Abstract
Objective:
This study aims to investigate the early and intermediate-term outcomes of otoendoscopic “bridging” tympanoplasty with tragal cartilage-perichondrium composite in patients with large pars tensa perforation of tympanic membrane (TM).
Methods:
A retrospective analysis was conducted on 45 patients with large perforations in pars tensa of TM who underwent otoendoscopic “bridging” tympanoplasty. TM closure rates and postoperative pure-tone audiometric improvement were evaluated.
Results:
The closure rate was 95.6%. All patients underwent pure-tone audiometry, and significant postoperative hearing improvement was observed. The primary postoperative symptoms were pruritus and aural fullness, and no major complications such as cholesteatoma or facial nerve paralysis were noted.
Conclusion:
The otoendoscopic “bridging” tympanoplasty using tragal cartilage-perichondrium composite is an effective and feasible technique for large pars tensa perforations.
Keywords
Introduction
Tympanoplasty is one of the most common surgeries in otologic department, and the repair methods used vary depending on the location and size of the perforation. Repairing large perforations in the pars tensa of the tympanic membrane (TM) is challenging due to insufficient residual edges. Both graft selection and surgical technique remain areas of intense research.
From an anatomical perspective, the anterior tympanomeatal angle hinders the complete observation of TM due to its forward and downward inclination. In addition, the tip of malleus handle is concave toward tympanic cavity, leading to insufficient support for the graft and increasing the risk of poor healing. Furthermore, compared to the posterior region, the blood circulation in the anterior TM is poorer, which is not conducive to healing. 1
Multiple surgical techniques have been recommended for subtotal and total TM perforation with inadequate anterior remnants, 2 including anterior tab flap technique, 3 “mucosal pocket” myringoplasty, 2 3-point fix tympanoplasty, 4 and window shade tympanoplasty. 5 In this study, we introduced a novel “bridging” tympanoplasty technique that combines underlay and overlay principles, and conducted a retrospective analysis of its efficacy.
Materials and Methods
Study Population
A retrospective review was conducted on 45 patients with large perforations in pars tensa of TM treated in otolaryngology department from January 2021 to November 2023, all of whom underwent otoendoscopic “bridging” tympanoplasty. The inclusion criteria were (1) chronic suppurative otitis media with subtotal and total TM perforation with inadequate anterior remnant; (2) patients who were able to undergo pure-tone audiometry; (3) primary surgery; (4) absence of middle ear cholesteatoma and nasopharyngeal mass; (5) no serious systemic diseases, such as cardiovascular and cerebrovascular diseases, liver and kidney dysfunction, hematopoietic system diseases.
Method
Preoperative Evaluation
All patients underwent routine otoendoscopy, temporal bone computed tomography (CT) examination, pure-tone audiometry (including 500, 1000, 2000, and 4000 Hz), acoustic impedance testing, and nasopharyngoscopy examination before surgery.
Surgical Procedure
Preparation of Graft
Local infiltration anesthesia was administered to the medial aspect of the tragus (lidocaine injection containing adrenaline). An arc-shaped skin incision was made ~2 mm from the edge and subcutaneous tissue was dissected. An appropriately sized tragal cartilage-perichondrium composite graft, which contained bilateral perichondrium, was harvested. The outer perichondrium was peeled off to be used as a “bridging” graft, while the remaining cartilage-perichondrium composite was used as an underlay graft. Finally, the tragal incision was closed.
Tympanoplasty Procedure
Approximately 1 mm of epithelium was removed from the edge of the perforation to create a fresh wound. Local infiltration anesthesia was injected into the inferior, posterior, and superior walls of the external auditory canal. An arc-shaped incision was made ~5 mm from the tympanic annulus, extending counterclockwise from the 2 to 5 o’clock position (right ear as example). The external auditory canal skin flap was elevated, the TM flap was lifted, the epithelium on the surface of malleus was removed, and the fibrous tympanic annulus was elevated 360° with the external auditory canal skin (Figure 1A). The incudomalleolar joint was fully exposed, and the tympanic cavity, ossicular chain, and eustachian tube were inspected.
Fully expose the incudomalleolar joint, explore the tympanic cavity, ossicular chain, and eustachian tube (patients with ossicular chain instability underwent partial ossicular replacement prosthesis implantation), gelatin sponge was then placed in the anteroinferior portion of the tympanic cavity.
