Abstract
This article addresses the question of how the belief that leprosy is curable became an accepted scientific statement during the processes of colonization and postwar nation-building. Following the use of chaulmoogra oil to the introduction of diaminodiphenyl sulfone (DDS) as a cure for leprosy, the article shows how medical workers in Hong Kong and Taiwan, two contact zones in the global leprosy network, helped transform leprosy medicine into a set of practices centered on the mass administration of DDS. Focusing on the idea of a cure and looking for all the transformations this idea went through afterward, this article argues that it was chaulmoogra oil that created the research and management infrastructure for DDS. DDS did work, but it worked because chaulmoogra oil had already produced the conditions for a cure. This article contributes to recent calls to “provincialize STS” and to investigate postcolonial technoscience by examining the situated production of the global leprosy infrastructure. It shows that by serving as laboratories and extending the network in which the belief of a cure was embedded, the colonies were key to each stage forming the cure for leprosy as an accepted scientific statement.
On May 6, 1915, as part of an Asian tour that began in the Philippines, Dr. Victor Heiser, Director of the Rockefeller International Health Board for the East, arrived at the Pulau Jerajak Leper Colony, which was the central colony for those affected by leprosy in the Straits Settlements. Dr. Heiser took a picture of the people (see Figure 1) and wrote in his diary that “[t]here were 390 inmates, all males” and that “[t]hey refuse to work. Very few are willing to take treatment. Dr. Dane’s attitude is typical of laisez (sic) faire” (Heiser 1915, 137). Heiser made a short address to the people about the chaulmoogra oil treatment used in the Philippines: “I explained the case and a number of volunteers immediately came forward and promised faithfully to take the treatment for two years” (Heiser 1915, 137). Later in 1915, Heiser visited Sir Leonard Rogers, who was working in India, and encouraged him to develop a treatment using chaulmoogra oil (Heiser 1936). A review in the British Medical Journal (1940) stated that “[t]he modern history of the leprosy problem really begins in 1915, when Sir Leonard Rogers first began to treat the disease by injecting the sodium salts of hydnocarpus and chaulmoogra oils in watery solution” (p. 1056).

Picture of people at Pulau Jerajak taken by Dr. Heiser. Rockefeller Archive Center.
It might be surprising to both medical workers and historians to mark 1915 as the start of the modern history of leprosy, given that leprosy is considered eliminated following the mass administration of diaminodiphenyl sulphone (DDS) that began in the mid-twentieth century. Chaulmoogra oil—derived from the chaulmoogra tree—seems to be irrelevant to DDS, a synthetic drug formulated in the early twentieth century by German chemists. However, a deeper examination of twentieth-century leprosy medicine reveals that the medical practices organized around DDS were a metamorphosed form of colonial leprosy medicine. Dr. Heiser’s Asian tour was among the earliest in a series of events ultimately leading to the introduction of DDS. The idea and practices of voluntary treatment, proposed mainly by leprosy workers using chaulmoogra oil, were realigned by DDS to become the prevention strategy of treatment as the measure of control. In this article, I show that chaulmoogra oil created the colonial research and management infrastructure for DDS. DDS did work, but it worked because chaulmoogra oil had already produced the conditions for a cure. 1
The leprosy eradication campaign using DDS mass treatment that followed the Second World War was unusual. There are few diseases for which population and individual levels of intervention are so deeply dependent on each other and on a single measure—drug administration. For instance, we can consider malaria and smallpox, two diseases for which global eradication became the goal during the postwar period. Smallpox and malaria eradication relied on administering interventions before the diseases occurred, such as vaccinations or spraying pesticides to kill the insect that spreads the disease. By contrast, leprosy eradication relied on controlling individuals who were already sick. Sociologist David Armstrong (1995) identifies the rise of surveillance medicine focusing on population health as a general trend in the twentieth century. Whereas hospital medicine conceptualizes disease in a symptom-sign-pathology framework, surveillance medicine sees disease as an evolving phenomenon distributed in the group that can be removed by early intervention—not as a phenomenon in an individual’s body. Modern leprosy medicine distinctively combines the characteristics of hospital medicine and surveillance medicine: it attempts to control diseases in the group by intervening in those showing symptoms and signs. As such, symptoms and signs are not just linked to the underlying pathology in the individual bodies but also to the welfare of the community.
Although the literature on the history of leprosy in nineteenth- and twentieth-century empires is rich, not enough is known about the relationship between colonial medicine and the development of postwar leprosy control measures. This literature reveals that in colonial societies, fear of racial degeneration and the perceived need to maintain a healthy labor force led to various policies to control leprosy, including compulsory segregation in some countries (Edmond 2006; Vongsathorn 2012; Snelders, van Bergen, and Huisman 2021). In the first half of the twentieth century, leprosy became an archetypal tropical illness that had to be defeated with the assistance of Western know-how. This encouraged colonial medicine to develop as a broad enterprise, including missionary activity, modernization, and the protection of health and welfare of indigenous peoples (Worboys 2000; Wang 2007). By the early 1930s, the transnational circulation of knowledge on leprosy and a global apparatus for leprosy research was common (Vollset 2013). However, studies on the formation of postwar leprosy eradication campaigns using the mass administration of DDS and its colonial origins are lacking. 2 This article provides an analysis of the role DDS played in the continuation between the colonial and postwar eras in a way that contributes to actor-network theory’s conceptualizations of how materials act (de Laet and Mol 2000; Latour 1988, 1993, 1999; Latour and Woolgar 1986).
