Abstract
Background. There is evidence from laboratory and animal studies that fish oil–containing intravenous lipid emulsions (FOC-IVLEs) have a beneficial effect on inflammation and the immune response, suggesting a possible clinical benefit. Clinical studies of FOC-IVLEs have reported mixed results. The aim of this review is to present findings from recent randomized controlled clinical trials and other quality clinical studies investigating the effects of administering intravenous fish oil alone or as part of a multilipid emulsion and to examine the quality of these studies in an objective, evidence-based manner. Methods. Studies comparing FOC-IVLEs with other IVLEs in adults were included. Thirty-four clinical studies were evaluated: 19 investigated levels of inflammatory and immune markers as an endpoint; 13 investigated rates of infection or sepsis; 3 investigated clinical outcomes in septic patients; and 29 investigated general clinical outcomes. Results. There was conflicting evidence for a beneficial effect of fish oil on levels of inflammatory and immune markers and some evidence that fish oil decreased the rate of postoperative atrial fibrillation. Studies generally reported few statistical differences in clinical outcomes and rates of infection and sepsis with FOC-IVLEs as compared with other IVLEs. The quality of reporting was generally poor, and the presented evidence for comparisons between FOC-IVLEs and other IVLEs was inconclusive or weak. Conclusions. There is very little high-quality evidence that FOC-IVLEs have a more beneficial effect than other IVLEs on clinical outcomes in adult patients.
Intravenous lipid emulsions (IVLEs) are an important component of parenteral nutrition (PN) as a source of essential fatty acids and as an energy source in patients who are unable to tolerate nutrition through other routes. Traditionally, IVLEs for PN were derived from soybean oil (SO).1,2 However, SO is high in ω-6 polyunsaturated long-chain triglycerides (LCTs), and concerns have been expressed that PN with SO may have an immunosuppressive effect. SO may also exacerbate the release of proinflammatory cytokines and prostaglandin 2, thus contributing to an increased risk of complications.3,4 To address this potential risk, newer IVLEs have been developed to contain lower levels of long-chain ω-6 polyunsaturated fatty acids (PUFAs) to minimize the possible adverse effects associated with LCTs (Figure 1). One strategy in creating these new IVLEs has been to replace a portion of the SO component in an IVLE with medium-chain triglycerides (MCTs) and/or with olive oil (OO) and/or fish oil (FO). 4 Another strategy has been to use an IVLE supplement derived solely from FO, which is high in ω-3 PUFAs.

Commercially available intravenous fat emulsion products by ω-3:ω-6 triacylglyceride ratio. 5 FO, fish oil; LCT, long chain triglycerides; MCT, medium-chain triglycerides; OO, olive oil; SO, soybean oil.
Evidence from laboratory studies indicate that ω-3 PUFAs may result in lower inflammatory responses. 1 ω-3 and ω-6 PUFAs have competitive metabolic pathways. The metabolism of linoleic acid, a dominant ω-6 PUFA, produces arachidonic acid (C20:4 n-6), which is further metabolized to products with proinflammatory activity.1,6 Therefore, in theory, PN high in ω-6 PUFAs may be detrimental for patients at risk of a pathologic inflammatory response. 2 In contrast, metabolism of eicosapentaenoic acid (C20:5 n-3) and docosahexaenoic acid (C22:6 n-3), which are the ω-3 PUFAs predominant in FO, may reduce the production of proinflammatory eicosanoids in a dose-dependent way: (1) by directly competing with the metabolism of arachidonic acid1,6 and (2) by the nature of eicosapentaenoic acid and docosahexaenoic acid, which are metabolized to products that are less proinflammatory than arachidonic acid. Based on in vitro and animal experiments, the immune and proinflammatory properties of IVLEs rich in linoleic acid, a precursor of arachidonic acid, could have an adverse impact on clinical outcomes (eg, by increasing risk of infection, sepsis, or systemic inflammatory response syndrome [SIRS]).2,7
Despite evidence from in vitro and animal models indicating that ω-3 PUFAs may have a dose-dependent beneficial effect on inflammatory responses, there have been conflicting reports from meta-analyses and systematic reviews regarding the clinical benefits of using FO-containing (FOC)–IVLEs over other lipids. While well-conducted meta-analyses can be key to demonstrating positive or negative effects, inappropriately conducted meta-analyses can lead to skewed results. Of the meta-analyses and systematic reviews conducted, some have reported a statistically significant clinical benefit for FOC-IVLEs.8-11 However, 1 meta-analysis included only 6 relatively low-quality trials, 10 and another not only imputed up to 50% of standard deviations not reported by the original trial but did not correct for possible type 1 errors. 8 Neither of these practices is recommended as good practice by the Cochrane Collaboration.12,13 Other reviews have reported either no significant clinical benefits with FOC-IVLEs or an overall poor quality of included studies with considerable heterogeneity, making it likely that any statistical benefit would be insufficient to recommend the use of FO over other IVLEs.14-17
Given the conflicting results from previous reviews and meta-analyses, we decided to investigate the current published evidence base for the use of FOC-IVLEs for PN. Because of the low quality of most studies and the substantial heterogeneity across studies, it was not feasible to conduct a meta-analysis of the current published evidence base. Therefore, we have instead focused on this qualitative narrative review, which presents the findings from randomized controlled trials (RCTs) and other clinical studies and examines the quality of these studies in an objective, evidence-based manner. We sought to address the following question: Does administering FO alone or as part of a multilipid IVLE improve clinical outcomes?
