Abstract
The history of US biotechnology oversight for genetically modified plants is analyzed in the context of policy process theories to derive insights for contemporary governance of gene-edited plants. The Advocacy Coalition Framework sheds light on how opposing coalitions with different policy beliefs struggled to influence oversight, along with coalition disputes over the scope of issues that should be considered in regulatory policy-making. The Multiple Streams Approach and Punctuated Equilibrium Theory explain how focusing events arising from these struggles opened “windows of opportunity” to put issues on the public policy agenda and force changes to oversight over time. For example, nongovernmental organizations had a prominent role in bringing legal challenges through federal courts or in raising attention to risk issues in the media—efforts that prompted advancements in federal regulations, guidance documents, or risk-mitigation practices for biotechnology oversight. These policy dynamics depended on public information to bring controversies to light and elicit a policy response. However, recent biotech regulations allow for gene-edited crops to enter the marketplace without requirements for public disclosure or tracking. Lack of transparency jeopardizes the public legitimacy of gene-edited crops, venues for public participation in biotechnology oversight, and ultimately responsiveness to adapt oversight to future biotech products and emerging risks.
Introduction
Currently, new policies for the oversight of gene-edited crops are emerging in the United States and abroad with uncertainty about the future impacts of these policies. This paper examines the history of oversight for genetically modified organisms (GMOs) to formulate hypotheses about the future impacts of the current gene-edited crop oversight system. I use policy process theories, specifically the concepts of “focusing events” and “scope expansion,” to examine the historical dynamics of US genetically modified (GM) crop oversight. Toward the end of the paper, I hypothesize about how the current structure of gene-edited crop oversight is likely to impact policy dynamics into the future.
Policy process theories help guide the study of complex policy systems by providing a consistent framework for analysis (Sabatier and Weible 2014). A retrospective analysis of the dynamics of policy systems (e.g., oversight of biotechnology development) and subsystems (e.g., regulation of GM crops), coupled with this theoretical lens, lays the ground for hypotheses about the behavior of policy systems in the future. Policy process theories have much to offer the study of GMO oversight at this crucial time when policies are being formulated or beginning implementation for gene-edited crops. Yet, scholarship on GMOs oversight has rarely intersected with policy process theory.
I begin by discussing the role of focusing events within the context of three specific policy process theories. Then, focusing events and other aspects of these theories, such as scope or issue expansion, are used to frame the retrospective analysis of major events and the resulting oversight changes in a forty-five-year history for GM crops. Finally, the paper suggests hypotheses to consider about the potential impacts of current and emerging regulatory approaches for gene-edited crops. The ability of the oversight system to be responsive to emerging risk issues and societal concerns is discussed.
Analytical Approach and Prior Work
Retrospective analysis is a cornerstone of public policy analysis and is used to analyze the past in order to project outcomes from current or future policies (see Dunn 2015; Bardach 2019). The historical analysis of GM crop oversight in this article relies on previously published work by the author 1 which analyzed interviews conducted with diverse experts and stakeholders involved in US biotechnology oversight to identify key themes, events, and historical periods of GM crop oversight (Kuzma et al. 2009; Kuzma 2014, 2020). This paper builds upon those papers and other published literature to explore important historical periods and events in GM crop oversight through several policy process theories. These periods are marked by major changes to US GM crop oversight, such as the establishment of new regulatory rules, frameworks, or federal agency guidance documents. Transitions from one phase to the next mark these significant policy changes.
Policy process theories from Sabatier and Weible’s (2014) seminal book were selected based on which were most applicable to the observed dynamics of US GMOs oversight. Focusing events were prominent in the selected theories as well as in the history of US GM crop oversight. Three of these theories also place importance on how coalitions of actors strive over time to influence the scope of issues considered in the policy process in order to influence policy dynamics. The science and technology studies (STS) literature has identified struggles over the scope of what should count as “knowledge” or expertise worthy of being considered during decision-making as aspects of power and control in the policy-making process for GMOs (see Bonneuil and Levidow 2012; Macnaghten and Habets 2020). This is referred to as “knowledge politics” across broader scientific areas (see Grundmann 2007; Stehr 2015). Similarly, policy process theories describe how coalition groups with power tend to scope policy issues narrowly in order to retain control of the policy agenda (Schattschneider 1960; Jenkins-Smith et al. 2014). Such overlaps between STS concepts and scope expansion/contraction from policy process theory are identified in the analysis of GMOs oversight history.
Policy Process Theories and Focusing Events
The history of US biotechnology oversight points to certain public events or controversies that forced changes to the oversight system, for example, through the adoption of federal policy, promulgation of agency rules, or revision of regulatory guidance documents (Kuzma et al. 2009; Kuzma 2014, 2020). Under policy process theories, key circumstances or occurrences that catalyze policy change are called “focusing events” (Sabatier and Weible 2014; Baumgartner and Jones 1993; Zahariadis 2014; Jenkins et al. 2014; Baumgartner and Mortensen 2014). Focusing events help to bring attention to issues and put them on the public policy-making agenda. For the analysis, three policy process theories were chosen that make “focusing events” a central focus of policy change.
Focusing events are a central element in the multiple streams approach (MSA; Kingdon 1995; Zahariadis 2014). MSA describes three streams flowing through the policy system: policy problems, policy ideas, and politics. At critical junctures, these streams are brought together by policy entrepreneurs to increase the chances that policy changes can be made. One key idea of MSA is that the “policy window” can open when problems, ideas, and politics come together as prompted by focusing events. According to MSA, a key limit to getting issues put on the policy agenda under complex and chaotic circumstances is whether one can garner the attention of policy makers who are under significant time constraints. Focusing events play a role in this process because they often involve public crises or societal conditions that enable policy makers to concentrate their time on the issue at hand and consider new policies or programs to address it.
Punctuated equilibrium theory (PET) arose out of MSA and adopts some of its key elements. In contrast to MSA, it holds that policy subsystems (like US GM crop oversight) are not always chaotic and complex but have periods of stability and incremental change (equilibria), or alternatively, larger-scale departures from past policies and programs and punctuated change (Baumgartner and Jones 1993; Baumgartner and Mortensen 2014). Under PET, periods of stability are maintained due to the conservative nature of political systems that favor the status quo and the interests of groups already in power (Baumgartner and Jones 1993). These “policy monopolies” can collapse when pressures for change build up and newly involved political actors or government institutions intervene. Like with MSA, “focusing events” play a role in dynamic policy change and often stem from these public pressures. However, PET expands the idea of focusing events beyond events in a temporal sense, to different ways of understanding a policy issue—such as a change in “policy image,” or new places for making policy, such as a change in “policy venue.” For example, a group without power in a static policy system may change the venue for a policy dispute to a more independent place (such as a judicial court or a media outlet) where they can win the dispute and effect change.