Underlay tympanoplasty was performed using the tragal cartilage-perichondrium composite (Figure 1B), followed by repositioning of the skin and TM flap (Figure 1C).
The prepared perichondrium was trimmed to an appropriate size, embedded on the medial side of the perforated edge, and advanced toward the anterior wall of the external ear canal with a crochet hook, so that it covered the potential gaps between the tragal cartilage-perichondrium composite and the bony surface of the anterior wall (Figures 1D and 2). In short, the perichondrium serves as a “bridge” connecting the composite with the anterior wall.
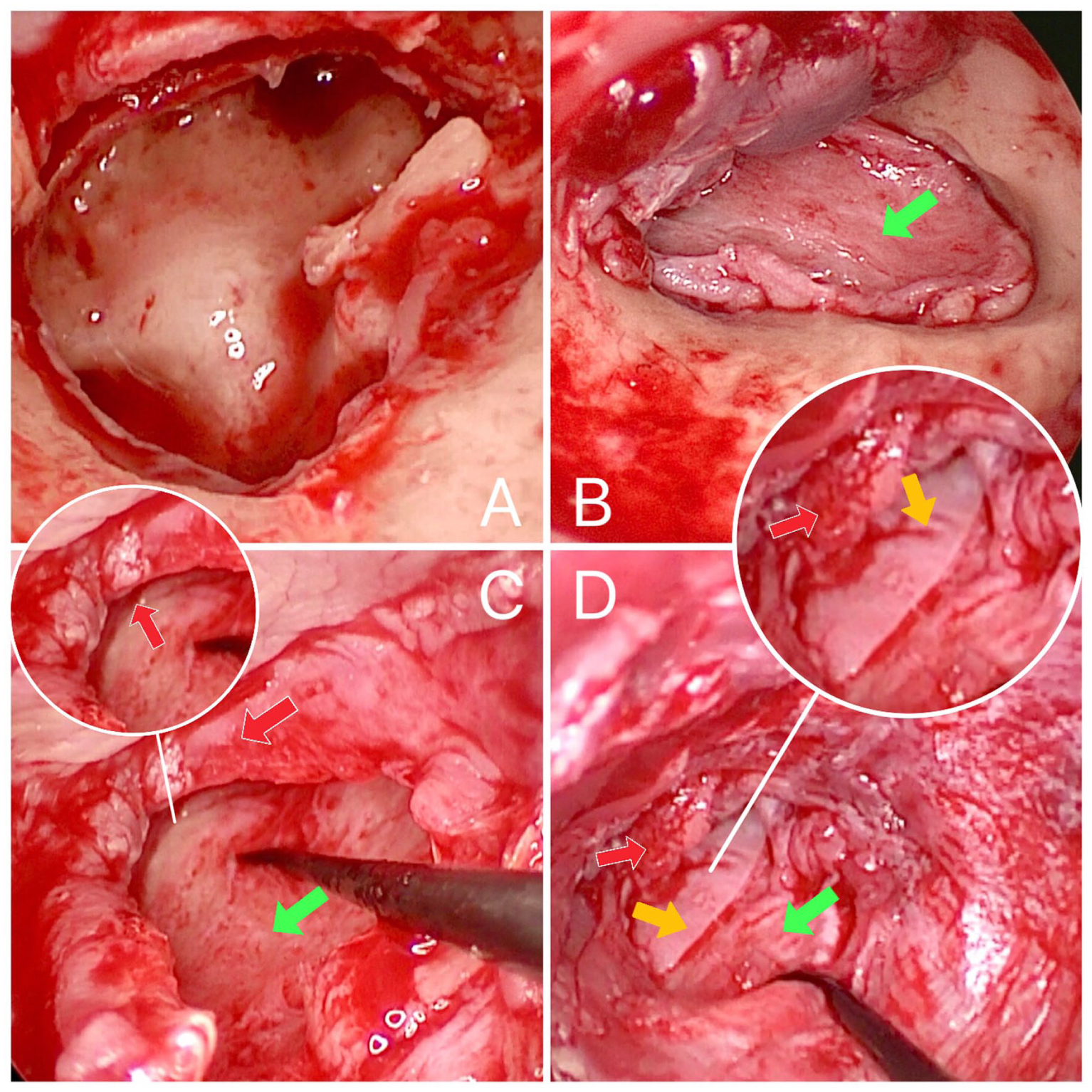
(A) The external auditory canal skin and tympanic membrane flap were elevated circumferentially (360°) to fully expose the tympanic cavity and ossicular chain. (B) The tragal cartilage-perichondrium composite graft (green arrow) was harvested and placed as an underlay graft for tympanic membrane repair. (C) The external auditory canal skin and tympanic membrane flap were repositioned. (D) The prepared perichondrium (yellow arrow) was inserted medial to the residual perforation edge (red arrow).