Methods
Literature Search and Assessment of Study Quality
This qualitative review of the literature on the use of FOC in PN used a comprehensive, preplanned search strategy to locate potentially relevant human clinical studies in English for inclusion. We conducted online searches of MEDLINE and EMBASE (inception to March 16, 2016), using the search terms “fish oil” and “parenteral nutrition.” Articles comparing FOC-IVLEs with other IVLEs with respect to clinical outcomes, infection and inflammation, or immune markers were eligible for inclusion in this review. Articles were excluded if they described results from studies of pediatric and neonatal patients or nonhuman or laboratory studies; if they were based on case studies or case series; if they exclusively used enteral nutrition (EN) in adults; or if they were not written in English. Additional studies were retrieved by searching the reference lists of relevant meta-analyses and systematic reviews.
Study quality was assessed with the Cochrane Collaboration assessment criteria for assessing risk of bias. 18 This tool grades studies as having a low, high, or unknown risk according to the following possible sources of bias:
Random sequence generation
Allocation concealment
Blinding of participants and personnel
Blinding of outcome assessments
Incomplete outcome data
Selective reporting
Other biases
Endpoints Assessed
Key endpoints identified for inclusion were the effects of FO on inflammation and immune response, sepsis and infection, mortality, length of hospitalization, length of stay in the intensive care unit (ICU) and other ICU-related or general outcomes, and cardiac surgery outcomes.
Results
Inflammation and Immune Response
Reported results
A total of 19 studies investigated the effect of FOC-IVLEs on markers of inflammation and immune response. Of these, 12 compared an FOC-IVLE with an SO-based IVLE,19-30 1 with an OO-based IVLE, 31 3 with a mixed MCT/LCT IVLE,32-34 2 with saline,35,36 and 1 with a control group in which patients received nutrition chosen at the discretion of the study clinicians. 37 None of the included studies compared 100% FOC-IVLE with other FOC-IVLEs. Therefore, for the purposes of this review, we henceforth simplify the text by referring to all FOC-IVLEs as FO. The inflammatory markers commonly reported in these studies were the proinflammatory cytokines interleukin 1 (IL-1), IL-6, and IL-8; the anti-inflammatory cytokine IL-10; the cell-signaling cytokine tumor necrosis factor alpha (TNF-α); and the ratio of T-cell lymphocytes CD4 and CD8 (cluster of differentiation). A CD4/CD8 ratio >2 is indicative of satisfactory immune status, whereas impairment in CD4 T-cell regeneration and persistent elevation of CD8 T-cell counts, leading to a lower CD4/CD8 ratio, are associated with immunologic dysfunction.
