Abstract
Amid the push for decarbonization and the rise of lithium-ion batteries, global demand for lithium urges an examination of its materiality. Drawing on Barry's chemical geography, which gathers various concerns related to the study of chemicals in the field, and Bachelard's meta-chemical proposal, which challenges a substantialist understanding of chemicals, we propose an experimental alliance between a chemist and an anthropologist concerned with different ways of problematizing lithium's materiality. Guided by a commitment to Latin American territories and embracing a slow science ethos, we seek to foster a sense of responsibility rooted in the material genealogy of chemical substances. Through ethnographic analysis of lithium extraction practices in the Salar de Atacama, Chile, and examination of lithium behaviors in materials chemistry laboratories in Argentina and Europe, we establish a partial connection between lithium chemical labs and underground ancestral lithium brines. Ultimately, we envision futures that acknowledge the ancestral origins of Latin American undergrounds, resisting the univocity of a future-oriented, battery-ion age. In so doing, we endeavor to cultivate a mode of attention concerned with place and deep-time materiality, challenging lineal illusions of progress while embracing the complexities of our planetary present and past.
The adoption of electrical energy storage, particularly in the form of lithium-ion batteries, is steadily increasing on a warming planet still waiting to be decarbonized. In a historical moment in which one size future-oriented responses to global warming aim to create an electric-battery-powered future on a global scale, and where achieving net zero emissions globally by 2050 “requires up to six times more mineral inputs in 2050 than today” (IEA 2021, 271), lithium demand is growing exponentially: in 2022 demand outpaced supply, notwithstanding the 180 percent increase in production since 2017 (IEA 2023). In narrative terms, this is like saying that the global thirst for lithium, driven by electric vehicle sales, is exponentially unquenchable: the quantity of lithium extracted and produced does not satisfy global demand.
Through our experimental alliance between a Chilean psychologist and anthropologist (Cristóbal) and an Argentinian chemist (Martina), in this article we show that lithium, as a chemical substance, is not only deeply entangled with the history of the planet, but also with technical processes, procedures and situated concerns which differently problematize its materiality. We explore, in a situated way, how different ways of problematizing the materiality of lithium make it possible to envision different futures stemming from different stabilizations of the past. Inspired by Donna Haraway's (2016, 12) insistence that “it matters what matters we use to think other matters with,” we argue that it matters what lithium matters we use to think other futures with. We suggest thinking of lithium as an ancestral material from the deep-time Andean underground, which helps us imagine an “ancestral future” rooted in the Earth's past. Drawing upon anthropologist Marina Weinberg's (2023, 12) concerns with lithium extractivist grammars, we emphasize that it matters which matters “are chosen, romanticized, recovered, cared [for], and even repaired, in order to give the illusion of a clean energy transition.” In a historical moment when “chemical compositions of atmospheres, landscapes, and bodies have become critical sites for politics, government, and everyday experience” (Barry 2017, 13) and a view of the Earth as a planetary system is becoming more and more central (Clark and Szerszynski 2021), we write this paper as an experiment in creating alliances between our disciplines of chemistry and anthropology, which tend to remain isolated within their niches. Thus, we write to practice a responsible “disciplinary alliance in the Anthropocene” (Blok and Jensen 2019).
To orient the reader to our experimental alliance, we begin by outlining the stories and situations that motivated this collaborative work. From an anthropological perspective, Cristóbal has been researching lithium extractivist practices—linked to globalizing techno-optimist projects aimed at decarbonizing the planet—for more than eight years. Since 2020, thanks to funding from the European Research Council, Cristóbal has assembled a team of anthropologists, who have been studying the technological replacement of combustion engines with lithium-ion batteries. 1 Together they traced lithium from extraction sites in Chile, one of the largest lithium producers; to widespread usage of lithium-ion batteries in the transportation sector in China, a leading lithium-ion battery producer; and to recycling practices, materials and industries for spent lithium-ion batteries in Norway, a country that is at the forefront of electric vehicle adoption. In this article, we focus on Cristóbal's ethnographic work in Chile, the author's home country.
Since 2017, Martina has been immersed in lithium research primarily in Argentina, working as a chemist while engaging with social scientists in interdisciplinary collaborations. Her current work is carried out at a scientific institute affiliated with CONICET (the National Scientific and Technical Research Council in Argentina) and the National University of La Plata. Here, Martina has studied lithium-conducting materials, collaborating with specialized groups in electrochemistry evaluating these materials in lithium-ion batteries. Additionally, Martina is a member of the Geopolitics and Common Goods Study Group 2 (Sp. Geocomunes), an Argentinian collective of researchers dedicated to investigating geopolitical lithium dynamics and socio-ecological transition processes, particularly in South America. Since 2020, she has been part of the Interuniversity Forum of Lithium Specialists of Argentina, 3 which brings together thirty lines of research with the aim of creating interdisciplinary collaborations, organizing conferences, generating educational material, intervening in legislative projects, and so on. Our collaboration initially arose from Cristóbal's engagement with the Geocomunes group, and his interest in learning about the chemical practices that Martina was developing within chemical laboratories.
That both our research agendas are closely linked to our biographies and to Latin American territories where lithium is being extracted, is not a coincidence. On the contrary, our profound sense of belonging to these territories, where lithium extraction generates often-disruptive social and material transformations (Bonelli and Dorador 2021; Fornillo and Gamba 2019), was one of the main motivations that led us to work together and to attempt to articulate, and partially connect, the different sensibilities and methodologies inherent in our disciplinary practices. And as we are both Latin American, it would be a betrayal not to explicitly consider the longstanding existing entanglements between scientific practice, the production of knowledge and our continent's violent history of colonialism.
Thinking about the materiality of lithium from a Latin American perspective also means inheriting a specific history of colonization: of how Europeans imported into Latin America a type of human agency capable of moving immobile things and re-channeling underground and surface flows on a large scale, transporting them across continents. 4 This large-scale Earth-transformation triggered by the colonial extraction of “natural resources” from the region (see Galeano 1971) has not ceased (see Gudynas 2018; Svampa 2019). We write this paper in a historical moment in which lithium is enacted as a critical mineral for the energy transition, and when, according to the US Geological Survey, about 67 percent of the world's lithium reserves, supplying about half of world's demand, are situated in the so-called lithium triangle formed by Argentina, Bolivia, and Chile (López-Calva 2022). This article expands on our earlier work on lithium extractivism in South America within our respective fields (Bonelli and Dorador 2021; Fornillo and Gamba 2019; Weinberg and Bonelli 2021; Zicari, Fornillo and Gamba 2019), and builds upon our longstanding commitment to create alliances with “the power to make concerned people think and act together” (Stengers 2018, 93). More particularly, we want to show how lithium, within an eco-modernist logic tied up with post-fossil capitalism (Fraser 2021; Szeman 2021), is produced as a chemical reagent “stripped of its natural history, the circumstances of its production and the techniques that intervened in its extraction” (Bensaude-Vincent and Simon 2012, 70). Against that background, and responding to the call of this Special Issue of Science, Technology and Human Values on Underground STS, we aim to craft a partial connection (Strathern 2004) between lithium compounds used in chemical labs, and the temporal planetary depths of South American underground brines.
