Abstract
Over the past decade, the International Commission on Radiological Protection (ICRP) has developed a comprehensive approach to environmental protection that includes the use of Reference Animals and Plants (RAPs) to assess radiological impacts on the environment. For the purposes of calculating radiation dose, the RAPs are approximated as simple shapes that contain homogeneous distributions of radionuclides. As uncertainties in environmental dose effects are larger than uncertainties in radiation dose calculation, some have argued against more realistic dose calculation methodologies. However, due to the complexity of organism morphology, internal structure, and density, dose rates calculated via a homogenous model may be too simplistic. The purpose of this study is to examine the benefits of a voxelised phantom compared with simple shapes for organism modelling. Both methods typically use Monte Carlo methods to calculate absorbed dose, but voxelised modelling uses an exact three-dimensional replica of an organism with accurate tissue composition and radionuclide source distribution. It is a multi-stage procedure that couples imaging modalities and processing software with Monte Carlo N-Particle. These features increase dosimetric accuracy, and may reduce uncertainty in non-human biota dose–effect studies by providing mechanistic answers regarding where and how population-level dose effects arise.
Keywords
1. Introduction
Several approaches have been developed, tested, and compared for the limitation of radiation dose in non-human biota (NHB). The International Commission on Radiological Protection (ICRP) has developed a comprehensive approach that includes the use of Reference Animals and Plants (RAPs) for the assessment of dose to NHB.
ICRP’s current approach to dosimetry calculations for NHB relies on simplified organism geometry. Organisms are modelled as ellipsoids made of homogeneous four-component soft tissue (ICRU, 1992). Some models also contain simple ellipsoidal organs that serve as targets for dosimetric calculations (ICRP, 2008). These rudimentary models are used to generate absorbed fractions that are, in turn, used to calculate dose conversion factors (DCFs) from radionuclides that are distributed uniformly throughout the organism. There are two scenarios that require consideration beyond that available with homogeneous models. The first is when comparing whole-body average dose rates for homogeneous and non-homogeneous distributions of incorporated radionuclides, and assessments of the uncertainties of whole-body doses due to those non-homogeneous sources. The second is when it is necessary or prudent to estimate organ dose rates from radionuclides that partition heterogeneously.
Voxel models are three-dimensional (3D) models created from radiological imaging modalities [e.g. computed tomography (CT) and magnetic resonance imaging (MRI)]. These models allow radioecologists to calculate organ-specific DCFs from heterogeneously distributed radionuclides. Voxel models are more robust and easier to defend than the previously used ellipsoidal models, but the additional effort required to make such models may not be justifiable. If simplified models are conservative, there is no need to create complicated models. However, if simplified models are not conservative, there is a need for the creation of voxel-derived dosimetric parameters.
2. Description of Homogeneous Models
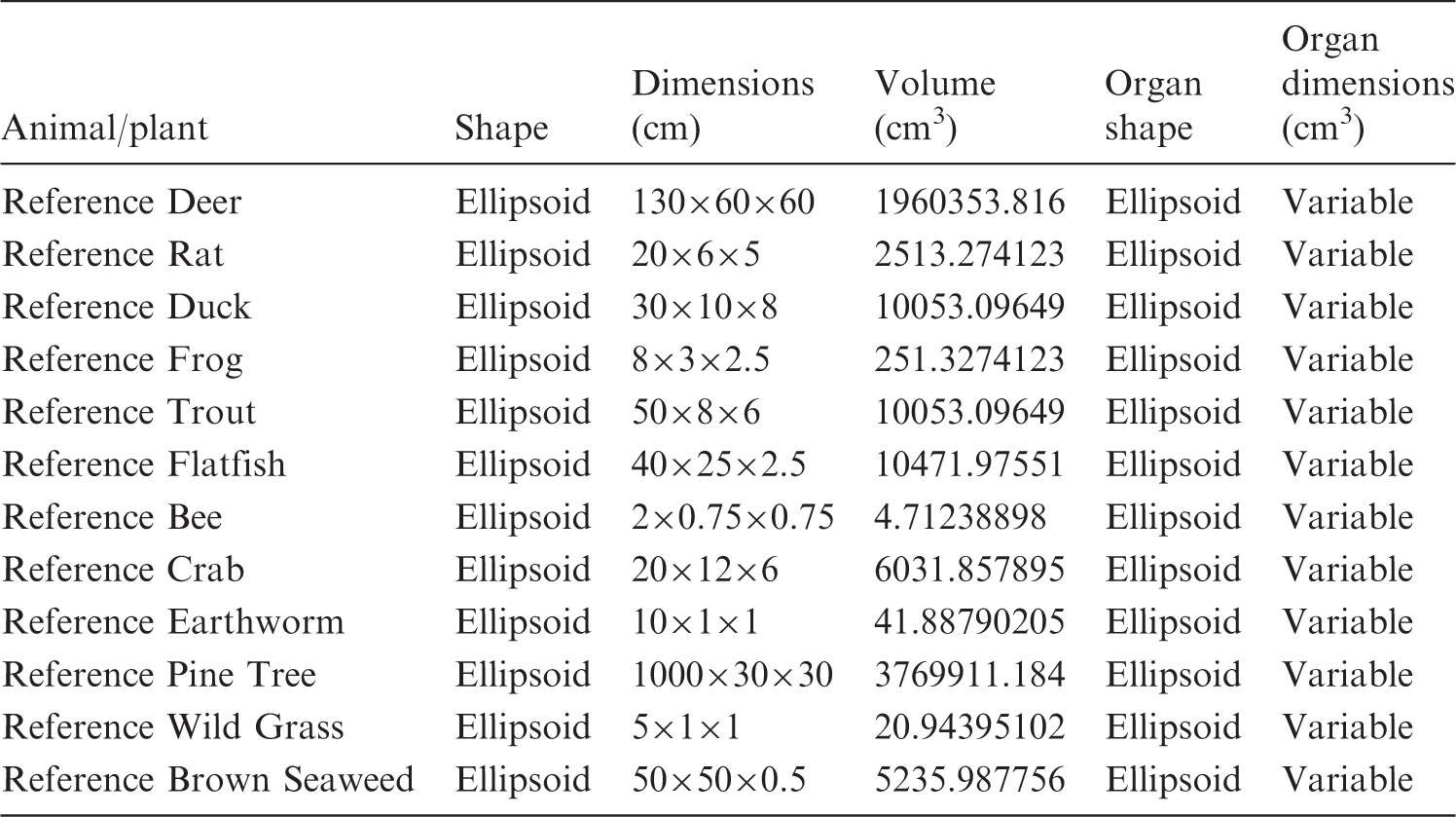
Since the 1970s, environmental dosimetry has relied upon the ‘uniform isotropic model’ as defined by Loevinger and Berman (1976). The uniform isotropic model consists of the following assumptions: the organism is in an infinite homogeneous medium; the activity is distributed uniformly throughout its body; and the densities of the medium and the organism’s body are the same (ICRP, 2008). Fig. 1 shows an example of the homogeneous models in use by ICRP today, and Table 1 provides details on the dimensions of each RAP.