Since the 1990s, scholars have described the mutually sustaining relationship between colonialism and medicine (Vaughan 1991; Lyons 1992; Arnold 1993; Marks 1997; Hunt 1999). Other studies see colonialism as an incidental background for the practice of medicine (Harrison 2005; Ernst 2007). Whether and how context matters to the practice of medicine and health experiences still requires further discussion. To this goal, there have been calls to provincialize science and technology studies (STS) (Law and Lin 2017), to integrate geographical differences into the narrative of the making of modern medicine (Hodges 2012), and to investigate “postcolonial technoscience” to reveal the patterns of the local transformations giving rise to global claims (Anderson 2002). I contribute to this line of discussion by identifying the colonial origin of the first effective drug for leprosy—DDS, illustrating how local workers in multiple places collectively made colonial medicine metamorphose into global health. I start with colonial leprosy doctors’ work in the early twentieth century and follow the actors to Hong Kong and Taiwan, focusing on the idea of a cure and looking for all the transformations this idea went through.
Instead of showing the influence of the center on the peripheries, I see Hong Kong and Taiwan as two contact zones in the global leprosy network that started to take shape in the early twentieth century and fully congealed after the Second World War. Since the introduction of DDS, patients were registered in treatment programs to promote adherence. The number of patients on treatment were reported and collated to produce national and global prevalence statistics. The concept of “the elimination of leprosy as a public health problem” was initially developed in East Asia, where the declining prevalence was noted in various countries in the late 1980s (Saunderson 2008). Highlighting the many forms of contacts, interconnections, and circulations that facilitated and were maintained by leprosy medicine, this article illustrates that the postwar East Asian context profoundly shaped the practice of medicine. Hong Kong and Taiwan’s joining in the global leprosy network showed that DDS worked independently from the researchers who first introduced it. As such, the fact that leprosy was curable with DDS became a statement embedded in a network long enough to make it difficult to be challenged. The efforts of the actors in Hong Kong and Taiwan—in Latour and Woolgar’s (1986) terms—helped stabilize a scientific statement. As a result, postwar leprosy control linked the lives of sick individuals to the welfare of the community.
Chaulmoogra Oil and the Colonial Infrastructure for a Cure
The use of chaulmoogra oil marked an important era of leprosy. For the first time, some leprologists were confident they had found a specific treatment for the disease. In the 1920s, when the debate about forced segregation versus voluntary treatment was salient, the belief in chaulmoogra oil as a cure became the rationale for voluntary treatment. This period also saw the globalization of the leprosy problem and formation of international collaborations.
In the late nineteenth century, intensified colonization activities made leprosy a visible threat, prompting a range of responses. In the British medical community, constitutional explanations for leprosy were gradually giving way to bacteriological ones (Edmond 2006). For the American colonies, embracing a bacteriological discourse was more natural (Anderson 1992). At the beginning of the twentieth century, we see the more scientifically oriented British physicians in India and pioneering American colonial doctors in the Philippines working together to find a cure for leprosy. A common focus for these medical workers was chaulmoogra oil, which came from the Eastern materia medica.
Having a long history in Burma, India, and China, chaulmoogra oil began to receive more attention from medical professionals in Europe and the United States at the turn of the twentieth century, when it was tried at the Louisiana Leper Home in Carville (Parascandola 2003). The remedy was reintroduced to the East through colonization, beginning in the Philippines. In 1902, Culion Island was chosen as the site for a leprosy colony that would accommodate 2,000-3,000 patients and would carry out a form of biological and civic transformation in which “savages” might become social citizens. In 1905, Victor Heiser, as Director of Health for the Philippines, urged all medical officers to take a census of the patients in their region and report their findings to him (Anderson 2006). In 1908, Heiser visited the Louisiana Leper Home and learned techniques for the oral administration of chaulmoogra oil, arranging for the Louisiana method to be given a thorough trial in the Philippines (Parascandola 2003). At Culion, for the first time, leprosy patients became the subjects of intensive surveillance and discipline rather than being unproductively confined, such as in Molokai, Hawaii (Anderson 2006). Internationally, the Culion leper colony became a famous nationalist model of leprosy management (Leung 2009).
Heiser experimented with different preparations of chaulmoogra oil. While oral administration tended to have a nauseating effect on the patient, hypodermic injection was also difficult because the oil was not satisfactorily absorbed. Eventually, Heiser made progress with hypodermic methods after several years of trying (see Figure 2). When Heiser visited the Pulau Jerajak Leper Colony in 1915, he brought enthusiasm about his hypodermic methods and recommended that doctors and patients to adopt the newest remedy.