Our alliance is inspired by geographer Andrew Barry's proposal (2017) to practice a “chemical geography” concerned with lithium in the field. For Barry, chemical geography not only rejects disciplinary hierarchies, treating field-based and abstract sciences equally, but it attempts to integrate ethical and political debates within the natural and social sciences, in a historical moment where chemical compositions are central to political and everyday experiences. To conceive and enact this non-reductionist intervention, we have drawn inspiration from Gaston Bachelard's (1968) proposed meta-chemical philosophy, wherein “true chemical substances are the products of technique rather than bodies found in reality” (Bachelard 1968, 45). Thinking with and through this meta-chemical sensibility has also allowed us to further experiment with our anthropological and chemical disciplinary knowledges in non-hierarchical ways, and to think of them as open and unstable products resulting from our methods, techniques, and epistemic traditions.
We build upon Bachelard's (1968, 45) proposal that “the real in chemistry is a realization,” a remark that resonates with Bruno Latour's musings on substances (Nordmann 2006). For Latour (1999, 151, our italics) the word substance “does not designate what ‘remains beneath,’ impervious to history, but what gathers together a multiplicity of agents into a stable and coherent whole…substance is a name that designates the stability of an assemblage.” Our alliance is inspired by this Latourian meta-chemical sensibility and, in particular, by Bachelard's commitment to displacing a metaphysical conception of chemical substances as stable foundations of the real. Indeed, in this paper, we think of lithium as a chemical substance that emerges as a product of technique through specific situated practices and procedures. This does not mean to relativize, from a human-centric perspective, the individual power lithium has to operate on its own terms, according to its affinities, to its “specific powers to decompose and enter into new compositions” (Stengers 2021, 23). Our aim is to show that there are different ways to problematize lithium's materiality, and that these different problematizations not only have profound implications on how we think of our possible futures, but also on the type of responses that we can generate in our engagement with lithium materialities and their interdependences. 5
Following from this, we wish to problematize how the thirst for lithium “stabilizes lithium” in the Latourian sense, as an isolated ionic resource without history—like ions uprooted from their planetary history. In doing so, we build upon historian of chemical science Evan Hepler-Smith’s (n.d.) concern with material genealogy and a chemical ethics of responsibility. Our interdisciplinary alliance embraces this in a more-than-chemical way: we are inspired by a slow science (Stengers 2011, 10) that accepts “what is messy not as a defect but as what we have to learn to live and think in and with.” And learning to live and think amid what is messy involves questioning the lineal, future-oriented ideal of technological progress and, instead, embracing the notion of living and dying with “the earth-wide precariousness” (Savransky 2022).
We propose a lithium analytics that builds upon two different meta-chemistries. The first is a future-oriented and uprooted resource metachemistry that stabilizes lithium (sensu Latour) as the lightest metal in the periodic table and, therefore, the most suitable for manufacturing electrochemical devices with the highest energy density. For this metachemistry, lithium atoms are all the same. It is not their material genealogy that matters, but their physicochemical properties: their high oxidation potential, small size, and low atomic mass—all properties that make them the star (i.e., critical, strategic) element in energy storage technologies. Its location in Mendeleev's Periodic Table gives it a metallic luster that we do not see in Earth, since lithium is never found in pure form. On the contrary, under normal conditions, lithium tends to lose an electron (forming an ion) and to exist relationally in combination with other elements.
Second we propose a deep-time meta-chemistry that allows us to think of lithium as an ancestral material, a deep-time material of the underground—in our case, Andean— realms. 6 Our work is inspired by Hepler-Smith’s (n.d.) material genealogy of chemical substance, and by indigenous writer and philosopher Ailton Krenak (2022). To speak about deep-time meta-chemistry allows us to imagine an “ancestral future” that relies on the Earth's past. In what follows, we show that these meta-chemistries express themselves in different ways in the different lithium worlds where we work, specifically through our ethnographic and chemical work in lithium extraction sites and lithium chemical labs.
We show how our transdisciplinary slow alliance can potentially contribute to thinking up futures from underground ancestral pasts. If the future can be imagined, it is ancestral, for it already permeates the deep-time of present underground brines. We aim to generate a transdisciplinary responsibility that critically questions the universal vision of a future-oriented, battery-ion age, wherein electrical energy storage in the form of lithium-ion batteries is considered indispensable for achieving net zero emissions.
The first section of the article is set in the Salar de Atacama (Atacama Salt Flat) in northern Chile, where Cristóbal has conducted ethnographic research and lived intermittently since 2015. We offer an account of the extraction of underground brines, illustrating how the extractive industries in this region mobilize a future-oriented resource meta-chemistry that treats these brines as a “chemical deposit box” devoid of a past. In doing so, we demonstrate how deep-time meta-chemistry is transformed or rendered implicit by the uprooted resource meta-chemistry inherent in extractivist grammars and practices.
In the second section we show how these meta-chemistries are also expressed (in different ways) in chemistry labs, located in Argentina and Europe, where Martina has worked. In certain laboratories, the objective categories that guide scientific practice are, to use Stengers’s (2018, 117) words, “the very ones which are relevant for industrial development, since both agree about ignoring the same type of complications.” In the context of lithium chemical research, this means working with materials that can improve batteries’ performance and discarding those that resist or complicate this ideal. In other laboratories—especially in public university settings—there is an opportunity for scientific practice to work, learn and co-exist with those substances that fast science and industry interests would normally discard.
Underground Brines: The Case of the Salar de Atacama in Northern Chile
More than half of the world's identified lithium deposits are found in the arid, other-worldly landscapes of South America's high-altitude salt lakes, straddling the Andean mountains range of Chile, Argentina, and Bolivia. Although lithium is abundant in other parts of the planet, there are economic advantages to mining it in South America: extracting it from underground brines in salt lakes is far cheaper than mining the hard-rock deposits found in places like Australia. As such, the consolidation of lithium-based energy storage technologies is leading to more and more controversial mining projects in the region (Blair et al. 2022; Jerez, Garcés and Torres 2021).
