Abstract
Ecological risk assessments for radioactive substances are based on a number of inference rules to compensate for knowledge gaps, and generally require the implementation of a weight-of-evidence approach. Until recently, dose (rate)–response relationships used to derive radioprotection criteria for wildlife have mainly relied on laboratory studies from a limited number of species as representatives of biodiversity. There is no doubt that additional knowledge, combined with advanced conceptual and mathematical approaches, is needed to develop general rules and increase confidence when extrapolating from test species to complex biological/ecological systems. Moreover, field data sets based on robust sampling strategies are still needed to validate benchmark values derived from controlled laboratory tests, and to indicate potential indirect ecological effects, if any. This paper illustrates, through several examples, the need for implementing a combined laboratory–field-model approach to obtain science-based benchmark doses (or dose rates) (e.g. screening benchmarks for ecological risk assessments or derived consideration reference levels), based on robust meta-analysis of dose–effect relationships covering ecologically relevant exposure time scales, species, and endpoints.
1. INTRODUCTION
1.1. International background in brief
Over the last 15 years, there has been considerable international effort to investigate the issue of protection of the environment from radioactive substances. In Europe, much of the focus has been on collating relevant information and developing different approaches to enable regulatory assessments and protective actions to be undertaken [FASSET (Williams, 2004), ERICA (Larsson, 2008), PROTECT (Howard et al., 2010)]. These developments were well aligned with the European framework for managing environmental risk from chemicals. The major operational outcomes were: an ecological risk assessment (ERA)-type tiered approach and associated tool for characterising and managing the environmental risk for radioactive substances [the ERICA Integrated Approach (Larsson, 2008)]; the derivation of screening ecological benchmarks needed for implementation of the tiered approach (Garnier-Laplace et al., 2008); and the FREDERICA database collating primary data on effects of ionising radiation on non-human species (Copplestone et al., 2008). In the meantime, the United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR) reviewed the literature to update its estimates of effects of ionising radiation on non-human biota (UNSCEAR, 2008). Through