Picture of patients at Culion waiting to receive injections of chaulmoogra oil. Rockefeller Archive Center.
At the same time that Heiser was experimenting with hypodermic injections, British doctor Sir Leonard Rogers, working in India, was making progress in treating patients with acids isolated from chaulmoogra oil. The two men corresponded frequently about their clinical observations and preparations of chaulmoogra oil. In July 1919, Heiser wrote to Rogers to tell him of recent treatment results and express his optimism about eradicating leprosy: I am just in receipt of a report from Hawaii, of which I enclose you a copy, giving the results of treating leprosy by the use of an ester of chaulmoogra oil. This work also looks encouraging. It is good news to learn that you still maintain your interest in the treatment of leprosy. If we could only find out something about the transmission of the disease perhaps it would not be too much to hope for its ultimate eradication. (Heiser to Leonard Rogers, July 8, 1919)
Rogers reconceptualized the leprosy problem, believing it could be addressed by discovering and treating new infections and enabling the full use of remedies. He came up with a “most rapid method of reducing leprosy incidence,” which consisted in “the examination of the households and other contacts of known cases every few months for five to ten years to enable the great majority of infections arising from them to be detected and cleared up by treatment in the early amenable stages” (Rogers 1934, 3-4). This plan was first attempted on Nauru Island and in Sudan, and Rogers gained a favorable impression of the results. Later, he reiterated that “it is clear that the nearer the successful Nauru plans of regular thorough examination of all the inhabitants can be carried out in any infected area, the more rapidly will leprosy be reduced and eventually eliminated” (Rogers 1945, 5).
Although Rogers was a strong proponent of voluntary treatment, segregation was the prevalent approach in the 1920s. In 1928, the first official meeting of the Leprosy Commission of the League of Nations was held in Paris. Medical advisors expressed their doubts about the effectiveness of chaulmoogra oil and dismissed Rogers’s views about therapeutics as unsound. The committee members also argued that leprosy was a “technical” issue, which excluded missionaries. In the second meeting of the Leprosy Commission in Bangkok in 1930, the commission was again split, with one group focusing on special treatment and the other on isolation. The final report was a compromise, recommending simple and elastic laws focusing on notification and isolation of bacteriologically positive cases in state facilities in accordance with the laws of that country (Vollset 2013). After the Bangkok meeting, the commission members traveled together to Manila to attend the Wood Memorial Conference. Again, the conference made unprecedented efforts to reach a compromise. The conference also marked the establishment of the International Leprosy Association as well as a medical periodical, the International Journal of Leprosy (Long 1967; Vollset 2013).
Until then, leprosy had mostly been a concern for missionaries, who treated leprosy patients isolated at colonial institutions for evangelism purposes (Vaughan 1991; Kipp 1994). From 1915 to 1930, leprosy became a scientific object requiring global collaboration and state intervention. In the 1920s, British medical missionaries devoted a lot of effort to developing a cure for leprosy. Their initiatives were doubted by leprologists advocating compulsory segregation. Some leprologists even proposed excluding medical missionaries from formal power. The main meetings around 1930 sought to reach a compromise between the two camps of voluntary treatment and compulsory segregation. All of the main developments—leprosy as a contagious disease, therapeutics for leprosy, the nationalist model of leprosy prevention exemplified by Culion, and dispensaries—constituted elements that would later be reorganized by DDS into a new set of practices. By the mid-twentieth century, the compromise position of the 1930s had moved on to treatment as the key control measure.
DDS and Treatment as the Control Measure
DDS began being used to treat leprosy in the mid-1940s and was soon accepted by the international community. Much like the French hygienists expecting something important from Pasteurism (Latour 1988), the leprosy community immediately accepted early DDS trials in India and Africa as favorable. With DDS, ideas of treatment as control and dispensaries aiming at early detection became consensus. Leprosy also became a more standardized disease, and its population aspect was highlighted.
As medical workers started to doubt the value of chaulmoogra oil, some workers turned to sulphone drugs, which drew the attention of medical workers for its antibacterial effects. 3 In 1941, Dr. Faget initiated Promin treatment at the Carville Leprosarium in the United States. The first to use DDS, the parent material of Promin, in leprosy were Robert Cochrane and his colleagues at the Lady Willingdon Leprosy Settlement in Chingleput, India. Learning of its use in cattle, they obtained a 25 percent suspension of DDS in peanut oil, using it on a small group of patients by subcutaneous injection in a dosage of 5 cc. twice weekly (Doull 1963). In 1947, Dr. John Lowe at the Uzuakoli Leper Settlement in Nigeria began to administer DDS orally (Lowe 1950), a practice that later became widespread.