Current International Commission on Radiological Protection geometric model of the Reference Animals and Plants with inner ellipsoid representing the internal structure (ICRP, 2008). Parameters of the International Commission on Radiological Protection’s homogeneous model.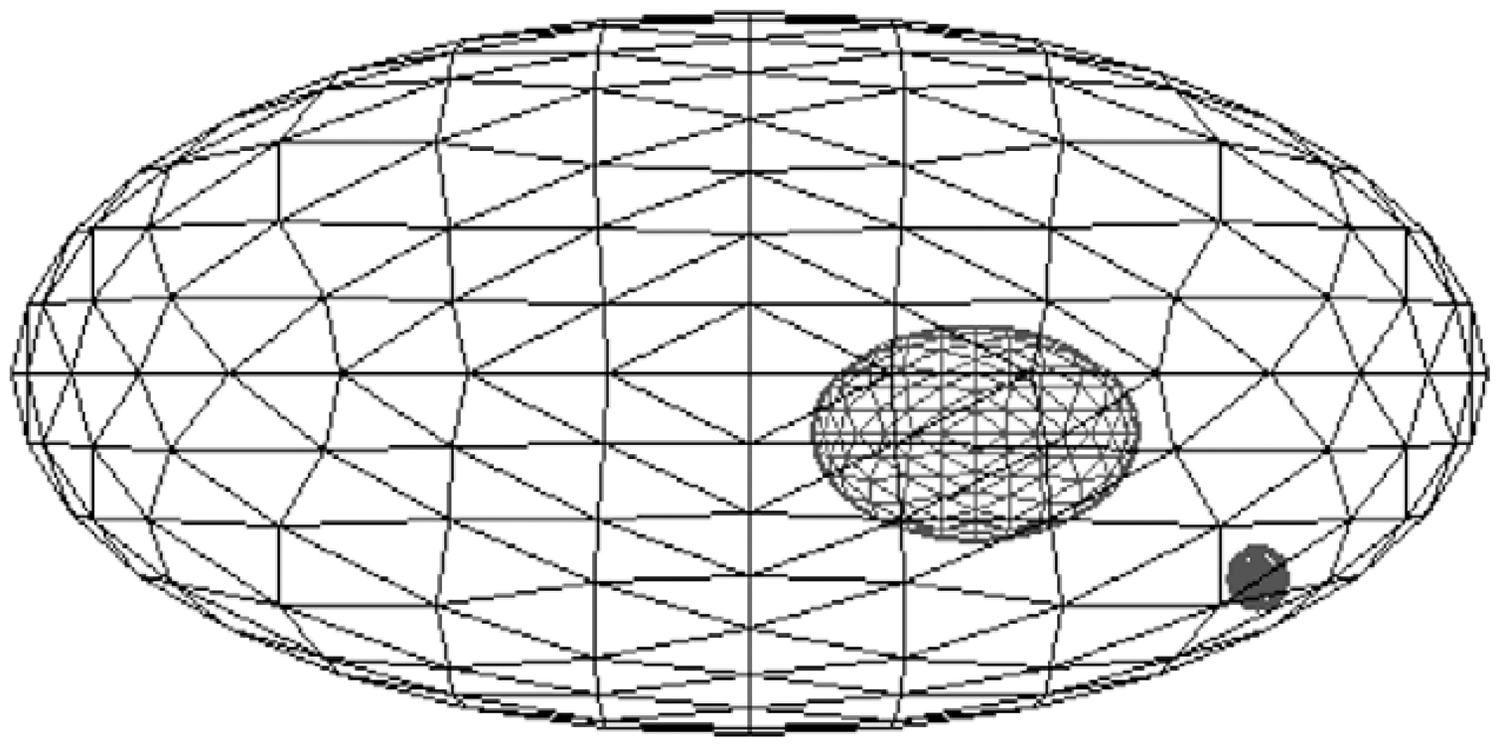
3. Concept of Voxel Phantom
A voxel phantom is constructed with a series of image slices formatted in pixels with a given thickness, forming a (a) Example of a voxelised object inside a solid ellipsoid; (b) representation of a volume pixel or voxel unit.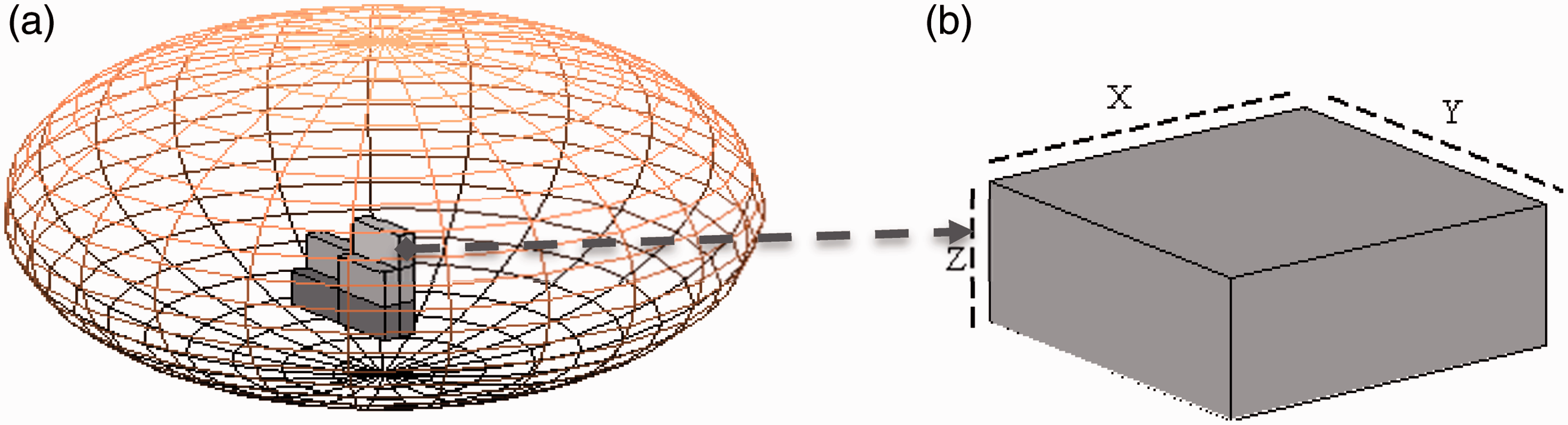
4. Construction of a Voxel Phantom
4.1. Selection of an imaging modality
CT is a non-invasive imaging technique that allows for visualisation of the internal structures of an organism without the superposition of over- and underlying structures (Hendee, 2002). CT scans are ideal for organisms where tissues have differing effective atomic numbers or density. In cases with these factors, contrast suffers and it may be difficult to resolve internal structures. MRI is well suited for these cases, as it is able to provide excellent contrast resolution in even those tissues with similar density and atomic number. In general, CT scans are acceptable for organisms with more bony structures and/or differentiation in tissue composition (e.g. Reference Crab). MRI scans are recommended when the organism consists primarily of soft tissue) (e.g. Reference Flatfish).
4.2. Required software suites
4.2.1. 3D-Doctor (or any other segmenting software)
3D-Doctor is a powerful and user-friendly image processing software for medical, scientific, and industrial imaging, developed by Able Software Corp. It specialises in processing, rendering, modelling, visualisation, and quantitative analysis of images from MRI, CT, positron emission tomography, and other industrial uses (Able Software Corp., 2012). Organism images are segmented on each image slice, outlining each identifiable organ. The software exports boundary (.bnd) files that are needed for input into a lattice generation tool (e.g. Voxeliser, discussed below).
4.2.2. Voxeliser
Voxeliser was developed in the python coding language by the Human Monitoring Laboratory of Canada (Kramer et al., 2010). This program reads boundary file geometry (.bnd) and creates repeated structures (e.g. universes) for Monte Carlo simulations. The limit on the number of voxels in MCNPX 2.5 or MCNP5 is 50 million. In order to stay within the limit, Voxeliser can adjust the array by using a compression factor or array reduction that will combine several voxels into one (Kramer et al., 2010). If a compression factor is applied, there will be a reduction of resolution of the final lattice.
4.2.3. Monte Carlo N-Particle (MCNPX 2.5, MCNP5)
MCNP is a radiation transport code that is capable of solving the transport equation in three dimensions for many particle types, including neutrons, photons, and electrons. It is also capable of coupled electron/photon/neutron transport calculations. The code generates a random number that describes the statistical life of each particle, including scattering (coherent or incoherent), fission, leakage, absorption interaction, and other particle–material interactions that may occur; thus, it can take quite some time for the code to run (X-5 Monte Carlo Team, 2008). MCNP has built-in cross-section libraries for different particles that allow the user to specify the elemental composition and density of each object.
4.2.4. Overall process
First, a CT or MRI image of the organism of interest is obtained. Those images are uploaded into the 3D-Doctor program and segmented. The boundary file is exported and subsequently uploaded into Voxeliser, which created the lattice geometry for input into MCNP. Absorbed fractions are obtained from the MCNP output.
5. Model Comparisions
Parameters of various organs on voxel-based models.
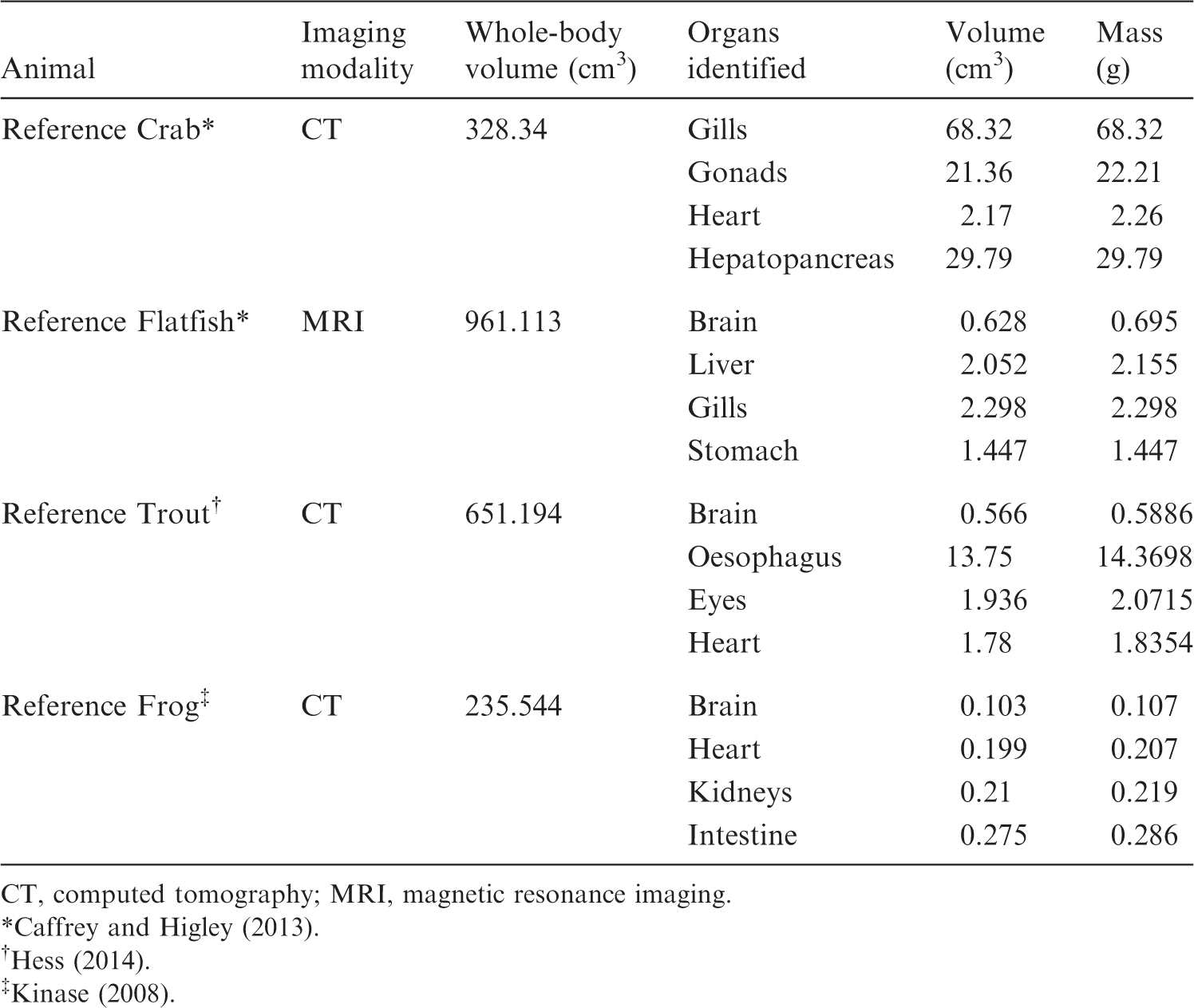
CT, computed tomography; MRI, magnetic resonance imaging.