Over the past eight years, Cristóbal has made several visits to a place called Aguas de Quelana, at the edge of the Salar de Atacama, where one of the biggest lithium deposits in the region is located. This place is known among locals for its lagoons and as a breeding ground for flamingos. Activists, geologists, Indigenous Atacameños, and researchers all agree that to see the impact of lithium mining in the Atacama, you must look at the lake systems on the eastern edges of the Salar. This was confirmed by an inhabitant of the Salar, who we will call Felipe, who, in a conversation with Cristóbal in March 2017, noted that the freshwater “that came down subterraneously from the mountain range, and that arrived here forming lagoons, is no longer there.” After pointing out a freshwater well from the nearest lithium industrial site in Aguas de Quelana, Felipe added: “we are clear about what happened…these lagoons have been dried up by the lithium mining industries…the people of the Salar are very worried because as the water levels go down, the flamingos are leaving…meaning the tourists who generate income for the local population could stop coming.” Felipe associates the extraction of lithium from the salt flats with the disappearance of Quelana's lagoons. Leaving aside these dynamics, it is important to note that other aquatic ecosystems on the edge of the Salar de Atacama, such as La Punta and La Brava lagoons, have also been affected by brine extraction. This environmental damage is currently part of a lawsuit brought by the Chilean State Defense Council against three mining companies seeking reparations for the continued extraction of water resources (Figure 1). 7
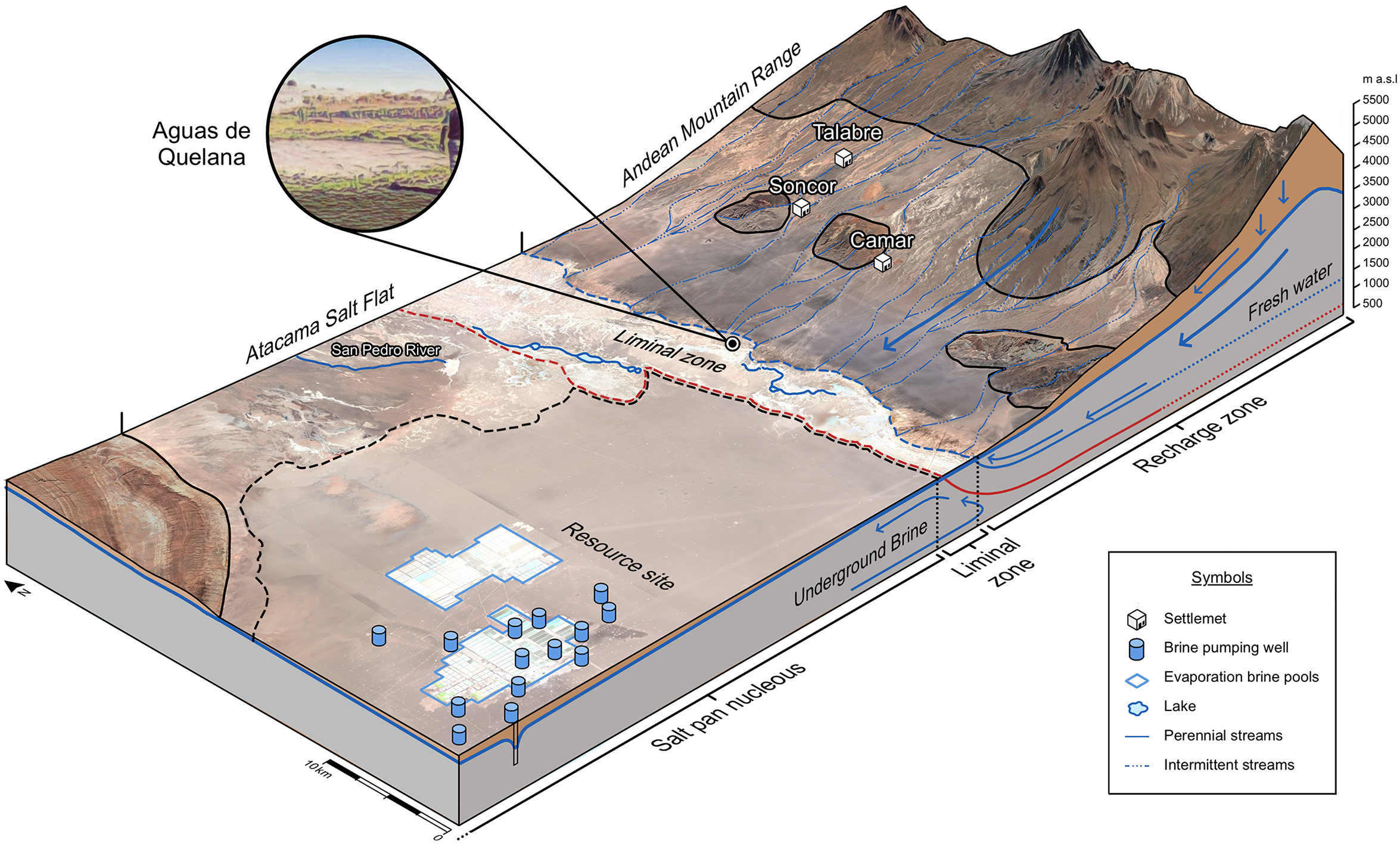
Illustration of Atacama Salt Pan.
In hydrogeological terms, these lagoons emerge where freshwater coming from the mountain range meets underground saltwater from the Salar. The abrupt slope in this area and the collision of freshwater and dense saltwater causes these lake systems to emerge, generating a border zone between the freshwater and saltwater of the Salar. These subterranean flows were an inherent part of the ancient processes that formed the Salar. The geological temporal scales are difficult to imagine: mountain water gradually infiltrated porous land over millions of years, converging in a closed basin that was once below sea level. Today, this region is more than 2,000 meters above sea level, has very low precipitation rates, is extremely arid, and is formed by an endorheic (closed) basin where the little available water flows toward the central area, which is topographically the lowest. As it descended over time, the mountain water flowed toward the deeper parts, dissolving elements that modern chemistry calls sodium, potassium, lithium, among others. These ions travelled diluted in the water to the bottom of the basin, dissolving and precipitating over millions of years to form these evaporite systems. This millennia-long descent has favored the accumulation of underground brines in the subsoil of the Salar de Atacama, and in the Altiplanic Puna of Argentina.