DDS was immediately accepted and belief in its effectiveness facilitated the idea of treatment as a control measure. At the Rio de Janeiro meeting in 1946, there was already a general tone of hopefulness, despite the feeling that the time had not yet come to fully assess the healing power of the new drugs (Muir 1947). In 1948, the Fifth International Leprosy Congress in Havana recommended the use of DDS to treat leprosy. In 1953, the first WHO expert committee on leprosy was held. The committee proposed the idea of treatment as a control measure, pointing out that “modern treatment, which effectively reduces the infection in leprosy patients, and therefore their infectiveness, is regarded as the most potent generally applicable weapon now available in the control of the disease” (WHO 1953, 7). The report also recommended establishing dispensaries aiming at the early detection and treatment of cases. Isolation was still seen as important in fighting leprosy, given that it was applied with discrimination and combined with education and effective treatment. The ideas of treatment as a control measure and dispensaries aiming at early detection and treatment were not new. They had been proposed by Leonard Rogers, who advocated chaulmoogra oil. Now DDS, cheap and easy to administer, became the new actor.
Robert Cochrane, a pioneer of DDS, returned to England in 1951 and assumed the role of an expert by providing training and advice. In 1953, using his salaries from the various emoluments he received, Cochrane established the Leprosy Research Centre, which provided briefings and instructions for students and scientists. The training courses used a demonstration set composed of 108 color transparencies illustrating the whole pattern of the disease and showing the clinical variations in the African, Mongolian, and Caucasian races. Students were taught that leprosy is an example of a disease in which the host–parasite relationship is of paramount importance, showing how histopathology always mirrors the clinical condition (Cochrane 1964). The Leprosy Research Centre was active in promoting leprosy research through its various capabilities. By offering a diagnostic service, the Centre facilitated the trial of drug B.663 carried out by Dr. Stanley Browne of the Research Station at Uzuakoli (Cochrane n.d.). In addition, with the Head of the Department of Dermatology at the Slade Hospital, Cochrane discussed building a unit for treating leprosy cases diagnosed in the UK, which enhanced scientific studies for four decades starting in the 1960s (Robertson and McDougall 2005). By the 1960s, there were visible flows of personnel, drugs, and biopsy sections among the research centers in the United States and the UK and various leprosaria in Asia and Africa.
The laboratory evidence for the existence of Mycobacterium leprae started to appear after the leprosy eradication plan based on understanding of bacteria had already been in practice in many countries. Explanations about how DDS works came even later, in the 1970s (e.g., Mier and van den Hurk 1975). The postwar history of leprosy conforms to Latour’s observation of the construction of scientific fact. The experimenters begin by claiming that there is an actor whose competence is defined by the performances it undertakes. Then, they manipulate the environment to allow the actor to work (Latour 1999). Based in the metropoles, leprosy workers with experience in the colonies, such as Robert Cochrane, assumed the role of experts providing advice for research and prevention work, especially in East Asia. There, once DDS mass administration began, leprosy became an entity that existed not in individual bodies but in the population and that could be removed over time through intervention.
The First Coming Through of DDS in Hong Kong
In Hong Kong, since the 1870s, deportation had defined the colony’s management of leprosy. An exception was the Kennedy Town Leprosarium, which was established in 1938 as an undesirable and temporary solution to the influx of leprosy, pending the return of peace on the mainland. Immediately after the war, deporting all Chinese leprosy cases was proposed again but rendered unfeasible by the closure of the mainland borders following the establishment of the People’s Republic of China (PRC). The government responded by erecting a temporary leprosarium on the western edge of Hong Kong Island, ultimately paving the way for the Hay Ling Chau Leprosarium (Ham 2012). Hay Ling Chau was different from previous leprosy facilities in Hong Kong, which were temporary practices put in place only when deportation was not feasible. It was a well-designed facility that aimed to solve the leprosy problem. Functioning between 1951 and 1975, Hay Ling Chau was seen as an experience other East Asian countries could learn from.
Recalling the establishment of Hay Ling Chau, Neil Fraser highlighted 1949 as a turning point. As secretary for China in the Mission to Lepers, Fraser toured mission leprosaria throughout China, along with the Secretary of the American Leprosy Missions Inc. in 1948. At this time, the possibility of Hong Kong becoming a center for mission work was still mooted. Following the coming to power of the communists in China in 1949, refugees fled to Hong Kong, and the government was no longer able to send leprosy patients back to China. Leprosy work in China also had become very difficult because local officials had used their newly won authority to close down work in the homes established by the Mission to Lepers. During the turmoil sweeping aside existing leprosy work in the mainland, the Mission to Lepers started work in Hong Kong. In December 1950, 161 patients were transferred from the crowded wards at Tung Wah Hospital to the newly built Sandy Bay Centre. Fraser took over as medical superintendent (Fraser n.d.).