The ability to model realistic shapes and sizes with proper elemental compositions can improve the accuracy of calculations to determine dose rate in NHB as the range of particles is highly dependent on the physical properties of the tissue (e.g. effective atomic number and density). Fig. 3 shows a 3D rendering of the trout model, shown without the muscle tissue; this illustration underscores the level of detail present in a voxelised model.
Three-dimensional rendering of the trout model, shown without muscle tissue, from side (top) and above (bottom) (Hess, MSc thesis in progress).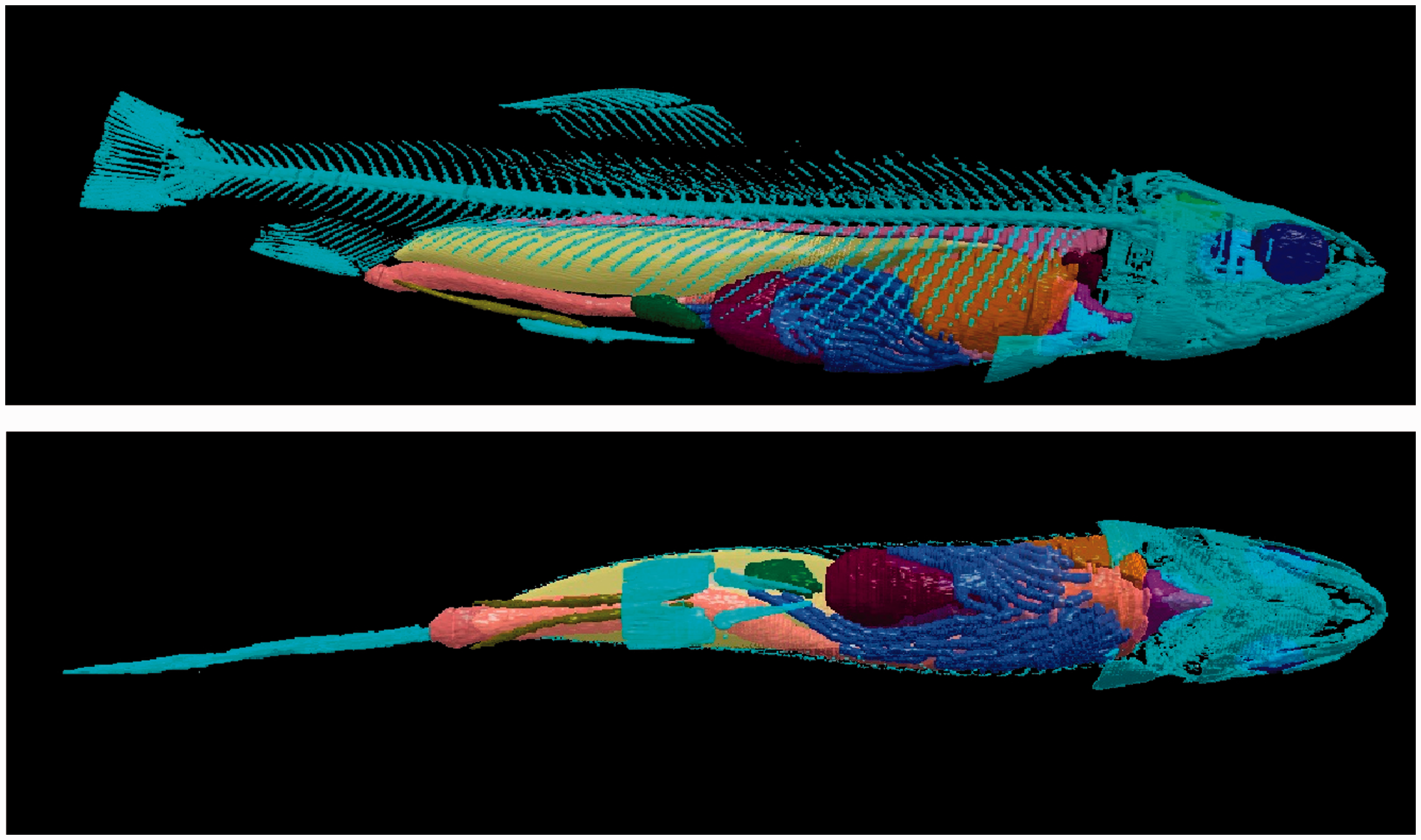
It is evident from Fig. 3 that using a voxel-based model can be beneficial for many types of simulations, such as simulating a source distributed in multiple organs, or radionuclide transport through organ systems, to name a few. Table 2 shows the anatomical characteristics of four voxel phantoms (note that not every organ is listed, due to the large quantity of organs identified in each model).
5.1. Dose conversion factors
In order to determine if the efforts expended in creating voxel phantoms is justifiable, it is useful to assess the uncertainty in a simple model by calculating doses in a voxelised phantom. In order to conduct this comparison, two sets of absorbed fraction data were used: AFs from simplified, homogeneous models, and AFs from voxelised models for specific source:target pairs. In instances where the radionuclide of interest is distributed homogeneously throughout the organism (e.g. tritium), it is expected that the simplified model and voxel model will show reasonably consistent results in dose rate calculations. For the comparisons detailed below, it was assumed that the radionuclide in question was perfectly partitioned into a single organ; this represents the most extreme case, and if the simplified model is conservative for this case, it can be assumed to be conservative for all cases. In order to assess the conservatism of the homogeneous model, dose rates were calculated for a theoretical 1-MBq source. The source was partitioned perfectly into each voxelised organ to obtain voxel-derived dose rates, and this was compared with dose rates arising from the same 1-MBq source distributed uniformly within the homogeneous model (Ruedig et al., 2015).
5.1.1. Dose conversion factor calculations
RAPs and radionuclides were selected based on the availability of dosimetric data. To this end, Reference Rat, Reference Flatfish, Reference Trout, and Reference Crab were included in this analysis (Mohammadi et al., 2011, 2012; Caffrey, 2012; Caffrey and Higley, 2013; Hess, MSc thesis in progress). Seven radionuclides were selected for analysis as they represented a wide range of radiation types and energies, and are commonly encountered: 14C, 36Cl, 60Co, 90Sr, 131I, 137Cs, and 210Po. Note that 137Cs AF data were not available for Reference Rat. Biological behaviour is left for further study; as such, some organ:radionuclide pairs may not be indicative of reality.
Dose rates were calculated as follows:
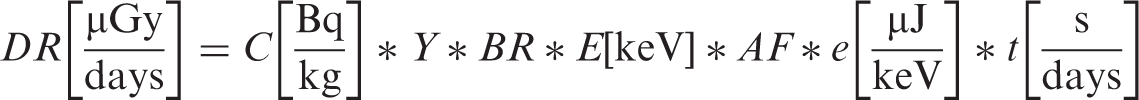
The following assumptions were made (consistent with ICRP methodologies).
Any radiation occurring in <1% of decays was omitted. As there are no AFs for positrons, the ɛ/β+ decay (1.9%) was omitted in the 36Cl calculations. The subsequent annihilation photons were included, and assumed to originate in the same organ as the parent nuclide. Daughter product emissions were included when the daughter half-life was >10 days (90Sr/90Y and 137Cs/137mBa). Alpha emissions were assumed to be absorbed completely in the originating organs. Beta emitters with energies >100 keV were assumed to be absorbed completely in the originating organs. For beta emitters, average energy was taken to be one-third of the maximum.
AF data were obtained from published literature in the case of Reference Rat (Mohammadi et al., 2011, 2012) and Reference Crab (Caffrey and Higley, 2013), and from the Master’s theses of Oregon State University students in the case of Reference Flatfish (Caffrey, 2012) and Reference Trout (Hess, MSc thesis in progress; Ruedig et al., to be published). DCFs for simplified models were taken from
There is no consensus on the summation of individual organ doses (e.g. tissue weighting factors) to obtain a whole-body dose rate for NHB; as such, no attempt was made to sum the individual organ dose rates, and whole-body dose rates from simplified models were compared with individual organ dose rates from the voxelised model.