The Advocacy Coalition Framework (ACF) is the last policy process framework used in this analysis. Like PET and MSA, ACF applies at the level of the policy subsystem (in this case, US GM crop oversight), but it pays more attention to the competing coalitions within the system and how they catalyze policy change (Jenkins-Smith et al. 2014). Variables in the ACF include: at least two competing coalitions in a policy subsystem using various strategies to influence decisions made by government authorities, relatively stable parameters such as the macro-level constitutional structure of a political system, and dynamic external events.
This last category of dynamic external events is analogous to the focusing events described in PET and MSA. In ACF, existing policies and programs reflect the beliefs of the predominant coalition with greatest influence on the policy system. The ACF posits that focusing events provide short-term opportunities that can be exploited by coalitions with different policy beliefs than those in power. Coalitions hold three levels of beliefs according to the ACF: deep core beliefs (normative, cultural worldviews), policy beliefs (related to the fundamental policy issue at hand), and secondary policy beliefs (specific policy proposals regarding the issue at hand; Jenkins-Smith et al. 2014). The ACF also hypothesizes that coalitions form from individuals or groups that share deep and core policy core beliefs; that major controversies between opposing coalitions in a policy subsystem occur when deep and policy core beliefs are in dispute; and that coalitions of allies versus opponents tend to be stable over periods of at least a decade.
Another important hypothesis related to the ACF is that coalition groups with power will tend to confine policy issues narrowly in order to retain control of the policy agenda, whereas those without power will attempt to expand the issues to recruit additional members to their coalition and increase their power (Schattschneider 1960; Jenkins-Smith et al. 2014). How this scope expansion and contraction play out in the history of GMO oversight will be discussed below, along with their links to similar concepts from the STS literature.
It should be noted that as these three theories are not mutually exclusive, they are used together to interpret dynamics in US GM crop oversight over time. For instance, the ACF sheds light on how opposing coalitions with different policy beliefs and ways of scoping GM crop issues struggled to change US biotechnology oversight over the course of its forty-five-year history. Meanwhile, MSA and PET help explain how focusing events arise from such coalition struggles and work to open windows of opportunity, disrupt power arrangements, and bring attention to GMO oversight issues from decision makers, which put issues on the public policy agenda and catalyze changes to the oversight system over time.
Analysis of Policy Dynamics of GMOs Oversight
Evolution (1973-1986)
The laboratory development of recombinant DNA (rDNA) methods (aka modern biotechnology) in the early 1970s is the beginning of the US biotechnology oversight story. As rDNA techniques emerged, scientists proposed a temporary moratorium on laboratory work using them due to concerns about the transfer of pathogenic sequences to workers from rDNA vectors (Berg et al. 1975; Talbot 1980; Hurlbut 2015). Shortly thereafter, they convened the Asilomar Conference, which brought together prominent scientists working on molecular biology along with a few lawyers and journalists to discuss whether rDNA experiments warranted precaution and to put some consensus restrictions on GM technology (Berg et al. 1975; Talbot 1980; Hurlbut 2015).
The development of biotechnology and this temporary pause to reflect on biotechnology safety can be thought of as the first “focusing event” for biotechnology oversight that sparked national policy change (Figure 1). The Asilomar Conference resulted in voluntary guidelines for physical and biological containment of GMOs in the laboratory. It outlined the types of experiments that should not be conducted, such as transferring genes to and from human pathogens (Talbot 1980). After Asilomar, this temporary self-imposed moratorium of the scientific community was lifted, and soon thereafter, the National Institutes of Health (NIH) published guidelines for rDNA research for laboratory experiments tied to NIH funding (NIH 1978).
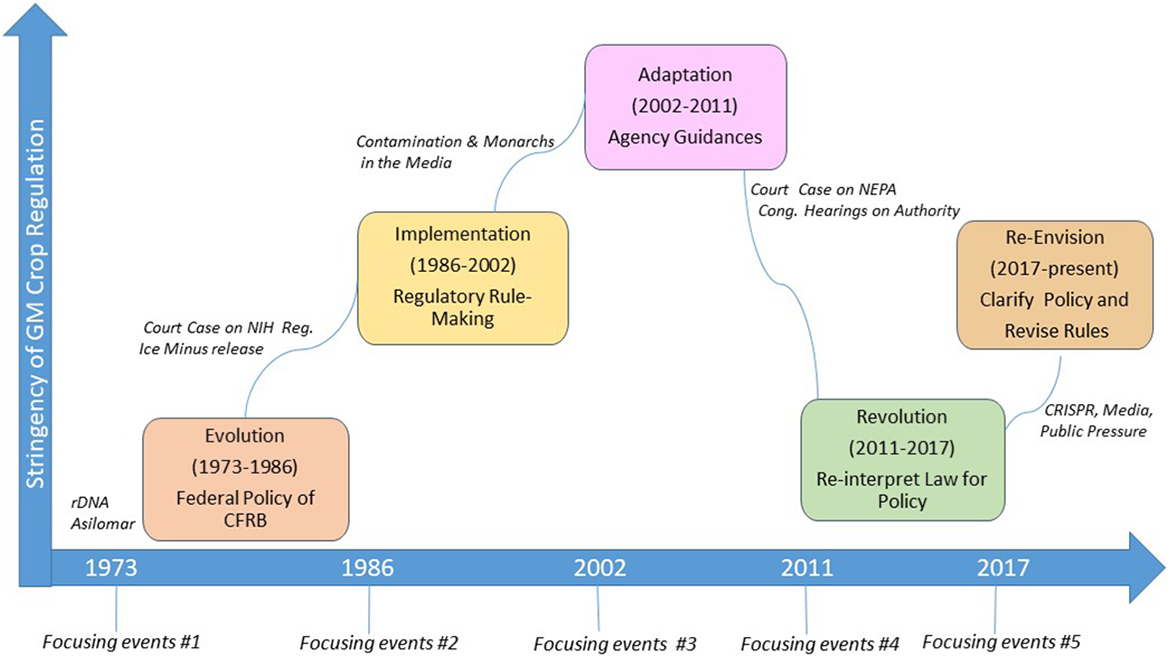
Dynamics of the US genetically modified organisms (GMOs) oversight system. The x-axis depicts the 45+ year time line of US GMO oversight with focusing events prompting changes in GMO oversight as described in the text. The y-axis is an estimate of the degree to which GM crops (including gene-edited crops after 2010) would undergo formal premarket regulation by US federal agencies. For example, historical phases higher on the y-axis mean a greater number of regulations, guidance documents or policies to regulate GM crops were enacted as a result of the focusing events. Those lower indicate a relaxation of policies or their implementation to loosen premarket regulation of GM crops, such as when the definition of “plant pest” was narrowly interpreted for gene-edited crops after 2011 and most were exempt from US Department of Agriculture review under the Plant Pest Act.
