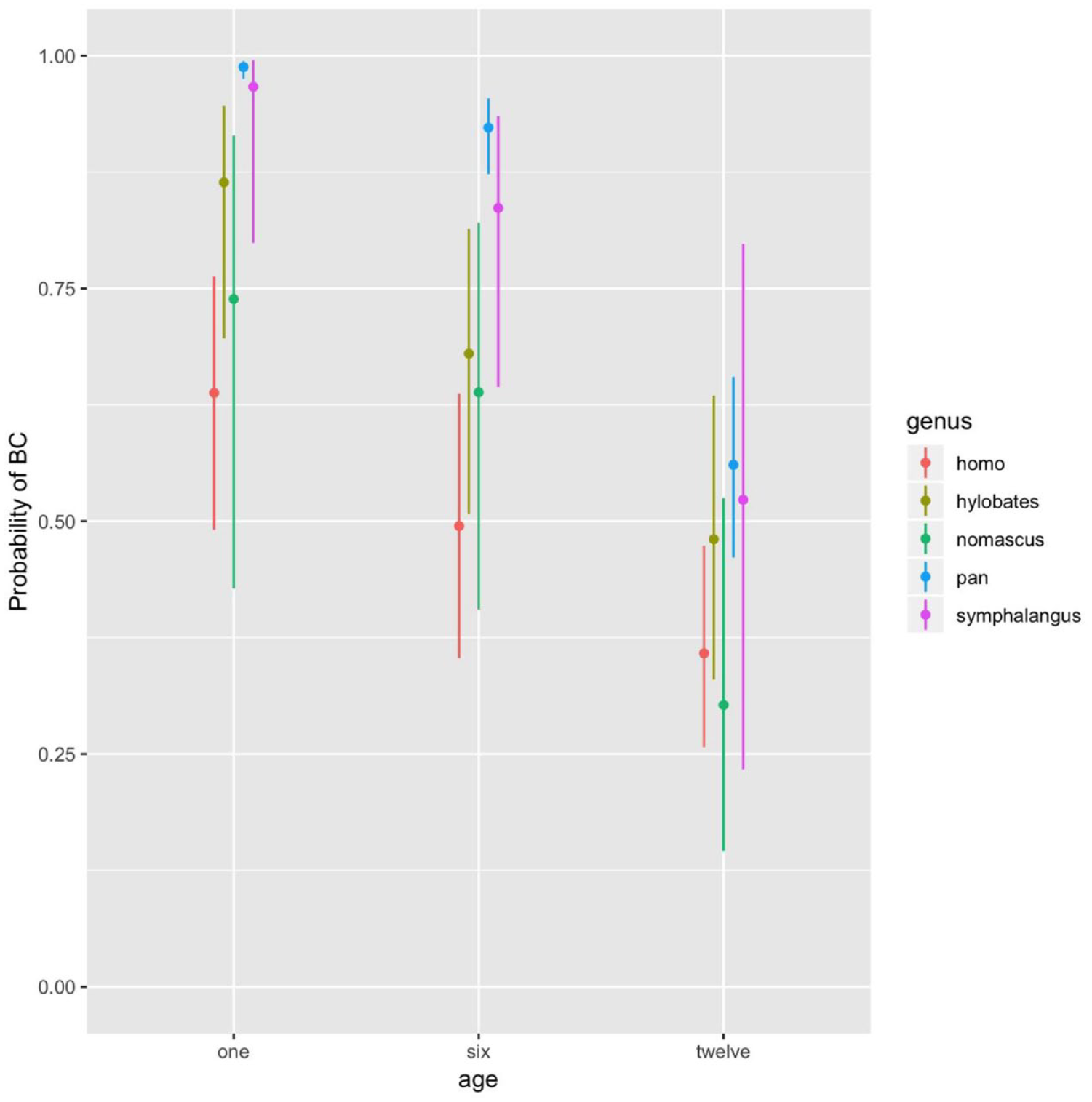
For Each Genus Observed (Homo: N = 11, Pan: N = 21, Symphalangus: N = 5, Hylobates: N = 9, Nomascus: N = 6), Estimated Probability of Body Contact (BC) With 95% Confidence Intervals, at One, Six, and Twelve Months of Infants’ Age.