Along with Olaf K. Skinsnes, a newly arrived member of the University of Hong Kong’s Department of Pathology, Fraser designed a leprosarium that would administer the new sulfone therapy. Skinsnes and Fraser considered that the situation in Hong Kong needed a specialized institution as opposed to general hospitals. The best leprosaria of the time were institutions built as asylums and evolving into treatment centers. They found that there was little information about the needs and design of the institution they were considering. For the design, they identified the main questions as those “relating to the size of the institution needed, the relationship of effectiveness to size and problems of management, and the proportion of infirmary beds to ambulatory patient housing” (Skinsnes and Fraser 1969, 183). Skinsnes and Fraser conducted a two-year survey regarding the magnitude of the leprosy problem in Hong Kong. They concluded that there were perhaps 6,000 persons with leprosy in the Hong Kong population, which was estimated at 2.36 million. Based on the study, Skinsnes and Fraser designed the Hay Ling Chau Leprosarium as a facility with 540 beds, which liaised with fourteen outpatient clinics. They later described their experience as “the design of a leprosarium in the context of its functions as visualized under therapy potentials available in the mid-twentieth century” (Skinsnes and Fraser 1969, 193).
Nun Island was chosen as the site for the leprosarium (see Figure 3). The island was renamed Hay Ling Chau (喜靈洲), a choice that appeared to the Hong Kong Auxiliary to be felicitous due to its assonance and meaning because it readily suggests “healing” (Hong Kong Auxiliary 1951a). By May 1951, a detailed examination of all patients was undertaken. In the same year, a plan for the foundation of the Maxwell Memorial Hospital (see Figure 4) was drawn following the death of Dr. J. L. Maxwell in Hangchow, China, from whom the Hong Kong Auxiliary had hoped to receive help in the development of Hay Ling Chau (Hong Kong Auxiliary 1951b).

Map of Hay Ling Chau (originally “Nun Island”). The Leprosy Mission (TLM).
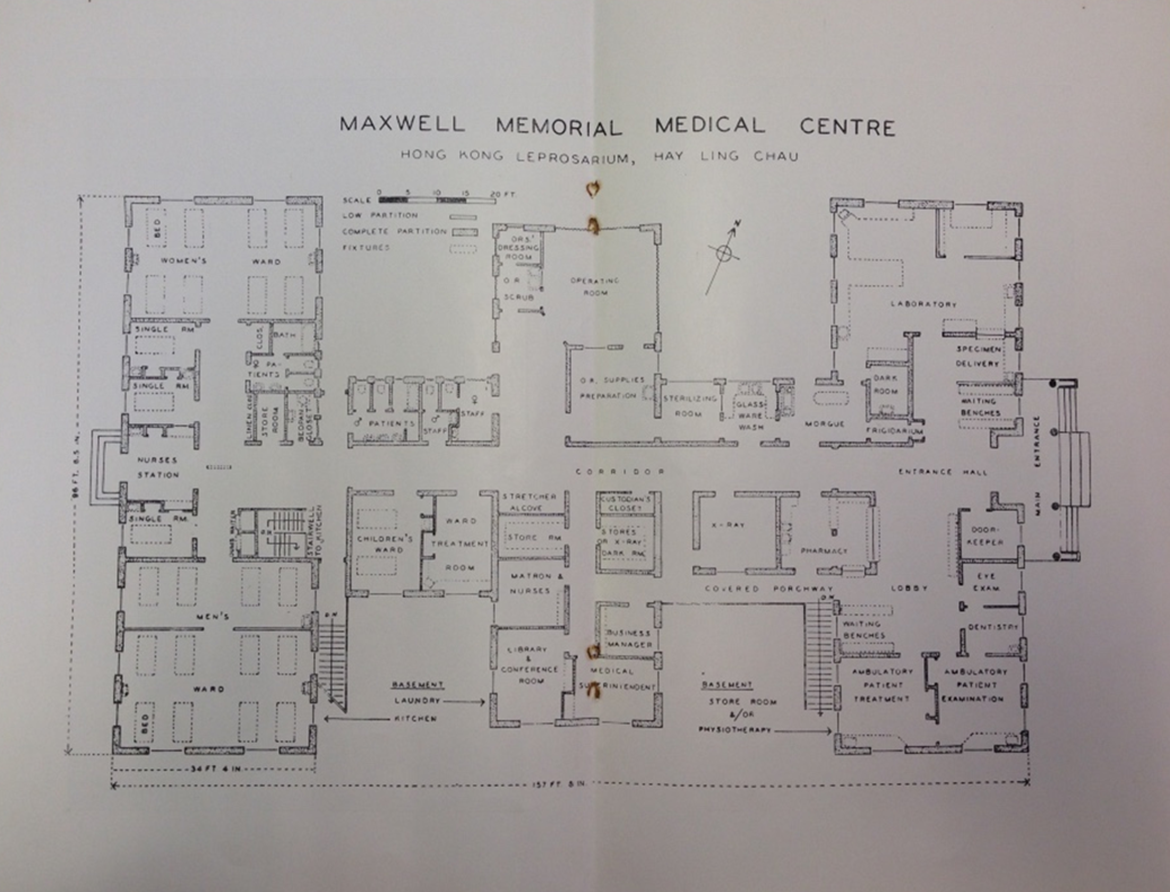
The plan of the Maxwell Memorial Medical Centre. TLM.