Policy process theories help understand these early events in US biotechnology oversight history. First, the Asilomar Conference was narrow in scope and participants by design, with attendees largely falling into one advocacy coalition: an elite set of early developers and proponents of biotechnology, along with a few media representatives to publicize the event, while the general public was excluded (Hurlbut 2015). From policy process theory, and in particular ACF, one hypothesis is that coalition groups with power (in this case, the molecular biologists developing rDNA method) will tend to confine policy issues narrowly in order to retain control of the policy agenda (Schattschneider 1960; Jenkins-Smith et al. 2014). Asilomar seems to fit this hypothesis, as the biotechnologists’ coalition excluded critics from the conference and tried to stave off onerous top-down government regulations for biotech by giving themselves a mandate for self-regulating laboratory safety. One of the biotechnologists leading the conference explained that it was an attempt at “avoiding governmental responses” which would be “too rigid, too hard to reverse, and too hard to work within” (Weiner 2001, 208). Hurlbut (2015, 11) describes the meeting as “an expression of not only scientific responsibility, but also of control,” where the scientific community assumed authority to “determine what risks warrant worry, and what technological futures are possible, desirable, and good.” In the terms of PET, issue confinement also helped to maintain the status quo, power relations, and a stable policy equilibrium (Schattschneider 1960; Baumgartner and Jones 1993).
The struggles over the scope of the risk issues and the people whose expertise counted in Asilomar can also be interpreted through the STS concept of “knowledge politics” (Grundmann and Stehr 2003; Grundmann 2007; Stehr 2015). Knowledge politics creates an inside/outside distinction for the “specific bodies of knowledge used as justification for policy” and the “experts chosen by politicians to perform their role of advisors” (Grundmann 2007, 417). In this early evolution phase of GMOs oversight, the dominant coalition of early developers of molecular biology exerted power by confining the risk issues to be considered at Asilomar and limiting who could participate in the conference. More recent studies illustrate how knowledge politics continues to play an important role for maintaining power and control in the policy process for GMOs (Bonneuil & Levidow 2012; Macnaghten & Habets 2020).
For several years in the early development phases of rDNA technology, the molecular biology community succeeded in staving off top-down mandatory government regulation and avoiding “punctuations” that might have otherwise come through focusing events, new policy images or venues. By 1978, several bills were introduced into Congress to expand the NIH guidelines and make them mandatory for all laboratory work, including work in the private sector and funded publicly by other agencies (Weiner 2001). However, research universities and scientific organizations lobbied to stop national legislation that they regarded as a threat to their control, while scientists assured policy makers that they had previously overstated the risks during Asilomar and could now ensure the safety of biotechnology (Weiner 2001). The NIH guidelines for laboratory use remained the main policy vehicle for biotech oversight until the early 1980s.
However, in the ensuing years, another advocacy coalition was emerging with different core and policy values than the biotechnology development coalition. This coalition was composed of nongovernmental organizations (NGOs), including environmental groups, and concerned academics from ecology, social science, and ethics, who voiced opposition to moving forward so rapidly with rDNA research. This emerging advocacy coalition worked to expand the issues associated with biotechnology policy by raising broader concerns beyond direct harm caused by laboratory pathogens (Weiner 2001). The NGOs and concerned scientists’ coalition expanded the issues from narrow definitions of biotechnology risk to human health from the use of pathogenic sequences in the laboratory to larger societal and ecological impacts. This broadening of the scope of debate may have helped to put biotech policy on the national legislative agenda.
Issue expansion is a tactic described in policy process theories to recruit additional members to a coalition and thus disrupt power relations (Schattschneider 1960; Zahairidis 2014; Baumgartner et al. 2014). With a wider coalition, groups initially without power who opposed to the status quo (in the GMOs case, the NGOs coalition) are more likely to spark a focusing event, for example, by having additional capacities or resources to move the struggle to a more neutral policy venue. This occurred in the early 1980s when researchers proposed to move GMOs out of the laboratory and into the environment. In response to this proposal, NIH approved small-scale field tests of a GMO, the Ice Minus bacterium, that was to be sprayed on strawberry plants to prevent frost damage (Weiner 2001). However, in 1983, three environmental NGOs and two individuals filed suit against the federal officials responsible for this decision, arguing that NIH did not have the requisite ecological expertise to make such a decision, and that it had not complied with the requirements of the National Environmental Policy Act (NEPA) in approving the release of Ice Minus ( FET v. Heckler 1985). Congruent with PET and ACF, the NGO coalition, initially the coalition without power, shifted the policy venue to the more independent federal courts, where they had a chance of winning (Figure 1, Table 1).
Focusing Events, Theoretical Elements, and Associated GM Crop Oversight Changes.
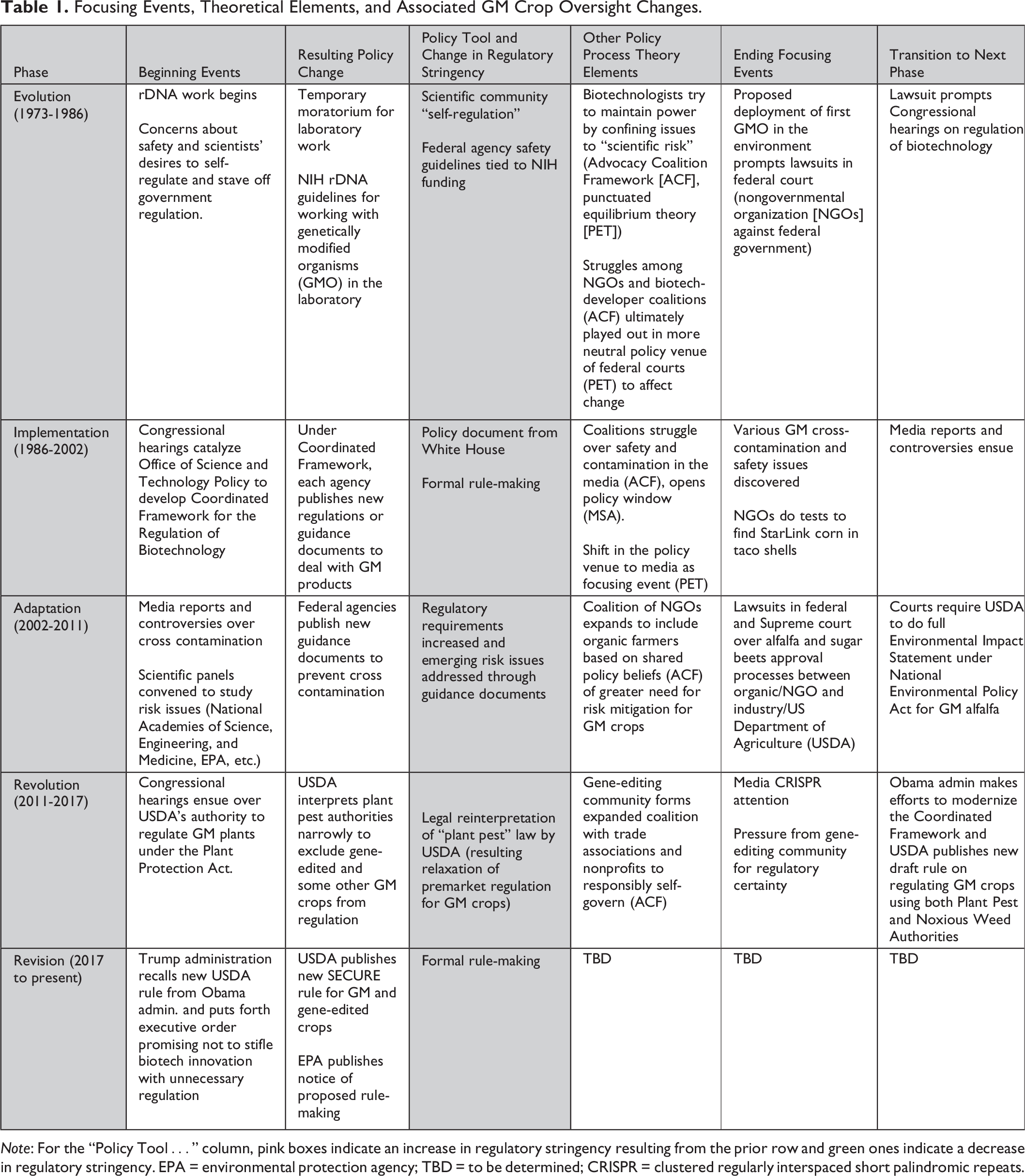