For the body stimulation system, the probability of body stimulation was generally higher in Homo than in ape genera, which rarely showed it (Figure 2). LRTs revealed the best fit for Model 4, including the interaction between genus and age. The probability of body stimulation decreased with age in Homo (between months 6 and 12 and months 1 and 12; p = .001 and p < .001, respectively), while across ape genera, there was no significant change with infants’ age, remaining at very low levels, despite high inter-individual differences in body stimulation at month 1 and 12 of infants’ age in Symphalangus (Supplemental Table S3).
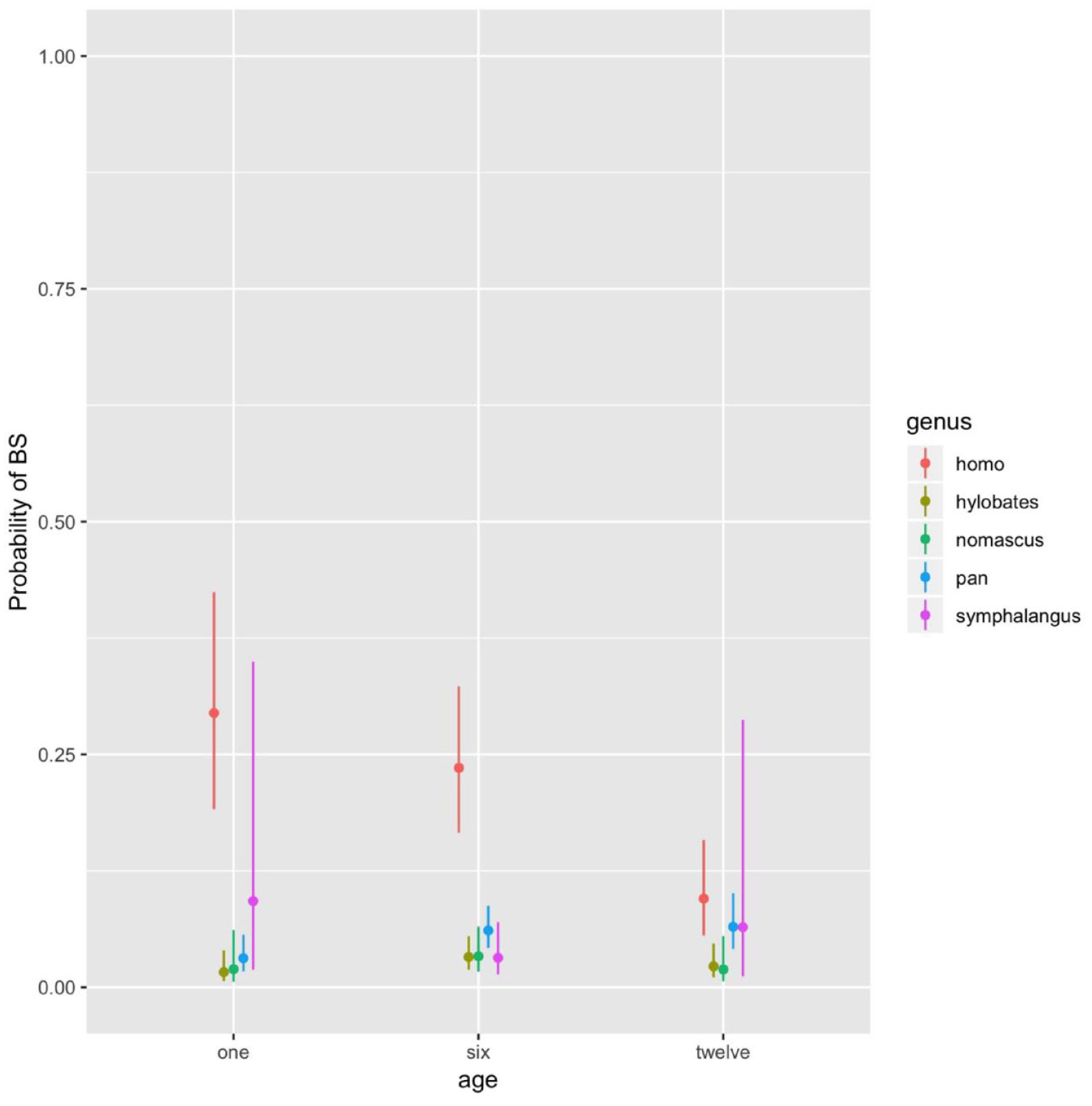
For Each Genus Observed (Homo: N = 11, Pan: N = 21, Symphalangus: N = 5, Hylobates: N = 9, Nomascus: N = 6), Estimated Probability of Body Stimulation (BS) With 95% Confidence Intervals, at One, Six, and Twelve Months of Infants’ Age.
