Abstract
Systemic or locoregionally administered alpha-particle emitters are highly potent therapeutic agents used in oncology that are fundamentally novel in their mechanism and, most likely, overcome radiation resistance as the alpha particles emitted have a short range and a high linear energy transfer. The use of alpha emitters in a clinic environment requires extra measures with respect to imaging, dosimetry, and radiation protection. This is shown for the example of 223Ra dichloride therapy. After intravenous injection, 223Ra leaves the blood and is taken up rapidly in bone and bone metastases; it is mainly excreted via the intestinal tract. 223Ra can be imaged in patients with a gamma camera. Dosimetry shows that, after a series of six treatments for a 70-kg person with an overall administered activity of 23 MBq, 223Ra results in an absorbed alpha dose of approximately 17 Gy to the bone endosteum and approximately 1.7 Gy to the red bone marrow. During administration, special care must be taken to ensure that no spill is present on the skin of either the patient or staff. Due to the low dose rate, the treatment is normally performed on an outpatient basis; the patient and carers should receive written instructions about the therapy and radiation protection.
Keywords
1. INTRODUCTION
Alpha-particle emitters are highly potent therapeutic agents that are fundamentally novel in their mechanism and, most likely, overcome radiation resistance as the alpha particles emitted have a short range (50–100 µm) and a high linear energy transfer (LET). Alpha particles induce numerous DNA double-strand