Abstract
Fundamental estimates of radon-associated health risk have been provided by epidemiological studies of miners. In total, approximately 15 studies have been conducted worldwide since the 1960s. These results have contributed directly to radiological protection against radon. The present article summarises the main results, with a focus on analyses of miners exposed more recently, estimates of radon lifetime attributable risk, and interaction between radon and smoking. The potential for the upcoming Pooled Uranium Miner Analysis project to further improve our knowledge is discussed.
1. Miner studies
Large-scale uranium mining began in the 1940s in Czechoslovakia during the Second World War (Tomasek et al., 1994) and in US mines in 1946 (NRC, 1999). The first epidemiological cohorts of miners began to be assembled in the 1960s. In total, more than 12 cohorts provided estimates of the exposure–risk relationship between radon and radon decay products and the risk of lung cancer among miners: in Australia (Radium Hill), Canada (Ontario, Newfoundland, and Eldorado, which combines the Beaverlodge and Port Radium cohorts), China (Yunnan), Czech Republic (Western Bohemia), France (CEA-COGEMA), Germany (Wismut), Sweden (Malmberget), and the USA (Colorado Plateau, New Mexico). Most cohorts included uranium miners, but some were based on tin (Yunnan), iron (Malmberget), or fluorspar (Newfoundland) mines. Most cohorts were composed essentially of males and considered mortality data alone.
Numerous reviews of miner cohorts have been published since the 1980s, especially by the United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR, 1982, 1988, 2000, 2009, 2019), the International Agency for Research on Cancer (IARC, 1988), and the International Commission on Radiological Protection (ICRP, 2010). Several combined analyses have also been performed (NRC, 1988, 1999; ICRP, 1993; Lubin et al., 1994; Tomasek et al., 2008a; Tirmarche et al., 2010; Leuraud et al., 2011; Hunter et al., 2013; Lane et al., 2019). The largest cohort, assembled at the end of the 1990s in Germany, is the Wismut cohort, including nearly 59,000 male workers (Kreuzer et al., 1999, 2018; Walsh et al., 2015). Notable combined analyses include Publication 65 [including more than 31,000 miners from seven cohorts (ICRP, 1993)], the BEIR VI report [including more than 60,000 miners from 11 cohorts (NRC, 1999)], and the Alpha-Risk European project [including more than 50,000 miners from three cohorts (Tirmarche et al., 2010)].
These miner studies have had a significant influence on the understanding of radon risks. They consistently demonstrated a positive association between cumulative radon exposure and lung cancer death. On that basis, radon was classified as a recognised lung carcinogen in 1988 (IARC, 1988). In a recent review performed by UNSCEAR, the combined excess relative risk (ERR) estimated from these cohorts was 0.60 (95% confidence interval 0.34–0.87) per 100 working level month 1 (WLM) (UNSCEAR, 2019). In addition, these cohorts allowed quantification of the impact of factors that modify the exposure–risk relationship, such as attained age, age at exposure, time since exposure (TSE), and exposure rate. Several studies also considered the impact of other exposure factors present in the mines on the estimated risk of lung cancer, such as external gamma exposure and uranium ore dust (Marsh et al., 2012; Rage et al., 2012), arsenic, silica, or fine dust (Sogl et al., 2012; Walsh et al., 2015). The association between radon and lung cancer generally persisted after adjustment of these factors.
Outside of lung cancer, excesses have been reported in several miner cohorts for non-Hodgkin’s lymphoma, multiple myeloma, larynx cancer, kidney cancer, liver cancer, and stomach cancer, and associations with cumulated radon exposure have been suggested for leukaemia, cancers of the extrathoracic airways, and cerebrovascular disease (Darby et al., 1995; Tomasek and Malatova, 2006; Kreuzer et al., 2014, 2017; Drubay et al., 2015). Nevertheless, all these observations are not consistent, and, at the present time, lung cancer is the only known health effect of radon exposure (ICRP, 2010).
2. Analyses of miners exposed in recent periods
Mining conditions have changed drastically over time, with improved working conditions, the introduction of forced ventilation, and the implementation of radiological protection measures from the late 1950s onwards. Fig. 1 shows the changes in mean radon exposure levels over time in the three cohorts of uranium miners (Czech, French, and German) involved in the Alpha-Risk European project (Tirmarche et al., 2010). Concentrations of radon progeny in the first underground mines were several orders of magnitude higher than what has been commonly encountered since the 1960s. It should be noted that these changes in mining conditions over time not only led to a reduction in WLM, but also to changes in the parameters involved in the exposure assessment (e.g. increase in ventilation, decrease in equilibrium factor, change in attached fraction, etc.) and an improvement in the air quality of the mining environment.