Moreover, the Aguas de Quelana, where Felipe took Cristóbal in 2017, is a liminal zone between the indigenous Atacameños territory and what geologists call the nucleus of the salt flat, from where the underground brines with a high concentration of lithium are currently pumped out. Determining the (presumed) permeability or impermeability of this zone—which defines whether lithium underground brines are connected with the Quelana lagoons—is crucial to adjudicating responsibility for environmental damage. This is a zone that configures something akin to what geographer Nigel Clark (2017) has called “strata-relations,” where underground geophysical worlds converge with human worlds on the surface, and where geological strata themselves (transformed by extractive activities) are triggers for political controversy. But how these geological and human worlds converge depends on the knotted social and political interests and concerns at stake (Greim, Solomon and Breyer 2020; Latour 2004). In fact, since Felipe shared his thoughts on the Quelana lagoons in March 2017, this liminal zone has become a pole of unusual attraction for European and American researchers who visit Atacama for a few weeks—or even days—looking for “fast evidence” they can mobilize in their future publications about lithium extraction.
An example: in March 2023, a team of Nordic European researchers requested a meeting with Cristóbal and his local collaborators, a team of Atacama resident academics working for a Chilean university. Their objective? To ask for help in obtaining chemical samples of the Salar waters to be analyzed in Europe by chemists and geologists interested in the environmental deterioration of the Salar due to lithium extraction. In that meeting, Cristóbal and his collaborators tried to explain that obtaining a sample in indigenous Atacameño territory could take years, because it depended on the authorization of the Council of Atacameño Peoples, the communities involved and their interests. 8 The geologist, however, was reluctant to understand this logic, arguing that her object of study “was the whole planet,” and that her results could have a “global” impact. Just when the conversation was getting interesting, as Cristóbal and their team of collaborators problematized this premise, humorously inviting the geologist to consider studying energy transition controversies in her own country, the geologist asked to be excused, and left. The reason for her hasty departure was that their assistants had contacted an Atacameño inhabitant who knew the waters of Quelana, and had, for a price, agreed to take them to the requested place. Strata relations, therefore, are not immune to the fast science tendency driven by the thirst for lithium, in this case within academia.
We’re interested here in exploring how resource meta-chemistry operates at industrial scales. Indeed, the lithium industries refer to the nucleus of the Salar as the “resource site,” different to the “conversion site,” where lithium is treated to produce chemical compounds, such as lithium carbonate or lithium hydroxide, and also different to the “off-site,” an empirical and conceptual place defined by extractive industries existing “beyond the economic and productive realms” (see Weinberg 2023). What concerns us here is the resource site, a deep underground deposit where various salts lie dissolved underwater, and complex relations of ions lie under the surface, awaiting to be pumped out, and eventually, isolated, or realized into (in Bachelard's terminology) discrete and uprooted ionic compounds. 9
The grammar of the so-called resource site functions through particular technologies and definitions. Diamond drilling machines, diesel-powered brine pumping wells reaching as deep as 200 meters below ground, and brine pools that receive brine from the wells, are used to put the salt pan “to work” (Daggett 2019), enacting the salt pan aquifer as a “chemical deposit box” consisting of the basin and its subterranean flows. From this box, underground brine is extracted as resource. The technologies used to extract lithium-as-resource also include well-pools that receive brine with a higher concentration of lithium and leave behind salt crops (bischofite, halite, lithium carnallite, and sylvinite, among others); connecting pipes between wells; conceptual and numerical models fed by stratigraphic mapping; and other hydrogeological analysis tools (Figure 2).
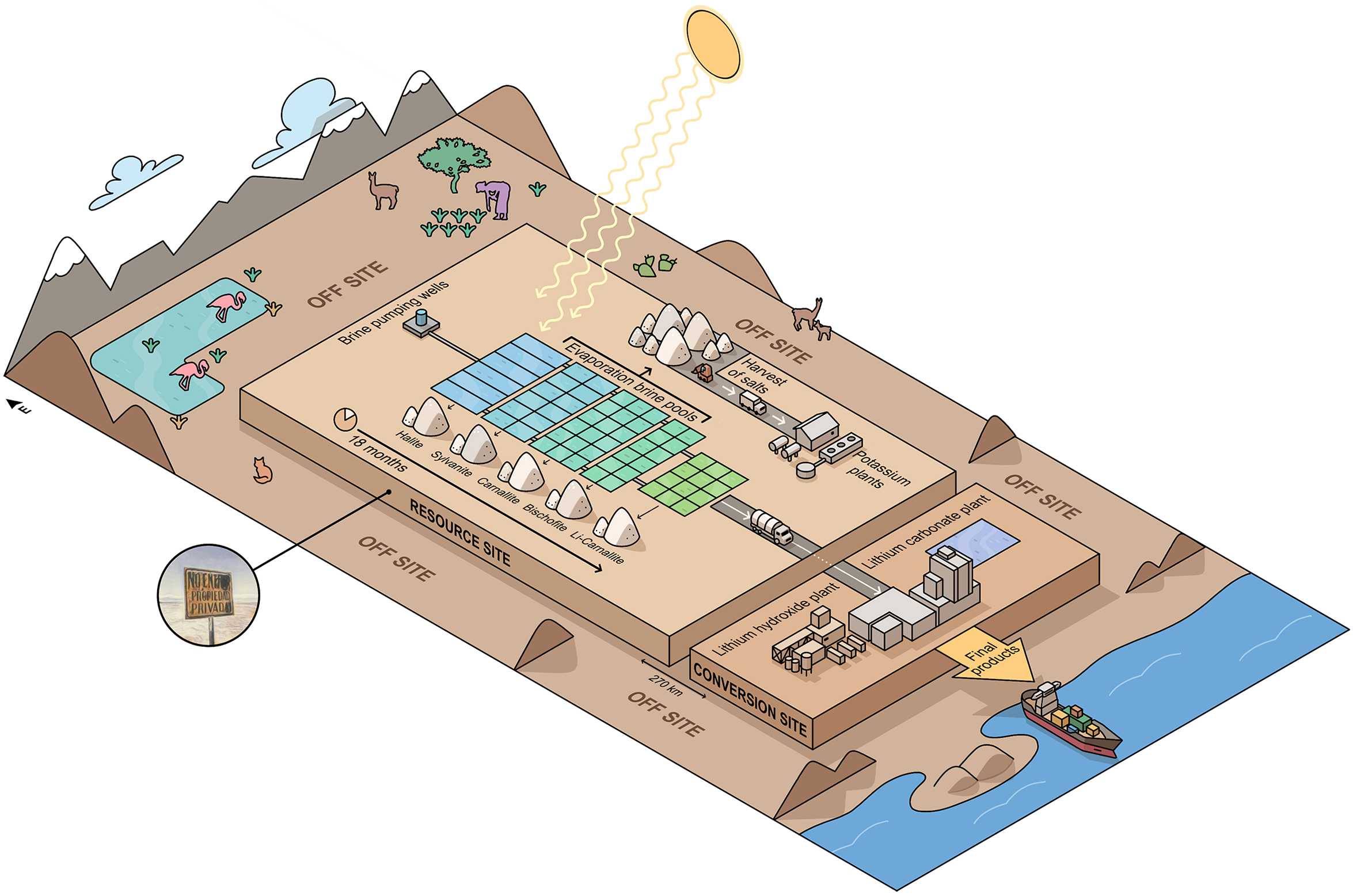
Illustration of the lithium extractive grammar.
