Abstract
Recent record-linkage studies of cancer risk following computed tomography (CT) procedures among children and adolescents under 21 years of age must be interpreted with caution. The reasons why the examinations were performed were not known, and the dosimetric approaches did not include individual dose reconstructions or account for the possibility for missed examinations. The recent report (2013) on children by the United Nations Scientific Committee on the Effects of Atomic Radiation concluded that the associations may have resulted from confounding by indication (also called ‘reverse causation’), and not radiation exposure. The reported cancer associations may very well have been related to the patients’ underlying health conditions that prompted the examinations. Reverse causation has been observed in other epidemiological investigations, such as a Swedish study of thyroid cancer risk following I-131 scintillation imaging scans, and in studies of brain cancer risk following Thorotrast for cerebral angiography. Epidemiological patterns reported in the CT studies were also inconsistent with the world’s literature. For example, in a UK study, teenagers had a higher risk of brain tumour than young children; in an Australian study, cancers not previously linked to radiation were significantly elevated; and in a Taiwanese study, the risk of benign tumours decreased with age at the time of CT examination. In all studies, solid tumours appeared much earlier than previously reported. Remarkably, in the Australian study, brain cancer excesses were seen regardless of whether or not the CT was to the head, i.e. a significant excess was reported for CT examinations of the abdomen and extremities, which involved no radiation exposure to the brain. In the UK study, the significance of the ‘leukaemia’ finding was only because myelodysplastic syndrome was added to the category, and there was no significance for leukaemia alone. Without knowledge of why CT examinations were performed, any future studies will be equally difficult to interpret. It is noteworthy that two recent studies of children in France and Germany found no significant excess cancer risk from CT scans once adjustment was made for conditions that prompted the scan, family history, or other predisposing factors known to be associated with increased cancer risk. Nonetheless, such studies have heightened awareness of these relatively high-dose diagnostic procedures, and the need to reduce unnecessary examinations and lower the dose per examination commensurate with the desired image quality.
1. INTRODUCTION
Computed tomography (CT) examinations for imaging have increased markedly over the last 20 years (NCRP, 2009). In the USA alone, over 85 million CT examinations were performed in 2012, and the overall use per year, while slowing, continues to rise (PRWeb, 2012). These examinations are associated with absorbed doses that are much higher than those associated with chest x-rays and most other diagnostic procedures. A CT examination to the abdomen could result in a stomach dose as high as 10 mGy, whereas a neonatal abdominal CT could result in a stomach dose of the order of 20 mGy (Brenner and Hall, 2007; Huda and Vance, 2007; Huda and Mettler, 2011). A single examination, however, only carries a small theoretical cancer risk, and this future risk must be balanced with the immediate clinical benefit. If an imaging scan is clinically warranted in young adults, the immediate benefits far outweigh a very small theoretical long-term risk (Zondervan et al., 2013). Diagnostic scans also reduce the number of invasive procedures, unnecessary hospital admissions, and the length of hospital stays. Nonetheless, because of the possibility that multiple CT examinations of children and young adults could result in organ doses in the range where epidemiological studies might be able to reveal risk, studies of CT examinations in childhood have the potential to provide new information on the risk of cancer following exposures in childhood (Boice, 2014).
Unfortunately, the methodological deficiencies of recent CT studies from the UK (Pearce et al., 2012), Australia (Mathews et al., 2013), and Taiwan (Huang et al., 2014) limit their relevance to both clinical practice and understanding low-dose radiation health effects. The critical problem is that the reasons for performing the CT examinations were not known, and these reasons (rather than the CT radiation dose) were the probable cause of the subsequent cancer diagnoses. This is called ‘confounding by indication’ or ‘reverse causation’. No individual dosimetry was performed, no information on the actual CT scanners that exposed the studied populations was available, and the acknowledged problem of incomplete documentation of the number of CT examinations performed raises serious questions regarding the validity of the organ-specific dose estimates, and the purported dose–response relationships. These criticisms have been raised by scientific committees (NCRP, 2012; UNSCEAR, 2013) and others (Walsh et al., 2013, 2014). Recent CT studies from France and Germany confirm that confounding by indication can play a major role in distorting reported risk estimates if not properly taken into account (Journy etal., 2015; Krille et al., 2015).