Based on the results of these included studies, there is conflicting evidence for a beneficial effect of FO on the immune response and inflammation in adults. Six studies cited significantly lower levels of IL-6 with FO than with SO,19,27,29 MCT/LCT,25,33 or saline. 36 In contrast, 10 studies found no significant differences in levels of IL-6 with FO vs SO,20-22,26 MCT/LCT,24,28,32,34 OO, 31 or control nutrition. 37
One study reported significantly lower levels of IL-1 and IL-8 with FO than with MCT/LCT. 33 Another study cited lower levels of IL-1 with FO as compared with SO. 27 In contrast, 2 studies found no significant difference in levels of IL-1 vs MCT/LCT.24,34 One study indicated higher levels of IL-10 and HLA-DR (human leukocyte antigen–antigen D related) with FO than with SO. 30 Another study cited a lower level of IL-10 with FO and no significant difference in HLA-DR. 19 In contrast, 3 studies reported no significant difference in levels of IL-10 with FO than with MCT/LCT28,34 or saline. 36
Three studies observed significantly lower levels of TNF-α with FO than with MCT/LCT 33 or SO.27,29 In contrast, 5 studies found no significant differences in TNF-α levels when compared with placebo, 35 SO,20,22 MCT/LCT, 34 or OO. 31 Two studies found significantly higher CD4/CD8 ratios in patients using a FO/SO combination vs those using SO alone,23,29 whereas another study reported no significant difference for the same endpoint. 30 One study found no significant differences in T lymphocytes, CD3, CD4, CD8, and CD4/CD8 ratio between patients using FO and those using SO. 27
Importantly, 4 of the 7 studies that recorded differences in inflammatory or immune markers with FO over other IVLEs reported no corresponding differences in clinical outcomes19,25,30,33; 1 additional study did not examine clinical outcomes. 23 However, 1 included study noted a lower rate of postoperative infections with FO vs SO. 27 Three studies investigated patients with SIRS, and of these, 2 cited significantly fewer instances of SIRS with FO than with SO.20,29 The third study found no difference in outcomes for patients with SIRS in the intent-to-treat population when FO was compared with SO (length of stay in ICU). 32
Study quality
Although significant differences in the levels of some proinflammatory markers were reported in some studies, it is not possible to determine whether these changes are clinically relevant. None of the studies reported which standard thresholds, if any, were used to assess whether the reported levels of proinflammatory markers were within or outside the clinical reference range for the laboratory in which they were measured.
The quality and strength of evidence were low in most studies, and it is not possible to determine whether studies were adequately powered. All studies had small sample sizes and were likely to be underpowered for an assessment of proinflammatory markers (Table 1). Only 2 of 19 studies cited a sample size calculation based on levels of a proinflammatory marker (IL-6). Of these, 1 study recruited a sufficient number of patients, based on the sample size calculation, but found no significant differences in levels of IL-6 between FO and MCT/LCT. 32 The investigators in the other study predicted that a sample of 95 was required to demonstrate a statistical effect, but only 38 patients were recruited for their study. 33
Findings From Clinical Studies of FO-Containing IVLEs in Adults.
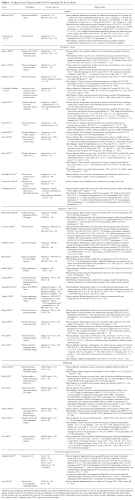
APACHE, Acute Physiology and Chronic Health Evaluation; CD, cluster of differentiation; EN, enteral nutrition; FO, fish oil; GI, gastrointestinal; HLA-DR, human leukocyte antigen–antigen D related; ICU, intensive care unit; IL, interleukin; IVLE, intravenous lipid emulsion; LCTs, long-chain triglycerides; MCTs, medium-chain triglycerides; NR, not reported; NS, not significant; OO, olive oil; PN, parenteral nutrition; SAPS II, Simplified Acute Physiology Score; SIRS, systemic inflammatory response syndrome; SO, soybean oil; SOC, standard of care; TNF-α, tumor necrosis factor alpha.
The majority of studies did not report statistical methods in sufficient detail to allow for an appropriate quality assessment. In general, most authors poorly described their methodology; therefore, the quality of each study was difficult to determine (Figure 2). Most studies included multiple endpoints, but none used tests to assess or correct for possible effects of multiplicity on significant outcomes. Based on the Cochrane Collaboration Assessment Tool, which ranks quality as high, low, or unknown, 18 14 of the 18 included studies with an RCT design were classified as having an unknown to high risk of bias (Figure 2).20-25,27-31,33-35
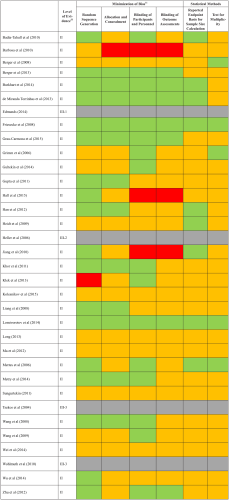
Assessment of quality of included studies. Red, high risk; yellow, unknown risk / not reported; green, low risk; gray, not available.
Conclusions
There is insufficient high-quality data investigating inflammatory and immune markers, which has resulted in inconclusive evidence for a beneficial effect of PN with FO on inflammatory and immune markers as compared with other IVLEs (Table 2).