Hay Ling Chau was established with the confidence that DDS worked. Patients were regularly counted, measured, and examined so that they could be assigned a status as negative or positive for bacteria. By the early 1960s, it was believed that with timely treatment, leprosy could be cured after one to five years (Fraser 1962). During Hay Ling Chau’s first ten years, more than 300 patients were deemed as having recovered and returned to their homes ( Wen Wei Pao 1962). The leprosy work at Hay Ling Chau is a good example of the concept of the third trial in Latour’s analysis of the construction of scientific reality (Latour 1999). After pointing out to the scientific community that there are characters to be explained and demonstrating that a certain actor is in action, what remained for scientific reality to take shape was a third trial—to convince the scientific community that an event took place independently of their wishes (Latour 1999a, 123). Hay Ling Chau served this function of proving that DDS worked even outside the carefully monitored trials led by Cochrane and other leprosy researchers. According to their observation in 1954, Skinsnes and Fraser found that acid-fast stains of bacilli from patients under sulfone therapy for four or more months demonstrated marked morphologic changes. A decrease in the number of bacilli in the tissues of treated patients reinforced their belief (Skinsnes and Fraser 1969, 184).
The routinization of treatment and discharge of patients at Hay Ling Chau took place at the same time when the Leprosy Research Centre in London was established. This was not a coincidence, rather the result of the consolidation of an international leprosy community. In 1955, Robert Cochrane visited Hay Ling Chau during his trip to Korea and was impressed with the set-up (Fraser to Donald Miller, March 25, 1955). Fraser went to London and attended training courses at the Leprosy Research Centre. Hay Ling Chau was a laboratory for experimenting with the modern method as well as a place for scientists to confirm the correspondence between the world and the representations they had constructed in their research centers. 4 When the Hong Kong Auxiliary reviewed the process through which Hay Ling Chau was established, it noted that the usual pattern of developing research and teaching facilities about leprosy after the establishment of a leprosarium regarding providing clinical material had been reversed (Hong Kong Auxiliary 1951a, 10).
Skinsnes and Fraser (1969) saw the experience at Hay Ling Chau as potentially useful to other countries (p. 183). Fraser actively promoted the Hay Ling Chau experiences, such as connecting East Asia with the UK and the United States. In 1955, both Fraser (as Secretary for Hong Kong and the Far East of TLM) and Cochrane visited South Korea and made recommendations to the government, which was trying to segregate patients at the time. Fraser submitted plans to the Minister of Health for a mobile teaching and research unit, with the ultimate goal of segregating patients in suitable provincial colonies. Fraser and Cochrane also met with Western medical workers in South Korea to discuss what could be done to follow up on the meetings. They agreed to invite representatives of the American Korean Foundation, the Mission to Lepers, and the American Leprosy Missions to form a Leprosy Committee that would keep in close touch with the Ministry of Health and do its best to implement Cochrane’s recommendations (Fraser to Donald Miller, April 30, 1955). Following the same pattern, in postwar Taiwan, the collaboration between government, missionaries, and organizations related to US Foreign Aid also shaped the transformation of leprosy prevention.
Mass Campaign in Taiwan, a Society That Must Be Defended
In Taiwan, systematic and large-scale intervention started after the Second World War. With the collaboration of government and missionaries, and with funding from US aid, the leprosy prevention system in Taiwan came into shape by 1960. It comprised two leprosaria and thirteen local clinics. This system was designed to execute DDS mass administration and actively reach out to patients. It extended both the colonial initiatives for leprosy prevention and the new mode of leprosy prevention that the Republic of China (ROC) established in the early twentieth century, manifesting the particular context of postwar Taiwan.
Leprosy had been a concern for missionary medicine and for the Japanese colonial government. In 1925, medical missionary George Gushue-Taylor became the superintendent of the Presbyterian MacKay Memorial Hospital in Taihoku. Having previously treated leprosy patients in Tainan, he now started his own antileprosy enterprise. In 1927, with the help of the Mission to Lepers, a leprosy dispensary was formally opened opposite the hospital. With both donations and government funds, Gushue-Taylor established the Happy Mount Leprosy Colony in 1934. A simultaneously medical and religious environment, the Happy Mount was designed to be a model village promoting contemporary Western Christian civilization as well as an alternative to the compulsory segregation policy adopted by the Japanese government (Wang 2007). The Japanese government passed its first leprosy law in 1907, seeing leprosy sufferers as a special kind of vagrant to be confined and leprosy treatment as a form of social welfare (Burns 2019). Compulsory segregation was not introduced to Taiwan until the late 1920s (Fann 2009). In 1930, the government’s Happy Life Hospital (Rakusei-in, 樂生院, Losheng Sanatorium) opened, with a capacity of one hundred permanent patients and fifty outpatients (Wang 2007).