For the FC system, LRTs revealed the best fit for Model 3, including genus and age, but not their interaction. Human mothers were more likely to engage in FC than all other ape genera, which showed very low probabilities across all ages (Figure 3). Despite the main effect of age, contrasts revealed no significant differences between specific ages (Supplemental Table S4).
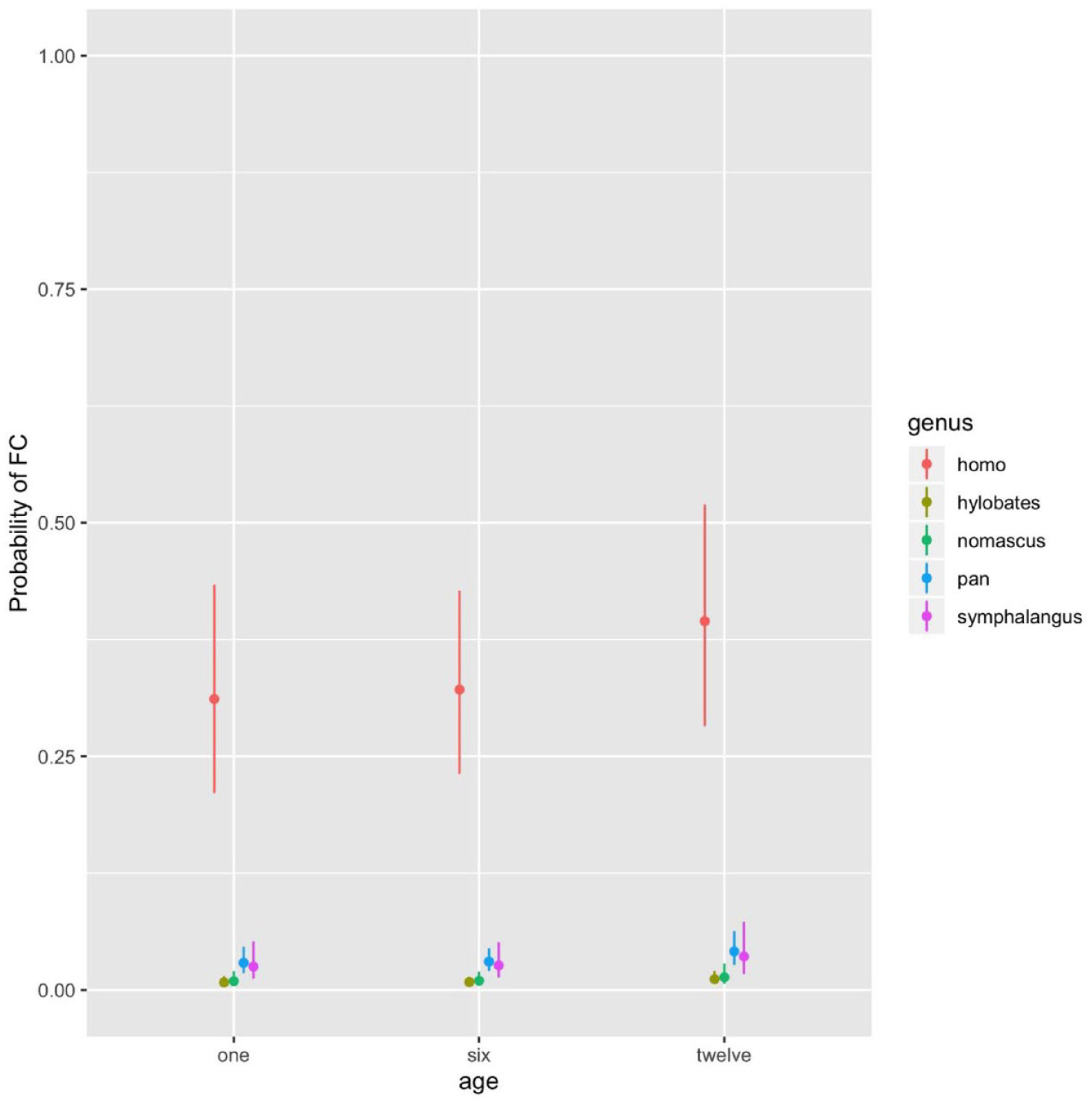
For Each Genus Observed (Homo: N = 11, Pan: N = 21, Symphalangus: N = 5, Hylobates: N = 9, Nomascus: N = 6), Estimated Probability of Face-to-Face Contact (FC) With 95% Confidence Intervals, at One, Six, and Twelve Months of Infants’ Age.