breaks along the respective tracks. Eberlein et al. (2015a,b, 2016) have shown that the DNA damage repair of double-strand breaks in beta-emitting radionuclides can be visualised in vitro and in vivo in human lymphocytes by the γ-H2AX assay. Fig. 1, which shows the track of an alpha particle traversing a human lymphocyte, illustrates that this assay can also be applied to alpha emitters.
DNA damage caused by the track of an alpha particle (shown as a green streak) through a human lymphocyte visualised by the γ-H2AX assay (image courtesy of H. Scherthan, Bundeswehr Institute of Radiobiology, Munich, Germany).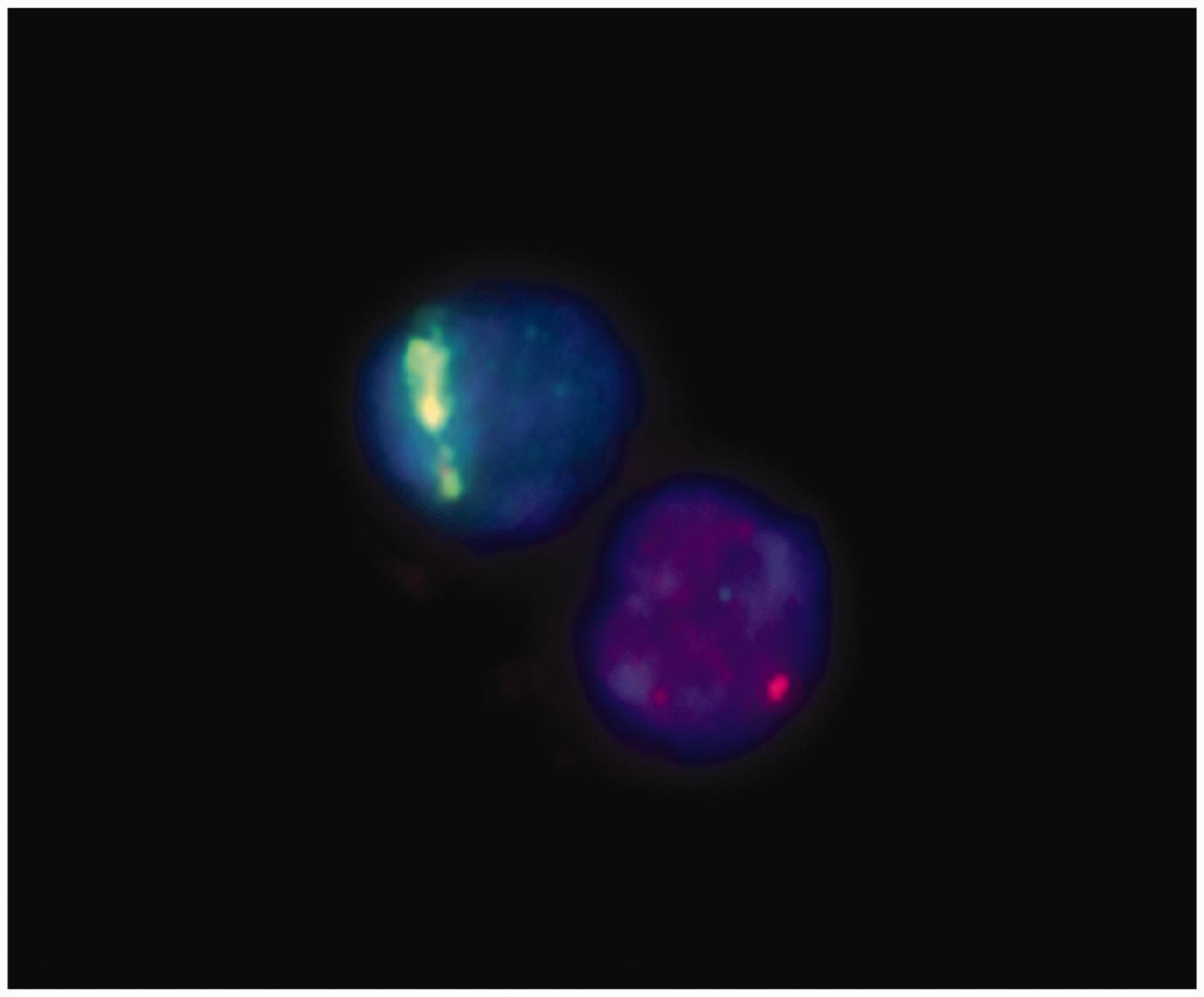
The high LET of alpha particles and the limited ability of cells to repair DNA damage from alpha radiation explain their high relative biological effectiveness (RBE) and cytotoxicity. Therefore, alpha emitters are becoming of increasing importance for both systemic and locoregional application in therapeutic nuclear medicine in oncology.
Alpha-emitting isotopes for therapeutic applications in nuclear medicine.
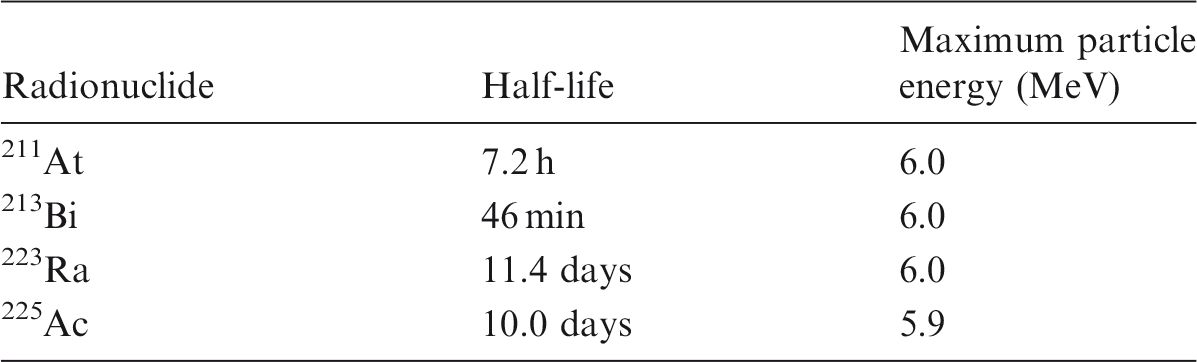
Treatment with alpha particles is mainly focused on targeted cancer therapy and bone pain palliation. Examples for targeted cancer therapy are described by Sgouros et al. (2010). These include, but are not limited to, the treatment of melanoma or leukaemia with 213Bi-labelled antibodies, or the treatment of ovarian carcinoma with 211At-labelled antibodies. The most recent example is the treatment of metastatic prostate cancer with 225Ac- or 213Bi-PSMA ligands (Kratochwil et al., 2017, 2018).
