Abstract
Non-cancer effects and risks at low doses from ionising radiation are controversial topics within the field of radiation protection. These issues are discussed in International Commission on Radiological Protection (ICRP)
1. Introduction
Several publications by the International Commission on Radiological Protection (ICRP) outline risk assessment for cancer in great detail. Most recently, this was addressed in
In 2012, an extensive update to the recommendations regarding deterministic effects (now tissue reactions) was issued in
Recent evidence suggests that uncertainties exist between the risk of cardiovascular disease and the dose–response curve, calling an assumed linear relationship (as in
ICRP publications such as
2. Epidemiological analysis (dose–response curve)
Estimated excess risks for ‘all circulatory disease’ and cataracts in
2.1. Variations in dose-response for circulatory disease subtypes
While broad categories for circulatory disease (e.g. ‘all circulatory disease’, ‘heart disease’, or ‘stroke’) seem to indicate a positive dose–response relationship, closer inspection reveals some discrepancies. When examining the dose–response relationship for circulatory disease subtypes, the shape of the curve and even the apparent positive dose-response relationship may be questionable. For example, Ozasa et al. (2012) and Suzuki (2012) examined excess risks calculated using a linear model fit to atomic bomb life span study (LSS) data for well-defined subtypes of heart disease over discrete time intervals. The data show no significant excess risk for ischaemic heart disease over the subject time periods. In contrast, a significant excess risk is indicated for hypertensive heart disease for most time periods. In neither case is a linear curve necessarily indicated when the excess risks are plotted against absorbed dose.
Both studies suggest that a seeming linear dose–response relationship for the broad category of all cardiovascular disease may be an artefact of the combination of disease subtypes and time periods. In fact, certain well-defined disease subtypes tend to indicate non-linear responses with a potential threshold (e.g. rheumatic and hypertensive heart diseases). Suzuki (2012) demonstrated that in several studies pertaining to circulatory disease risks at low doses, the apparent dose–response relationship can vary when evaluated against additional factors (e.g. population, irradiated volume, etc.). ICRP suggested for non-cancer risks lean heavily on the LSS data. While these data may indicate a potential linear dose–response relationship for circulatory disease when taken as a whole, they may not support an argument for linearity when disease subtypes are examined individually.
Although the data from the two studies illustrate some uncertainties in the apparent positive dose–response relationship, the data also show the importance of considering the overall effect for lifetime data. The data for later time periods may indicate that there is some trend towards a positive dose–response relationship in later time periods for ischaemic heart disease, as might be expected for a late manifesting effect. However, in short, the analyses by Ozasa et al. (2012) and Suzuki (2012) demonstrated that a non-linear dose–response relationship with possible threshold may be a more accurate model for cardiovascular disease.
Like Suzuki (2012), the National Council on Radiation Protection and Measurements (NCRP) noted that only certain subcategories of heart disease demonstrate excess risks (NCRP, 2012). NCRP concluded that evidence supporting a reduced threshold for cardiovascular disease in the range of 0 to 0.5 Gy is weak (NCRP, 2012). Specifically, NCRP stated the following (NCRP, 2012).
In a review of several studies on the circulatory disease risks following low-dose radiation in medical settings, ‘inconsistent results…limit the confidence in establishing a single value for a low-dose threshold for cardiac effects following therapeutic radiations’ (p. 217). Similarly, a review of worker studies on the association between heart disease and radiation dose did not provide consistent evidence for a positive dose–response relationship, nor was it possible to exclude effects on the same order of magnitude as reported for the LSS cohort. NCRP supports the 2008 (United Nations Scientific Committee on the Effects of Atomic Radiation) conclusion by UNSCEAR that there is little evidence to ‘establish a causal relation following heart doses <1 to 2 Gy’ (p. 222).
2.1.1. Questions of linearity
Several other studies have illustrated an apparent non-linear dose-response relationship for circulatory disease risks. For example, Schöllnberger et al. (2012) used a multi-model inference method whereby various dose–response models were combined mathematically so that risk estimates could be based on several possible dose–response models. Using this approach on data for cerebrovascular disease yielded a weak dose–response relationship below approximately 0.6 Gy with a risk estimate approximately one-third of that estimated using a linear non-threshold model. Similarly, for circulatory disease, the combined model approach indicated zero risk below approximately 2.2 Gy. Takahashi et al. (2012) analysed stroke incidence data from the atomic bomb Adult Health Study, which also suggests a potential threshold for stroke.