All these techniques are intertwined with definitions of the extraction process as “natural” because it uses solar energy that “naturally” evaporates water, as well as working with gravity to precipitate heavier salts. 10 Crucial to the grammar of lithium extraction is the definition of underground brine as a fluid that is not water, a definition based on the ISO standard and classifications. According to this standard, freshwater has a total dissolved solids content of 5 milligrams per liter, while brine from the Salar de Atacama has more than 300 milligrams per liter. Lithium thirst appears here as a thirst for ions—not for water.
In the words of a sustainability professional working in one of the Salar's lithium industries: The debate over whether brine qualifies as water is ongoing. According to ISO standards, water is defined by its total dissolved solids: freshwater contains 5 milligrams per liter, seawater 35, and Salar lagoons range from 30 to 150. So, we don’t consider the salt flat to be water…even though, yes, basically, [extraction] can affect the dynamics of the water resource within the Salar basin in some way because it does have an impact on the water levels in the nucleus, but we are not reducing the amount of water available for human consumption or for agriculture. Because—the truth is—brine is an aqueous solution but it is very different from water. Nobody is going to drink brine, and nobody is going to irrigate their plants and crops with brine. (Interview with Cristóbal 2021)
The nucleus of the Salar is transformed into a “harvest” of salts (cosecha de sales) that precipitate as the underground brines evaporate in the pools after being pumped. When the brine reaches a higher concentration of lithium, it is transported in tanker trucks to the “conversion site,” where the boron, magnesium, and calcium in these brines will be separated. We seek to reveal here how the industry disconnects the nucleus of the Salar and human communities to occupy the “chemical resource site:” the brine underground non-water, undrinkable, whose mobility is controllable and, therefore, extractable, is separated from the ancestral territory of human settlements that need water to live. Thus, the industry establishes the stability of the extracted underground brine as possessing economic value, as an object of property rights.
The practices and infrastructures dedicated to extracting underground brines, harvesting salts, and generating lithium-rich brines can be thought of as a mechanism for making deep-time meta-chemistry implicit (sensu Hepler-Smith). For resource meta-chemistry, how deep-time lithium affinities configure South American underground brines within wider, Earth-forming evolutionary processes, does not matter at all.
Undergrounds Lithium Brines: A Deep-Time Metachemical Realization
To imagine how lithium ended up under the surface, entailing deep-time geological and evolutive processes, we build upon the work of Norwegian mineralogist Victor Goldschmidt, who in the first half of the twentieth century systematically visualized what nineteenth century chemists had baptized “the discipline of geochemistry.” As Andrew Barry (2017, 8) recalls, “CF Schönbein, Professor of Chemistry in Basel, first used the term in 1838. Subsequently Christoph Bischof, Professor of chemistry and technology at the University of Bonn…came to regard the Earth as ‘a vast chemical laboratory.’”
It is this understanding of the Earth as chemical laboratory that Goldschmidt developed to understand and predict the distribution of elements on Earth. This helps us to imagine a deep-time geo-meta-chemistry capable of understanding the presence of lithium in underground brines. Goldschmidt was one of the first to extend Mendeleev's classification of the elements to account for the immense variety and complexity of the Earth's materials (Vance and Little 2019). In his monumental textbook on Geochemistry…Goldschmidt defined Geochemistry as a field that was fundamentally concerned with the verteilung [the distribution] of the elements: “Modern geochemistry studies the distribution and amounts of chemical elements in the minerals, ores, rocks, soils, waters, and the atmosphere, and the circulation of elements in nature, on the basis of the properties of their atoms and ions.” (Goldschmidt 1954, 1, in Barry 2017, 9)
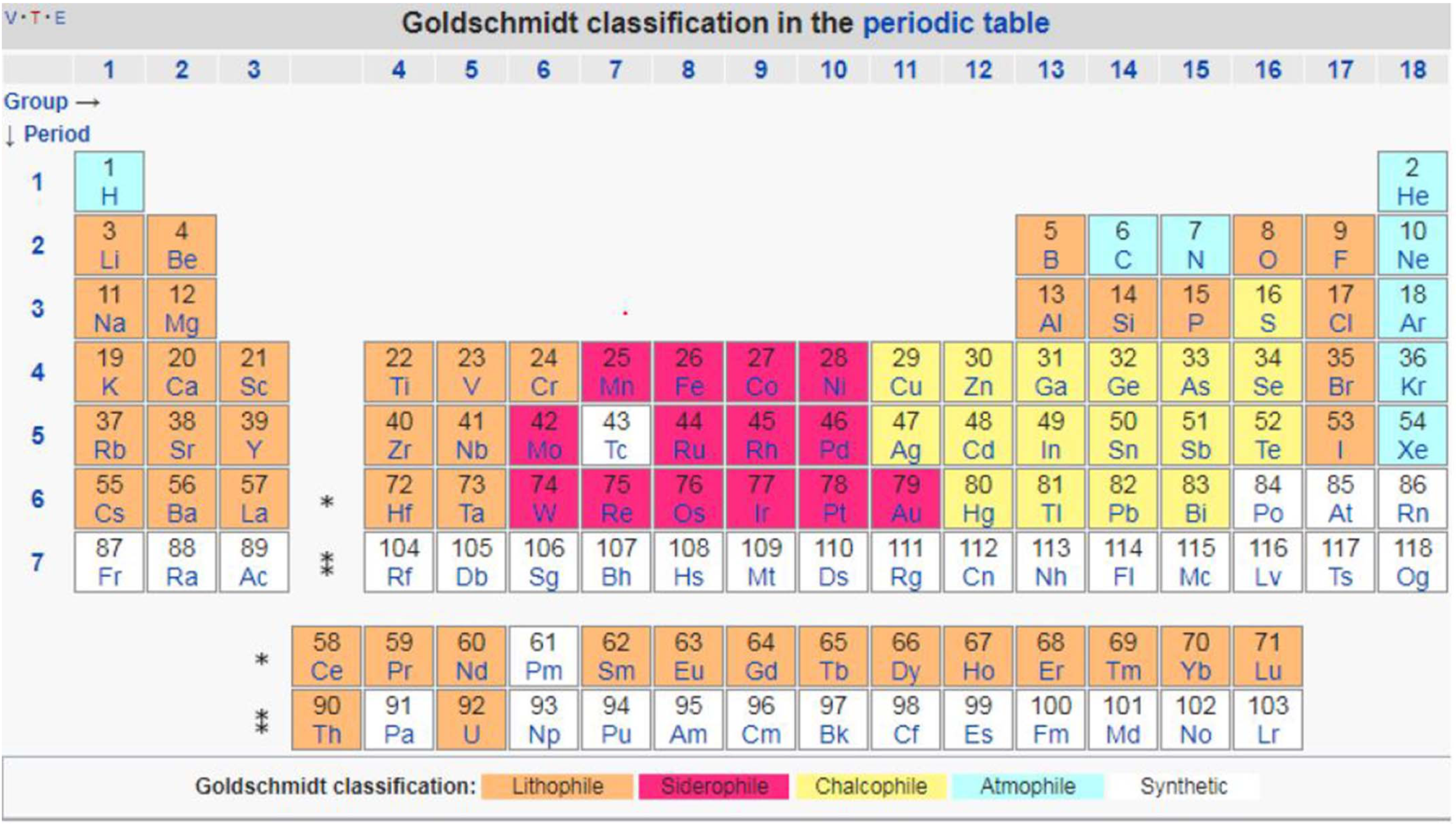
Goldschmidt classification of the periodic table. Downloaded from: https://lms.su.edu.pk › download.