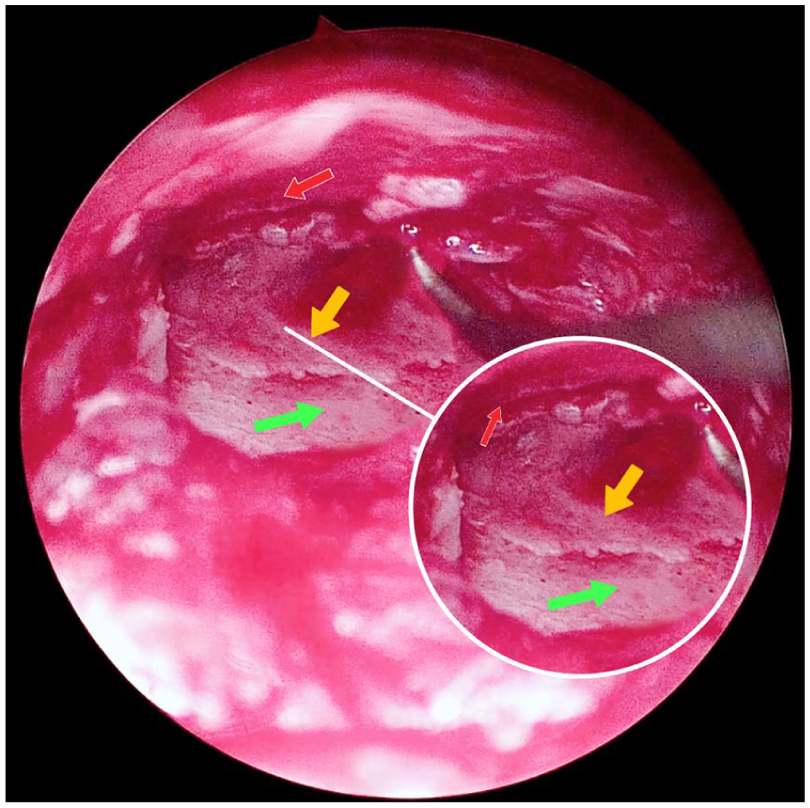
The prepared perichondrium (yellow arrow) was inserted medial to the residual perforation edge (red arrow) and advanced toward the anterior canal wall to cover any potential gaps between the tragal cartilage-perichondrium composite (green arrow) and the bony anterior external auditory canal wall.
External Auditory Canal Packing
The deep external auditory canal was packed with gelatin sponge fragments soaked in ofloxacin otic drops, and the superficial portion was packed with gelatin sponge cylinders, then the tragus incision was then covered with sterile gauze.
Postoperative Management and Follow-Up
Gauze strips were removed 3 days after surgery, and surgical sutures were removed 1 week postoperatively. At 3 weeks postoperatively, gelatin sponge cylinders were removed, and ofloxacin otic drops were administered. At 1 month postoperatively, residual gelatin sponge fragments in the external auditory canal were debrided. All patients were followed up for 1 year postoperatively.
Efficacy Evaluation
Patients were stratified into implanted and no implanted groups based on artificial ossicle implantation. Monthly follow-up was performed for 1 year postoperatively to assess TM closure, hearing changes, and other ear-related symptoms. Pure-tone audiometry was performed at 3 months postoperatively, and the air conduction (AC) threshold and air-bone gap (ABG) were calculated at 0.5, 1, 2, and 4 kHz. Mean AC thresholds and ABG values were calculated using standard 4-frequency method (0.5, 1, 2, and 4 kHz). If subjective hearing changes were reported within 1 year, pure-tone audiometry was repeated. Preoperative and postoperative data were compared.
Statistical Analysis
Statistical analysis was conducted using statistical package for the social sciences (SPSS) 26.0. Pure-tone audiometry results were normally distributed, and normality was assessed using the Shapiro–Wilk test. Paired t-test was used to compare preoperative and postoperative data within implanted and no implanted groups, with P < .05 considered statistical significance.