In contrast to the other parenting systems, object stimulation was only observed in humans, so the model comparison was only based on human data and only included two models with no genus as a test predictor (Table 2). LRTs revealed the best fit for Model 1, with the probability of object stimulation in humans significantly increasing between months 1 and 12 and between months 1 and 6 (Figure 4; both p < .001) but not between months 6 and 12 (Supplemental Table S5).
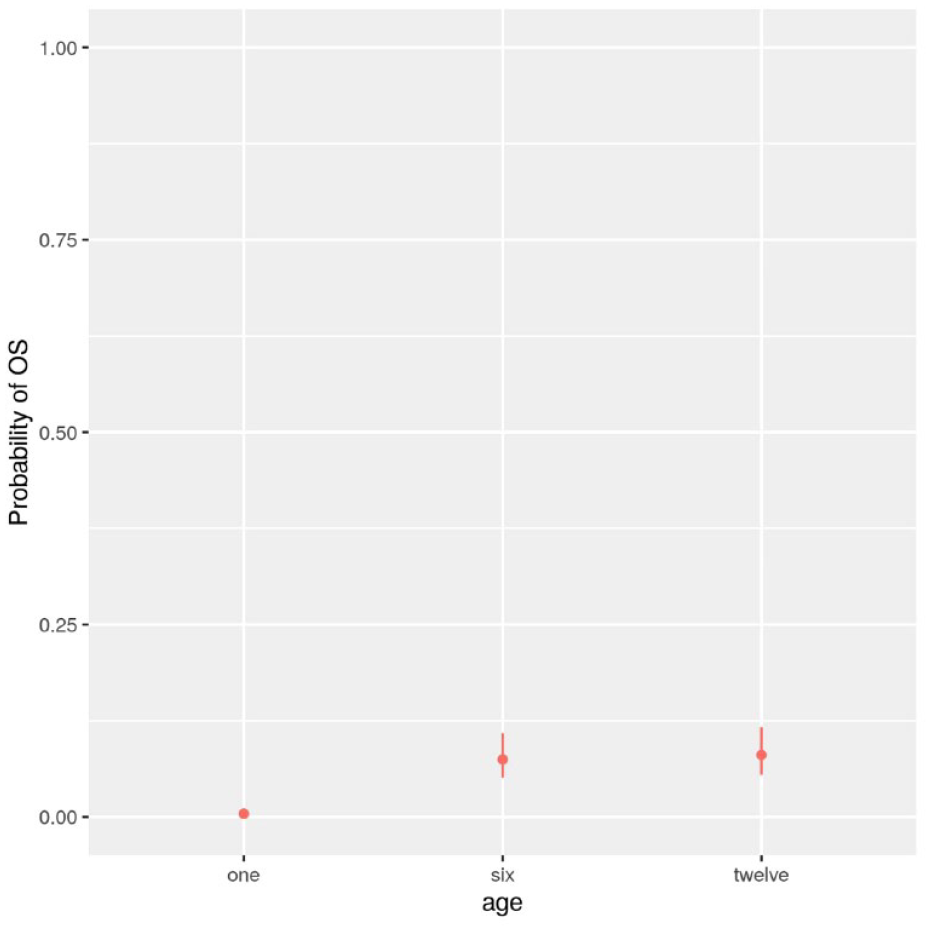
Only for Humans (N = 11), Estimated Probability of Object Stimulation (OS) With 95% Confidence Intervals, at One, Six, and Twelve Months of Infants’ Age.