To explain how the elements were distributed in the different layers that today make up the Earth, Goldschmidt identifies different “stages” in the planet's history (Vance and Little 2019). Affinity properties govern the first stage of geochemical evolution, while “ionic potential” (the ratio of the elemental charge to the radius) governs the tendency of ions to remain in solid mineral phases, or to dissolve in a liquid medium. Lithophile elements that have low and high ionic potentials tend to form ionic bonds that are easily disrupted by water in weathering processes, solubilizing readily in underground waters. 11 Lithium is in this group. By contrast, lithophile elements with intermediate ionic potentials are often insoluble. The latter are immobile in weathering and tend to be concentrated in soils.
During the development of the underground brines, lithium's affinity for the type of rocks found in Andean basins, coupled with its tendency to solubilize in the magmatic hot springs circulating in the subsoil, made it possible for it to be transported in solution to the aquifers of the Salar's subsoil, where it accumulated. As a result, the lithium present in the region's minerals (such as ignimbrite rocks or smectite clays) is extracted and dissolved by weathering, explaining its presence in high concentrations in the underground brines of extremely arid regions, like the Andean salt flats. In this deep-time meta-geo-chemistry, the processes of weathering, transport of lithium to the salt basin, evaporation, infiltration of the brines and, finally, accumulation in the underground aquifers of the salt flats, occurred over long periods of time—up to several million years, a realization that is ignored in resource meta-chemistry.
In Atacama, we see how the vast chemical laboratory on Earth is being produced as a chemical deposit box in which the initial natural earthly distribution of chemicals, as stated by Goldschmidt, is literally turned inside-out (Hecht 2021). Let's now explore how lithium's individuality is realized far away from these earthly geo-chemical underground labs. Let's dive into how lithium is problematized differently within different chemical labs.
Chemical Labs for Lithium-Based Materials
Co-author Martina works in the Advanced Ceramics Group of the Mineral Resources and Ceramics Technology Center, a public institute in Buenos Aires, Argentina. In 2017, at a time when Cristóbal was meeting Felipe in the salt flats on the other side of the Andes, Martina's research group decided to open a new line of research into lithium-based ceramic materials. Why? The Argentinian government had paid a lot of attention to lithium since 2012, when the National Ministry of Science and Technology began to regard lithium research as “strategic” 12 in the context of a global lithium-ion battery boom. In the Argentinian context, it meant promoting the primary exploitation of the Andean salt flats, and some attempts to link scientific research in the field of lithium batteries with state-led lithium industrialization projects (Fornillo and Gamba 2019).
The group found a way to engage the lithium question by synthesizing materials that we will call LZO, which could be obtained through a chemical reaction between a mineral called zirconia, widely studied in the research group, and lithium carbonate, one of the main reagents used to make battery materials, which is produced in the country. The work of reacting lithium carbonate with zirconia was taken on in 2017 by a postdoctoral fellow we’ll call Pedro. The synthesis method Pedro chose was the solid-state reaction. This involves gently mixing the starting materials (zirconia and lithium carbonate), then heating them at a certain temperature for a specific length of time, giving energy to the system that ultimately transforms the starting materials into LZO.
For a year, Pedro studied lithium carbonate and zirconia through a series of experiments consisting of thermal treatments, examination by microscopy, using lasers to determine particle size, and grinding to decrease particle size and improve reactivity. 13 After finding the optimum grinding time and heat treatment time and temperature, Pedro was able to obtain the LZO and publish his results.
In 2018 Martina was working in an electrochemistry lab at the same university, which had an agreement that enabled her to test materials made by Pedro for use in batteries. Martina found a paper showing that LZO could be used as an “active anode material” (Dong et al. 2015) in a lithium-ion battery. Recall that batteries are basically composed of three elements: cathode, anode, and electrolyte. When the battery is on, lithium ions migrate from the anode to the cathode and, in that process, electrical energy is produced.
To test the LZO as an anode, Martina followed the standard procedure: she mixed the powder with a conductive agent (carbon) and a binder (a polymer used in the electrode formulation) into an organic solvent to obtain a thick, black paint, which she applied to a copper foil. When the paint dried, she cut the sheet into circles 1 cm in diameter, to be used as anodes in the test battery. These anodes went into a so-called glove box, a workspace without oxygen or water, where the test battery cell is assembled. Assembling a battery cell involves placing the working electrode (copper painted with LZO) in contact with the separator (a porous plastic moistened with electrolyte) and the counter electrode (in this case lithium metal), in a sandwich structure.
Martina performed several tests simultaneously under different conditions, to learn about the properties of this material and how much energy it could generate. But, from the start, the LZO generated very little power—only about 40 mAhg−1, less than half of what was reported in the paper she was working from.
Months later, in 2019, Martina decided to continue investigating the material. In the literature, she found that partially changing the zirconium ion (Zr4+) for another element could facilitate lithium transit and improve its behavior in batteries. Looking at Mendeleev's Periodic Table to see which atom could work, she realized it had to be smaller and have less charge than the Zr4+ ion. Before the pandemic prevented laboratory access in March 2020, she succeeded in obtaining a pure LZO-based material with a slightly different composition, in powder form.