Results
Clinical Data
Patient characteristics are summarized in Table 1. A total of 45 patients (ears) were included in the study, including 20 males and 25 females. Patients’ ages ranged from 15 to 63 years, with a duration of disease ranging from 3 months to 40 years. Nine cases (20.0%) were wet ears. Ten cases (22.2%) had calcified plaques on the TM. Based on the intraoperative conditions, we performed artificial auditory bone implantation on patients with abnormal movement of the ossicular chain and 13 cases (28.9%) underwent artificial auditory bone implantation (all were partial artificial auditory bones).
Demographic and Intraoperative Data.
Perforation Closure Rate
Among the 45 cases, 43 (95.6%) had TM closure, and 2 cases developed fungal infection and recurrent perforation 3 months after surgery, resulting in surgical failure.
Audiological Examination Results
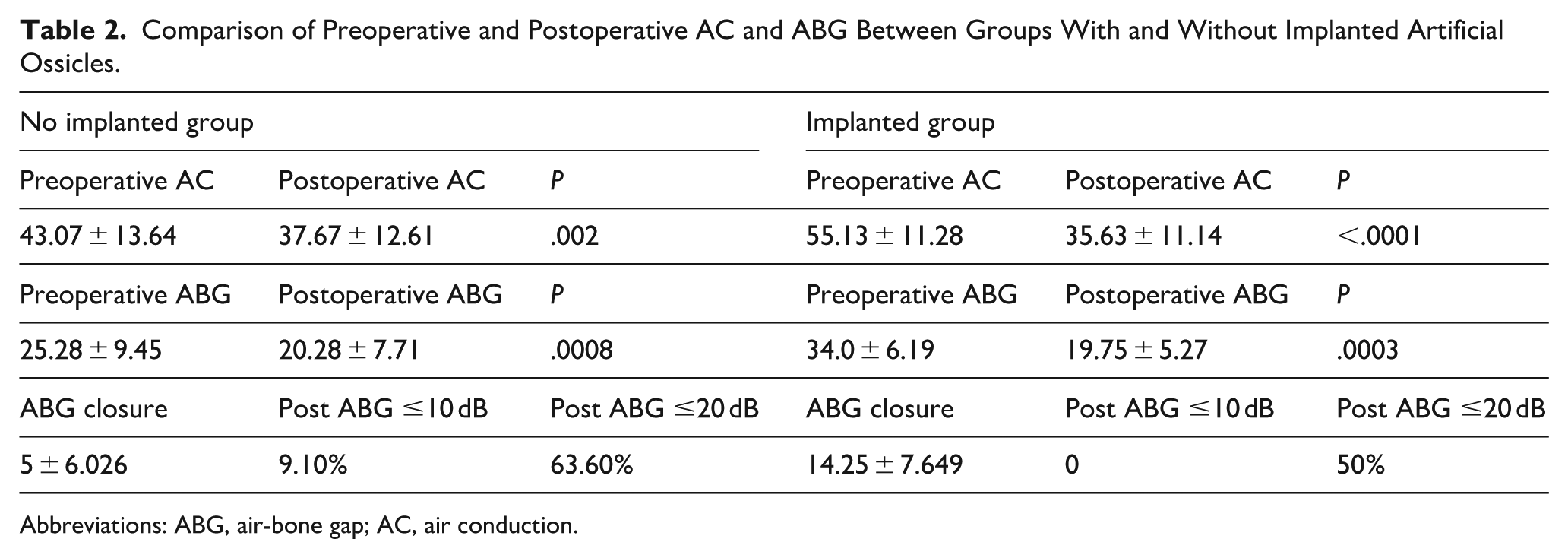
Three months after surgery, all patients underwent pure-tone audiometry, and no patients reported significant changes in hearing during the 1 year follow-up. Therefore, we analyzed the pure-tone audiometry results at 3 months postoperatively. Postoperative AC (Table 2 and Figure 3) and ABG (Table 2 and Figure 4) values were significantly improved compared to preoperative levels (P < .05), with more pronounced improvement in the implanted group.
Comparison of Preoperative and Postoperative AC and ABG Between Groups With and Without Implanted Artificial Ossicles.
Abbreviations: ABG, air-bone gap; AC, air conduction.