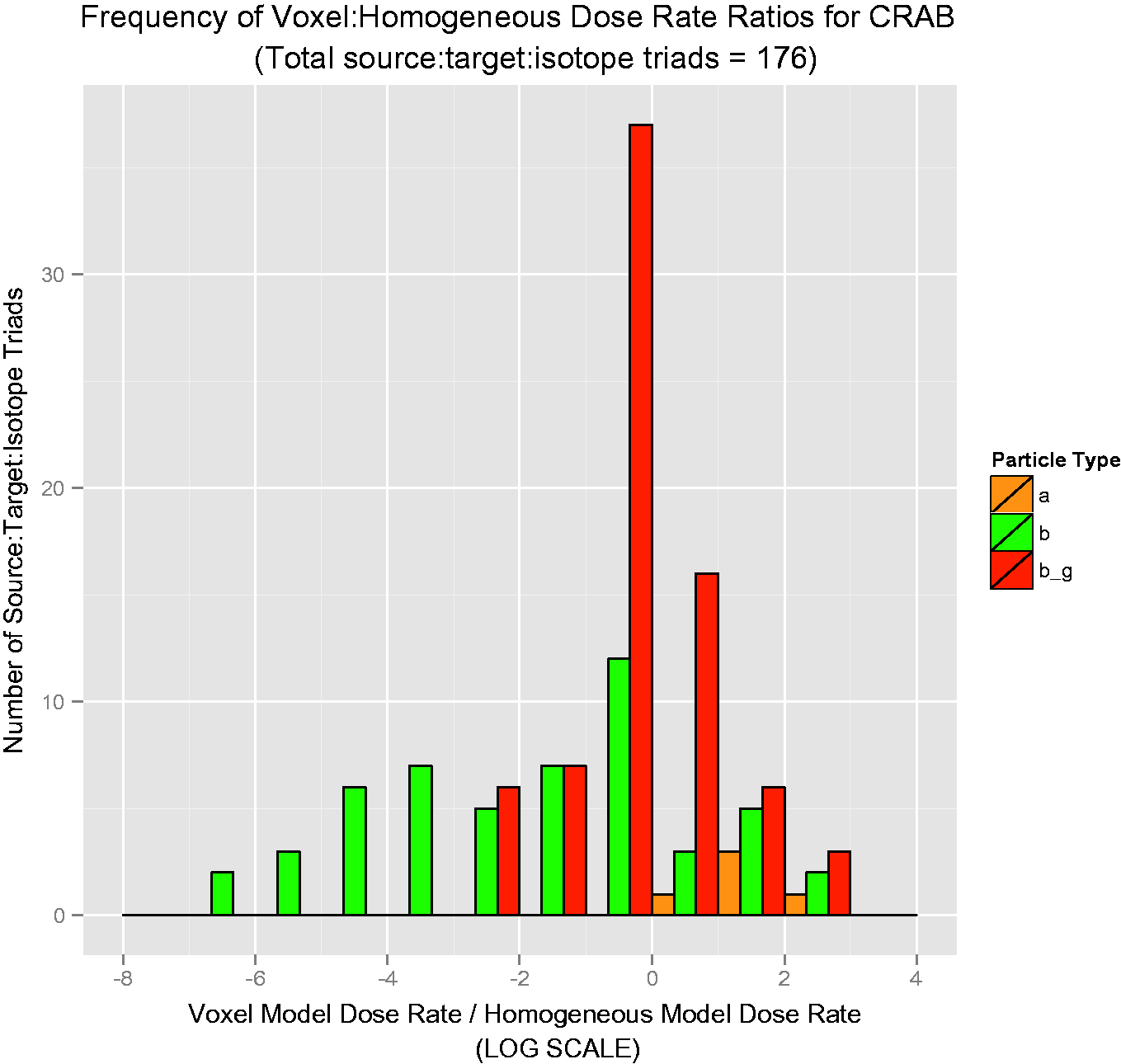
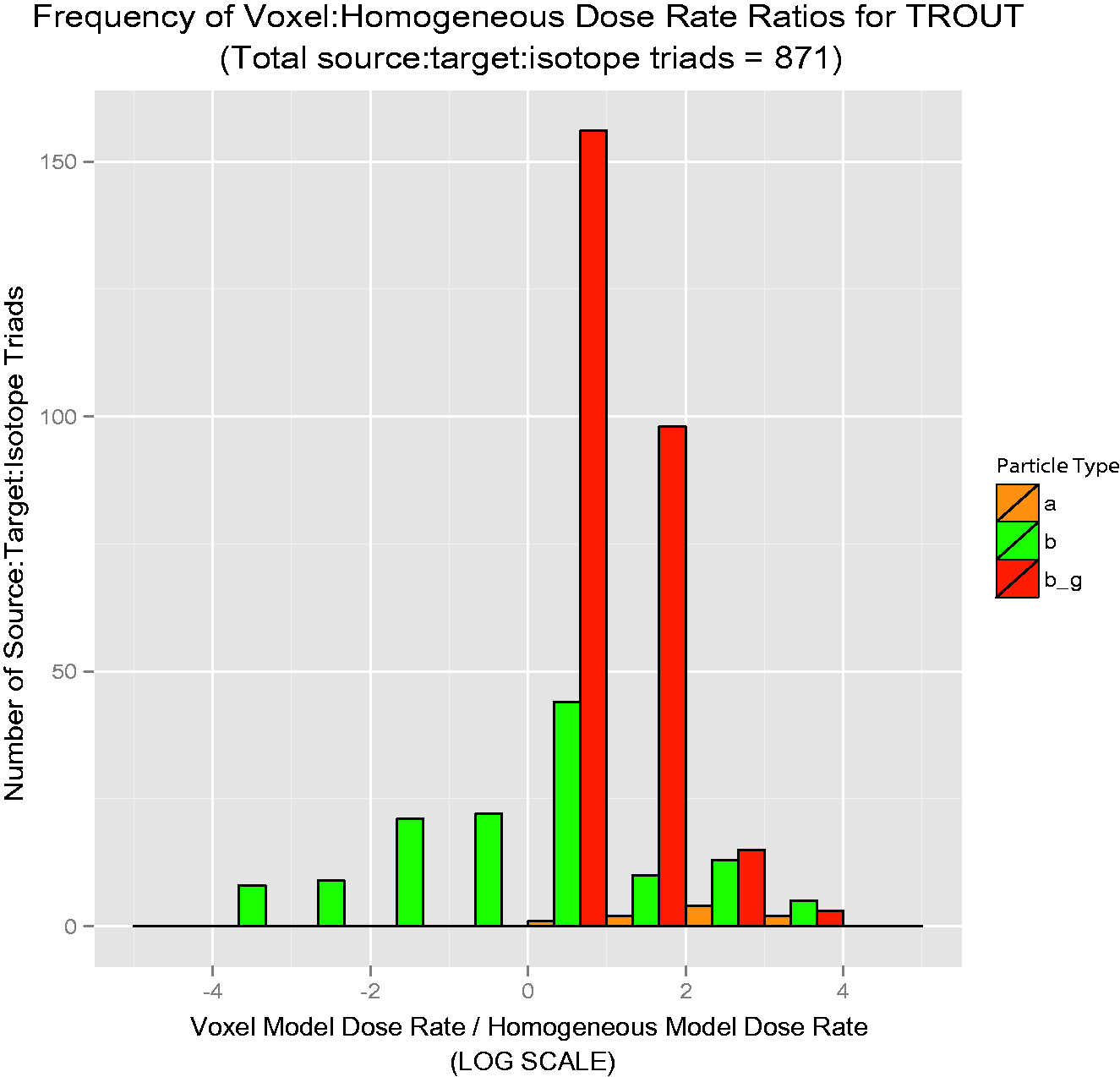
The results are offered as ratios of voxelised:simple dose rate. Thus, if the ratio is unity, the predicted dose rates are equivalent. For ratios greater than unity, voxelised dose rates are thus higher, and the simplified model is not conservative; for ratios less than unity, simplified dose rates are higher, and the simplified model is conservative. Each bar in Figs 4–7 is colour-coded for each source:target combination. The line at unity indicates where simplified and voxelised dose rates are equivalent. Bars to the right indicate source:target pairs where voxelised dose rates are higher; bars to the left indicate that the simplified model dose rate is higher. Bars to the left that appear to run off the chart are cases where the voxelised dose rates were essentially zero, and could not be plotted properly on a log scale.