For metastatic bone pain palliation, 223Ra dichloride has obtained – as the first and, to date, only alpha emitter – marketing authorisation in Europe and the USA for treatment of patients with castration-resistant prostate cancer and symptomatic bone metastases (Parker et al., 2013). All other studies reported in the literature (Sgouros et al., 2010; Kratochwil et al., 2017, 2018) were single centre, mainly compassionate use applications, which are not suited for deriving general rules for the use of alpha emitters in patient treatment and developing general radiation protection rules.
The use of alpha emitters in a clinic environment requires new and different measures with respect to imaging, dosimetry, and radiation protection compared with the use of beta emitters. As 223Ra is now in use worldwide, this paper will focus on these topics, particularly for 223Ra. Most of the results, however, can also be applied to other therapies with alpha emitters.
2. USE OF RADIUM 223
2.1. Physical properties
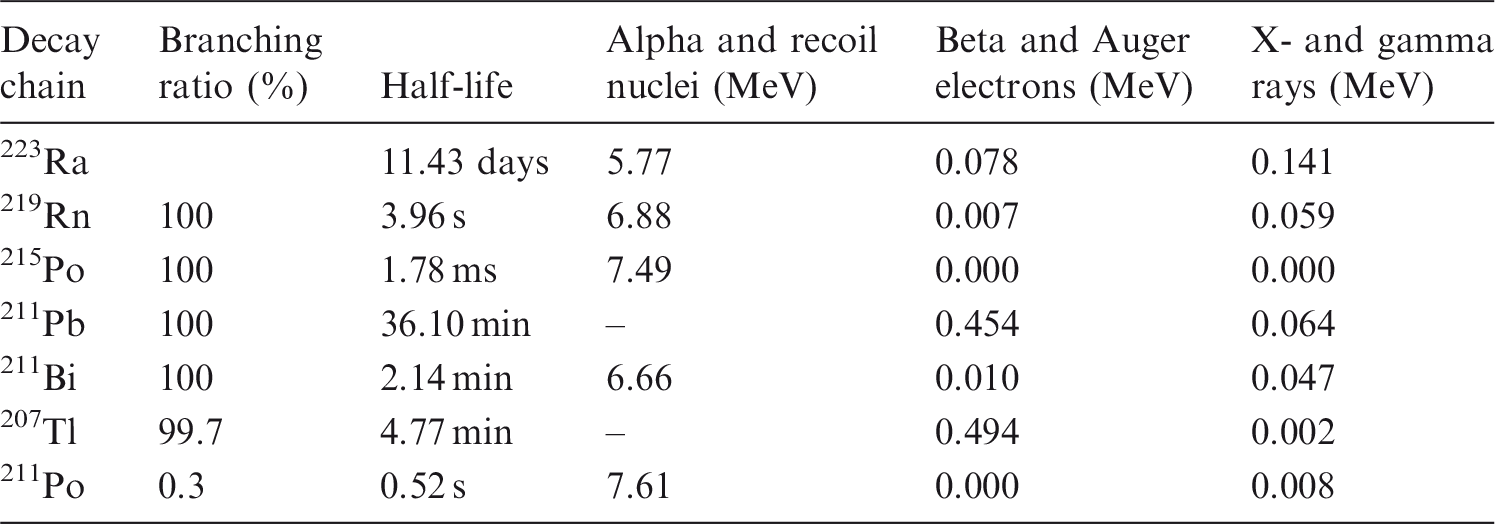
223Ra is a short-lived (half-life 11.4 days) alpha emitter that decays through a cascade of short-lived alpha- and beta-emitting progeny, with the emission of approximately 28 MeV through complete decay of the progeny to stable lead. A schematic drawing of the decay chain is shown in Fig. 2. A listing of the decay chain, branching ratios, half-lives, and energies deposited by alpha and recoil nuclei, beta and Auger electrons, and x- and gamma rays for 223Ra and its progeny is provided in Table 2 for 223Ra and the corresponding progeny with branching ratios >0.3%.