2. THE UK STUDY
The UK CT study (Pearce et al., 2012) was a record-linkage study of leukaemia [and myelodysplastic syndrome (MDS)] and brain cancer incidence following CT scans to 178,000 individuals at 0–21 years of age. Collection of the scan data for individual patients was not possible, and thus average CT machine settings from two national surveys were used to estimate organ doses. In the absence of individual dosimetric information, the authors conducted dose–response analyses for leukaemia and MDS, and combined and estimated the excess relative risk per Gy to be 36, which is extremely high. Similarly, for brain cancer, a significant dose–response relationship was reported with an excess relative risk per Gy of 23. A recent United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR) report concluded, ‘… There are concerns about the risk estimates because of lack of information about indications for the CT scans and the consequent potential for ‘reverse causation’ (i.e. cancers may have been caused by the medical conditions prompting the CT scans rather than by the CT dose), and lack of individual dosimetry’ (UNSCEAR, 2013). Similarly, the National Council on Radiation Protection and Measurements (NCRP) concluded that, ‘Children who receive frequent examinations may have some underlying disability related to the outcome of interest. That is, a child who receives multiple CT examinations of the head may have a central nervous system disorder that is prompting such examinations’ and it is these underlying disorders that are related to the cancer diagnosis and not the CT radiation dose (NCRP, 2012).
Risk of brain tumour related to age at computed tomography examination among patients in the UK paediatric study (Pearce et al., 2012). Note that the Japanese atomic bomb survivor study had the opposite pattern: ‘The risk of glioma is highest at <5 years at irradiation and seems to largely disappear at the age of 20 years or more at irradiation, suggesting that susceptibility decreases as brain development nears completion’ (UNSCEAR, 2013).

ERR, excess relative risk.
The UK study excluded patients who developed a brain tumour within 5 years of their first CT examination, but this does not strictly address the issue of a short latency or possible dose-response relationship. The risk of brain tumour was concentrated among children who received multiple CT examinations, so brain tumours within 5 years of the last CT examination were included in the analysis, and may very well have distorted the results. Regardless, latency or time from exposure to occurrence was much shorter than observed in other studies (NA/NRC, 2006; UNSCEAR, 2008). Radiation-induced brain cancer is an example of the substantial variance with other studies of high-quality dosimetry and epidemiological rigor. The effect of age at CT exposure for radiation-induced brain tumour was the opposite of what has been observed in studies of Japanese atomic bomb survivors, Israeli children treated with radiation for ringworm of the scalp, and survivors of childhood cancer (Ron et al., 1988; Neglia et al., 2006; UNSCEAR, 2008). The risk following CT exposure in the UK increased with age at exposure, which is the opposite pattern seen in all previous studies where children under 5 years of age at exposure have the highest risk, and teenagers and young adults have minimal risk, if any.
The UK study appears to be the first radiation epidemiological investigation to combine leukaemia and MDS, and little if any justification was given for doing so. The UNSCEAR Committee noted, ‘For 74 observed leukaemias, they found an excess relative risk Gy−1 of 36 … However, they included MDS with the leukaemias, and the MDS cases had an extremely high relative risk. Without the MDS cases, the estimated risk was still high, but no longer statistically significant’ (UNSCEAR, 2013). NCRP stated, ‘MDS accounted for 12% of the 74 leukemic conditions in the analysis’. MDS may be related to radiation, and this has recently been reported among long-term survivors from Nagasaki (Iwanaga et al., 2011). There is no evidence in any study that MDS occurs shortly after exposure. Further, in contrast to studies of atomic bomb survivors (Hsu et al., 2013), the leukaemia excess in the UK study was due to acute lymphocytic leukaemia, and no increase was seen for acute myelogenous leukaemia.