1.2. Basic definitions and extrapolation issues
Published effects benchmarks (dose rate in µGy h−1) for chronic exposure situations.

Garnier-Laplace et al
Reference ‘organism type’ refers to the International Commission on Radiological Protection’s Reference Animals and Plants (RAP). For any RAP, derived consideration reference levels are given in the last column as a band of dose rates within which there is likely to be the same chance of deleterious effects occurring to individuals of such type of organism.
2. Materials and methods
2.1. Overview of the method for estimating population effects for a given species by combining Leslie matrices and dose-rate–effects relationships
Generally, protection of the environment targets populations and ecosystems. However, most laboratory effect tests are performed at individual and subindividual levels. One of the most important challenges is to extrapolate measured effects on individual endpoints to the population. Focusing solely on individual endpoints can lead to inaccurate estimates of risk to populations due to the complexity and non-linearity of the relationship between effects on individual survival, reproduction, or growth, and population dynamics. The proposed method is based on the use of a Leslie matrix, where the population of a species is represented as an age-structured vector the survival rate the fecundity rate,
Based on dose-rate–effect relationships established for individual endpoints, responses to ionising radiation are expressed as reduction coefficients applied to each variable (Fig. 1). Such dose-rate–response relationships were built according to a well-defined procedure explained in detail in Garnier-Laplace et al. (2010). The method, initially proposed by Lance et al. (2012), has been generalised to representative wildlife species (Alonzo et al., 2013). Here, only two terrestrial species, a mammal ( Extrapolation approach proposed for predicting population-level response by combining dose-rate–effect relationships and an age-structured population matrix.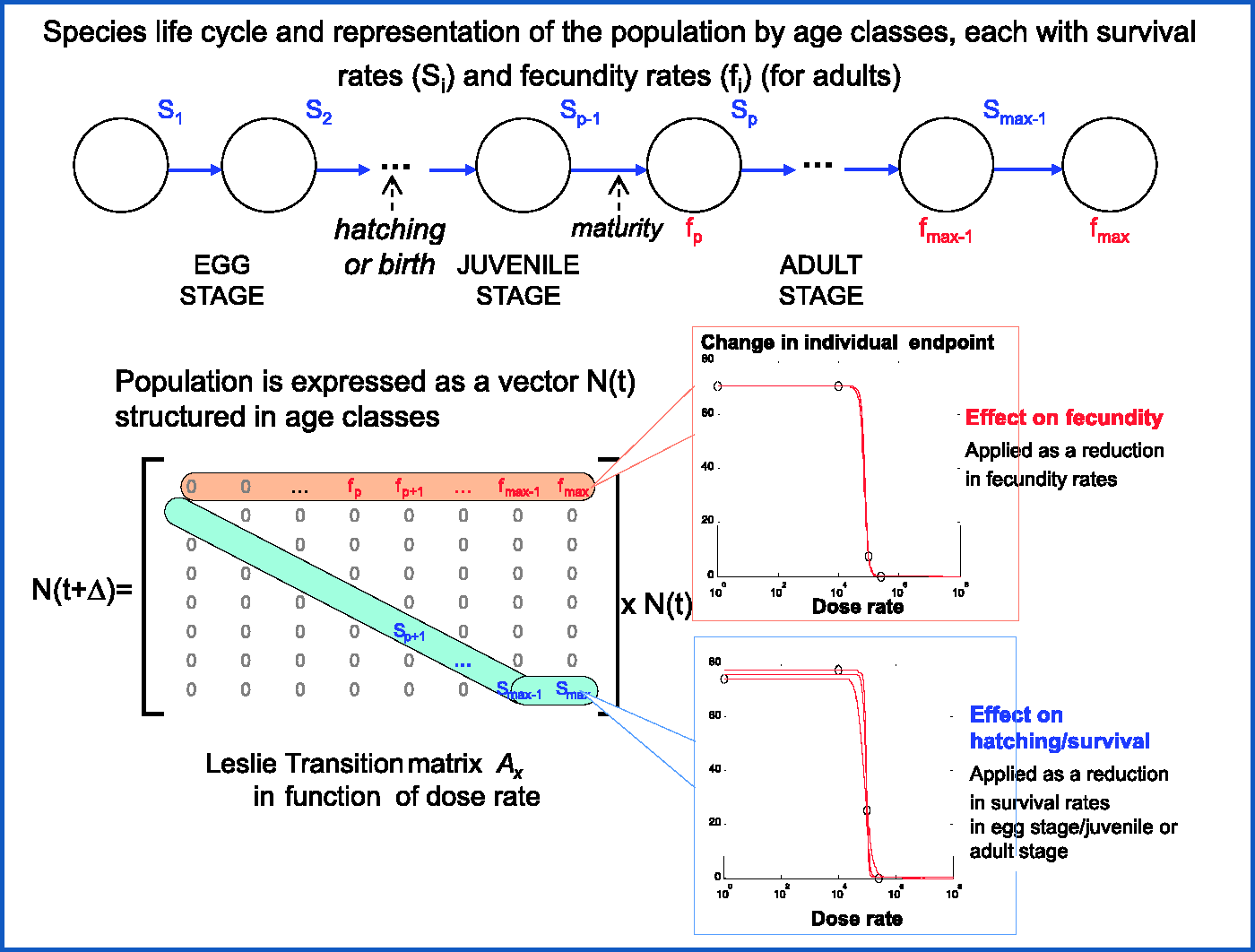
2.2. Overview of the method for estimating the range of variation of radiosensitivity among species
The method aims to represent the range of variation in radiosensitivity among species using a statistical extrapolation model named ‘species sensitivity distribution’ (SSD). Using the FREDERICA database, focusing on terrestrial species and endpoints directly relevant to population demography, the analysis was restricted to chronic external exposure situations acquired under controlled conditions, and to chronic effect data derived from field studies within the Chernobyl exclusion zone. For each species and endpoint, the dose-rate–effect relationship was built, and the