Discussion
In this study, we applied one of the existing cross-cultural frameworks used to study parenting behavior in humans to other primate species. In particular, we used the component model of human parenting to directly compare developmental pathways of mother-infant-interactions across species, during infants’ first year of life, and to identify similarities and differences between humans and four genera of nonhuman primates, including chimpanzees and bonobos, and several species of small apes (Nomascus, Hylobates, and Symphalangus). Except for OS, which was only observed in humans, the other three systems (BC, BS, and FC) were also observed in the other genera. In line with our predictions, human mothers were more likely than other mothers to show FC (Prediction 2) and OS (Prediction 3). However, human mothers did not show less BC than the other genera (in contrast to Prediction 1), and their developmental patterns did not significantly differ from that of other primates. Largely in line with our predictions, BC decreased with infants’ age across all species (Prediction 4), although Symphalangus and Nomascus failed to reach significance. However, the probability of FC failed to increase through infants’ age, for all species (in contrast to Prediction 5). BC was the most frequently observed parenting system across species and age, without a clear difference between humans and other species. As predicted, BC decreased with increasing age across species, but to a different extent for each genus: while it significantly decreased for Homo, Pan, and Hylobates, it failed to reach significance for Symphalangus and Nomascus. Interpreting these inter-specific differences in developmental patterns is no easy endeavor because of the paucity of longitudinal studies with these species in literature. The steadier decrease in BC for Homo, Pan, and Hylobates cannot easily be explained in terms of differences in their socioecological characteristics or motoric development, and more studies with larger sample sizes are therefore needed to also account for the large variation across conspecific dyads. Previous studies with humans had mostly investigated this parenting system with infants up to 3 months of age, with a focus on play contexts (Kärtner et al., 2010). Our study shows that, despite a decrease in BC through age, this parenting system remains an important component of mother-infant interactions during the first year of infants’ life, not only in humans but also in apes. This is not surprising, as BC is considered an evolutionarily old system, which can be highly adaptive for all ape species by allowing mothers to provide warmth and protection to their infants but also to convey emotions, empathy, and a feeling of security (Hofer, 1987; Keller et al., 2009; Stack, 2001).
For BS, we did not have predictions, as little is known about nonhuman primates, apart from the reports of “physical exercises” in chimpanzees (Bard, 1994). Our results showed that human mothers engaged significantly more in BS than the other genera. In humans, BS decreased through age, while the probability for apes remained relatively low throughout the first year of life. Furthermore, the type of BS qualitatively differed between humans and apes. While human mothers often caressed and kissed their infants and supported them during their crawling, standing, or walking attempts, ape mothers stimulated their infants’ bodies mainly during grooming or play interactions. Other studies report tickling in chimpanzees (van Lawick-Goodall, 1968) and bonobos (Palagi, 2006) as a type of BS, which was not observed in the current study. These differences between humans and other ape species might be explained by the higher adaptive advantage that these behaviors might provide to humans, as this species is characterized by slow motoric development, and infants might especially benefit from BS, in contrast to infants in other apes that engage in independent motoric activities from earlier on (Bründl et al., 2021).