Mean annual radon exposures [in working level months (WLM) per year] in the three European cohorts of uranium miners.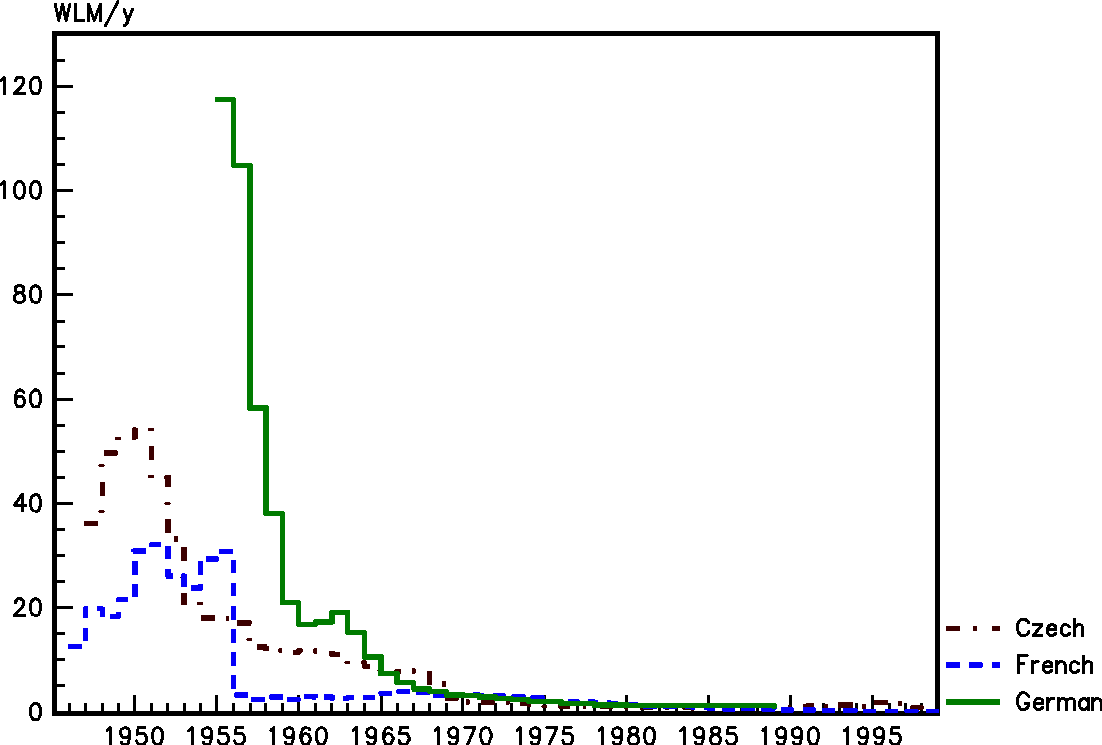
An inverse exposure–rate effect (or protraction enhancement effect) was observed in most analyses of miner studies (Lubin et al., 1994; NRC, 1999). Models have been developed to combine the modifying effects of TSE, age, and exposure rate. The first model was proposed in the BEIR VI report, in which exposure rate was an average calculated as the cumulated exposure divided by the number of years of exposure (NRC, 1999). An improved approach was later proposed in which radon exposure over time was distributed in different windows of annual exposure rate or different periods of exposure (Tomasek et al., 2008a; Tirmarche et al., 2010). This exposure rate effect is reflected by a lower ERR per 100 WLM estimated for high exposure rate categories (annual exposure of several WLM per year or higher) than for low exposure rate categories (annual exposure of several WLM per year or lower). The inverse exposure rate effect was attenuated or no longer observed when restricting analyses to miners exposed to low levels of cumulative WLM exposure or hired in recent periods (Lubin et al., 1995; Tomasek et al., 2008a; Kreuzer et al., 2015, 2018), but estimated ERR per 100 WLM was generally higher than that estimated in the full cohort (ICRP, 2010; Tirmarche et al., 2012; Kreuzer et al., 2015). This was confirmed by a recent review performed by UNSCEAR; compared with the ERR of 0.6 per 100 WLM estimated from the full cohort, an ERR estimate approximately two-fold higher of 1.53 (95% confidence interval 1.11–1.94) per 100 WLM was obtained when restricting the analysis to more recent work periods and lower exposures or exposure rates (UNSCEAR, 2019). UNSCEAR indicated giving preference to the latter estimate due to improved radon exposure assessments in more recent periods, and to these radon exposures being more reflective of current mining conditions. However, it was noted that this estimate was less precise due to smaller sample sizes (UNSCEAR, 2019).