Cl-36 source in Reference Crab. Co-60 source in Reference Flatfish. Sr-90 source in Reference Rat. Cs-137 source in Reference Trout.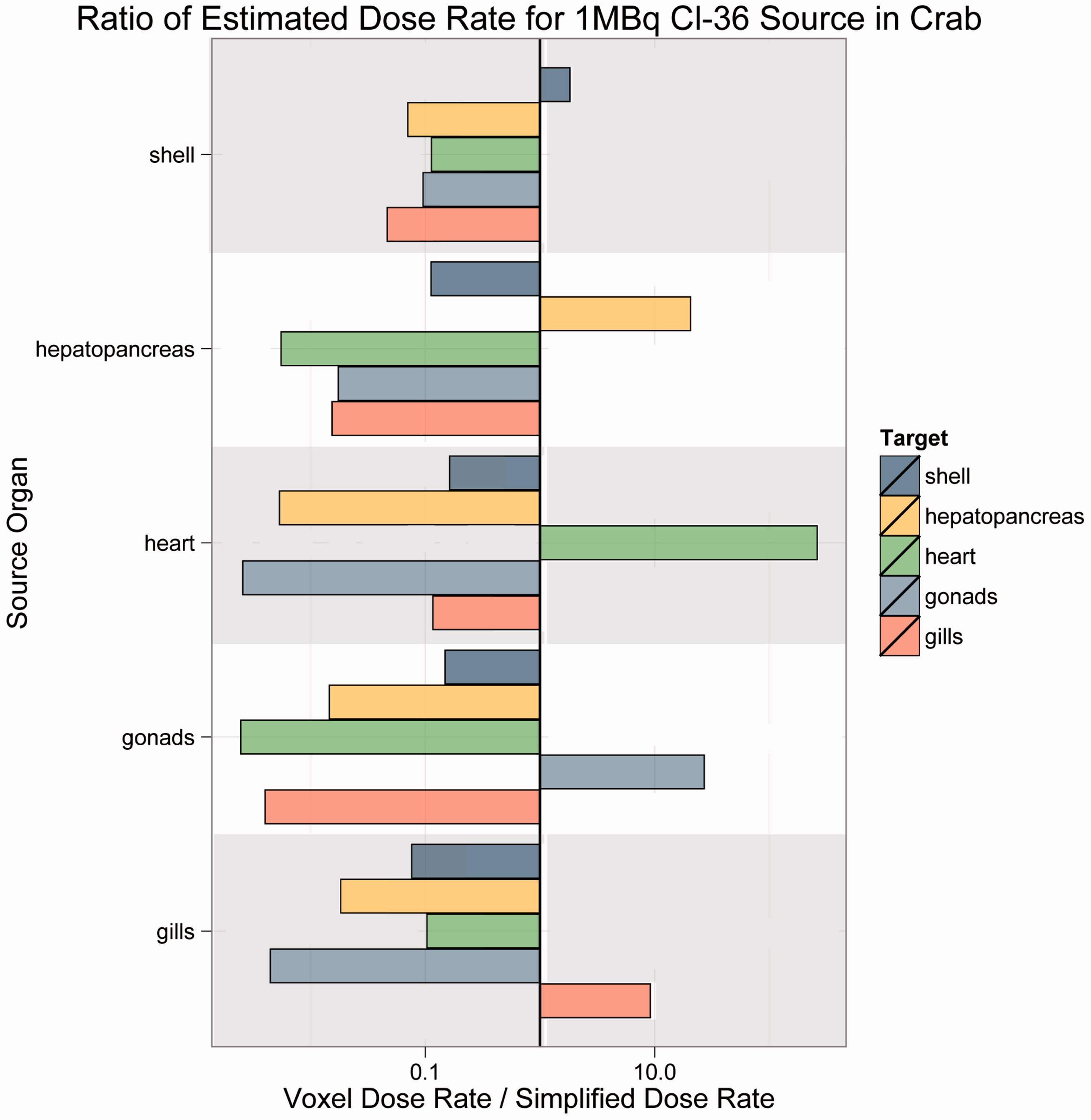
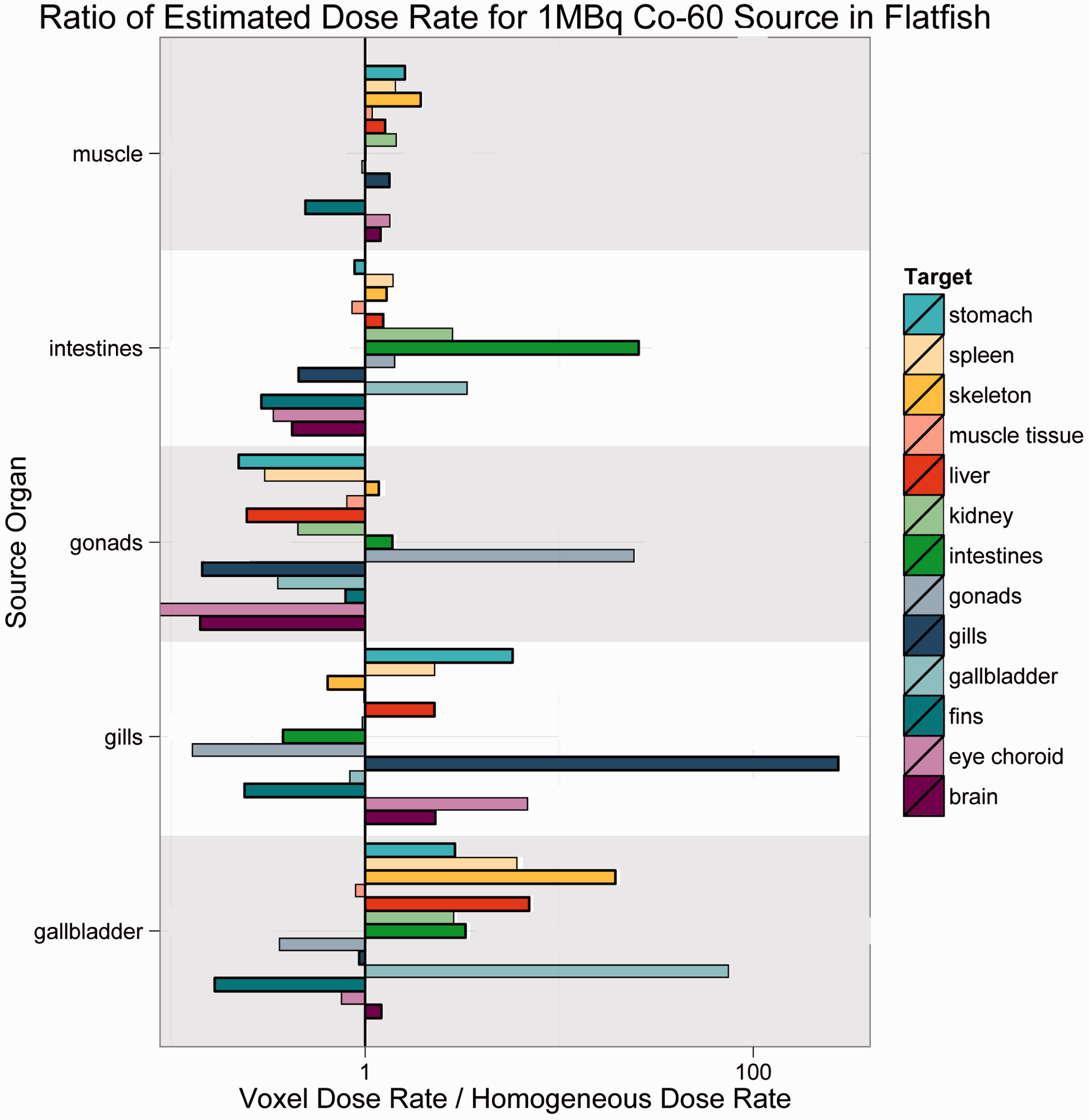
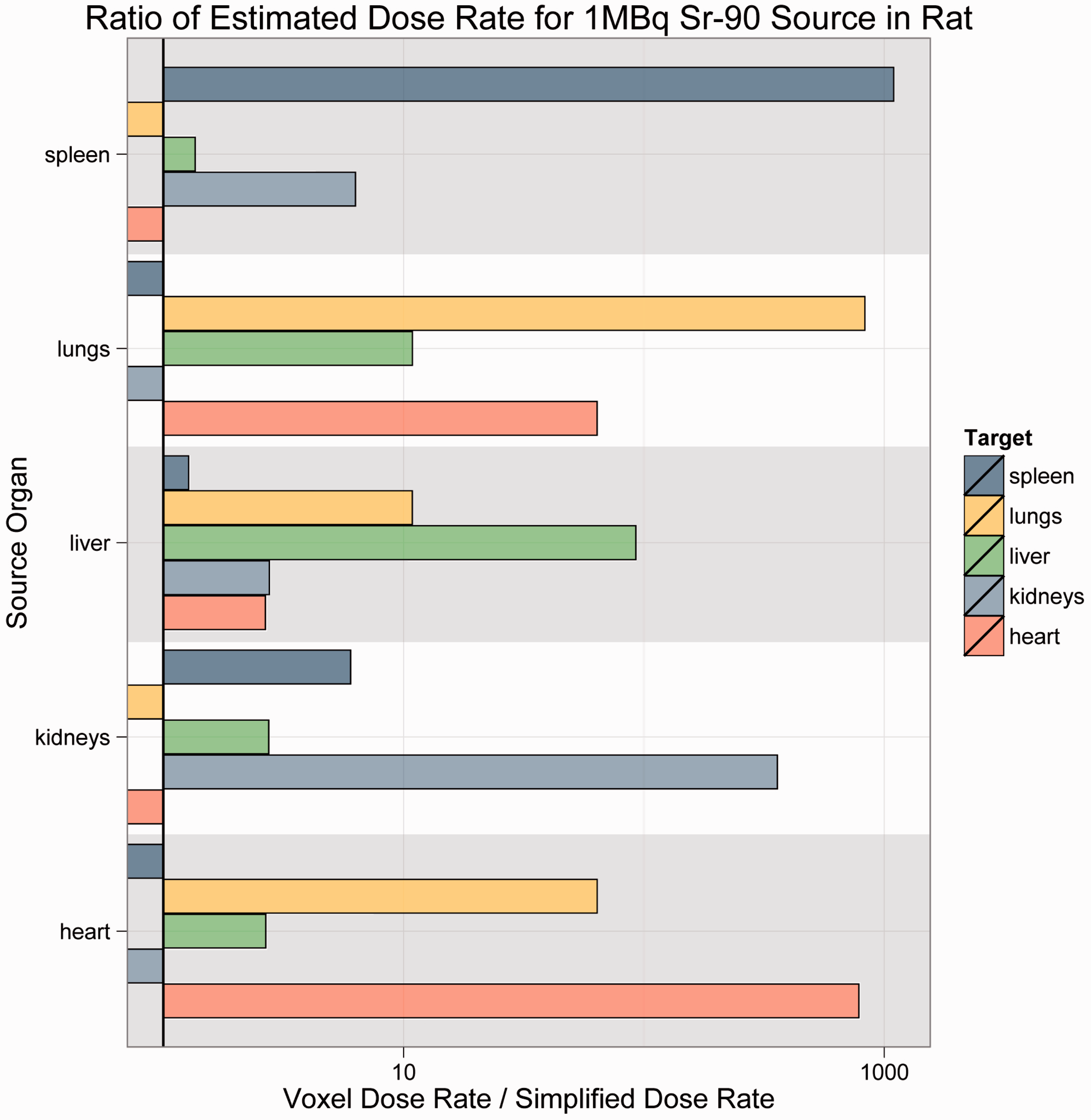
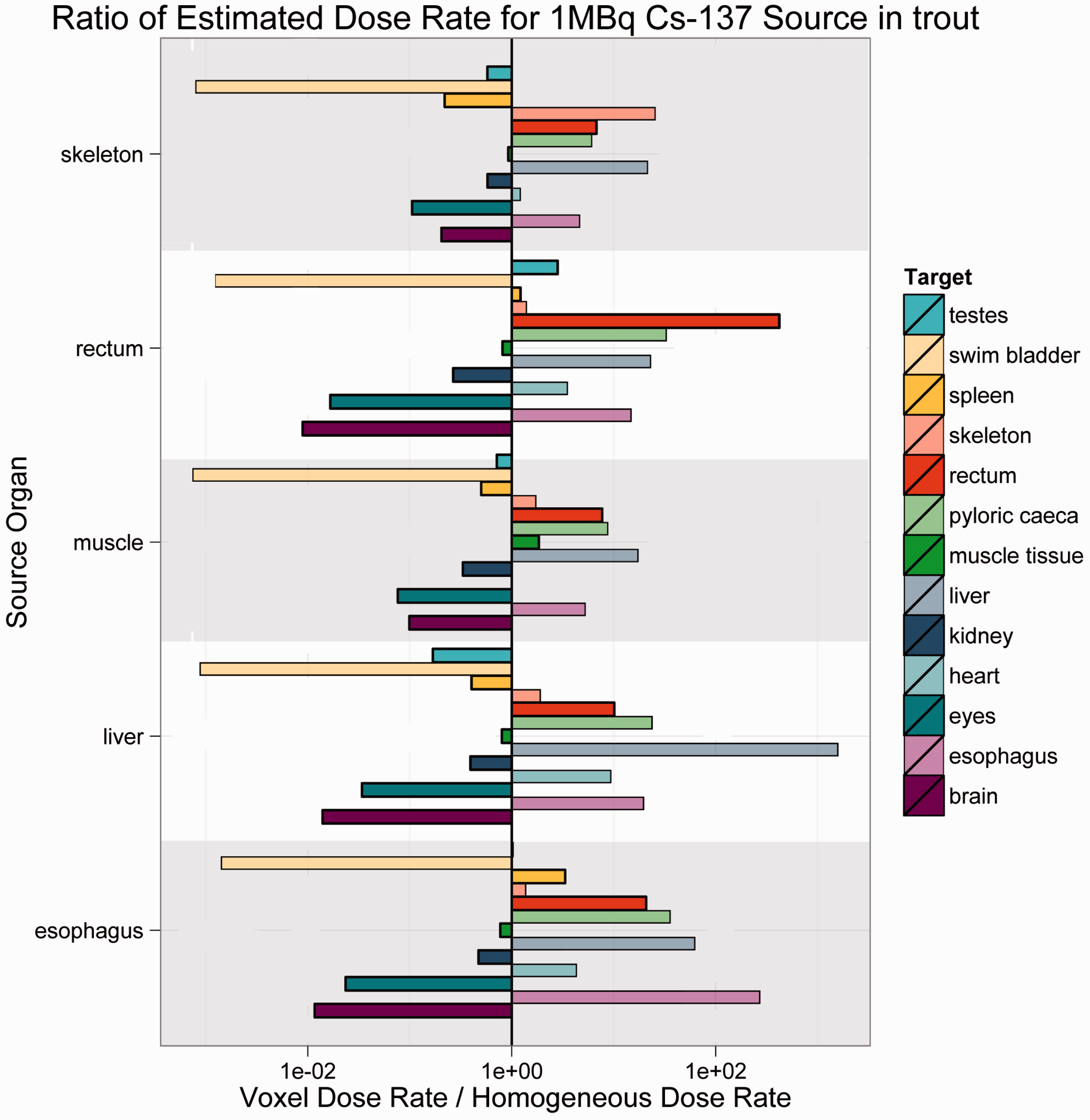
As Figs 4–7 illustrate, there was no consistent trend seen across the organisms and radionuclides examined in this study. Fig. 4 shows estimated dose rates for a 1-MBq Cl-36 source in Reference Crab. Cl-36 is a beta emitter with a short range. In this case, the homogeneous model generally predicts higher dose rates, and thus is largely conservative. Fig. 5 depicts a 1-MBq Co-60 source in Reference Flatfish. Co-60 is a beta/gamma emitter, and has been known to concentrate in the kidneys. In this instance, there are no immediately discernible trends, likely due to the mass and positioning of the source organ in the fish. Fig. 6 shows a 1-MBq Sr-90 (including contributions from Y-90) source in Reference Rat. Sr-90 is another radionuclide that is likely to partition in specific organs. In this case, the simplified model would likely underestimate the organ dose rate, meaning that it is not conservative. Fig. 7 represents Cs-137 in Reference Trout. Caesium distributes in the soft tissues, and again, no discernible trends are immediately obvious.
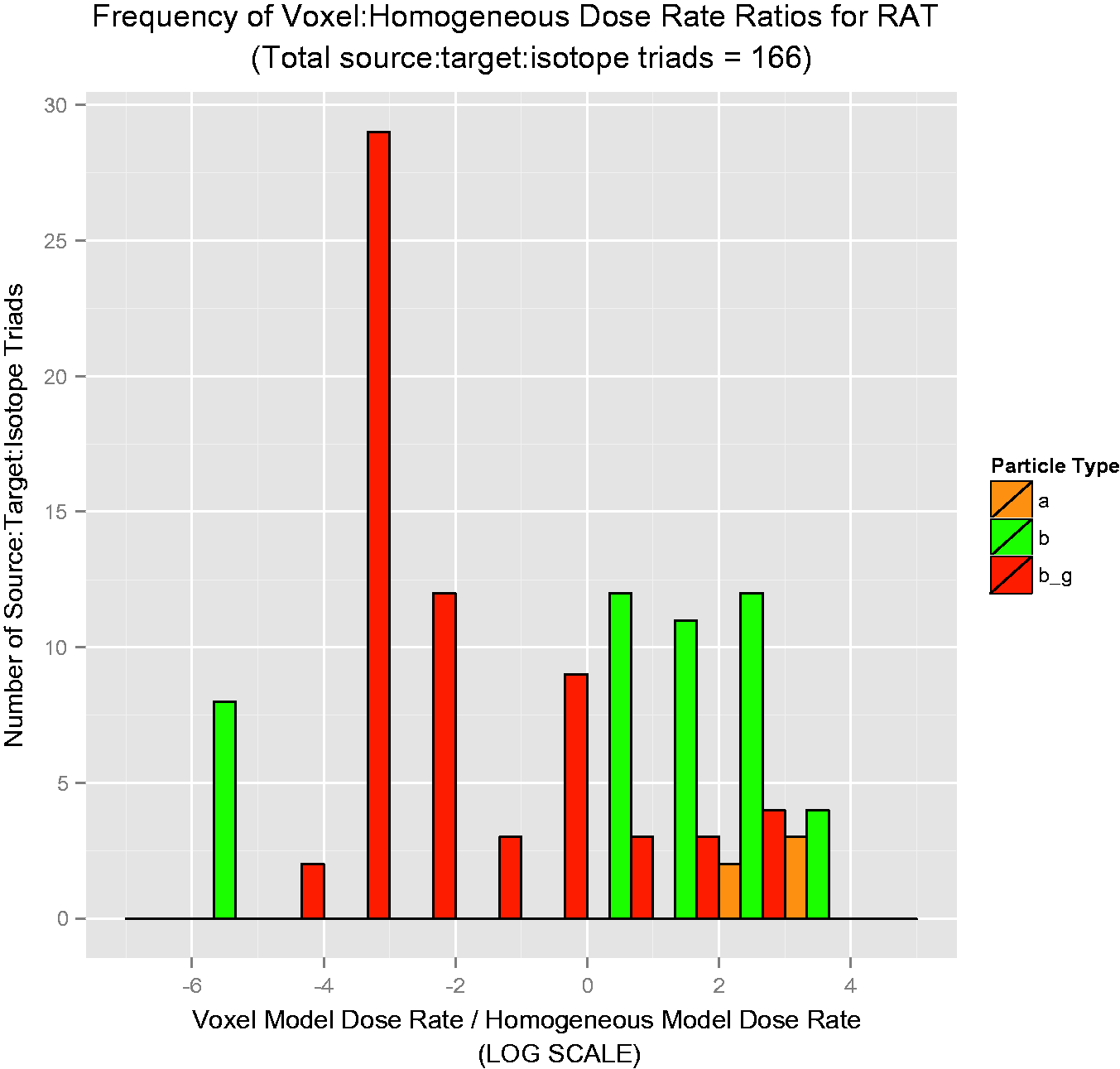