Note: For the “Policy Tool…” column, pink boxes indicate an increase in regulatory stringency resulting from the prior row and green ones indicate a decrease in regulatory stringency. EPA = environmental protection agency; TBD = to be determined; CRISPR = clustered regularly interspaced short palindromic repeats.
This federal case was a significant focusing event for the development of US biotechnology oversight at a critical juncture. Ultimately, the plaintiffs, the NGO coalition, scored a victory when the US Court of Appeals stated, “we emphatically agree with the District Court’s conclusion that NIH has not yet displayed the rigorous attention to environmental concerns as demanded by law” ( FET v. Heckler 1985, 2). This led to renewed interest in biotechnology at the US Congress, and Congressional hearings about US biotechnology oversight followed (see US Congress 1983). In response to Congressional interest and the lawsuit, the White House Office of Science and Technology Policy (OSTP) tasked an interagency working group with drafting an overall federal framework for biotechnology oversight (Marden 2002).
The NGO lawsuit and mounting struggles between advocacy coalitions with differing values (biotechnologists for self-regulation and technological progress; NGOs for “naturalness” and environmental protection) occurred. The biotechnologist coalition attempted to contract the issues to narrow set of biotech “scientific” risks, while the NGO coalition sought to expand the scope of the issues considered to broader ecosystem, social, and ethical harms. These struggles set forth a “punctuation” in policy-making according to PET or a political opening of a policy window according to MSA. These focusing events set the course for more formal, mandatory federal regulation for biotechnology for decades to come. The lawsuit by the NGOs played out in a neutral, public venue, unlike Asilomar that was a closed meeting largely limited to scientists. It also prompted a bigger policy change, and struggles between coalitions were now in the national and Congressional spotlight.
Implementation (1986-2002)
The Coordinated Framework for the Regulation of Biotechnology (CFRB) arose out of the White House OSTP working group. The CFRB was a policy document outlining how multiple federal agencies would work together to oversee the different products of biotechnology, including their environmental release (US OSTP 1986). This point marks the end of the evolution phase and beginning of the implementation phase of GM crop oversight (Figure 1, Table 1).
Although biotechnology developers were not successful in staving off formal regulation by the start of the implementation phase, they did partially “win” the policy struggle when the CFRB stated that: the risks of biotechnology products were the “same in kind” as conventionally bred organisms—in other words, there were no new categories of risk with GMOs; the product should be the focus of regulation, not the process by which it is made (e.g., biotechnology); and therefore, no new laws were needed for regulating biotechnology.
As a result, federal agencies were instructed to use existing laws to regulate biotechnology products. Yet the CFRB proved flexible enough to adjust over time with new biotech products and technologies given its weak legal grounding (Kuzma et al. 2009), and biotechnology developers were able to move forward within an oversight framework that could be reinterpreted over time.
Around the same time that the CFRB was established, US laws were enacted that allowed for closer relations between academia, government, and industry to capitalize on their research through intellectual property partnerships (see Adams et al. 2003; Winickoff 2013). Therefore, the biotechnology–developer coalition expanded to include government research agencies and private sector entities including the agricultural and chemical companies involved in early GM plant development and seed sales. Given its access to greater private resources and political processes, the expanded coalition grew in power considerably during the implementation phase of GMOs oversight. Since the late 1980s, biotechnology developers floated between private and public sectors, and the relationship between the two has remained close over time. 2
During the implementation phase, policy tools such as formal rules were needed for the federal agencies to implement the CFRB and apply existing laws to new biotechnology products. The laws were significantly stretched under the CFRB to promulgate agency regulations for diverse products of biotechnology in this phase. For example, in 1997, EPA passed rules to regulate GM microbes as “toxic chemicals” under the Toxic Substances and Control Act of 1976 (US EPA 1997). The Animal Plant Health and Inspection Service (APHIS) of US Department of Agriculture (USDA) was the lead agency for biotech crops. APHIS passed rules to regulate GM plants as “plant pests” under the Federal Plant Pest Act (FPPA) of 1957, initially under field trials (USDA 1987, rev. 1997) and then for full-scale market release (USDA 1993). During the implementation phase, rDNA methods to engineer plants used sequences from a bacterium that causes plant disease (Agrobacterium) in order to deliver genes into plants. Thus, the DNA fragments from these plant pests provided a regulatory hook for the USDA for all GM crops, even if the engineered gene had nothing to do with the “plant pest” risks on which the FPPA was based.
Later in the implementation phase, the EPA promulgated rules to regulate GM plants with pesticide-like substances (e.g., GM crops with Bt protein) under the Federal Insecticide, Fungicide and Rodenticide Act (FIFRA; US EPA 2001). Yet the Food and Drug Administration (FDA) did not promulgate formal rules for reviewing the safety of foods from GM crops. Instead, it issued voluntary guidance for industry for the review of GM foods with novel proteins under the Federal, Food Drug and Cosmetic Act (FFDCA; US FDA 1992).