Decay chain of 223Ra as provided by RadDecay Version 4 (Negin and Worku, 1993). Decay chain, branching ratios, half-lives, and energies emitted by alpha and recoil nuclei, beta and Auger electrons, and x- and gamma rays for 223Ra and the corresponding progeny with branching ratios >0.3%. The data are taken from the Medical Internal Radiation Dose tables (Eckerman and Endo, 2008).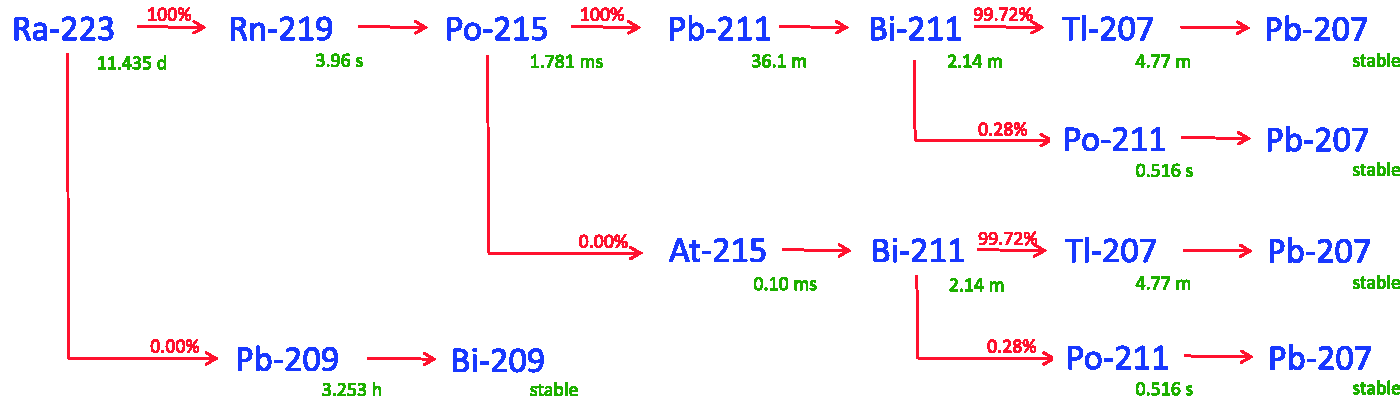
Due to its method of production and according to the manufacturer's specifications, 223Ra may be contaminated with up to 0.004% 227Ac (beta emitter, half-life 21.8 years) and up to 0.5 % 227Th (alpha emitter, half-life 18.7 days).
2.2. Imaging
Radium-223 and its progeny can be imaged with a gamma camera. Due to the poor energy resolution, only three major photon peaks that are of potential use for this purpose (∼82 keV, ∼154 keV, and ∼270 keV) can be distinguished. The results obtained by Hindorf et al. (2012) suggest that quantitative imaging can be performed as a whole-body scan at a low speed, with the camera equipped with either a medium- or a high-energy collimator, and with a 20% wide energy window centred on 82 keV. The authors showed, in a phantom study, that activity could be quantified with an accuracy of ±10% for a 200-mL volume placed within water containing background activity, and with an accuracy of ±40% for a 0.5-mL phantom for the 82-keV energy window. The authors concluded that, according to these results, uptake quantification in patients’ bones is possible with a sufficient degree of accuracy for a clinical situation. However, they assumed an activity of 100 kBq kg−1 to be administered. For the currently recommended treatment of metastatic prostate cancer, the patient will receive six administrations of 223Ra (55 kBq kg−1) at 4-week intervals. This corresponds to an overall activity of 33 MBq for a patient with a body weight of 100 kg.