Potential Effects of FO-Containing IVLEs in Clinical Studies.

CD, cluster of differentiation; FO, fish oil; HLA-DR, human leukocyte antigen–antigen D related; ICU, intensive care unit; IL, interleukin; IVLE, intravenous lipid emulsion; PN, parenteral nutrition; SIRS, systemic inflammatory response syndrome; SO, soybean oil; TNF-α, tumor necrosis factor alpha.
Wang et al 30 reported a significantly higher level of IL-10.
Grau-Carmona et al 40 allowed participants to receive up to 50% of lipids from enteral nutrition.
Tsekos et al 46 conducted an observational study.
Mertes et al 44 did not report statistical comparisons.
Heller et al 52 did not include a control group that did not receive FO. Comparisons were made among doses of FO, and this was an observational study.
Only 4.2% of participants in the Edmunds et al 48 study received FO, and this was an observational study.
Sepsis and Infection in Adults
Reported results
A total of 11 studies were included comparing the effect of FO and other IVLEs on rates of postsurgical sepsis and infection, with 3 additional studies comparing clinical outcomes in patients who had sepsis at study entry. Of these, 9 compared an FOC-IVLE with a SO-based IVLE,19-22,24,26,28-30 2 with an OO-based IVLE,31,38 3 with a mixed MCT/LCT IVLE,30,33,39 and 1 with control nutrition. 37 Sepsis and infectious complications were defined by standardized criteria in 4 studies.19,20,29,38 One study reported on catheter-associated infections, 21 3 studies on infectious events (eg, pneumonia, bacteremia, and abdominal abscesses),24,26,30 and 3 studies on infectious complications without providing details.22,28,33
The majority of studies concluded that administering PN with FO does not reduce the risk of postsurgical sepsis or infection when compared with other IVLEs. One study indicated significantly fewer patients in the ICU with nosocomial infections when parenteral FO was compared with MCT/LCT; however, 50% of the energy contribution from lipids was achieved through EN in this study. 40 Another study cited significantly fewer infections in adult postsurgical patients with FO vs OO (23.1% vs 78.6%, P = .007) 38 ; however, the difference in the percentage of patients with sepsis (the primary endpoint) between the FO and OO groups did not reach significance (7.7% vs 35.7%, P = .1). In addition, of the 23 endpoints investigated, the rate of infection was the only endpoint that showed a significant difference. Moreover, no appropriate statistical methods were used to correct for multiple endpoints. In contrast, 10 studies reported no significant differences in infection rates with FO vs other IVLEs.19-22,24,28-30,32,33
Based on the 3 studies that included septic patients at study entry, there was no significant difference in overall mortality with FO over other IVLEs.31,37,39 One study investigating markers of brain injury and sepsis-associated delirium found no differences between FO and control nutrition. 37 One retrospective study of septic patients cited no differences in relevant inflammatory markers with FO over standard nutrition therapy and no improvement in clinical outcomes. 41
Study quality
In general, the clinical studies were of short duration: 3 studies collected data for 4 weeks,21,32,40 whereas the remainder were completed within 5–8 days. Only 2 studies engaged in long-term follow-up to determine the long-term clinical effects of FOC-IVLEs.37,40
The quality and strength of evidence of most studies were low, primarily due to poor reporting of statistical methods. It is not possible to determine whether studies were adequately powered to assess rates of sepsis or infection. Only 2 of the 12 studies investigating rates of sepsis or infection indicated a sample size calculation that was specifically based on sepsis or infection (Figure 2).20,38 One of the 3 studies investigating septic patients cited a sample size calculation using an outcome relevant to septic patients (S100β level). This study found no differences in markers of sepsis-associated injury or inflammatory mediators between patients receiving FO and those given standard care. 37
The included studies generally did not report the study protocol or methods in sufficient detail to allow for an appropriate quality assessment. Most studies measured multiple endpoints, but none reported using a test to assess or correct for possible effects of multiplicity on significant outcomes (Figure 2). When study quality of RCT designs was assessed via the Cochrane Collaboration assessment tool, 18 10 of the 14 included RCTs were classified as having an unknown or high risk of bias (Figure 2). *
Conclusions
The majority of comparative investigations fail to demonstrate any differences in rates of sepsis or infection after administration of PN with FO as compared with other IVLEs. In addition, current evidence suggests no difference in rates of mortality or brain injury in patients with sepsis at study entry when FO is compared with other IVLEs. Consequently, based on the poor quality of reporting, it is not possible to determine whether the observed lack of differences between FO and other IVLEs is conclusive.