Returning to laboratory work in 2021, Martina set out to evaluate whether the modified material improved ionic conductivity compared to the original. To do this, she created solid ceramic samples from the powder and studied their electrical properties. She used the pure synthesized powder to press “tablets” of 1 cm by 1.5 mm (Figure 4), and placed them in an oven at 1050 °C.

Ceramics sample. Photo by Martina Gamba.
Martina then traveled to a laboratory in the south of Buenos Aires to learn how to “test” these samples. By heating them to a specified temperature and applying an alternating current, it's possible to measure the material's response to changing temperatures. Mathematical analysis of the data allows chemists to determine whether the ceramic tablet exhibits electrical conductivity or acts as an insulator. As these tests are carried out with temperature, a preliminary study must be conducted to understand the material's response to heating. The ideal—the expectation—is that no changes occur. What could 300 °C do to a material that, during its preparation, has spent 12 h in an oven at 1050 degrees?
Imagine Martina's (unhappy) surprise when the tablet appeared to be losing mass as it was heated. In other words, since its manufacture a month before, something in the material had changed state—something had taken over the ceramic. To verify that this loss of mass was related to the time passed since its manufacture, Martina took a material with a similar composition and preparation but obtained a year earlier, and found the loss of mass had increased. In other words, aging had triggered the decomposition of the ceramic material. Martina and her colleagues were able to carry out conductivity measurements, but their interpretation remains under discussion due to the material's curious behavior. Far from writing the results off as unreliable, they decided to carry out a systematic study to find out the mechanism through which the sample changed.
They found that the samples’ loss of mass began in the first week after manufacture. How was the material affected? The passing of time itself is not a reactant—it does not modify a material. Rather, the passing of time is an enabling condition for the samples, stored in a laboratory cupboard, to be affected by the environment. And the environment, in this case, is the Argentinian city of La Plata where average air humidity exceeds 70 percent: this humid air is a mixture of nitrogen, oxygen, carbon dioxide, and water in its gaseous phase, among other gases.
In Martina's material, lithium first forms part of a crystalline structure. Martina's hypothesis is that, over time, it diffuses toward the edges of the grains that make up the ceramic and reacts with water in the environment. Lithium's behavior in this material evokes its tendency (like other lithophilic elements with low ionic potentials), as demonstrated by Goldschmidt, to form ionic bonds that “are readily disrupted by water,” explaining its high concentration in seawaters and underground brines (Vance and Little 2019). Including lithium in ceramic materials introduces a tendency for these ceramics to react with water vapor, mirroring the natural solubility of lithium ions in water. Over time, exposure to water vapor can degrade the ceramic structure as the lithium ions within the ceramic matrix seek to hydrate and mobilize. LZO decomposes (modifies its chemical composition) when exposed to humid air: from the perspective and interests of fast science, this materials show “a complication” that must be ignored.
In 2022 Martina began a laboratory placement in a ceramic materials research group in Northern Europe. The aim of the visit was to synthesize a family of solid electrolyte compounds for solid-state lithium batteries. On the first day of her research stay, she was received by Dr. U, the chief investigator, who was already familiar with Martina's work in Argentina. Within their first few interactions, Dr. U noted: “we’re not going to talk about your Argentinian materials on the first day, so as to avoid starting your stay on a negative note.” As a result, during her placement, although Martina gave two seminars for the group and one for the institute, she never spoke about her work in Argentina.
At that time, Martina did not know what Dr. U's comment meant. Writing this article a year on from the comment, Martina thinks it could be related to the materials’ instability in air, or the idea that their performance as a material out-side lithium-ion batteries is not worth “wasting her time on” Either way, Stengers’s (2011, 4) proposal for practicing slow science, suggested by Cristóbal when he first heard this story, resonated strongly with Martina giving a retrospective account of her experience with Dr. U: “For many scientific researchers, to slow down and lose ones’ time with questions that do not directly contribute to the immediate and evaluable progress of their field is even something akin to a sin—to a temptation a true scientist knows he has to resist.” Echoing the European geologist's haste to obtain a Salar water sample without considering the Atacama's political history, Dr. U had no time to slow down. She had no time to embrace the potential of slow science to foster a profound reconnection between the chemical practices in a material laboratory with broader deep-time geodynamics. By allowing “time to speak” in the LZO study, and accepting the invitation to embrace the “messiness” of our world that the interdisciplinary dialogue introduced, Martina was able to observe and interpret lithium's individual behavior—although far from the lithium brines—within an environmental context. The slow, inherently complex process of interdisciplinary dialogue allowed her to link chemical behaviors with their geological roots, but Dr. U had no time for all this messiness.
A year on, Martina can see how a scientific logic centered on performance reinforces resource meta-chemistry. Traditional reviews and articles in the field of material science or electrochemistry often overlook the material genealogy of these ions, treating raw materials or starting reactants as historical blank slates, and attempting to stabilize “lithium” as a future-oriented and uprooted resource. Within this logic, if the materials do not work, there is no time to understand why: materials are discarded so no other scientist will ever know about it, and new materials will be evaluated until one responds to what is demanded.
Some months after returning from the research placement, Martina presented the results of her work on LZO decomposition at a meeting of the Argentinian Crystallographic Association. To her surprise, the paper was selected to be presented orally, and not in poster format. The questions she feared most were from colleagues involved with resource meta-chemistry and fast science, along the lines of why waste time and resources studying the decomposition of a material? Yet she was pleasantly surprised when she received comments that enriched her work and offers for future collaboration to understand the remaining questions. These questions are currently specific to the field of chemistry, but their very relevance emerged from the collaboration with Cristóbal: How do lithium ions move within the structure? What parameters affect the speed of the process? Can it be reversed, and under what conditions? “Beyond the objective categories that are relevant for industrial development” (Stengers 2018, 117), Martina was able to appreciate the potential for more questions to arise through interdisciplinary dialogue. Such emergent line of questioning can reconnect chemical materials to their deep-time roots and, through these pathways, generate new senses of responsibility, rather than the efforts (technological, energetic, material) to “stabilize” a chemical substance uprooted and removed from its history.
Presenting her results to the scientific community, Martina “discovered” that it is possible to “do” science without responding to the categories imposed by fast science. Combined with her interdisciplinary dialogue with Cristóbal, the public sector work context created the conditions for Martina to reflect on her experimentation with lithium and undertake one of slow science's central challenges: “to accept what is messy not as a defect, but as what we have to learn to live and think in and with” (Stengers 2011, 10). 14 In this case, the messiness was how lithium's individuality and geochemical affinity with its surroundings, including oxygen and water, reconnected—at least partially—in the lithium chemical laboratory with the ancestral Andean underground brines.