In a study by Pacilio et al. (2016a) on image quantification, the authors used static imaging of the 82-keV and 154-keV peaks with a medium-energy general purpose collimator for quantification of the uptake of 223Ra in bone lesions. Whole-body scans could not be performed due to technical limitations of the equipment used.
Fig. 3 shows an example of a series of whole-body scans for 223Ra in a single patient. It was taken during a phase I pharmacokinetics study of a single administration of 50 kBq kg−1 223Ra dichloride in Japanese patients with castration-resistant metastatic prostate cancer (Yoshida et al., 2016).
Biodistribution of activity, as detected via gamma camera, of 223Ra over time in a patient. The figure was originally published by Yoshida et al. (2016). The article by Yoshida et al. is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/).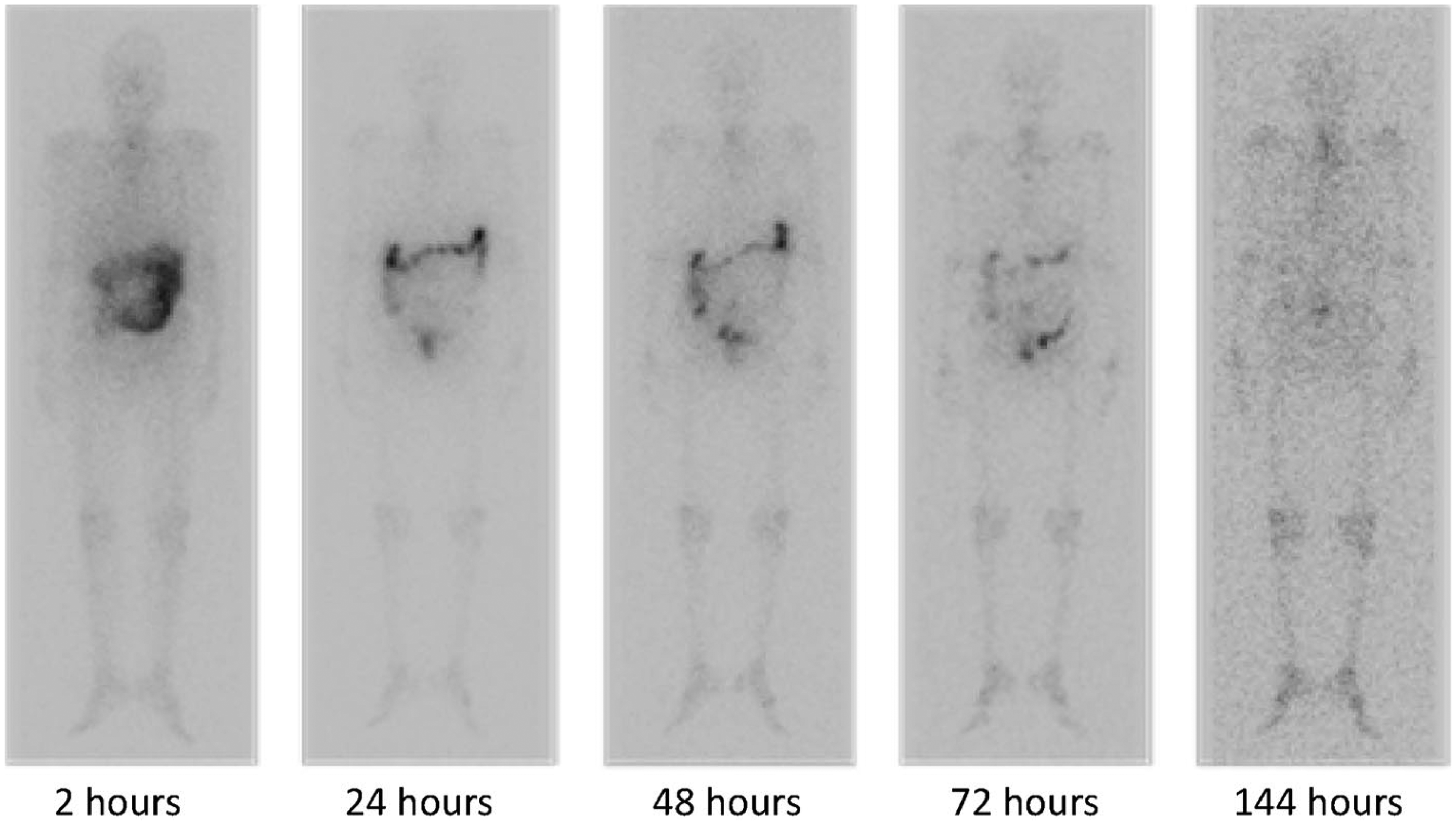
Overall, imaging is possible; however, quantification of the images is difficult due to technical limitations, and is not part of the clinical routine for patients treated with 223Ra dichloride.