Over 50 graphs such as those in Figs 4–7 were created. To show the data in a more concise manner, histograms describing the results were created. They are shown in Figs 8–11, and these log-scaled plots illustrate the distribution of deviations among all radionuclides for the four RAPs studied (Ruedig et al., to be published). Yellow bars indicate alpha emitters, green bars indicate pure beta emitters, and red bars indicate mixed beta/gamma emitters. Each of the histograms in Figs 8–11 contain data from all radionuclides included in the study: 14C, 36Cl, 60Co, 90Sr, 131I, 137Cs, and 210Po. Fig. 8 compares 871 source:target values for Reference Flatfish, and found that the homogeneous model is primarily conservative for pure beta emitters, but less so for mixed beta/gamma emitters. Fig. 9 illustrates comparisons for 166 source:target values, and in this case, the homogeneous model is primarily conservative for mixed beta/gamma emitters, but not conservative for alpha and most pure beta emitters. Fig. 10 portrays the comparisons for 176 source:target values in Reference Crab, with the same results as seen in Reference Rat: the homogeneous model is conservative for mixed beta/gamma emitters, but not conservative for alpha emitters and the majority of beta emitters. Fig. 11 compares 871 source:target values for Reference Trout, and has results akin to Reference Flatfish: the homogeneous model is conservative for pure beta emitters, but not conservative for mixed beta/gamma and alpha emitters.