Similarly to the CT-induced risk of brain tumour, the non-significant excess relative risk per Gy of 19 for leukaemia (excluding MDS) was higher than seen in previous studies, including that of the atomic bomb survivors which reported an excess relative risk per Gy of 6.5 for children and young adults exposed under 20 years of age (Hsu et al., 2013). Interestingly, there are statements in UNSCEAR (2013) that may be slightly misleading regarding age at exposure effects among atomic bomb survivors. First, it was stated that very high risks were seen shortly after exposure among children under 10 years of age at exposure; however, this analysis was based on a statistical model that did not describe the incidence data, as well as the more recent analysis by Hsu et al. (2013) which showed a lower risk. Second, UNSCEAR (2013) stated that there was a substantial excess of childhood leukaemia cases among those exposed at less than 10 years of age and followed to 5–14 years of age: 10 observed against approximately 1.6 expected. Unfortunately, the expected value was based on Japanese general population rates for 1950 which have been judged to be inappropriate or, at best, misleading for comparison with the Life Span Study. Computations of observed over expected values from population rates have been found to give high-sided estimates of risk, and the elevations appear to be ‘primarily due to non-radiation factors’ (Grant et al., 2009).
3. THE AUSTRALIAN STUDY
Cancers reported to be significantly increased in the data-linkage study of 680,000 Australian children (aged 0–19 years at exposure) who received computed tomography (CT) scans and 10,000,000 Australian children with no record of such exposures (Mathews et al., 2013).

These sites were not found to be increased following radiation exposure (UNSCEAR, 2008).
Brain cancer excesses were seen regardless of whether or not the CT was to the head, i.e. significant excesses were reported for CT examinations of the abdomen and extremities that involved no radiation exposure to the brain.
All other sites were reported to have significant or non-significant excesses.
These sites are highly radiosensitive and frequently found to be elevated in studies of childhood and young adult exposures.
Excesses were significantly elevated for melanoma and Hodgkin’s lymphoma; cancers that are not inducible by ionising radiation (UNSCEAR, 2008). Remarkably, the associations with brain tumour were found regardless of whether the CT was to the head or to another part of the body. In other words, a significant association for brain tumour was found for CT scans of the abdominal region and extremities that resulted in no radiation exposure to the brain, clearly suggesting reverse causation (confounding by indication) where the conditions being screened or the characteristics of the patient were the cause of the CT examinations, rather than the CT examinations being the cause of the subsequent tumour.
UNSCEAR (2013) concluded that the Australian study has methodological flaws that resulted in implausible risks. Reverse causation was again raised as an explanation for the associations, i.e. that the cancers were caused by the medical conditions prompting the CT scans rather than by the CT dose. This potential bias could not be examined as no documentation was available on the indications for the CT scans. The UNSCEAR Committee went on to conclude that the exceptionally high-risk estimates for ‘all cancers, excluding brain cancer after brain CT’ risk (excess relative risk Sv−1) were statistically incompatible with the data at comparable ages from the Japanese Life Span Study on atomic bomb survivors: 27 [95% confidence interval (CI) 17–37] vs 3 (95% CI 2–6).
The Australian findings were also at odds with previous studies in that two highly radiogenic sites were not elevated, i.e. breast cancer and acute lymphoid leukaemia were not increased. Breast cancer has been found to be elevated in several studies of childhood and young adult exposures to fractionated diagnostic radiation (Boice and Monson, 1977; Hoffman et al., 1989), and the absence of this tumour in contrast to the elevation of non-radiogenic sites raises caution in making any causal interpretations for the associations seen in this study (Walsh et al., 2013, 2014). Both acute lymphocytic leukaemia and acute myelogenous leukaemia were increased in the study of atomic bomb survivors (Hsu et al., 2013); in contrast, the UK CT study did not find acute myelogenous leukaemia to be increased, whereas the Australian CT study did not find acute lymphocytic leukaemia to be increased.