A statistical distribution was fitted to the selected sets of
3. results and discussion
3.1. Extrapolation from individual to population level for two species
The population response to chronic ionising radiation was explored in Extrapolation approach proposed for predicting population-level response by combining dose-rate–effect relationships derived from chronic toxicity data and Leslie matrices (top: results for 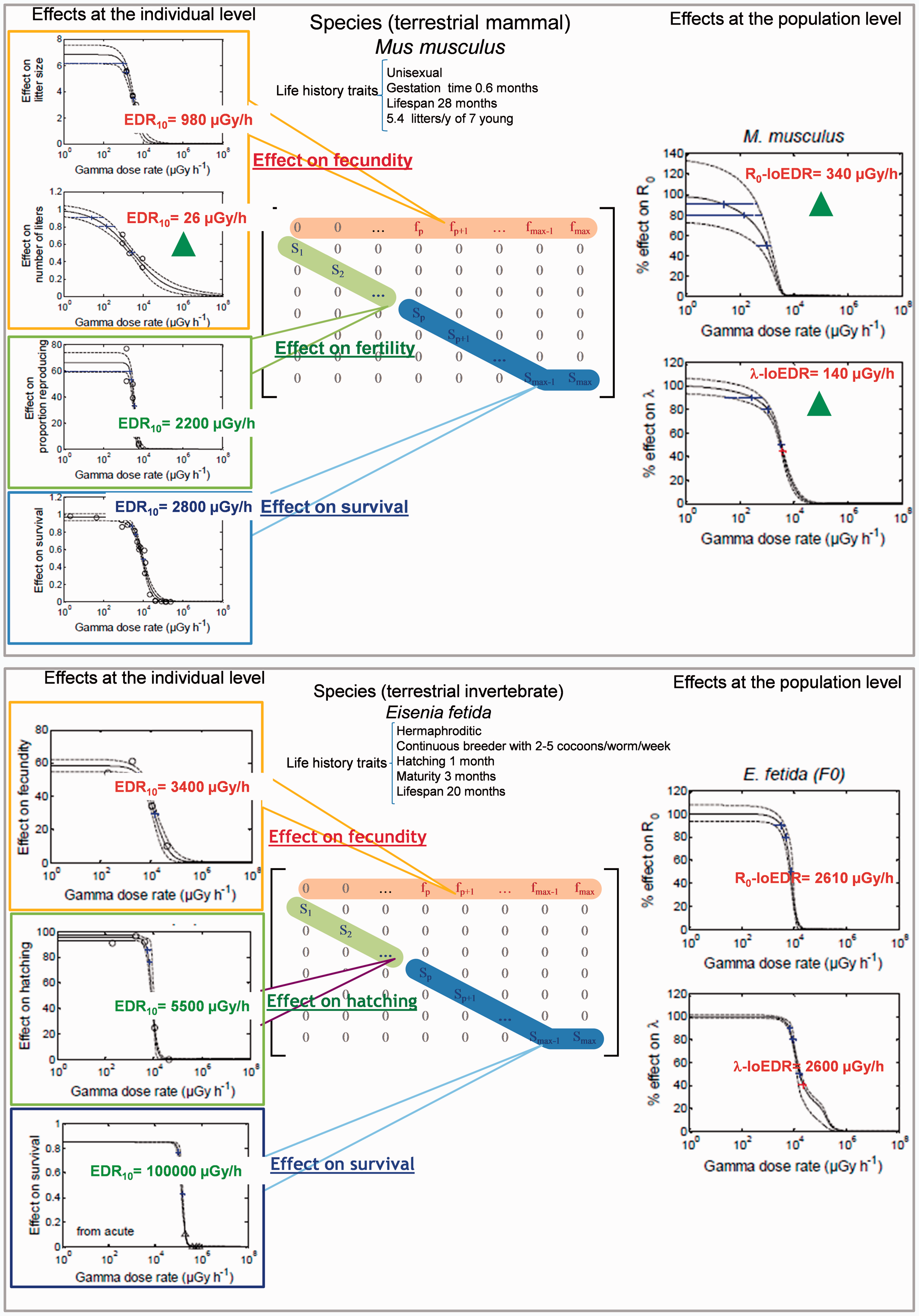
3.2. Extrapolation from one species to another, and from laboratory exposure conditions to the field
Based on laboratory data on radiation effects, species radiosensitivity varied over six orders of magnitude, with vertebrates being among the most radiosensitive organisms (Garnier-Laplace et al., 2010). This suggested that research on primary mechanisms of interactions between ionising radiation and living organisms (from biomolecules up to individuals) is still needed to extrapolate effects in terms of quality and intensity on a rational basis among the wide biodiversity. Additionally, two sets of chronic radiotoxicity data in terrestrial non-human species (one acquired under controlled external gamma irradiation in the laboratory or in the field, and another acquired from field studies in the Chernobyl exclusion zone characterised by a much more complex exposure situation) were compared. This comparison, discussed in detail in Garnier-Laplace et al. (2013), indicated that the best estimate of the median value of the distribution established for field conditions at Chernobyl (approximately 100 mGy h−1) was eight times lower than that from controlled experiments (approximately 850 mGy h−1), suggesting that organisms in their natural environmental were more sensitive (Fig. 3). However, this first comparison highlighted a lack of mechanistic understanding and confusion resulting from field sampling strategies that did not account sufficiently for confounding factors. The field exposure–effects relationship may be modified due to the combination of radiotoxicity effects on growth rate/reproduction and geographic gene diversity, competition, predation, and abiotic factors including pollutants other than radionuclides. Until more field data are acquired, this study calls for a precautionary approach when deriving any benchmark values.