At the same time, the elites of the newly established ROC were experimenting with a new mode of leprosy prevention first developed in the mainland—the “nationalist-hybrid model.” In the 1920s and 1930s, China saw heightened nationalism amid political turmoil following the development of the Chinese Communist Party. The elites called for compulsory segregation and saw Culion Island and the compulsory segregation policies used in Taiwan and in Australia as ideal. Lacking the necessary funds, the ROC government had to work with missionaries. During the Chinese Civil War between 1945 and 1949, the ROC government usually sought collaboration with foreign and native Christian organizations. The International Mission to Lepers not only helped with establishing a leprosarium but also sent medical professionals and the new drug Promin (Leung 2009).
After the ROC retreated to Taiwan, the Losheng Sanatorium became an organization under the Provincial Department of Health—the main public health institution in Taiwan, fusing the initiatives of Gushue-Taylor and those of the Chinese elites. In 1952, US foreign aid started to be directed to the Losheng Sanatorium to build accommodation for soldiers diagnosed with leprosy (Fann 2009). Starting in the 1950s, leprosy in Taiwan saw government and missionaries collaborating and being influenced by US aid.
One of the first things that the ROC government and missionaries worked together on was introducing the leprosy prevention method characterized by case finding and the mass administration of DDS that was pioneered in Hong Kong. In 1950, Gushue-Taylor, who was forced to leave Taiwan during the Second World War, was considering how to best use the funds he collected for leprosy work in Taiwan, along with how leprosy work in Taiwan should continue (Gushue-Taylor to Rev. H. C. Etter, May 22, 1951) (Figure 5). Hugh MacMillan, a colleague of Gushue-Taylor, insisted on the need for specialist leprosy workers for Taiwan and asked Dr. Fraser for help (MacMillan to Donald Miller, February 28, 1951). In reply, Fraser offered training a doctor (Fraser to George Gushue-Taylor, August 15, 1951). As a result, Dr. Chao Yung-fa, whose father was a physician at Happy Mount, was chosen to receive training at Hay Ling Chau for a year under the sponsorship of the Sino-American Joint Commission on Rural Reconstruction (JCRR), a government commission specifically established to direct US foreign aid. After returning to Taiwan in 1952, Dr. Chao worked at the Losheng Sanatorium as the head of the Department of Prevention. Applying the methods for treatment and examination he had learned in Hong Kong, Dr. Chao conducted slit-skin smears for all the patients to keep track of treatment results. He also helped establish an outpatient clinic (Losheng Sanatorium 1955).

Dr. George Gushue-Taylor with male patients in the waiting room at a leprosy dispensary in Taihoku Formosa in 1927. TLM.
In the early 1950s, more resources were directed to Losheng, and the various actors concerned with leprosy convened to modify the prevention policy. Chen Zong-ying, a military physician, assumed superintendency for the sanatorium. Being a Christian and having connections with the JCRR, he was instrumental in the transformation of the leprosy prevention system. With US aid funding, Losheng built more accommodation and consult rooms for various medical specialties (Fann 2009). On January 23, 1954, the 90th Meeting of the Coordination Committee on Foreign Aid in Medicine and Health was held in Taipei. Present were people from several international health organizations, members of the JCRR, Donald Miller, Neil Fraser, George Gushue-Taylor, and Chen Zong-ying. Fraser suggested that establishing village clinics for the treatment of early leprosy would be quite effective, that there had to be more doctors and nurses to treat patients, and that the segregation of patients should be reexamined. He also addressed the First Annual Meeting of the Taiwan Leprosy Relief Association (TLRA) in the same year, emphasizing the benefits of modern treatment and maintaining that leprosy can be cured.
In the mid-1950s, a leprosy prevention system designed for executing DDS mass treatment took shape. In 1955, Losheng hired US doctor Richard Hofstra as a consultant doctor. That same year, he started a clinic on the islands of Penghu (MacMillan 1994). In the following two years, four more leprosy clinics run by the TLRA were built in Southern Taiwan (Fann 2009). By 1963, there were thirteen leprosy clinics in Taiwan run by the government or the TLRA (Losheng Sanatorium 1963). These clinics dispatched DDS and referred serious cases to Losheng or the Happy Mount Colony. The missionaries working for the clinics were equipped with motorcycles so they could visit patients who lived too far away to attend the clinic. In 1960, with the help of the JCRR and WHO, Losheng organized two mobile teams that toured all the counties in Taiwan to discover new cases. In 1961, Losheng established a database of all the cases in Taiwan to be followed up on (Fann 2009).
The leprosy prevention system, consisting of a central leprosarium, local clinics, and mobile teams, became the main apparatus through which leprosy was eradicated in Taiwan. The years 1954-1963 were important for leprosy work in Taiwan. In 1954, the government launched its ten-year plan for leprosy prevention. It also established the Leprosy Prevention Committee in 1959 and conducted outreach to find new cases starting in 1960. The cases discovered in 1960–1988 totaled 3,753, and 1,184 entered the sanatorium. Many patients continued to stay in the sanatorium, even after they presented negative bacteria results. In 1981, there were 784 patients in the Losheng Sanatorium, 667 of whom (85 percent) had lived in the sanatorium for more than ten years. In that year, the average number of years living in the sanatorium was twenty one and a half (Losheng Sanatorium 1991).