Agencies also began to apply NEPA to their decisions about the release of GMOs into the environment. NEPA requires that federal agencies assess the potential environmental impacts of all “major Federal actions significantly affecting the quality of the human environment” (42 USC §4332(2)(C) 2011). However, under the CFRB and during the implementation phase, USDA decided to use the more limited environmental assessment and Finding of No Significant Impact options for GM plants under NEPA, rather than full environmental impact statements (EISs). In the subsequent adaptation phase, this decision proved to be controversial in federal oversight for GM plants
During the implementation phase, GM crops entered field trials in 1987, and within a decade, thousands of field trials were underway. Major commodity crops with engineered insect-resistant (Bt) and herbicide-tolerant (Ht) genes entered the market in 1995, and these GM crops grew to dominate US markets for corn, soybean, and cotton within just five years. During this same time, significant controversies over the safety of GM crops to humans and the environment began to emerge in the public sphere. As with the transition from evolution to implementation, the transition from implementation to adaptation phases was marked by struggles between two major coalitions: biotech developers (including those in industry) and consumer/environmental NGOs as interpreted by ACF. This time, there was a shift in the policy venue (PET) that sparked the focusing events. NGOs presented and amplified public stories in the media, which prompted public pressure and policy change (Figure 1, Table 1).
A key focusing event involved the public controversy over the contamination of human food with StarLink GM corn (Figure 1). StarLink corn was approved by EPA under FIFRA only for animal use, but not for human food use, because the Bt protein used in this GM corn variety (Cry9C) had some resemblance in amino-acid sequence homology and indigestibility to human allergens. However, in September 2000, the Washington Post reported that traces of StarLink GM corn DNA were found in Kraft taco shells and other products intended for human consumption. Interestingly, the tests for StarLink presence were ordered by a coalition of consumer and advocacy NGOs (Segarra and Rawson 2001). Kraft confirmed the finding and voluntarily recalled the shells from grocery stores. In November of the same year, the FDA recalled more than 300 corn products. The StarLink corn producer, Aventis, voluntarily withdrew its license for the variety from EPA, as foreign markets for US corn were threatened (Segarra and Rawson 2001).
After this public focusing event, several bills were introduced into Congress to require labeling of GM foods and to amend the FFDCA to require mandatory, premarket consultation and approval for GM foods (Segarra and Rawson 2001). Although these bills were not ultimately passed, it prompted the FDA to issue stronger, revised guidance for studies in the context of voluntary GM food safety consultations (US FDA 2006). After the StarLink corn focusing event, EPA would never again approve something for animal feed without human food approval. This illustrates the important role of NGOs in actively creating public focusing events. In this case, it started with NGOs testing taco shells on grocery shelves, which led to the media becoming the policy venue. In terms of PET, the venue change may have prompted greater public attention to the issue and brought the issues associated with GM crop cross-contamination to the national policy-setting agenda (Figure 1, Focus Event #3; Table 1). NGOs, wielding much less power in typical policy-making venues, took GM risk issues to the public venue of the media to raise awareness among the general public, expand their coalition, and thus grow their power.
Following the StarLink incident, GM crop safety was questioned by laboratory studies that attracted many media reports, about harm to Monarch butterflies from Bt corn pollen; gene flow between Bt and non-Bt maize in Mexico, the biodiversity center of maize; and contamination of soybeans for food use with corn grown to produce engineered pharmaceuticals (Segarra and Rawson 2001; Fox 2003; Quist and Chapela 2001). For example, the company ProdiGene failed to eliminate volunteer GM corn plants used for pharmaceuticals manufacturing, from a soybean crop planted later in the same field and destined for human food (Fox 2003; Byrne 2008). USDA imposed a fine of $250,000 on the company, and ProdiGene had to reimburse the federal government $3 million for the destruction of the contaminated soybeans. These controversies, like StarLink, were also marked by amplification in the media and struggles between consumer and environmental NGOs and biotech advocates over the interpretation of uncertainty in health and environmental safety studies (see Pew Initiative 2002).
Adaptation (2002-2011)
During the adaptation phase, federal agencies associated with the CFRB were prompted to adapt their regulatory procedures to address emerging cross-contamination and safety issues (Kuzma 2014), leading to a greater degree of regulatory stringency (Figure 1, Table 1). They did so in response to growing public discussion in the media and among scholars and scientists about the events surrounding StarLink, ProdiGene, monarchs, and Mexico. For example, during this time, the National Academies of Science, Engineering, and Medicine (NASEM, 2002, 2003, 2004) also convened several committees to review health and ecological safety issues associated with GM crops. These NASEM reports focused on specific emerging concerns, although they generally concluded like previous and future NASEM reports (NAS 1987; NASEM 2000, 2017) that the categories of risks posed by GM products were the same as non-GM products. Concerns grew about the potential allergenicity of GM foods, comingling of unapproved and approved GM varieties, and gene flow to wild relatives or neighboring crops, which may contaminate organic crops or cause risks to agriculture or the environment (e.g., through Ht genes passed on to weedy relatives).
Yet agencies did not promulgate new regulations for GM crops, and the interpretation of the laws under the CFRB remained largely unchanged. Instead, several guidance documents and regulatory policies were published to address these emerging concerns (Table 1, Figure 1). FDA put out a guidance document to improve early food-safety evaluation of GM crops (US FDA 2006). USDA published enhanced biosafety guidelines requiring more stringent confinement measures for growing GM plants containing pharmaceutical and industrial engineered proteins in field trials (USDA 2003).
The USDA did attempt to promulgate new regulations for GM plants, and in 2008, it went so far as publishing a draft rule. The agency tried to clarify its authority for GM crops under the newer Plant Protection Act of 2000 (PPA) and update its regulations to comply with the new law, which combined and replaced the 1957 FPPA and the Federal Noxious Weed Act of 1974. In this draft rule, USDA proposed to use its noxious weed authorities to regulate GM crops and change its environmental-release decisions to a tiered permit system based on categories of risk (USDA 2008). The rule would have subjected GMOs to oversight based on noxious-weed risks for the first time in addition to “plant pest” risks. However, USDA never finalized the 2008 rule because the Presidential administration changed just after the publication of the draft rule.
Around the same time, NGOs were able to expand their coalition to include the organic industry and organic farmers. Once again, like in the evolution to implementation transition, an expanded coalition that initially had less power than the opposing biotech developer coalition was able to expand the scope of risk issues and put forth legal challenges in a neutral policy venue (the federal courts) to contest federal government decisions, specifically USDA decisions for GM crops. These legal challenges were an important focusing event sparking policy change (Table 1, Figure 1).