General Clinical Outcomes in Adults
Reported results
A total of 29 studies were included comparing FOC-IVLEs with other IVLEs with respect to clinical outcomes in adults. Of these, 13 compared an FOC-IVLE with a SO-based IVLE,19-22,24-26,28-30,42-44 6 with MCT/LCT,32,33,39,40,45,46 2 with OO,31,38 3 with placebo or saline,35,36,47 and 1 with various control emulsions. 48 Three studies included a comparison group that received no intravenous therapy control,49-51 and 1 study supplemented all patients with FO. 52
Mortality
Only a few of the included RCTs that incorporated mortality as an outcome measure reliably reported a significant difference with FO vs other IVLEs. One prospective observational study indicated increased survival with doses of FO >0.1 and ≤0.2 g/kg/d as compared with doses of FO <0.1 and >0.2 g/kg/d. 52 One retrospective observational study cited a reduced rate of mortality when FO was administered preoperatively or postoperatively as compared with MCT/LCT. 46 In contrast, 15 studies reported no significant difference in the mortality of patients receiving FOC-IVLEs vs other IVLEs or placebo. † It is important to note that underlying rates of mortality were low in most studies; therefore, it is likely that the included studies had insufficient power to identify an effect of FO on mortality.
Length of hospitalization
Twenty-two studies reported results for length of hospitalization; of these, 17 indicated no significant differences between FOC-IVLE and other lipid emulsions, placebo, or other control. Three studies cited a shorter length of hospitalization with FO than with SO.20,29,42 One study reported a reduced length of hospitalization with FO than with MCT/LCT, which was significant after adjusting for age, glucose supply, and Simplified Acute Physiology Score II. 39 Finally, 1 study cited a significantly reduced length of hospitalization with FO >0.05 g/kg/d (P < .001). 52 In contrast, 16 studies found no significant difference in hospitalization with FO than with OO,31,38 placebo,35,50 MCT/LCT,32,40,45 SO,19,24,25,26,28,30,44 or other control.41,51 One study reported no significant difference among treatment groups (FO, SO, OO, MCT, and lipid-free) but did not directly report individual significance values between FO and the other treatment groups. 48
Length of stay in ICU and other ICU-related or general outcomes
A total of 14 studies reported no significant differences in length of stay in the ICU, duration of mechanical variation, or days of parenteral support between FOC-IVLEs and OO, SO, MCT/LCT, ‡ EN/PN,35,36,47 or placebo. 51 In contrast, 1 observational study cited a significantly faster discharge rate from ICU for patients who received FO when compared with those who received lipid-free PN. However, only 4.2% (19 of 451) of patients in this study actually received FO. 48 One dose-response study observed that doses of FO >0.05 g/kg/d were associated with a reduced duration of stay in ICU. 52 In addition, 2 studies investigated the effect of FO on total medical costs but reported no significant difference in cost between FO-containing PN and SO-based PN.20,29
Cardiac surgery outcomes
Four studies investigated clinical outcomes in patients who were given infusions of FO before cardiac surgery to prevent postoperative complications.36,43,47,49 Two of these studies reported no significant differences between treatment arms in general postsurgical clinical outcomes.36,47 Two studies cited a beneficial effect of FO on postoperative atrial fibrillation,43,49 but 1 study reported no significant improvement. 47 Of the 2 studies that cited a beneficial effect on atrial fibrillation, 1 did not incorporate a control group treated with an alternative IVLE. 49
Quality of studies
The quality and strength of evidence for most of the included studies were low, and it is generally not possible to determine whether studies were adequately powered. Most studies had small sample sizes and were likely to be underpowered for an assessment of general clinical outcomes (Table 1). In addition, only 3 of 29 included studies reported a sample size calculation based on 1 of the clinical outcomes investigated.
Most studies did not publish the full study protocol or methodologies in sufficient detail to allow for an appropriate quality assessment. Multiple endpoints were cited in most studies, but only 5 of 29 studies used appropriate tests to assess or correct for possible effects of multiplicity on significant outcomes.32,42,44,45,47 When the quality of RCTs was assessed with the Cochrane Collaboration assessment tool, 18 21 of 27 included RCTs were classified as having an unknown or high risk of bias (Figure 2). ‖
Conclusion
The majority of current published evidence suggests no significant differences in general clinical outcomes, such as length of hospitalization and length of stay in ICU, duration of mechanical ventilation, or mortality, after administration of PN with FO as compared with other IVLEs. Notwithstanding, there is a small amount of low-quality evidence to suggest that FO may have a beneficial effect on postoperative atrial fibrillation (Table 2). Overall, given the poor quality of reporting in the included studies, it is not possible to determine whether the observed lack of differences between FO and other IVLEs is conclusive.