4. THE TAIWANESE STUDY
For completion, a recent record-linkage study was conducted in Taiwan to evaluate CT examinations of the head and subsequent development of benign brain tumour (Huang et al., 2014). Approximately 24,000 children with head CT were matched to four times this number of non-exposed children, i.e. children who did not receive a head CT. Any child with a reported diagnosis of cancer or conditions predisposing to brain tumour was excluded. A significant association with benign, but not malignant, brain tumour was reported. The reasons why the CT examinations were performed were not known. Apparently, children with benign tumours were not excluded, leaving open the real possibility that the results were confounded by indication, i.e. the symptoms prompting the CT scan were indications of an underlying brain tumour. This bias is supported by the peculiar epidemiological patterns that the risk decreased with time since CT examination, and that the oldest, not the youngest, subjects were at highest risk. Also, the study is limited by the small number of subsequent tumour cases (only five malignant and 14 benign tumours). The reported associations would essentially disappear if only a few of these brain conditions were present at the time of the CT examination. No dosimetry was performed. Similar to the UK and Australian studies, reverse causation implies that it is the early symptoms of undetected tumour, or of factors that predispose to tumour, that are the indications for the CT scans, rather than the CT scans per se that are causing the apparent excess risk of cancer.
5. CHALLENGES OF INDIVIDUAL DOSE ASSESSMENTS IN THE ABSENCE OF INDIVIDUAL DOSIMETRIC DATA
Collection of the scan data for individual patients and organ doses for individuals based on their exposure characteristics was not possible in any of the studies; therefore, average CT machine settings from national surveys were used. The resulting organ dose estimates made are highly uncertain and of questionable validity. A somewhat arbitrary year (2001) was chosen in the CT studies as the demarcation of the high exposures from CT examinations in previous years (conventional CT) and the lower exposures in use nowadays (helical CT) (Kim et al., 2012). This distinction is important because the scan time for paediatric patients with a conventional CT scanner could be from 10 to 35 min in the 1990s compared with 50–60 s nowadays with a helical CT scanner (Kaste et al., 1997).
For children examined during the early years of CT imaging, there is the likelihood that any movement during the examination, which could take up to 35 min for conventional CT scanners, would result in a blurred image and prompt a repeat examination (NCRP, 2012). Unfortunately, evidence of repeat examinations was not available from the electronic databases accessed in either the UK or Australian studies.
Accounting for chronological changes in CT dosimetry is challenging, complex, and complicated. Machines changed, time per examination changed, and exposure factors for children changed with time. The rapid and continued changes in CT technology are remarkable (Frush et al., 2013). These changes, however, were not simultaneous across nations, and not even in single hospitals. One important factor that influenced patient dose is when the exposure technique for children was lowered from that used for adults. The desired image quality was achieved at a much lower exposure level when the examinations were ‘child-sized’ (Goske et al., 2008, 2012). This change meaningfully affected the organ dose to a child, but unfortunately, no data are available for the actual exposure parameters for individuals in the studied populations.
Missed CT exposures also include any examinations occurring outside the age or calendar year ranges of the study design, i.e. CT examination occurring after the defined paediatric ages and calendar years of study eligibility would not be recorded, yet cancer diagnoses after the paediatric ages would be included. This combination of missing doses would tend to inflate the estimate of risk per unit dose.
6. EXAMPLES OF REVERSE CAUSATION (CONFOUNDING BY INDICATION)
Reverse causation bias (confounding by indication) observed in a Swedish cohort study of thyroid cancer following I-131 scans (Dickman et al., 2003). Significant increases of thyroid cancer were observed only among patients examined with I-131 scans for suspicion of thyroid tumour, and this bias lasted for more than 20 years after the initial examination. The ‘other reasons’ for examination were mainly related to benign thyroid conditions such as hypothyroidism or hyperthyroidism.