Reference Flatfish: distribution of voxelised:simplified ratios for all radionuclides studied. Reference Rat: distribution of voxelised:simplified ratios for all radionuclides studied. Reference Crab: distribution of voxelised:simplified ratios for all radionuclides studied. Reference Trout: distribution of voxelised:simplified ratios for all radionuclides studied.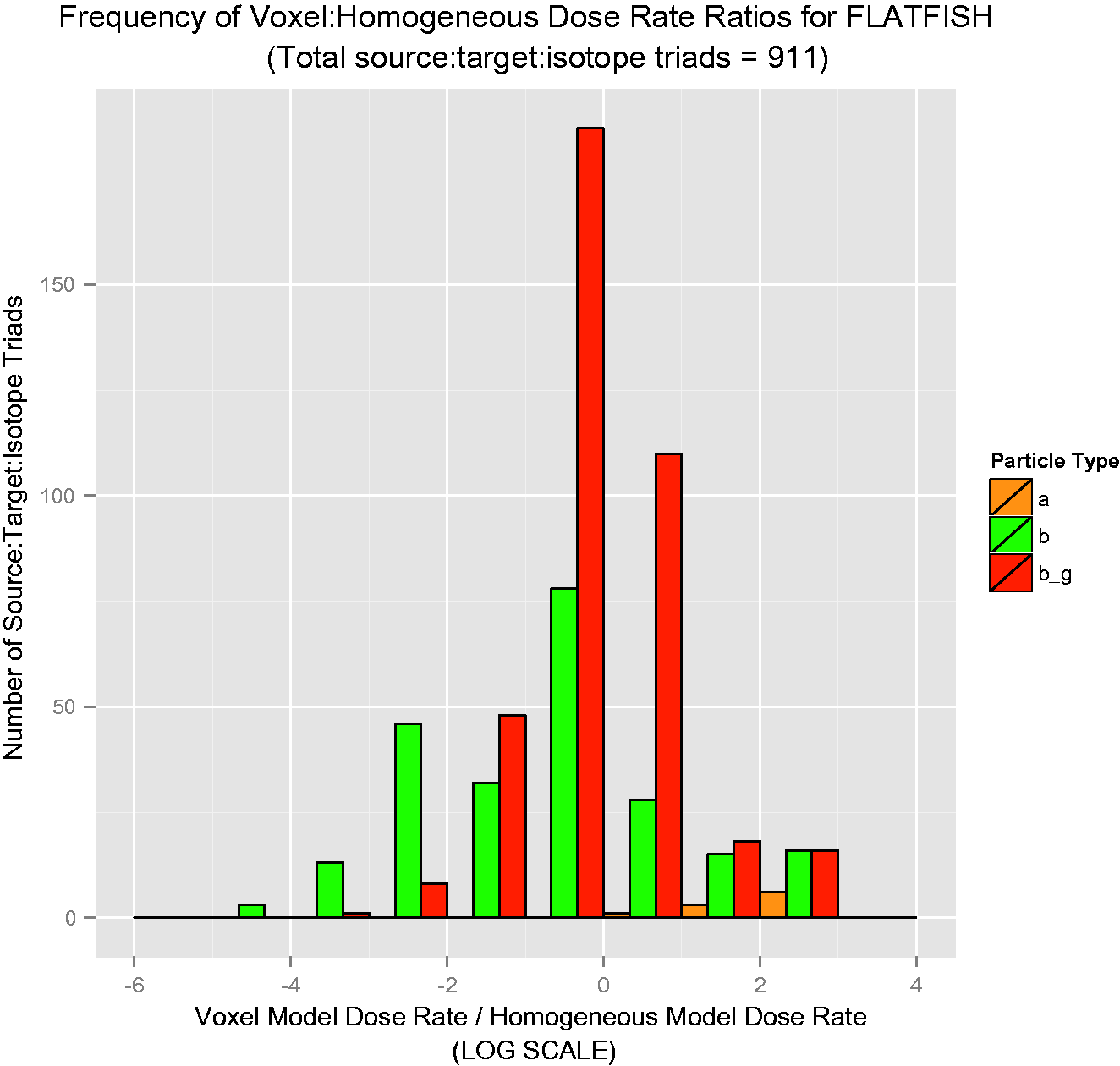
5.2. Voxel model dose conversion factors compared with mass ratios
In 2008, Gómez-Ros et al. derived a relationship that allowed for the estimation of organ dose rates from ellipsoidal geometries, assuming both a central and eccentric point source (Gómez-Ros et al., 2008). In order to calculate an organ-specific dose rate from whole-body absorbed fractions, Gómez-Ros et al. defined the mass ratio (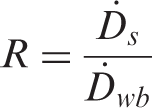
Gómez-Ros et al. (2008) showed that the maximum value of 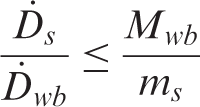
This ratio suggests that the dose rate to a small organ from a heterogeneously partitioned source can be large compared with the hypothetical case of the same source distributed homogeneously throughout an organism’s body. It also provides a rigorous methodology for estimating organ dose rates from existing dosimetric data for homogeneously distributed sources, by multiplying the whole-body dose rate by the ratio
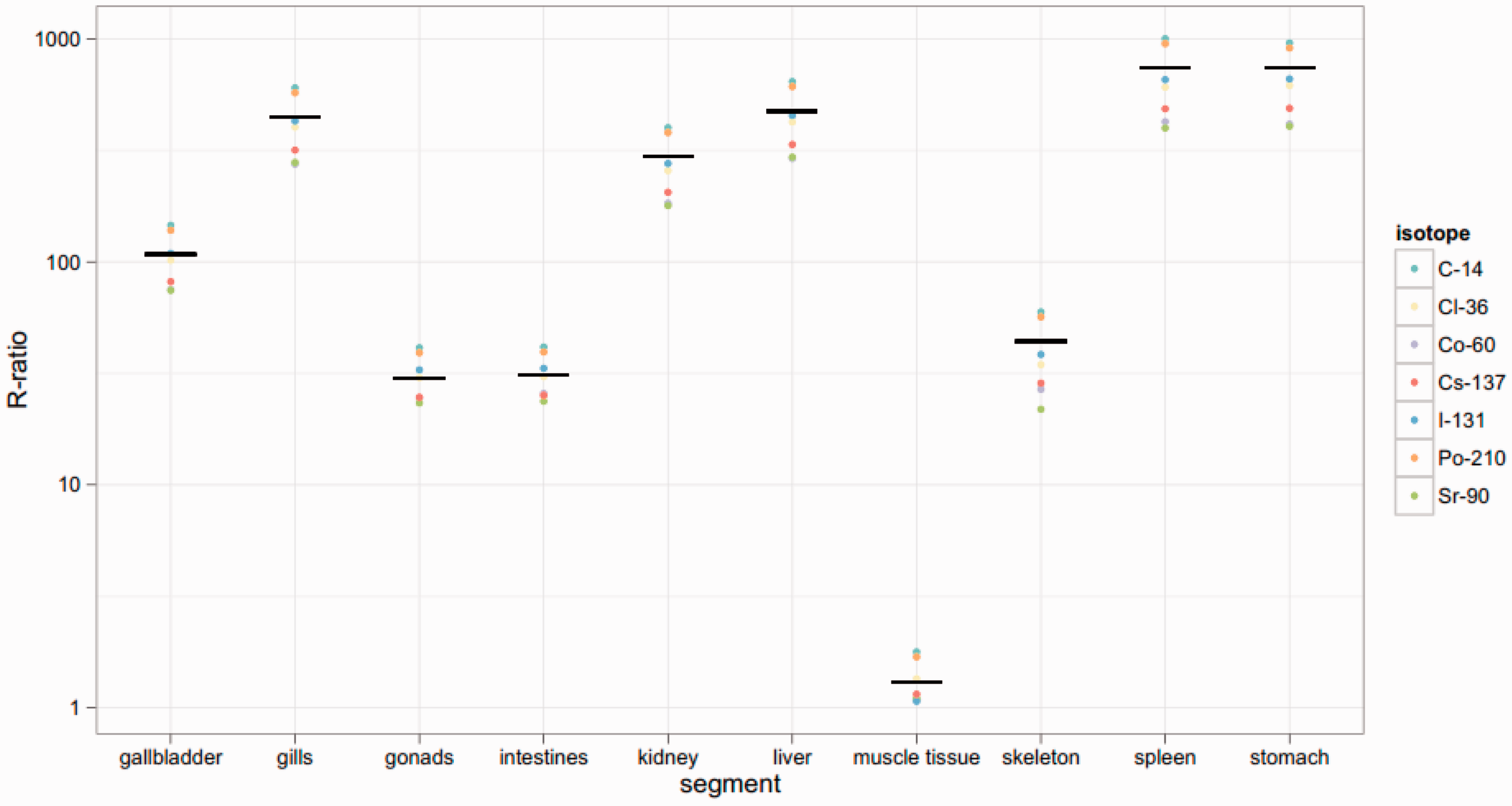
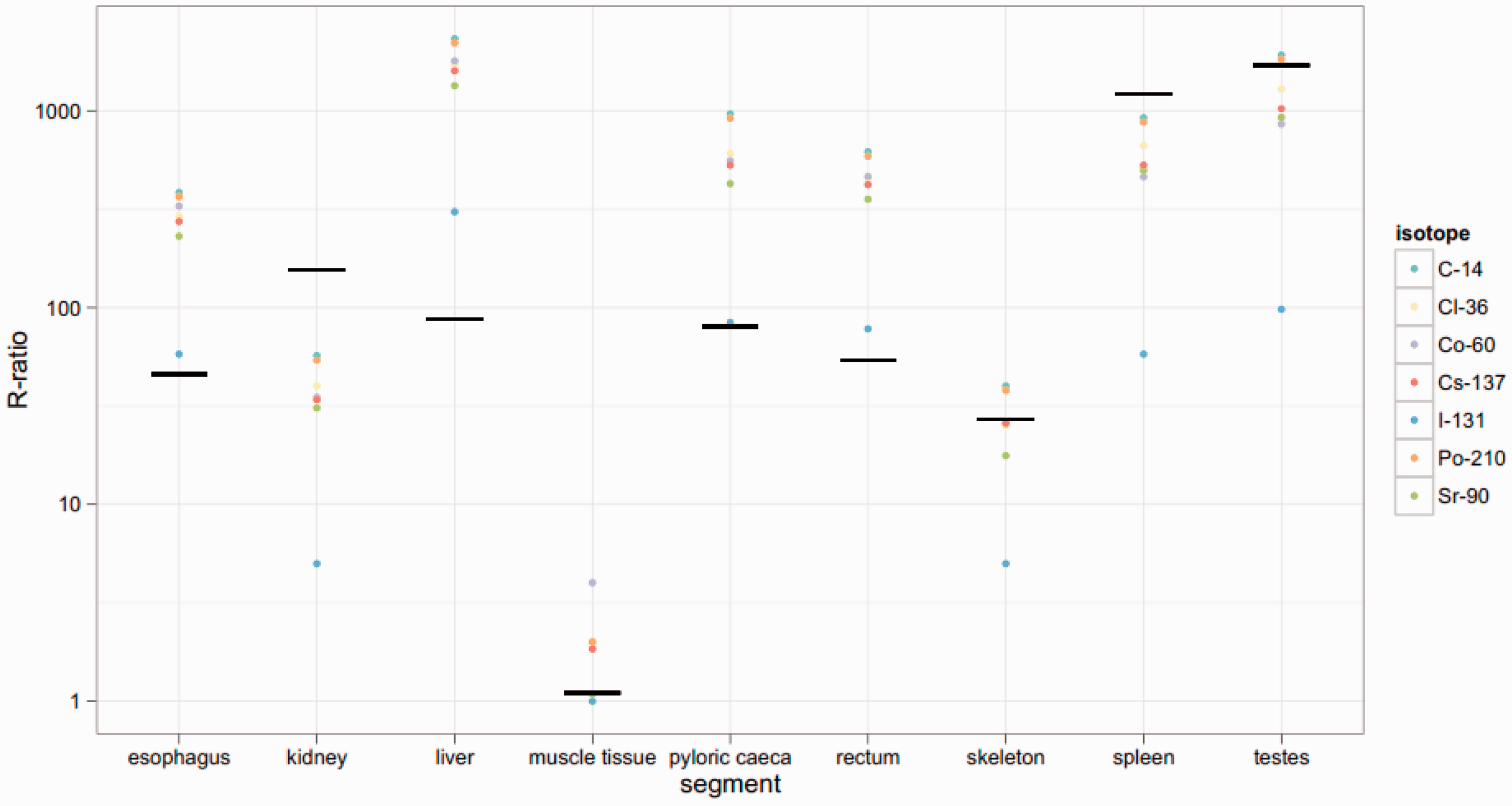
Figs 12–14 show dose rates calculated via mass ratios compared with those calculated for each segmented portion of the organism for Reference Crab, Reference Flatfish, and Reference Trout, respectively. It can be observed in Figs 12 and 13 that the mass ratio value is remarkably consistent with the voxelised model. However, as shown in Fig. 14, the mass ratios for Reference Trout match the voxelised model poorly. This suggests that there are some instances in which mass ratios may be lacking.
Reference Crab: Reference Flatfish: Reference Trout: 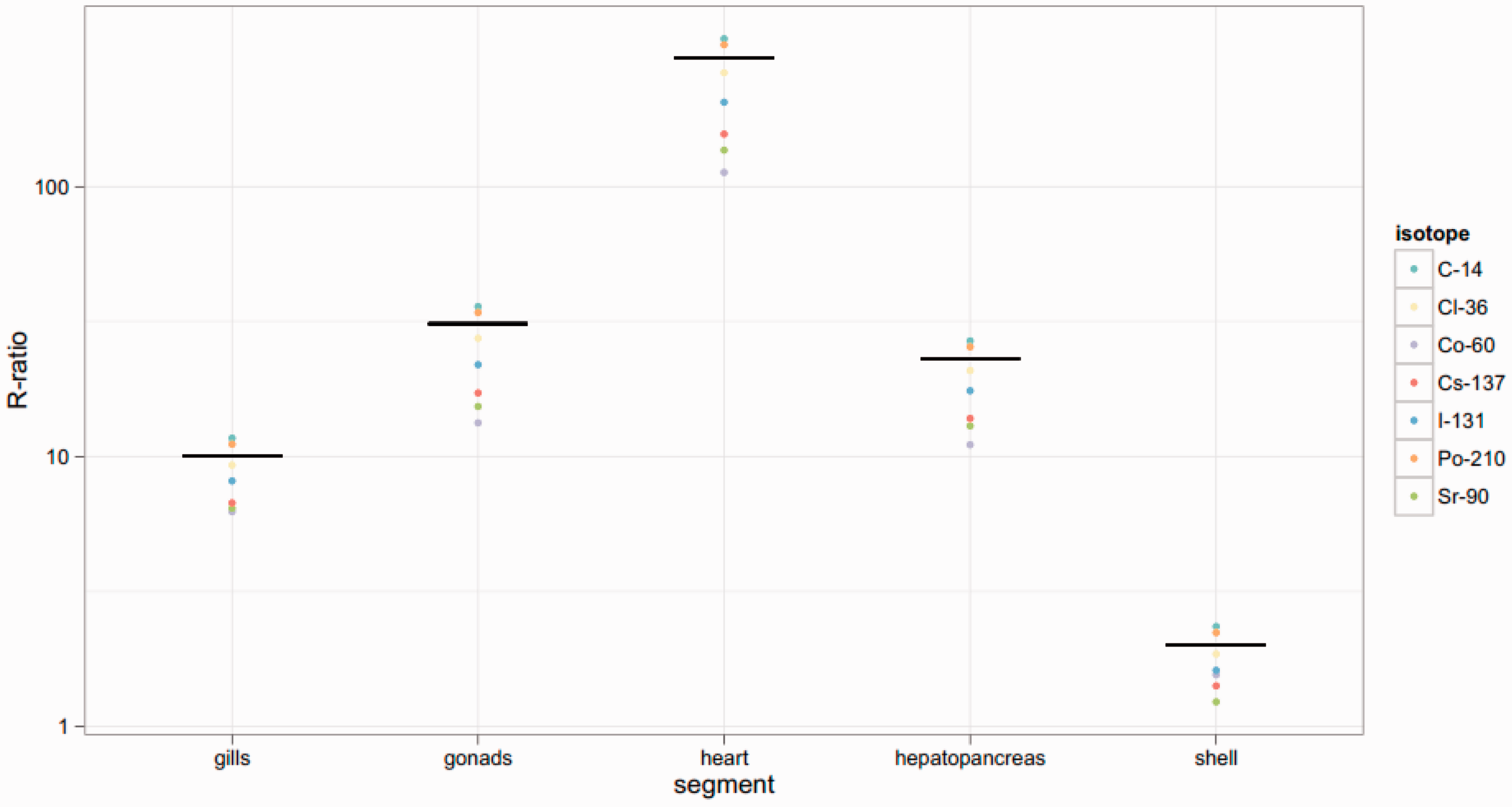