2.1.2. Possible treatments for data
While some circulatory disease subtypes indicate little to no dose–response relationship in the low-dose region (or, at the very least, a non-linear dose–response) relationship, it must be acknowledged that other types of circulatory disease clearly show effects that should be accounted for. In this regard, the treatment of the data is important. For example, (a) circulatory disease data could be aggregated according to the affected organ or tissue (e.g. brain vs heart); or (b) specific subtypes of disease could be examined individually, possibly even with separate methods or adjustments for detriment.
The first method would have the advantage of providing some further differentiation and possibly some alleviation of the aforementioned ‘artefact’ of linearity from blending multiple disease subtypes, as there seem to be significant differences between the dose–response relationships for heart disease vs stroke. A disadvantage of this method would be that the artefact effect would not be eliminated completely, as some heart disease subtypes demonstrate positive dose–response relationships while others do not. Option (b), above, would eliminate any question of an artificially imposed dose–response relationship, although examining several possible disease subtypes begs the questions as to which subtypes to analyse and how to incorporate the risks from several different diseases (some of which may show excess risks whereas others do not). Another consideration is the fact that, in general, when data are divided into subtypes, larger uncertainties can arise with the use of smaller sample sizes.
Ultimately, if the shape of the dose–response curve is not truly linear, an assumed linear non-threshold relationship provides a possibly overly conservative estimate of risk. In addition, there is not sufficient evidence to maintain that low-dose/dose-rate exposures cause significant non-cancer health effects. Recent studies have emphasised the importance of including Adult Health Study data and the heterogeneity amongst disease subtypes when determining excess risks for non-cancer effects. Such findings indicate that further research into the dose–response relationship may be warranted.
3. Detriment
This section reviews the application of detriment to the calculation of cancer risks and the potential importance of this concept to current estimations of non-cancer risks.
3.1. Detriment and cancer risks
Stochastic (e.g. cancer) risks are modelled such that the probability of occurrence increases with dose. Severity for stochastic risks is expressed by adjusting the probability of occurrence using factors to account for various components of detriment. According to Detriment adjustment for cancer risks. LSS, Life Span Study.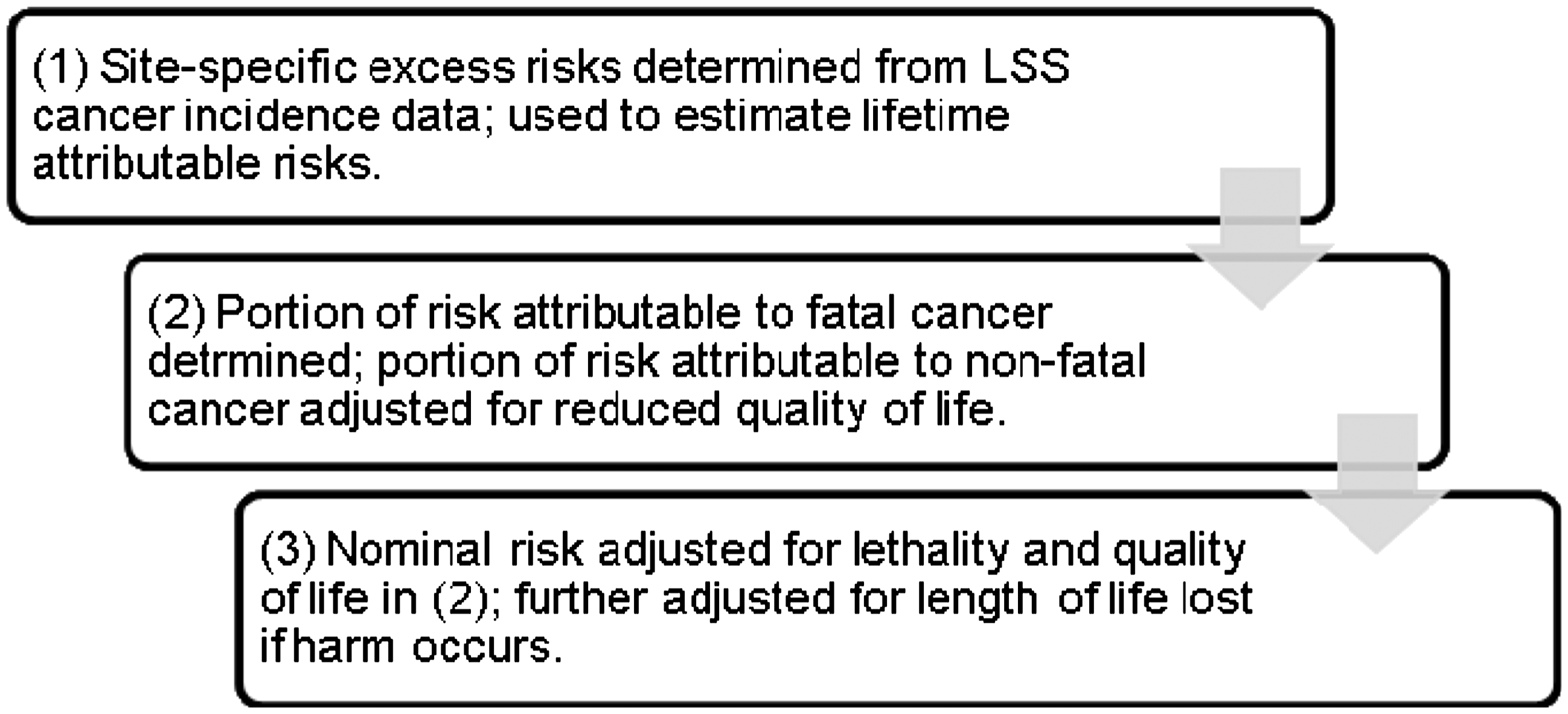
From here, dose limits are estimated based on a variety of considerations in addition to the calculated fatal cancer risk estimate.