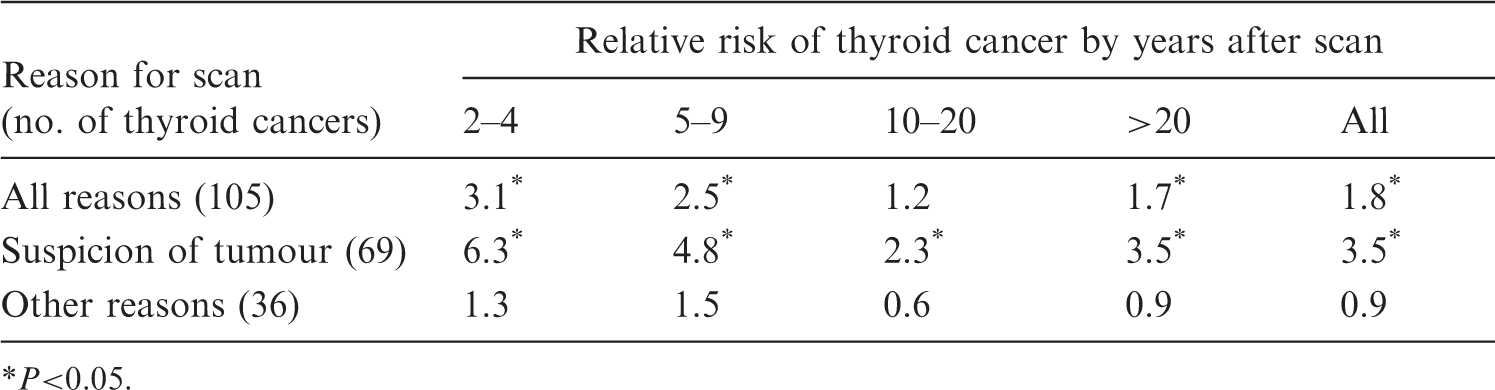
When the patients were divided into two examination categories, suspicion of tumour and other reasons, it was evident that the significant association was due entirely to the patients who were examined because of suspicion of tumour, with the relative risk overall being 3.5 (significant). For those patients who were examined for reasons other than suspicion of tumour, there was no evidence for an association between the I-131 scan and thyroid cancer (relative risk 0.9). The reverse causation bias lasted for more than 20 years after the initial I-131 examination, in that the suspicion of thyroid tumour was significantly associated with the occurrence of thyroid cancer even 20 years after the initial scintillation scan. Further, because the vast majority of these patients were over 20 years of age when examined, the observed high risk of thyroid cancer was not expected as the adult thyroid gland is relatively resistant to cancer induction by radiation (UNSCEAR, 2008).
Another example of reverse causation bias in radiation epidemiological studies is in: patients given radioactive Thorotrast (a colloid of thorium dioxide) as the contrast medium for cerebral angiography for suspicion of brain tumour (Dos Santos Silva et al., 2003; Travis et al., 2003). Patients in the USA (Travis et al., 2003) who had angiographic examinations because of suspicion of tumour were significantly more likely to develop brain cancer regardless of whether or not the examination included radioactive Thorotrast as the contrast medium [standardised mortality ratio (SMR) 33.6], or whether the examination involved a non-radioactive contrast medium (SMR 15.6). The relative risk of brain cancer comparing the Thorotrast group with the non-radioactive contrast medium group was 1.3 (95% CI 0.6–3.7). Similarly, in Portugal, the risk of dying from a nervous system disease was greatest in the subgroup of patients who received Thorotrast angiographic procedures because of suspicion of a nervous system condition (relative risk 7.6, 95% CI 1.22–317) (Dos Santos Silva et al., 2003). Again, it can be concluded that the preclinical symptoms of brain cancer prompted the clinician to request a diagnostic imaging scan, and it was the preclinical symptoms that led to the overt diagnosis of brain cancer, not the radiation dose received from Thorotrast.