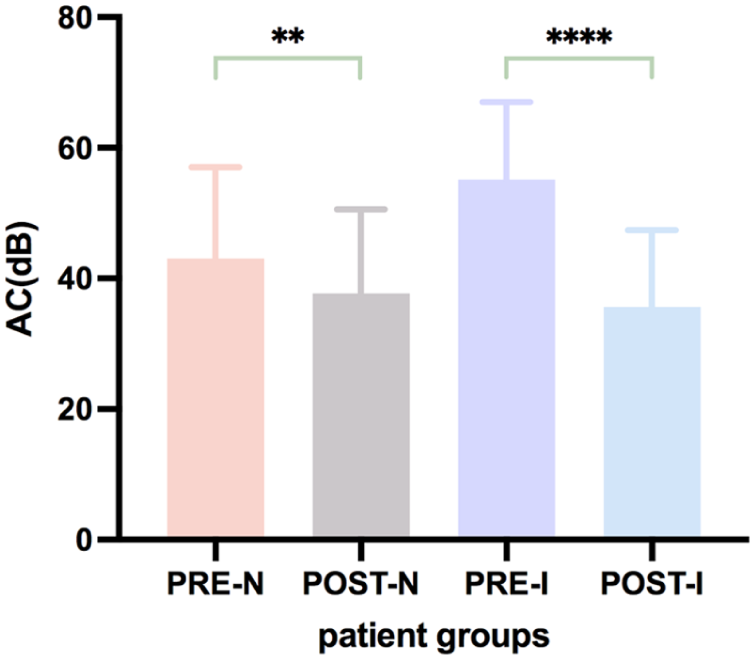
Preoperative and postoperative AC of no implanted group and implanted group. AC, air conduction.
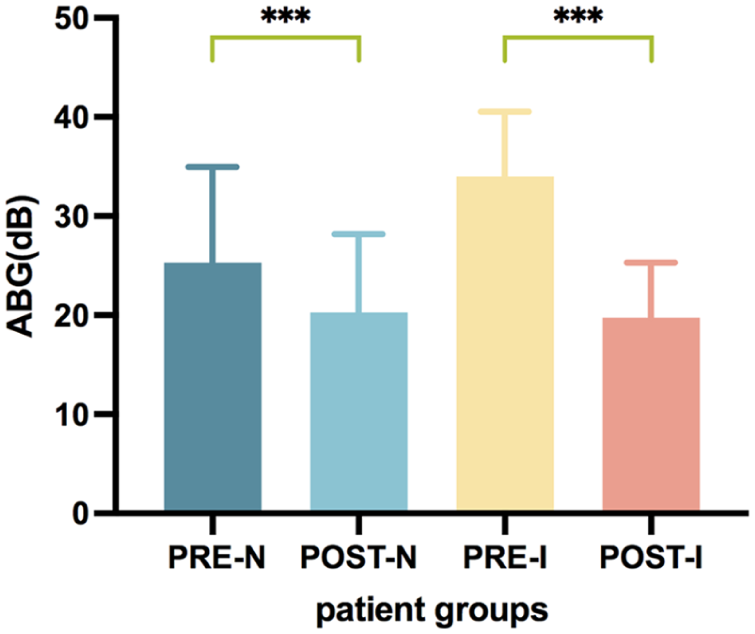
Preoperative and postoperative ABG of no implanted group and implanted group. ABG, air-bone gap.
Postoperative Complications
The primary symptoms of 45 patients within 6 weeks after surgery were pruritus and aural fullness, mostly caused by canal packing and the symptoms gradually decreased or even disappeared over time. During the follow-up period, 2 cases (4.4%) of recurrent perforation due to fungal infection were noted at 3 months after surgery, all of which occurred during hot summer months. After administering boric acid ethanol otic drops, the fungal infection was relieved but the perforation did not heal. One case of taste abnormality (2.2%) resolved spontaneously. No severe complications, such as cholesteatoma or facial nerve paralysis, were reported.