2.3. Dosimetry
To calculate absorbed doses to organs and/or lesions of a patient, the pharmacokinetics of the radiopharmaceutical in the respective organ/lesion need to be assessed by several quantitative measurements in order to obtain the respective time–activity curves. These measurements can be taken from quantitative imaging (Hindorf et al., 2012; Pacilio et al., 2016b) and from the activity in blood samples, measured by a well counter.
The respective time integral of the time–activity curves represents the number of decays occurring in the organ/lesion considered. Multiplication by a corresponding dose factor allows assessment of the absorbed dose. The dose factors need to account for all progeny of the decay chain.
Another option for calculating absorbed doses to normal organs and tissues could be to apply the kinetic model of the ICRP for 223Ra, as done by Lassmann and Nosske (2013). The bone endosteum was calculated to receive the highest absorbed doses at 7.5·10−7 Gy Bq−1 for alpha and 1.1·10−8 Gy Bq−1 for beta/gamma. The absorbed doses to the red marrow were 7.2·10−8 Gy Bq−1 and 5.5·10−9 Gy Bq−1, respectively. A dosimetric calculation based on compartment modelling (Lassmann and Nosske, 2013) results in an absorbed alpha dose of approximately 17 Gy to the bone endosteum after a series of six treatments for a 70-kg reference person with an administered activity of 55 kBq kg−1 223Ra each (overall 23 MBq 223Ra). The corresponding cumulative absorbed alpha dose to the red bone marrow is approximately 1.7 Gy.
Yoshida et al. (2016) confirmed these results in a series of measurements of the pharmacokinetics in six Japanese patients.
In an Italian multi-centre trial that based dosimetry on quantitative imaging, Pacilio et al. (2016b) found that the mean effective half-life of 223Ra in bone lesions was 8.2 days and the absorbed dose after the first injection was 0.7 Gy (range 0.2–1.9 Gy). The mean RBE weighted absorbed dose (RBE = 5) was 3.5 Gy (range 1.1–9.3 Gy). The almost 10-fold range reflects the individual pharmacokinetics in patients. These values cannot be compared directly with the results of compartment modelling calculations, as tumour dosimetry is not included in these models.
Several studies have measured the disappearance of 223Ra from the blood and excretion pathways (Carrasquillo et al., 2013; Chittenden et al., 2015; Yoshida et al., 2016). All studies showed rapid blood clearance; the major excretion pathway, however, is faecal excretion (see Fig. 2). This is in contrast to most therapeutic radiopharmaceuticals labelled with beta emitters, which are mainly excreted via the kidneys and the urinary tract (e.g. Luster et al., 2008; Bodei et al., 2013).
At present, neither treatment planning nor post-therapeutic dosimetry are performed in clinical practice due to the technical limitations and the lack of a well-established dosimetric methodology for 223RaCl2.