The ACF theory highlights issue expansion as a way to recruit additional members to a coalition and gain more leverage to get issues on the national agenda to effect policy change. Environmental and consumer NGOs were able to combine their interests in consumer choice, health, and environmental safety with the organic industry’s concerns about cross-contamination of their crops with GM pollen or GM crops—a socioeconomic risk issue to the industry. The National Organic Program rule of 2000 prohibits the intentional use of GM crops in certified organic foods (USDA 2000). As GM crops flooded the market in this phase, organic farmers became increasingly concerned that GM crops would cross-pollinate or comingle with their crops and that they would no longer be able to fulfill their contractual obligations or assert that their products were GM-free. They saw the harm from GM crops as lost revenue and competition. According to PET, the policy image for biotechnology risk issues changed, expanding from a narrow definition of environmental and health risks, to include socioeconomic risk and harm to a US industry. The NGO group recruited an industrial sector of farming and food (organics), at a time when organic consumption was increasing among US consumers. By aligning their interests to those of the organic farming industry, NGOs’ broader concerns began to be taken more seriously by US policy makers—particularly those at USDA who are tasked with representing all of US agriculture. According to STS theory, the “knowledge politics” shifted (Grundmann and Stehr 2003; Grundmann 2007; Stehr 2015) as more people were allowed to participate in policy-making and the scope of the risk issues that should be considered in the GM debates broadened (Jasanoff 2005b; Macnaughten and Habetts 2020). According to policy process theory, the scope expanded (Schattschneider 1960) and the coalition broadened to gain power (Jenkins-Smith et al. 2014).
This issue expansion (“harm to a US industry”) and the expanded NGO–organic industry coalition disrupted power relationships and static policy states. Through the independent policy venue of the courts, lawsuits against industry and the USDA were introduced by the NGO–organic coalition to challenge the adequacy of USDA’s assessment of GM crops under NEPA (Cowan and Alexander 2013). The plaintiffs asserted that USDA did not comply with NEPA because it used a less rigorous environmental assessments than a full-scale EIS process. They also argued that socioeconomic risks should be considered in USDA’s decisions about GM crops under NEPA.
Courts were a more open policy-making venue for the NGO–organic coalition, whose members had fewer opportunities for participating directly in federal regulatory decision-making than the more powerful biotech industry–developer coalition. Participation in the regulatory decision-making process was largely open only to biotech developers and federal agency representatives reviewing GMOs (Kuzma et al. 2009; Kuzma 2021). Federal agency “knowledge politics” insisted that stakeholders and publics adhere to a limited scope of issues surrounding GMOs, and thus only the concerns of a limited set of experts were deemed as legitimate (Wynne 2001; Jasanoff 2005a, 2005b; Thompson 2007; Bonneuil and Levidow 2012; Macnaghten and Habets 2020). The dominant discourse for US risk analysis for GMOs was that it should be separate from broader concerns and values (Jasanoff 2005a, 2005b; Wynne 2001; Bonneuil and Levidow 2012; Macnaghten and Habets 2020), despite the inherent values and ethical issues embedded in the risk analysis process (Wynne 2001; Thompson 2007; Kuzma and Besley 2008). In the more neutral and open venue of the US court system, the coalition of NGOs and organic industry put forth their broader ecological and socioeconomic concerns, which had been outside the scope of policy-making discussion until then.
From these lawsuits, USDA lost two federal district court cases because it presented inadequate data to support its decision to allow deregulation of herbicide tolerant (Ht) GM alfalfa, and for ignoring evidence of potential environmental harm in field trials of GM bent grass. The USDA was forced to complete its first-ever EIS document for genetically engineered (GE)-crop deregulation in 2010 (Cowan and Alexander 2013; USDA 2010). Interpreted by PET and ACF, a shift in US regulatory policy was prompted by an expanded advocacy coalition and focusing events playing out in an independent policy venue (i.e., the courts) to disrupt power relationships, the knowledge politics, and the static policy system. The decisions on these cases mark the end of the adaptation phase (Table 1; Figure 1, Focusing Events #5).
Revolution (2011-2017)
In December 2010, USDA published the final EIS for GM alfalfa as mandated by the courts. USDA outlined three decision alternatives in the document: (1) ban the commercial planting of RR GM alfalfa (no deregulation); (2) approve it with planting restrictions such as greater isolation distances from other crops (partial deregulation); or (3) approve it with no planting restrictions (full deregulation; USDA 2010). USDA indicated that it was seriously considering the latter two options. In the document, USDA (2010) argued that “[b]ecause Congress has mandated a science-based approach in APHIS regulations and because there is no basis in science for banning the release of GT (glyphosate tolerant) alfalfa, a blanket prohibition of the release of GT alfalfa would contravene Congressional intent and must be rejected” (p. 14). In this statement, the USDA seemed to dismiss the consideration of potential socioeconomic harms to the organic industry by confining or contracting the issues to “science-based” and maintaining the narrow knowledge politics associated with their regulatory purview.
After the EIS for GM Ht alfalfa was published, both advocacy coalitions (NGO–organic versus biotech developers) put pressure on USDA to adopt a specific regulation model. The organic–NGO coalition argued for no or only partial approval, whereas the agricultural commodity–biotech coalition lobbied for full approval (Cowan and Alexander 2013). The US House Committee on Agriculture became interested and hosted a meeting on January 20, 2011, to discuss the case with USDA Secretary Tom Vilsack (US Congress 2011). At this meeting, members of the House of Representatives intensely questioned the Secretary about the partial deregulation option in the EIS. The vast majority of Congressional members on the committee voiced their support for full deregulation—that is, unrestricted planting of GE alfalfa—and several of these members came from states associated with agricultural biotechnology companies and industrial farming systems. Even more important to the oversight story in this revolution phase, the House members broadly challenged whether USDA had authority for regulating GE crops at all under the Plant Protection Act (US Congress 2011). Just a week after, on January 27, 2011, Secretary Vilsack announced that USDA was granting GE alfalfa full approval with no restrictions. Organic farmers vehemently opposed this decision given their need to keep GE pollen out of their fields to maintain organic certification. These events illustrate how even though the less powerful coalition (NGO–organic) was able to open up a policy venue, initiate a focusing event, and expand the scope of the issues, it could not overcome the knowledge politics of federal agencies and political influences of the more powerful coalition (biotech developers).
Although the policy process was affected by the NGO–organic coalition, the policy outcome was not favorable to their interests and their efforts ultimately backfired. After the alfalfa case and Congressional pressures, USDA started to make decisions that several GM crops, including gene-edited crops, did not fall under its plant pest based regulations at all. They began to interpret their regulatory authority narrowly to include only GM plants that contained plant pest DNA sequences. By contrast, in the earlier days of the CFRB, USDA signaled their intent to capture all GM plants under their plant pest regulations regardless of presence of plant pest DNA (see NASEM 2000). By 2011, however, newer biotechnology methods like gene editing and particle gun transformation were becoming common, and this allowed biotechnologists to avoid plant pest sequences from Agrobacterium in the making of GM plants or end products derived from GM plants. Around 2011, USDA began to exempt many gene-edited and GM crops from their regulations (Kuzma 2016a). Biotech developers embraced this approach, arguing that the risks of GM products (as a general category) were no greater than conventionally bred products (NASEM 2016).