FC was significantly more common in humans than in apes, in line with other studies showing that this parenting system is an important component of the distal parenting style characterizing German mothers in urban settings (Keller et al., 2009; Keller, Lohaus, et al., 2004). These findings are in line with previous work comparing captive chimpanzees and Western humans, which showed that, although face-to-face interactions with mutual gaze events were common in both species throughout the infant’s first year of life, mutual gaze events were more frequent and longer in humans than in chimpanzees (Amici et al., 2023). The importance of FC in several human communities is further supported by cross-species comparisons using eye-tracking, suggesting that adult humans looked longer at others’ faces than chimpanzees (Kano & Tomonaga, 2010). As compared to other species, humans have faces with less hair (Schmidt & Cohn, 2001), white sclera in the eye that enables better detection of eye movements (Kobayashi & Kohshima, 2001), everted lips and a rich repertoire of facial expressions that might draw infants’ attention toward the face (Burrows, 2008), and might represent an evolutionary adaption to better detect faces and the corresponding communicative signals, which might be especially important for human mothers monitoring infants’ behavior (Stern, 1977). However, it is important to emphasize that there is significant variability across human communities, as mothers from many non-Western communities spend less time in FC with their offspring than mothers from Western industrialized societies (Keller et al., 2009; Wörmann et al., 2014).
Regarding longitudinal changes in FC, its increase was evident as the main effect across all genera, but it failed to reach significance for each genus. Therefore, it is not clear whether all genera included in our study increase FC as a result of decreasing BC when infants gradually improve their locomotive skills and spend more time away from their mothers (Adolph & Franchak, 2017). In humans, mothers may rather increase the narrative envelope, with linguistic communication becoming increasingly more important. Moreover, the frequency of FC may vary across cultural contexts, following different developmental patterns. For example, an increase in FC was not found in rural non-Western communities, where mothers often carry their young infants in contrast to more distal caring routines in Western communities (Scheidecker, 2017; Wörmann et al., 2014). In apes, the probability of FC did not vary much over time. This seems to contradict previous research reporting that chimpanzee mothers frequently gazed at their infants (Bard, 1994) and that the gazing rate increased with decreasing BC in the infants’ first months of life (Okamoto-Barth et al., 2007). However, these differences might be explained by different coding procedures. In the present study, FC was only coded when it lasted at least 5 s, following the coding procedures for human FCs (e.g., Keller et al., 2009). However, chimpanzees process faces differently, scanning the whole face faster (Kano & Tomonaga, 2010) and engaging in shorter mutual gaze events (Amici et al., 2023). Therefore, the current coding procedure might not have captured FCs with very brief durations in apes, underestimating the importance of this system.
Finally, OS was only observed in humans, where it significantly increased in the first 6 months. The manipulation of objects is common and quite sophisticated in apes, including small apes (e.g., Cunningham et al., 2006), and it has been frequently observed during mother-infant interactions in humans and apes (e.g., Bakeman et al., 1990; Cunningham et al., 2006; Takeshita & Walraven, 1996). However, only human mothers seem to use objects to attract their infants’ attention. Human infants learn to not to explore objects in their familiar environment but also to practice the intended use of culturally created objects (Keller, Lohaus, et al., 2004). In some communities, including Germany, mothers are highly motivated to train their infants’ ability to handle objects from an early age on, frequently functioning as a substitute for unmediated dyadic exchanges (Keller & Greenfield, 2000; Keller, Lohaus, et al., 2004; Tomasello et al., 2005).
Overall, our study shows that mother-infant interactions partially differed between humans and apes, both qualitatively (as humans uniquely incorporate objects into interactions with their infants) and quantitatively (as humans more likely engaged in BC and FC, as compared to apes). These findings would suggest systematic differences between mother–infant interactions in humans as compared to apes, with differences across ape genera being instead less pronounced. However, while for each ape genus we included samples from different locations, the human sample only included Germans from urban areas. However, previous cross-cultural studies using the component model of parenting have clearly shown that human parenting styles and mother-infant-interactions show considerable variability across communities, depending on the specific cultural values and socialization goals (Kärtner et al., 2013; Keller et al., 2006; Keller, Lohaus, et al., 2004). For example, the rural-living Cameroonian Nso spend more time in BC with their infants and less time in FC, as compared to German mothers (Keller et al., 2009; Keller, Yovsi, et al., 2004). For depicting the obvious variety of mother–infant interactions in different human cultural communities, future studies should include mother–infant dyads from different cultural contexts to investigate if and how parenting systems differ systematically between apes and humans.