6. Discussion
In a general sense, dose rates from voxelised models and simplified models only agree well for pure beta emitters. Beta-/gamma-emitting radionuclides show the greatest deviations in dose rates. These comparisons serve to illustrate the discrepancies seen in the comparison of homogeneous with voxelised models. In Reference Rat, the simplified model was generally conservative, but in Reference Trout, voxelised dose rates were generally higher, regardless of radionuclide. The only generalisations that can be drawn from the data are that voxelised dose rates were consistently higher than homogeneous dose rates for alpha emitters. This is likely due to the short track length of alpha particles in tissue; alpha emitters are more likely to deposit all their energy in the source organ. In the homogeneous model, the energy is distributed over a much greater mass, and thus the overall dose rate is lower than that seen in the voxelised scenario. It is relevant to note that alpha emitters are among those radionuclides most likely to partition preferentially into specific organs, and thus the use of voxelised models for scenarios in which alpha emitters are present may be suggested.
7. Conclusions
The comparisons described above serve as a first-run examination of voxelised vs homogeneous models. The only clear conclusion that can be drawn with any level of certainty is that the models do not agree well, nor is one model consistently conservative.
This study should be taken for what it is: a first examination of voxelised vs homogeneous data. It neglects biology, and therefore organ:radionuclide pairs that are unlikely/impossible are considered in the analysis. In addition, most radionuclides rarely partition themselves perfectly, so the scenario above represents the most extreme case. More realistic comparisons that consider some of these nuances represent the next step in the process of determining the best method for expending resources on environmental protection.