The leprosy prevention system enacted a new conceptualization of leprosy and how to eradicate it. Leprosy now existed as an evolving phenomenon that could be removed by early intervention. The prevention system attempted to control leprosy in the group by intervening in bodies presenting symptoms and signs. The rationale was that, by identifying and treating them, leprosy would be gradually removed. This prevention measure had to be practiced during a very narrow time frame in individual cases—after the individuals were already sick and before they were crippled. Behind this measure was confidence in DDS. Losheng’s superintendent, Chen Zong-ying, for example, argued for treatment as a way of prevention. He claimed, “today medicine is very advanced and leprosy can be cured if treated early” (Losheng Sanatorium 1963, 7). Each year, the Losheng Sanatorium produced patient statistics, including the number of inpatients and outpatients, cases newly discovered, patients’ age groups, duration of hospitalization, and number of inpatients with deformities. The result of slit-skin smears for each patient was also recorded. As the numbers of patients treated and results of their bacterial examinations were collated into yearly national statistics, the individual body was connected to the health of the group. Ironically, whereas leprosy became eradicated, many patients, whom DDS promised to cure, never left the sanatorium.
In the early twentieth century, when China was pursuing modernization, Chinese elites did not challenge the racialized view that associated leprosy with China and, instead, hoped to control leprosy through segregation (Leung 2009). After the Chinese Civil War, the elites from the mainland brought to Cold War Taiwan the nationalist project and their connections with the United States. Thus, leprosy prevention in postwar Taiwan extended the elites’ attitude and aimed at removing a disease symbolizing backwardness. At the same time, the influence of missionaries continued, connecting Taiwan with Hong Kong, with the latter providing know-how of leprosy prevention. Unlike in the Japanese colonial era, when leprosy workers were divided into compulsory segregation and voluntary treatment camps, in postwar Taiwan, the government and missionaries agreed on DDS mass treatment. The transformed leprosy prevention system actively looked for patients and, by identifying and treating sick individuals, aimed to eradicate leprosy from Taiwan. For this to happen, many elements had to be in place: collaboration among various actors, and the colonial missionary initiative, the ROC nationalist-hybridization model of leprosy prevention, the postwar condition, and the availability of DDS. However, DDS was highlighted as the single actor that worked.
Connected through DDS
Leprosy has become a disease belonging to the past. I have illustrated how the formation of a research and management infrastructure along colonial lines relegated leprosy. Despite the actors’ sincere beliefs, DDS as a cure for leprosy did not act alone but worked in combination with other factors. I trace the proposition that leprosy can be cured to the conditions constructed by early twentieth-century leprologists who used chaulmoogra oil. In the 1950s, when Western researchers were setting up research centers in the metropoles, the Hong Kong leprosy prevention system was designed based on the assumption of DDS as a cure, and that Hong Kong could be a training center in Asia. Using what they learned from Hong Kong, leprosy workers in Taiwan assembled practices, ideologies, and infrastructures previously associated with chaulmoogra oil, realigning them with DDS. By the late 1950s, leprosy workers in Hong Kong, Taiwan, South Korea, and Japan were visiting each other. Rather than its inherent properties, DDS gained its ability to act because patients, laboratories, professions, interest groups, and technologies were tied to the fate of the proposition of an effective drug. Following this process in which the proposition became stabilized reveals three important aspects of the production of a global claim.
Leprosy control maintained a particular kind of biopolitics in East Asia. Examining the biomedical discourse in Africa, Vaughan (1991) finds that there are real differences between the nature of the colonial power/knowledge regime and that described by Foucault (1965, 1980). As Vaughan has found of Africa, the knowledge/power regimes in Hong Kong and Taiwan are both distinct from their Western counterparts. Facing the rise of the PRC, each developed leprosy management practices to produce a healthy population. In both Hong Kong and Taiwan, the medical discourse’s effect of distancing the “Other” within society had not been central to control because the problem of defining a normal population was more important. Leprosy medicine enabled both governments to see patients previously to be segregated as having the potential to become productive social members again. Being able to control leprosy marks a society as civilizing and catching up with the West—an imperative originating in colonial life politics and motivating modern leprosy medicine to evolve. The history of leprosy demonstrates that the proposition “we have never been modern” (Latour 1993) keeps raising the question about whether and how we are always already decolonizing. 5
Footnotes
Acknowledgment
The author thanks Wen-Hua Kuo, Sean Hsiang-lin Lei, Stefan Timmermans, Harry Yi-Jui Wu, and the two anonymous reviewers for their helpful comments. The author also thanks the archivists of the Leprosy Mission International and the Rockefeller Archive Center for their invaluable help.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was funded by the Ministry of Science and Technology, Republic of China (MOST 106-2628-H-007-002-MY2).