Moreover, our study included a very limited sample size, which clearly cautions against the generalizability of our results, across, and within human cultural communities. Even within the same cultural community, for instance, there may be important variations linked to different socioeconomic factors, like maternal education, income, and occupation, and these might clearly affect how mothers relate to their infants. Taking these factors into account will thus be necessary to ensure the generalizability of our findings, which at the moment have to be considered preliminary. Thus, rather than proposing a systematic difference between humans and nonhuman primates, we suggest that within the human species parenting is characterized by much more variability than within and possibly between ape species. To support such a strong claim, however, future studies will need to further include humans with different socioeconomic status and from different cultural backgrounds (e.g., from cultures that are expected to use a more proximal parenting style) and more populations of apes, including individuals from wild and captive settings. This will enable us to study the extent of variability in mothers’ interactions with their infants both within and between primate species in a more exhaustive and systematic manner. In line with this, this study combined a cross-species perspective and a longitudinal approach to compare developmental patterns across species (Rosati et al., 2014), but this approach came with certain costs. Most importantly, we were only able to follow a limited number of individuals over their first year of life, and for some of them, it was not possible to collect data at each of the three ages. Thus, it is possible that larger sample sizes, possibly also including other ape species like gorillas and orangutans, might reveal higher variability within and between ape genera. Finally, there are many individuals other than mothers who are crucial during infants’ development, especially in species with extensive cooperative breeding like humans (Hrdy, 1999). Exploring their roles and behavior from a strictly comparative perspective would also be highly informative (Hrdy, 2009).
Despite these limitations, using the component model of human parenting with a comparative and longitudinal approach allowed us to demonstrate that humans and other primates share at least three systems of parenting behavior (BC, BS, and FC), whereas OS was only present in humans. Possible qualitative and quantitative differences between humans and other ape genera will need to be confirmed by further studies, not only including a greater diversity of primate species but also more human communities, to get a better understanding of the extent of variability in parenting behavior between and within nonhuman primate species.
Supplemental Material
sj-docx-1-jbd-10.1177_01650254241255675 – Supplemental material for Applying the human component model of parenting to other primates: Developmental patterns of mother-child interactions across primate species
Supplemental material, sj-docx-1-jbd-10.1177_01650254241255675 for Applying the human component model of parenting to other primates: Developmental patterns of mother-child interactions across primate species by Katja Liebal, Manuela Ersson-Lembeck, Federica Amici, Martin Schultze and Manfred Holodynski in International Journal of Behavioral Development
Footnotes
Acknowledgements
The authors are especially grateful to the zoo directors, curators, and keepers at Leipzig Zoo, Tierpark Berlin, Tierpark Hellabrunn, Tiergarten Wilhelma, Zoo Osnabrück, Tierpark Jaderberg, Tiergarten Ulm, Zoo Eberswalde, Zoo Zürich, Zoo Mulhouse, Dyrepark Kristiansand, Randers Regneskov, Parken Zoo, Burger’s Zoo Arnhem, Twycross Zoo, Belfast Zoo, Drusillas Park, and Noah’s Ark Farm. They are also grateful to all the mothers and babies for participating in the study. They also thank Christel Schneider, Josep Call, and Bridget Waller for providing access to additional video footage. They are grateful to the research assistants Laura Thomsen, Suska Nolte, and Sonja Ebel for supporting our data collection and to Diana Griebsch for reliability coding. They are finally also very grateful for the highly constructive comments of the editor and two anonymous reviewers on an earlier version of this manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Deutsche Forschungsgemeinschaft (DFG) within the Excellence initiative Languages of Emotion.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