With legal challenges from NGOs and organic groups expected to continue into the future, USDA would be forced more frequently to use the intensive EIS process for NEPA review of GM crops, for as long as these came under the agency’s jurisdiction. The EIS process was seen as more burdensome for an agency with only limited staff and resources, as well as imposing greater costs on the biotech industry. USDA, faced with potentially more legal challenges from coalitions of NGOs and the organic industry and with increasing Congressional pressure against their authority to regulate GM crops at all, made a radical choice to back off from GM crop regulation, marking the new policy equilibrium in the Revolution phase.
This revolution phase returned to the policy tool of national policy-making, but this time, unlike the hearings and public working groups that generated the initial CFRB (Figure 1, Focusing Events #2), policy was quietly made behind closed doors and only captured in letters between GM crop developers and agency staff (which were eventually requested under the Freedom of Information Act by interested lawyers and scientists; Kuzma 2014). According to this policy, USDA would interpret its statutory authority to regulate “plant pests” under the Plant Protection Act more narrowly. Only after the Freedom of Information requests did USDA (2020) began to make these letters publicly accessible on their website under their “Am I Regulated?” (AIR) web archive of these decisions to exempt GM plants. From 2011 to 2020, more than hundred GM crops had undergone this abbreviated review process and were exempted from formal USDA (2020) oversight.
In summary, the political events that followed the federal court cases initiated by the NGO–organic coalition at the end of the Adaptation phase ultimately pushed the oversight system in a less rigorous regulatory direction during the Revolution phase (Figure 1, see y-axis). The efforts of the NGO–organic coalition succeeded in getting issues on the policy agenda, and the coalition even won its cases in federal court (Figure 1, Focusing Events #4; Table 1). However, what they set in motion backfired, as Congressional interest increased in the alfalfa Supreme Court case, powerful interests re-emerged, and Congress began to question whether USDA should be regulating GM crops at all. The NGO-led coalition won the battle in the courts, but it may have lost the war in hoping for greater oversight of GM crops. The oversight system came to a new policy equilibrium in the Revolution phase, which ran against their interests.
Toward the end of the revolution phase, the use of CRISPR-based gene-editing methods and the lack of regulation for gene-edited crops garnered significant media attention and was becoming more of a public issue (see Waltz 2016; Kuzma et al. 2016). Around the same time, the Obama administration set forth its willingness to revisit the CFRB to see whether it was well positioned to keep pace with the advances in biotechnology, such as CRISPR-based gene editing (Kuzma 2016b; Figure 1, Focusing Events #5; Table 1). Concerns were also growing among the gene-editing community about the increasing proportion of consumers choosing non-GM foods (Kuzma 2018), which might have increased the political feasibility of the Obama administration’s effort. Industry groups expressed desires to do better with gene-edited crops in light of controversies surrounding the first generation of GM foods (Kuzma et al. 2016), and a nonprofit and industry coalition formed to consider the responsible stewardship of gene-edited crops (CFI 2020). This marks the end of the revolution phase and beginning of the revision phase. While the revolution phase involved significantly weakening GM crops regulation, the revision phase reexamined this state in light of advances in gene editing and the trends described above, and set forth efforts to promulgate new rules.
Revision (2017 to Present)
In its final days in January 2017, the Obama administration published a change in USDA’s rules to regulate GM crops that would have captured more gene-edited crops and incorporated the noxious weed provisions of the PPA (in addition to the plant pest authorities, like the failed rule-revisions of 2008 had attempted to do). To many, changes to the USDA GMO rules made scientific sense because a demonstrated category of risks from the first generation of GE crops concerned the evolution of resistant weeds. For example, by this time, there had been reports of glyphosate-resistant weeds resulting from GE herbicide-tolerant grasses fertilizing wild relatives (Rosen 2018; Zapiola and Mallory Smith 2017). Yet, like the 2008 rule, the proposed 2017 rule was withdrawn under the next Presidential administration. The Trump administration revoked the 2017 GMO Obama rule, stating that “the proposed requirements would be too burdensome and had the potential to stifle innovation” (USDA 2020b, 4).
In 2019, the Trump administration put forth an Executive Order asking US federal agencies to streamline their approval processes for biotechnology products and remove undue regulatory barriers to innovation (White House 2019). Then, the USDA promulgated the current rule for GM crops in 2020 that exempts many categories of gene-edited crops, does not invoke the noxious weed provisions for reviewing risks under the PPA, and shifts away from plant pest DNA sequences as the regulatory hook and toward a review of plant pest risks from GM crops (USDA 2020b). This 2020 Sustainable, Ecological, Consistent, Uniform, Responsible, Efficient (SECURE) rule represents the first major change in USDA GM crops formal regulation since they first entered the market in the 1990s.
In one sense, SECURE is a step toward greater regulation of GM crops by replacing the abbreviated AIR review process (Kuzma and Grieger 2020). However, SECURE provides exemptions for several types of gene-edited crops and allows for developers to self-determine whether their crop is exempt without consulting with regulators. This self-exemption pathway also creates a situation where no public information would be available about the market entry of many gene-edited crops unless the developer chooses to voluntarily disclose it on their own public website or venue. 3 Furthermore, even if a gene-edited crop is reviewed by USDA, public information about the traits, purposes, and market uses of that crop may not be disclosed on the USDA’s regulatory lists to protect confidential business information in regulatory submissions or lack of regulatory obligations to publish this information (Kuzma and Grieger 2020).
In addition, the USDA’s new National Bioengineered Foods Disclosure Standards (NBFDS) exempt GM foods from being labeled as “bioengineered” if they do not contain foreign DNA in the final product (Jaffe 2022; USDA 2018). In other words, current labeling standards do not apply to most gene-edited crops or ingredients refined from gene-edited or GM crops. In addition, in 2020, the EPA proposed to draft rules to exempt most gene-edited crops from its oversight under FIFRA (EPA 2020). Finally, FDA’s review for food safety of gene-edited crops remains voluntary, and the agency has not yet released an updated voluntary guidance for reviewing gene-edited foods and their safety. In summary, although the revision phase saw a step toward greater regulation of GM crops through SECURE, there are significant gaps in regulatory review and public information about gene-edited crops.