Several explanations have been proposed to explain this effect:
Measurement errors associated with exposure assessment in the early years. Before the 1960s, radon exposure was generally not recorded for each miner based on measurements, and the errors associated with exposure estimates were very large. Also, some type of systematic overestimation of the real exposure levels may have occurred (Allodji et al., 2012a). These errors may lead to underestimation of the estimated ERR per unit exposure associated with early-year exposures and high exposure rates. Similarly, epidemiological studies that correct for measurement errors have shown an increase in risk estimates (Stram et al., 2000; Allodji et al., 2012b; Hoffmann et al., 2017). Overall, data quality was generally much better in recent periods, during which prospective monitoring of the workers was implemented in the mines, for both exposure and follow-up data. Studies of miners restricted to more recent work periods therefore allow analyses based on data of much better quality. Decreased risk of lung cancer at high radon levels. As illustrated in Fig. 1, before the implementation of ventilation and exposure control in the mines, some miners could receive several hundreds of WLM per year. If we apply the recent dose conversion coefficient of 10 mSv (effective dose) per WLM proposed by ICRP (ICRP, 2017), such levels of radon exposure correspond to effective doses of several hundreds or thousands of mSv per year. Estimated equivalent doses to the lung should be approximately eight times higher. At such dose levels, some health effects due to cell killing effects are expected. It is probable that elevated risks have been encountered by early miners for many different health effects, and the risk of lung cancer may be underestimated due to competing risks. Lower TSE effect due to shorter duration of follow-up. As stated above, a strong modifying effect of TSE has been observed in most analyses on miners. This TSE effect is reflected by a higher ERR per 100 WLM estimated 5–15 years after exposure than >25 years after exposure. Miners employed since the 1960s, but not before, obviously have a shorter duration of follow-up than miners employed before the 1960s. Consequently, this shorter duration of follow-up could prevent the TSE effect from being fully expressed, and thus give rise to the impression of a higher coefficient in the cohort of miners hired in recent periods. Nevertheless, this hypothesis is unlikely as miners hired since 1960 had, in 2010, a possible follow-up duration of almost 50 years, with a potential attained age of approximately 70 years. Today, the characteristics of these cohorts of miners hired in recent periods make them perfectly capable of taking a possible TSE effect into account. Conversely, it is possible that the strong TSE effect observed in the complete cohorts may be due to underestimation of the risk among miners hired in earlier periods.
In conclusion, it appears that, for the purposes of radiological protection, the most relevant studies on miners are those with low cumulative exposure levels, long duration of follow-up, and good-quality data. Today, the cohorts restricted to miners hired in recent periods are capable of providing good estimates of the risk of lung cancer associated with cumulated radon exposure. Even if estimates of ERR per unit exposure of radon are associated with wider confidence intervals due to a restriction of population size, exclusion of miners employed in the early years appears to be the best way to reduce bias. Recent analyses of cohorts of miners hired in recent periods generally demonstrate a significant association between cumulated radon exposure and risk of lung cancer, with estimated ERR per 100 WLM approximately two-fold higher than the values that were estimated in the 1990s (ICRP, 2010; UNSCEAR, 2019). Furthermore, the heterogeneity between cohorts of ERR per 100 WLM estimated from analyses restricted to miners exposed to low levels of cumulative WLM exposure or hired in recent periods is much lower than from analyses of full cohorts (ICRP, 2010; Tirmarche et al., 2012).