Discussion
In the past decade, otoendoscopy has been widely adopted in middle ear surgeries due to its advantages, including a clear field of view, minimal trauma, and rapid recovery. Otoendoscopy provides full exposure of the tympanic cavity via the natural channel of the external auditory canal under a high-definition imaging system, particularly aiding in the observation and treatment of anterior-inferior TM perforations, thereby improving the closure rate. However, its shortcomings mainly lie in 1-handed operation, easy contamination of the mirror surface, small operating space, and 2-dimensional vision which require surgeons to have sufficient clinical experience. Fortunately, these issues are gradually being addressed through advancements in surgical instruments and equipment.6 -8
Graft materials for tympanoplasty can include temporalis fascia, cartilage, perichondrium, periosteum, adipose tissue, and various biomaterials. With the development of minimally invasive concepts, tragus cartilage and perichondrium have become highly regarded. Özdemir et al 9 performed tympanoplasty on 104 patients using tragus cartilage with otoendoscopy, achieving a high success rate regardless of perforation size and location. Mohanty et al 10 found that the success rate of cartilage repairing for perforation in pars tensa of TM was 91.9%, higher than the 79% achieved with temporal fascia. Additionally, cartilage exhibits strong anti-infective properties and can achieve high success rates even in wet ear conditions.11,12 In a meta-analysis, Jalali et al 13 found that the success rate of tympanoplasty with cartilage was higher. Gozeler and Sahin 14 also recommend tympanoplasty with cartilage, especially in complex cases such as eustachian tube dysfunction, middle ear negative pressure and inward pocket formation.
Due to the deficient or absent anterior residual edge in the anterior part of TM, it is difficult for cartilage—perichondrium composite to fit tightly into tympanic anulus in patients with large perforations in pars tensa of TM. There is a risk of graft retraction or collapse into the tympanic cavity, resulting in gaps or small perforations between the anterior of TM and the anterior wall of external auditory canal. Therefore, we use an additional perichondrium as a “bridge” to connect the tragal cartilage-perichondrium composite with the anterior wall of external auditory canal. This is an innovative surgical approach that not only increases the contact area between the 2 structures but also reduces the pulling force during the healing process, which is more conducive to maintaining the stability of the composite.
Multiple factors can affect the closure rate of TM perforations, including the surgical approach, perforation location and size, choice of graft material, surgical techniques, anti-infection measures, middle ear moisture levels, and the condition of the mastoid process. 15 Carr et al 16 found that the location of TM perforation is an important factor affecting healing, with the lowest closure rates observed for anteroinferior quadrant perforation and subtotal perforation of TM. Studies by multiple scholars have shown that dry ear and wet ear have no effect on the closure rate of TM perforation,17,18 and our findings support this perspective. In our study, the closure rate was 95.6%, with the majority of patients achieving closure within 6 weeks postoperatively, which is encouraging compared to the 91.9% closure rate achieved with cartilage, 10 2 cases (4.4%) of recurrent perforation due to fungal infection were noted at 3 months postoperatively, all of which occurred during the hot summer. This finding may remind us that enhanced management of the postoperative external auditory canal environment for patients should be implemented to ensure the surgical area remains cool and dry.
Zahnert et al 19 found that grafts with a thickness of 0.5 mm or less can improve sound transmission quality and achieve optimal hearing recovery; however, the average thickness of human tragus cartilage is 1.016 mm. 20 Aarnisalo et al 21 believed that cartilage thickness has no significant effect on hearing. Dornhoffer 22 believed that using full-thickness cartilage can also achieve good levels of hearing. In this study, postoperative pure-tone audiometry showed significant improvements in hearing. Nevertheless, most postoperative ABGs were not completely resolved, and the exact underlying mechanism remains unclear. This may be related to the thickness of the composite material affecting sound transmission, as we observed a smaller ABG in patients who underwent TM repair using temporal fascia. The thickness of the composite material may gradually decrease over time; therefore, longer-term observation is needed to clarify the subsequent changes in ABG. In addition, completely lifting the TM from malleus handle may cause a change in the shape of TM, which also affects the effectiveness of postoperative hearing improvement.
We acknowledge that although the results are encouraging, this is a retrospective single-arm study without a control group, which limits the conclusions about the superiority or added value of the proposed technology. We plan to conduct cohort or randomized controlled trials in the future to further validate the results.
Conclusion
Otoendoscopic “bridging” tympanoplasty is an effective and feasible technique for the management of large perforations in pars tensa of TM. The early and intermediate-term outcomes of surgery demonstrate favorable closure rates and significant hearing improvement.
Footnotes
Acknowledgements
The authors thank Dr Yuzhang Zhou and Miss Rikee Liu for their editing of the manuscript.
Ethical Considerations
This study was approved by the Ethics Committee of the Clinical Medical College of Xiangyang No 1 People’s Hospital and conducted in accordance with the Helsinki Declaration.
Consent to Participate
Obtained informed consent from all patients.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Innovative Research Program of Xiangyang No 1 People’s Hospital (grant no XYY2023NT01).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