In our previous interviews with developers of gene-edited crops, most agreed that mistakes were made with the first generation of GM crops and acknowledged that stakeholders and citizens should be better informed and consulted as they move forward with gene editing (Kuzma et al. 2016). They also feared the increasing consumer demand for non-GM food products and hoped gene-edited foods would not be rejected as such. However, in this revision phase, policies enacted through SECURE and the NBFDS are likely to decrease consumer transparency and choice, violating ethical principles of autonomy and informed consent (Kuzma and Besley 2008; Thompson 2007; Kuzma 2018) and thus reduce the public’s ability to participate in decision-making about GMOs (Gordon et al. 2021; Kuzma 2021). Although transparency and choice are not enough to earn the trust of consumers, they seem to be a prerequisite for doing so, and these new policies could further erode consumer trust in gene-edited foods (Kuzma and Grieger 2020; Gordon et al. 2021). In addition, they preclude the inclusion of a wide range of expertise and special and localized knowledge to benefit regulatory decision-making and risk analysis (Wynne 1989; Funtowicz and Ravetz 1993; NRC 1996; Meghani and Kuzma 2011).
Discussion
Through the lens of policy process theories, this analysis illustrates how the US GM crop oversight system evolved to deal with emerging risk issues and technologies over time. Changes to oversight seem to be, at least in part, catalyzed by focusing events, as congruent with several policy process theories. These focusing events were aided by changes in scoping GM risk issues, expanding coalitions, and efforts to change the policy venue. They helped to put GM crop oversight issues on the national policy agenda and prompt changes to regulatory systems. Struggles among advocacy coalitions and focusing events played out in public venues, catalyzing new equilibria or phases in regulatory oversight. Some phases increased oversight capacity to deal with emerging GM crop risk issues and societal concerns (Figure 1, Evolution, Implementation, Adaptation), while others removed barriers to the development of gene-editing technology (Figure 1, Revolution, Revision).
What do these observations from policy process theory mean for the future of oversight for gene-edited crops? One hypothesis arising from this retrospective analysis is that public struggles among coalitions and public focusing events are necessary to force change in oversight systems over time, so that they are more responsive to emerging conditions and issues. Whether the current oversight arrangements for gene-edited crops will allow for this responsiveness in the future is an important question. With low transparency for the market entry of gene-edited crops currently, will public struggles leading to public focusing events be less likely?
Due to the limitations posed by the interpretative, historical, and theoretical approach taken in this article, there is not a clear-cut answer to this question. However, the analysis suggests that there is a connection between the public nature of policy processes and the ability of the GM crop oversight system to adapt to new technologies and risk issues over time. Thus, there are legitimate concerns that the lack of transparency associated with the current oversight system for gene-edited crops may hamper socially responsive and robust policy dynamics. This lends weight to the argument that without public knowledge of gene-edited crops and their uses in the market, opposing coalitions cannot struggle openly about specific risk or societal issues in the public arena. If these struggles are absent or even reduced, emerging risk issues and other societal concerns are likely to go unnoticed. Without public pressure and focusing events to advance gene-edited crop oversight, the oversight system is likely to stagnate.
As gene-edited crops enter and permeate markets, we could be setting ourselves up for significant, delayed impacts, given the absence of public coalition struggles and continual oversight adjustments over time. Over the course of the history of GM crop oversight, public struggles between coalitions allowed the system to adapt to new technologies and newly discovered risk issues, like the early environmental releases of GMOs, StarLink corn, cross-contamination of pharma crops, Monarch butterflies, or concerns about gene flow raised by the organic industry. Without these struggles, it is entirely possible that greater risk issues and controversies would have materialized, such as the detection of foodborne allergies in people consuming GM foods, harm to nontarget ecological species, and more inadvertent comingling.
Many gene-edited crops and their food products are poised to enter the open environment and the market without public disclosure of their location, purpose, or uses (Kuzma and Grieger 2020). The maturation of the GM crop oversight system depended on public struggles among advocacy coalitions, who were able to gather information about what GMOs were poised to be released, publicly contest those decisions in court, openly study the risk issues (like knowing to look for StarLink corn in taco shells), and raise their voices in the media or Congressional hearings (Table 1).
Although the future impacts of current oversight approaches for gene-edited crops are difficult to predict from the retrospective analysis presented in this article, a recommendation for greater transparency in oversight seems warranted. Voluntary initiatives for increasing transparency for gene-edited crops in the marketplace have been suggested (CFI 2020; Kuzma & Grieger 2020; Jaffe 2022; Gordon et al. 2021). However, voluntary initiatives rely on the goodwill of industry organizations and other biotech developers to participate—and usually there are market disincentives for doing so. Requiring the formal registration of gene-edited crops through USDA, even if that gene-edited crop is exempt under the SECURE rule, seems more likely to foster the transparency needed to enable public scrutiny of the regulatory framework to oversee gene-edited crops. A publicly accessible government database that contains at least the basic characteristics and market uses of gene-edited crops is more likely to allow for independent watchdogs to fulfill their important role of catalyzing policy change when needed.
However, transparency will not be enough if opportunities to participate and truly have a voice in GMO decision-making are not available to various experts, stakeholders, coalitions, and publics. Throughout the US oversight history for GM plants, the courts and the media have been the main venues for coalitions critical of biotechnology because there were no formal opportunities to participate in regulatory processes (Wynne 2001; Jasanoff 2005a; Bonneuil and Levidow 2012; Macnaghten and Habets 2020; Kuzma 2021). Although less powerful coalitions were able to get their issues onto the national agenda through these venues, they ultimately could not overcome the political forces dominating the biotechnology policy arena, which left coalitions often unable to expand the risk issues and set of values considered for federal decision-making. The dismissal of public and stakeholder concerns outside of “sound science” (which generally means dismissal of concerns outside of limited health and environmental toxicity that can be measured in laboratory studies prior to commercialization) remains in the GM policy sphere and has been entrenched in the knowledge politics of GM oversight for decades (Wynne 2001; Jasanoff 2005a, 2005b; Thompson 2007; Bonneuil and Levidow 2012; Macnaghten and Habets 2020; Kuzma 2021). Therefore, it is likely that participation in regulatory politics and federal agency decision-making for GMOs will continue to be closed, and broader concerns delegitimized, even with greater transparency (Kuzma 2021). If so, risk analyses will suffer from a lack of diverse perspectives and expertise, and the local and specialized knowledge of interested and affected publics (Wynne 1989; Funtowicz and Ravetz 1993; NRC 1996). Although transparency would allow for watchdogs and critics to bring issues to the agenda through media and the courts, it will also be important to create independent policy venues through which diverse groups and publics can voice broader concerns and values, as well as have them legitimized and taken into account for making rules regarding gene-edited crops.
Footnotes
Acknowledgments
The author would like to acknowledge the support of the Genetic Engineering and Society Center at NC State University and the Gene Edited Foods project at Iowa State University for organizing and guest editing the special volume in which this article appears. The author also thanks the anonymous reviewers for their helpful comments in improving the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