3. Estimated lifetime excess risk of lung cancer attributable to radon
Due to variations in the characteristics of the study populations (e.g. attained age, duration of follow-up), a direct comparison of estimates of ERR per unit exposure of radon obtained from different cohorts may be misleading. The calculation of the cumulated risk up to a given age (often called ‘lifetime excess risk’) in a specific exposure scenario can take such variations into account (Thomas et al., 1992) to reflect the risk attributable to radon exposure. Calculation of lifetime risk requires:
a risk model derived from an epidemiological study, with modifying factors such as attained age, age at exposure, TSE, and exposure rate; a projection model, enabling extrapolation of risk outside the range considered by the epidemiological study and transport to other populations; baseline reference rates for all-cause and lung cancer mortality, and age distribution of the reference population – this allows calculation of the baseline lifetime risk of lung cancer in the absence of additional radon exposure; and a scenario of exposure.
Since the 1990s, several calculations of radon-induced lifetime excess risk have been performed (ICRP, 1993, 2010; NRC, 1999; EPA, 2003; Tomasek et al., 2008b; UNSCEAR, 2019). Most of these calculations used the same methodology and scenario of exposure as proposed in Publication 65 (ICRP, 1993): a constant low-level exposure to 2 WLM per year during adulthood from 18 to 64 years of age, with the risk of lung cancer cumulated up to 90 or 94 years of age, and the same source of baseline rates [ICRP reference rates from Publications 60 or 103 (ICRP, 1991, 2007)]. Therefore, these calculations can be compared.
Publication 65 (ICRP, 1993) estimated a lifetime excess risk of lung cancer of 2.8 × 10−4 per WLM for radon exposure. This result was based on a risk model taking account of modifying effects of age and TSE, but not exposure rate. New lifetime risk estimates were calculated in Publication 115 (ICRP, 2010). Considering all available models derived from different single cohorts, estimated lifetime excess risks varied from approximately 3 to 7 × 10−4 per WLM, according to the model used. Priority was given to estimates based on models derived from combined analyses that were able to consider a modifying effect of exposure rate or hiring period; namely, the models from the BEIR VI report and the combined analysis of the Czech–French cohorts (NRC, 1999; Tomasek et al., 2008a). Results obtained from these two models were very similar, with estimated values between 4.5 and 5.5 × 10−4 per WLM. Based on these calculations, a rounded lifetime excess risk value of 5 × 10−4 per WLM was recommended by ICRP as the nominal risk coefficient for radon exposure for radiological protection purposes (ICRP, 2010).
Recent lifetime risk calculations performed by UNSCEAR gave similar results (UNSCEAR, 2019). Lifetime excess risk values were obtained by applying the BEIR VI exposure age–concentration model individually to the updated Czech Republic, Wismut, and Eldorado miner studies, and to the combined 11 miner studies used in the BEIR VI report. Values ranged from 2.4 to 7.5 × 10−4 per WLM for the Wismut and Eldorado studies, respectively. For the BEIR VI studies, the estimated lifetime excess risk was 5.5 × 10−4 per WLM (UNSCEAR, 2019).
In conclusion, miner cohorts provide a quantitative basis for estimating the excess risk of lung cancer attributable to radon exposure. Results appear to be very consistent, with variations within a factor of approximately 2. In the future, use of risk models derived from miners hired in recent periods may allow a reduction in this range of variation.
Calculations of lifetime excess risk of lung cancer attributable to radon exposure contribute directly to radiation protection. In 1993, ICRP adopted a nominal risk coefficient of 2.8 × 10−4 per WLM for radon exposure. Comparing this value with the total radiation detriment per Sv for adults given in Publication 60 (ICRP, 1991), a dose conversion convention of 4 mSv for 1 WLM was derived for workers (ICRP, 1993). In 2010, based on an updated review of epidemiological results, a nominal risk coefficient of 5 × 10−4 per WLM was recommended by ICRP for radiological protection purposes as a replacement for the previous value (ICRP, 2010). With this revised nominal risk coefficient and the detriment value of Publication 103 for adults (ICRP, 2007), a dose conversion convention of 12 mSv per WLM was obtained (Marsh et al., 2010, 2017; ICRP, 2017). Taking account of both epidemiological and dosimetric approaches, Publication 137 (ICRP, 2017) recommended the use of a single rounded value of 3 mSv per mJ h m−3 (approximately 10 mSv per WLM) in most circumstances for workers in buildings and underground mines.
4. Interaction between radon and smoking
Although smoking is by far the strongest risk factor for lung cancer, most studies of miners did not take account of smoking habits. Nevertheless, available results indicate that the relationship between lung cancer mortality and radon exposure generally persists when smoking habits are taken into account, with only marginal changes in the risk of radon-associated lung cancer (Tirmarche et al., 2012). Application of an indirect adjustment method on the Colorado miner cohort suggested no confounding by smoking of the association between radon and lung cancer (Schubauer-Berigan et al., 2009; Richardson et al., 2014).