Discussion and Conclusions
The findings from this qualitative narrative review do not demonstrate that FO or FOC-IVLE significantly improves any of the clinical outcomes assessed. There is very little high-quality published evidence that FOC-IVLE has a beneficial effect on clinical outcomes. Although there was some evidence of a positive effect for FO on inflammatory and immune markers, these findings did not translate to improved clinical outcomes. The majority of studies reported little difference in mortality, length of hospitalization, ICU-based clinical outcomes, postoperative complications, or rates of infection and sepsis with FOC-IVLEs as compared with other IVLEs. Most findings were weak and inconclusive, and the published evidence from these studies was poorly reported. These serious quality deficiencies limit the interpretation of the research data and assessment of the applicability of the findings to clinical practice.
A major impression from this review of all adult RCTs was the overall inferior quality of reporting of clinical studies in the field. The absence of published details on study protocols, methodology, and statistical methods means that it was not possible to accurately assess study quality. However, it is likely that most included studies were underpowered and used inappropriate statistical methods. We submit that this variable quality on the collection and interpretation of individual clinical data is a major contributor to the conflicting reports from meta-analyses designed to investigate the benefits of FO.8-10,14-17 It is also important to note that analysis of all adult RCTs according to the type of FOC-IVLE used did not demonstrate any further benefits of FO. All FOC-IVLEs appear to be safe and efficacious energy providers, but evidence for a beneficial effect of FO on infection and other clinical outcomes remains limited and inconclusive. Thus, based on the current published evidence, it is not possible to recommend ≥1 of the available FOC-IVLEs for any specific group of patients.
Recently published joint guidelines from the Society for Critical Care Medicine and the American Society for Parenteral and Enteral Nutrition provide no recommendations regarding the use of 1 IVLE over another. 54 Although the guidelines suggest that FOC-IVLE may be a suitable choice, they concede that the data are derived primarily from observational studies. 54 Notably, a systematic review and meta-analysis conducted by the European Society for Pediatric Gastroenterology, Hepatology and Nutrition Committee on Nutrition 55 concluded that in the neonate and pediatric populations, no recommendation regarding the choice of IVLE could be made, owing to the inadequate evidence. Thus, it appears that the key medical societies are challenged to provide recommendations on the use of FOC-IVLE based on the current levels of evidence.
The strengths of our qualitative review are that we approached the literature search and data collection in a systematic manner and that the evidence gleaned from studies was assessed for its quality per the Cochrane Collaboration assessment criteria. 17 Limitations of this review are that most studies did not publish the full study protocol or methodologies in sufficient detail to allow for an appropriate quality assessment and did not use appropriate tests to assess or correct for possible effects of multiplicity on significant outcomes. Additionally, only English-language articles were included.
Based on this review, it is clear that high-quality and adequately powered RCTs are necessary to determine the true effect of PN with FOC-IVLEs and to investigate whether FOC-IVLEs can result in measurable clinical benefits as compared with other IVLEs.
Footnotes
Financial disclosure:
This study was sponsored by Baxter Healthcare, manufacturer/licensee of Clinoleic. Medical writing assistance was provided by Elise Magatova, PhD, and Tania Dickson, PhD, CMPP, of ProScribe–Envision Pharma Group, and was funded by Baxter Healthcare. ProScribe’s services complied with international guidelines for Good Publication Practice (GPP3).
Conflicts of interest:
A.P.-A. is an employee and shareholder of Baxter Healthcare. O.A. and G.H. have received financial reimbursement from Baxter Healthcare for consultancy work, advisory boards, and speaking engagements. W.M. has no conflicts of interest to declare. Baxter Healthcare was involved in the study design, data collection, data analysis, preparation of the manuscript, and the decision to submit this study for publication.
Statement of Authorship
All authors equally contributed to the conception and design of the research; equally contributed to the design of the research; equally contributed to the acquisition and analysis of the data; equally contributed to the analysis of the data; contributed to the acquisition, analysis, and interpretation of the data; drafted the manuscript; critically revised the manuscript; agree to be fully accountable for ensuring the integrity and accuracy of the work; and read and approved the final manuscript.