3. RELATIVE BIOLOGICAL EFFECTIVENESS
For calculation of dose coefficients, Howell et al. (1997) have suggested, based on their experimental data, a radiation weighting factor of 5.4 for 223Ra. MIRD Pamphlet 22 reports, based on a review of experimental literature, a recommended RBE value for alpha emitters between 3 and 5 for cell killing (Sgouros et al., 2010). In more recent publications, an RBE of 5 is assumed for therapies with alpha emitters for comparing deterministic effects with those of beta/gamma emitters.
The radiation weighting factor of 20 as recommended by Publication 103 (ICRP, 2007) is strictly based on RBE values for stochastic (cancer) risks, and not for deterministic effects or tissue reactions.
4. RADIATION PROTECTION
With respect to radiation protection, only two studies are available (Dauer et al., 2014; Wanke et al., 2015) that rely on experimental data, and one study that describes a theoretical assessment of contamination scenarios (Stabin and Siegel, 2015).
Dauer et al. (2014) found that dose rates from patients administered 223Ra dichloride were typically less than 2 μSv h−1 MBq−1 on contact, and averaged 0.02 μSv h−1 MBq−1 at 1 m immediately following administration. Removal was primarily by faecal excretion, and whole-body effective half-lives were highly dependent upon faecal compartment transfer, ranging from 2.5 to 11.4 days.
Dauer et al. (2014) assessed the extremity doses of the worker preparing the syringe and performing the administration of 223Ra. They stated that ‘based on the measured mean shallow dose rates of 4.8 μGy min−1 MBq−1 and 0.6 μGy min−1 MBq−1 from the syringe and vial, respectively, a vial activity of 6 MBq and typical syringe activity of about 3.5 MBq, and further assuming that preparation of the dose takes 2 min of vial handling and 1 min of syringe handling while administering the activity takes an additional 1 min of syringe handling, it was estimated that a worker might receive approximately 41 μSv for each administration’. According to these observations, a worker could perform over 1200 administrations of 223Ra dichloride before reaching the threshold for monitoring of the extremities, which is 50 mSv (i.e. one-tenth of the annual extremity dose limit of 500 mSv). Dauer et al. therefore state that 223Ra is safe and straightforward to administer using conventional nuclear medicine equipment. El Mantani Ordoulidis and Backlund (2018) report that the Hp(0.07) dose rate at the surface of an unshielded syringe is between 13 mSv hr−1 MBq−1 (217 μSv min−1 MBq−1) and 27 mSv hr−1 MBq−1 (450 μSv min−1 MBq−1); the authors recommend the use of adequate syringe shielding.
Wanke et al. (2015) observed, in a multi-centre study, that the potential exposure of patient family members and carers by external irradiation and incorporation of radioactivity exhaled or excreted by the patient in saliva or sweat is well below 1 mSv. Therefore, the authors see no objections regarding treating patients as outpatients.
Stabin and Siegel (2015) analysed possible dosimetric consequences of various potential contamination events involving 223Ra dichloride: (1) inhalation dose to an individual cleaning up a spill due to 219Rn and its progeny; (2) external exposure of an individual cleaning up a spill; and (3) skin dose of a worker due to contamination. They concluded that, under extremely conservative assumptions, the skin dose is estimated to be 72 mSv, mainly caused by beta-emitting nuclides in the decay chain. The doses due to inhalation and external exposure are less than 10−2 mSv and 10−3 mSv, respectively.
After administration, the patient should be given radiation safety instructions and should be informed about the rules of conduct and hygiene that must be observed; this should be documented accordingly. Note that specific recommendations may differ based on local regulatory guidance, which may differ from one facility or region to another.
Overall, the medical use of 223Ra does not pose any significant radiation safety issues with respect to external irradiation, contamination, and incorporation that go beyond the use of beta emitters for radionuclide therapies in a hospital environment.