A final example of confounding by indication cropping up in epidemiological studies involving diagnostic radiation is a recent report of meningioma associated with dental x rays (Claus et al., 2012). Putting aside for the moment the fact that the study was based on patient recall of dental x rays many decades before the diagnosis of meningioma, and that the high-dose dental examinations were associated with the lowest risk, confounding by indication was likely because of the association of jaw pain with both meningioma symptoms and presentation for dental x rays. Meningiomas can cause referred pain to the orofacial region. A patient with such pain may receive dental x rays during the course of his/her care. It may be that radiographs do not cause meningiomas, but rather the presence of tumours triggers the need for radiographs (White et al., 2013). In other words, the dental x rays did not cause the meningiomas; the meningiomas caused the dental x rays.
7. CONCLUSIONS
Epidemiological studies have value in drawing attention to diagnostic CT examinations and the need to reduce unnecessary examinations, as well as to reduce the dose per examination commensurate with desired image quality for clinical evaluation. Programmes and initiatives exist within the medical community to raise awareness about the need for dose reduction and for considering alternative modalities among both children and adults: Image Gently (Goske et al., 2008, 2012); Image Wisely (Brink et al., 2010); and Choosing Wisely (Rao and Levin, 2012, 2014). These efforts should be encouraged even in the absence of direct evidence that CT radiation exposures are causally linked to excess risk of cancer. The epidemiological evidence to date is limited as summarised below.
Current studies of paediatric CT examinations and subsequent cancer risk do not provide evidence that low doses are causally associated with cancers in children, nor are they of much clinical relevance. Statistical associations can result from study biases as apparent in the UK, Australian, and Taiwanese studies. Reverse causation (confounding by indication) is a likely reason for the observed associations, i.e. the condition being evaluated prompted the CT examinations, and was also the cause of the subsequent cancer diagnosis. It is thus noteworthy that two recent studies of nearly 70,000 children in France (Journy et al., 2015) and nearly 45,000 children in Germany (Krille et al., 2015) found no significant excess cancer risk from CT scans – once adjustment was made for conditions that prompted the scan, family history, or other predisposing factors known to be associated with increased cancer risk.
Inconsistency with the world's literature on radiation-induced cancers following childhood irradiation include: age at exposure effects being highest for exposures at older and not younger ages (most notably for brain tumour); latency or time from exposure to occurrence being much shorter than observed previously; tumour sites that have never been associated with radiation were increased whereas some of the most sensitive organs were not increased (Australian study); and the computed radiation risks per Gy were so extremely high as not to be believable (in the author’s view).
Collection of the scan data and associated exposure information for individual patients was not done. Using national survey data to estimate scanner characteristics for individual procedures over a period of decades is of questionable validity. Doses from conventional and helical scanners are very different, and the type of scanner was not known for any individual patient. Exposure techniques changed over time, notably when it was recognised that adult techniques were providing unnecessary radiation dose to children of much smaller size. Excess relative risk per mGy is very difficult to interpret based on substantial uncertainties in organ doses associated with type of CT scanner, type of procedure, changes in scanners and technique over calendar year, missed examinations due to movement and blurred images prompting repeat examinations, and missed examinations that occurred outside the timeframe of the study age and calendar year eligibility criteria.
Unfortunately, these criticisms and methodological limitations have not discouraged recent media articles from claiming that epidemiology has now demonstrated directly that cancer risk is associated with CT examinations, concluding, ‘ … we need to find ways to use them [CT examinations] without killing people in the process’ (Redberg and Smith-Bindman, 2014). Such exaggerations based on highly questionable epidemiological observations serve little purpose, and may very well discourage patients from seeking the medical care needed for their children to diagnose life-threatening injuries, prevent unnecessary surgery and medications, and identify treatable causes of suffering (Ellenborgen et al., 2014). Theoretical future risks must always be balanced against the immediate benefits of these CT examinations. Epidemiology is yet to provide convincing evidence of future cancer risks following diagnostic CT examinations.
Unless the reasons for the examination can be determined and dosimetry improved in future studies (Einstein, 2012; Thierry-Chef et al., 2013; Walsh et al., 2014), they too will be equally ambiguous.