3.2. Severity and non-cancer risks
For deterministic (non-cancer) effects, both the probability and the severity of an effect increase above some threshold doses. Traditionally [i.e. in
In
The concept of severity plays an important role in both cancer and non-cancer risk estimations. In
4. Mechanisms
The understanding of the mechanisms behind non-cancer effects will also contribute to a better interpretation of the dose–response relationship, although the study of biological mechanisms for radiation-induced cardiovascular effects is still nascent. There is even some indication that mechanisms for damage may be different at low doses compared with high doses (ICRP, 2012). Little et al. (2012) acknowledged that postulated mechanisms for cardiovascular disease are still not well understood due to several factors. For one, target tissues for circulatory system effects at doses less than approximately 0.5 Gy ‘remain uncertain’ (Little et al., 2012: 17). Secondly, although certain animal models (such as the ApoE (Apolipoprotein E) mouse) are used to study cardiovascular disease effects, there are no current animal models for cardiovascular disease that can be used confidently to translate results from animal studies to human application.
Recent research indicates that the mechanisms for both cancer and non-cancer effects likely include non-targeted effects (genomic instability and bystander effects) and inflammatory processes. Such processes have been primarily observed in in-vitro systems at low doses. However, if the fundamental mechanisms for radiation-induced damage are similar for cancer and non-cancer effects, it is worth exploring whether differences between the methodologies for cancer and non-cancer risk determinations can be resolved. Research into non-targeted effects is still relatively new for both cancer and non-cancer effects, although ongoing research into mechanisms of response could contribute to a better interpretation of dose–response curves for circulatory diseases and may even provide a basis for the future harmonisation of cancer and non-cancer risk estimates. At the very least, emerging results indicating that non-targeted effects may play a role in circulatory disease induction following exposure to ionising radiation suggest that much work is needed to reconcile epidemiological data with radiobiology at low doses.
Efforts to understand underlying biological mechanisms for cardiovascular disease are complicated by the existence of confounding factors and noises. Of particular interest in the current framework of radiological protection is the effect of stress on the development of cardiovascular disease. While the risks of physical health effects directly caused by ionising radiation decrease according to reductions in exposure dose, the same cannot necessarily be said of psychological trauma. Striving to quantify the separate contributions of ionising radiation and stress to the development of cardiovascular disease, while continuing to inform the public on the scientific results, is particularly important in light of the 2011 accident at the Fukushima Daiichi nuclear power plant. Eventually, it may even be possible to develop a radiological protection system that accounts for the social aspect of risks as well as physiological damage.
5. Conclusions
Due to the fact that recommendations and publications from ICRP carry great weight within the radiation protection community, discussions regarding the treatment of epidemiological data, shape of the dose–response curve, and definitions of severity and detriment should be ongoing. Potential benefits could also be realised by exploring the harmonisation of cancer and non-cancer risk estimates. For non-cancer risks, specifically, this will involve examination of the importance of including data from the Adult Health Study, recognising heterogeneity among disease subtypes, exploring the appropriateness of an incidence value, creating metrics to account for severity and detriment, and further researching mechanisms for damage.
The radiation protection community has begun to recognise the importance of this topic, even proposing at the 2012 Science and Values Workshop in Tokyo, Japan, an ICRP working group to address non-cancer detriment. This topic was also mentioned at a February 2013 workshop on ‘Radiation and cardiovascular disease’ hosted by the Radiation Effects Research Foundation. As further insights are made into the mechanisms of cancer and non-cancer effects from ionising radiation, it is important to explore potential paths forward towards definitions of severity and detriment, and the potential harmonisation of risk assessments.