Extrapolation approach proposed for modelling interspecies radiosensitivity variation. Right: species sensitivity distribution (SSD) fitted to minimum values of 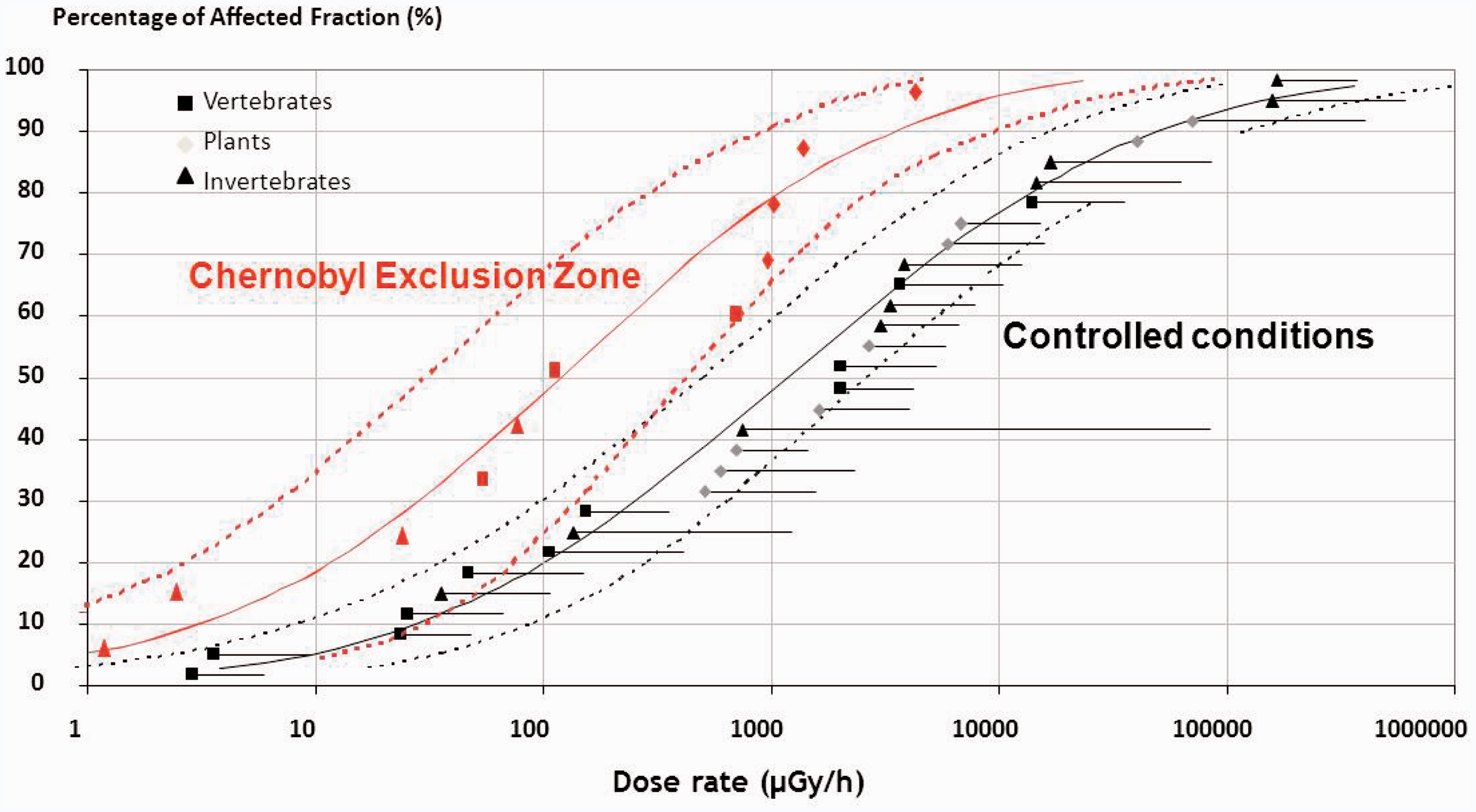
4. CONCLUSIONS
Population models proposed in this study integrate available radiation effect data measured at the individual level, and infer effects at the population level. They can be used to derive dose rates above which population growth is significantly affected when chronically exposed to ionising radiation. This modelling approach is useful to identify critical endpoints for population sustainability in any species, and to prioritise future research. Additionally, the discrepancy between controlled tests and Chernobyl effects data on wildlife is intriguing, mainly due to a shift to a greater radiosensitivity of wildlife in the field. This strongly highlights the need for fundamental research to understand elementary mechanisms.