Toward an Ancestral Future Beyond Fast Substantialism
Inspired by Barry's chemical geography as a performative intervention that “mobilizes or gathers together a set of concerns with the study of chemicals in the field,” and by Bachelard's (1968, 45) meta-chemical proposal and his critical attempt to “disperse the chemical substantialism,” we have sought to problematize lithium's individuality in the salt flats and underground brines where it's extracted and the chemical laboratories where it's used as a reagent. Analytically, we have considered lithium's materiality through two meta-chemical analytics: an uprooted resource meta-chemistry that stabilizes lithium as the most suitable metal for manufacturing energy-dense electrochemical devices, and a deep-time meta-chemistry, which allows us to think of lithium as an ancestral material of the Andean undergrounds. In an Stengerian spirit, we suggest that these meta-chemical analytics can act as conceptual tools to slow down reasoning (Stengers 2018, 114) in a historical moment where the “knowledge economy,” in relation to lithium, seems to dictate “the imperative not to slow down, not to waste time or else.”
We are aware that, as with any analysis, our distinction between meta-chemistries has limitations and conceals risks. One is the illusion that our own disciplinary practices can be associated with one meta-chemistry and not another. While we have associated resource meta-chemistry with specific figures in our story, such as Dr. U and the European geologist, among others, we wish to conclude by explicitly stating that these meta-chemical trends also operate in our roles as university researchers dedicated to understanding and intervening in lithium worlds. In this spirit, we invite our readers to think of Dr. U and the European geologist not as targets of denunciation, but as presences that make perceptible the special power of fast science ruled by epistemic and industrial productivity concerns. We are not immune to that power, which might tempt us to discard that which doesn’t adhere to the material and epistemic industrial production for which lithium's individuality appears solely as a re-agent resource, without considering its ancestral origin, how it was made, and the methods used to extract it.
Against that temptation we have shown how paying attention to different meta-chemistries at stake in problematizing lithium's materiality, can help us to partially connect scientific laboratories to their surroundings, and chemical reagents to their material genealogy, and to the ancestral underground brines. What we have sought to demonstrate is how these connections can be cultivated and “realized,” to borrow again Bachelard's terminology, through an experimental alliance of the coauthors’ anthropological and chemical practices. Our practices are concerned with fostering alliances that empower concerned people to think and act together, facilitating connections among divergent concerns (Stengers 2018, 93), and also with how to construct senses of responsibility in interdisciplinary practice that reconnect us with our Latin American territories.
Practicing a slow, interdisciplinary, experimental alliance in the Anthropocene has allowed us to imagine that laboratories and ancestral underground brines are indeed connected through a materiality of lithium made perceptible through a deep-time meta-chemistry; our experimental alliance facilitated an understanding of geochemical affinities present in both the underground brines and in the laboratory, a process that unfolded slowly, in dialogue. In contrast to the fast science approach, which does not pause to address complexity, slow science enabled us to delve into the temporality of materials and their territorial relations. We believe this is important: it has enabled both of us, Martina and Cristóbal, to think more slowly about the material genealogy of lithium and the role of our own practices in understanding lithium's individuality through “humble stories capable of powerful effects within a vast interscalar journey” (Hecht 2018; Neale, Addison and Phan 2022), notwithstanding the fast substantialism of lithium industries. We have shown how chemicals are realizations that depend, in part, on the time we can give ourselves to think and experiment collectively—from our different disciplines and sensitivities.
At present, there are more pressing collective tasks than reproducing fast substantialism; one task is resisting uprooted resource meta-chemistries by imagining, as Krenak (2022) proposed, an Ancestral Future. Needless to say, Krenak's invitation is a kind of oxymoron, as it suggests imagining the future from a planetary past, thereby displacing the modern obsession with lineal, progressive time and the messianic promises of modern technology (see Viveiros de Castro and Hui 2021), while emphasizing the very importance of place: contemplating an ancestral future helps redirect our modes of attention to the places we inhabit here and now. Therefore, our invitation to explore that “it matters what lithium matters we use to think other futures with” is also a material-spatial formula, in which the deep-time materiality of reagents and salt flats complicates any illusion of lithium as an isolated substance without a material past. In conclusion, in a planet still awaiting decarbonization, our experimental alliance aspires to cultivate a profound sense of responsibility, drawing from our ancestral Latin American undergrounds. It is from these underground roots of our past that we envision shaping our ancestral future.
Footnotes
Acknowledgements
Previous versions of this article were presented as joint lectures at the Netherlands Graduate Research School of Science, Technology, and Modern Culture (WTMC) in November 2022, and later deliberated upon in international workshops hosted by the Department of Anthropology at the University of Amsterdam. We extend our gratitude to all participants of these events, as well as the thoughtful reviewers of this journal. We offer special thanks to the editors of this special issue, Alessandro Rippa and Matthäus Rest, as well as to Courtney Addison for their patience and ongoing encouragement. Special thanks to Suzana Sawyer and Evan Hepler-Smith for being a source of inspiration in leading a metachemical project titled Experiments in More-Than-Chemical Worlds. Martina would like to thank her colleagues at the Mineral Resources and Ceramics Technology Center for their collective way of doing science and constant support even when facing the “unexpected behaviors of lithium materials.” Cristóbal would like to thank Esteban Castillo Gutiérrez for the preparation of Figures 1 and ![]() . This article was possible thanks to funding received from the European Research Council (ERC) under the European Union's Horizon 2020 research and innovation program (grant agreement No. [853133]).
. This article was possible thanks to funding received from the European Research Council (ERC) under the European Union's Horizon 2020 research and innovation program (grant agreement No. [853133]).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the H2020 European Research Council (grant number ERC Starting Grant Worlds of Lithium 853133).
Notes
Author Biographies
![]() ). He holds a PhD from the University of Edinburgh, and has been trained at the intersections of clinical psychology, social anthropology and Science and Technology Studies (STS) in Chile, Italy, UK and the Netherlands. Worlds of Lithium builds on Cristobal's 10-year research trajectory on the transformation of social and environmental realities when co-existing modes of existence and technologies meet.
). He holds a PhD from the University of Edinburgh, and has been trained at the intersections of clinical psychology, social anthropology and Science and Technology Studies (STS) in Chile, Italy, UK and the Netherlands. Worlds of Lithium builds on Cristobal's 10-year research trajectory on the transformation of social and environmental realities when co-existing modes of existence and technologies meet.