Most analyses are consistent with a submultiplicative or multiplicative interaction between radon exposure and smoking status (NRC, 1999; Leuraud et al., 2011; Kreuzer et al., 2018). When the smoking status is known, the estimated ERR per unit exposure of radon generally appears to be larger (even if not significantly) among non-smokers than among smokers (Lubin et al., 1994; Tomasek et al., 2002; Leuraud et al., 2011; Hunter et al., 2013). Nevertheless, an analysis of the Czech miner cohort concluded that an interaction close to additive between radon and smoking was observed (Tomasek, 2013). The explanation for this discrepancy is not known. It could be partly related to differences in the classification of smoking behaviour, or in the consideration of age and TSE in the modelling of the interaction between smoking and radon. Further analyses are needed to improve the characterisation of the joint effect of radon and smoking.
It is noticeable that almost all models derived from miner studies since the 1990s are ERR models (also referred to as ‘multiplicative risk models’). Such models suppose that the excess risk of lung cancer associated with radon exposure is proportional to the baseline rate of lung cancer. As baseline rates are highly dependent on smoking habits, it is recommended that excess absolute risk (EAR) models (also referred to as ‘additive risk models’) should also be developed to better understand the interaction of radon and smoking on the risk of lung cancer. Assessment of the impact of changes in smoking prevalence on the baseline rate of lung cancer, and on the risk attributable to radon, is also worthwhile.
5. Perspectives
Most recently, the Pooled Uranium Miner Analysis (PUMA) study was launched. This assembles information on cohorts of uranium miners in North America (Canada and the USA) and Europe (Czech Republic, Germany, and France). Data include individual annual estimates of exposure to radon decay products, duration of employment of each worker, and information on vital status, date of death, and cause of death. The PUMA study constitutes the largest study of uranium miners conducted to date, encompassing 124,507 miners, 4.51 million person-years at risk, and 54,462 deaths, including 7825 deaths due to lung cancer (Rage et al., 2020).
The PUMA study provides opportunities to evaluate new research questions and to conduct analyses to assess potential health risks associated with uranium mining that have greater statistical power than can be achieved with any single cohort. Planned research topics include analyses of associations between radon exposure and mortality due to lung cancer, cancers other than lung cancer, and non-malignant disease. For the risk of lung cancer, analyses will consider both ERR and EAR models; specific analyses of the impact of uncertainties, exposure rate, and smoking on the estimated exposure–risk relationship; and quantification of overall mortality excesses and lifetime risks. This international study will improve our understanding of radon-related diseases and strengthen the basis for radon radiological protection.
6. Conclusion
Miner cohorts have made a major contribution to the understanding of radon-associated risks and the consolidation of radiation protection against radon exposure. Miner cohorts have provided consistent results on the existence of an increased risk of lung cancer associated with radon exposure, even at relatively low exposure rates. They have also provided results on the modifying effect of age and TSE on this association, and on the interaction with smoking. One recurrent criticism is on the uncertainties associated with these results. It now seems evident that a good way to reduce uncertainties is to focus analyses on cohorts of miners employed in more recent years, after implementation of ventilation and individual exposure control in mines. Analyses focused on such populations generally have data of much higher quality to provide risk estimates that are more consistent between cohorts, and consider exposure levels that are more pertinent for current radiation protection purposes.
In the near future, the PUMA study will provide consolidated information on quantification of the risk of lung cancer associated with radon, and on potential risks other than lung cancer. Important questions that cannot be answered by miner studies remain, especially regarding risks among females and risks associated with radon exposure during childhood. Results from miner cohorts need to be complemented by epidemiological studies of indoor radon, dose and risk modelling, and experimental research in order to further improve our knowledge in this area.
Footnotes
This paper does not necessarily reflect the views of the International Commission on Radiological Protection.
1
Working level is defined as any combination of the short-lived progeny of radon in 1 L of air that will result in the emission of 1.3 × 105 MeV of potential alpha energy. 1 working level = 2.08 × 10−5 J/m3. Working level month is defined as the cumulative exposure from breathing an atmosphere at a concentration of 1 working level for a working month of 170 h.
