Abstract
Objective:
To evaluate the prevalence and predictors of no residual disease on wide local excision (WLE) specimens in patients with shave biopsy-confirmed head and neck basal cell carcinoma (BCC).
Study Design:
Retrospective cohort study of patients in an integrated healthcare system referred from dermatology to otolaryngology—head and neck surgery.
Setting:
Otolaryngology—head and neck surgery clinic.
Methods:
We reviewed medical records of adults with shave biopsy-confirmed BCC referred for WLE between January 2022 and December 2023. Data included patient demographics, comorbidities, histologic subtype, and timing of excision. Bivariate and multivariate analyses assessed associations with residual disease status on final pathology.
Results:
Among 243 cases, 66 (27.2%) WLE specimens had no residual tumor found on pathologic analysis. Patients with no residual disease were younger on average compared to those with residual cancer (65.4 ± 14.2 years vs 72.6 ± 13.0 years, P < .001). Of the 97 female patients, 39 (40.2%) had no residual disease and female sex was independently associated with no residual disease (odds ratio 3.398, 95% confidence interval 1.80-6.42, P < .001). Histologic subtype, diabetes, transplant history, and prior radiation showed no significant associations.
Conclusion:
Over one-quarter of patients had no residual tumor at excision, particularly younger individuals and women. These findings underscore the value of individualized risk assessment during preoperative counseling, especially for elderly or frail patients and those with lesions in cosmetically sensitive regions. Predictive models incorporating patient-level factors may help reduce overtreatment and improve decision-making in BCC management.
Keywords
Introduction
Basal cell carcinoma (BCC) is the most common cancer, with an incidence of 3.6 million cases per year in the United States. 1 Risk factors include advancing age, smoking, fair skin, cumulative and high-intensity ultraviolet radiation exposure, and genetic syndromes such as basal cell nevus syndrome. 1 Diagnosis is most commonly established via shave biopsy. Standard treatment involves surgical removal, typically through Mohs micrographic surgery or wide local excision (WLE), with adjuvant or neoadjuvant medical therapy, radiation, or immunotherapy reserved for more advanced cases. 2
Diagnoses of nonmelanoma skin cancers (NMSCs) like BCC have increased by 77% between 1994 and 2014. 3 However, there has been no corresponding increase in disease-specific mortality. 4 Therefore, there is growing concern for overdiagnosis and overtreatment of BCC—an indolent cancer with a metastatic rate below 0.1%—resulting in scar, disfigurement, and even hospital admissions, especially in frail, older patients.4 -8 Unnecessary medical expenditure is another concern: approximately $6.5 billion was spent on the treatment of NMSCs in the United States annually between 2016 and 2018. 9 Other recent studies have shown a substantial proportion of biopsy-proven BCC cases with no residual tumor on subsequent excision. 5
Identifying predictors of residual disease can help classify higher-risk cases while minimizing overtreatment in patients with greater potential for no residual disease. However, there is a dearth of research on these factors. This study aims to assess the prevalence of no residual disease in patients surgically treated for BCC in an otolaryngology—head and neck surgery (OHNS) clinic and to identify associated patient-level factors.
Methods
Patient Selection
A retrospective cohort study was conducted within Kaiser Permanente Northern California, a large integrated healthcare system. The study included patients referred to OHNS departments at Oakland and Richmond Medical Centers between January 2022 and December 2023. Eligible patients had shave biopsy-confirmed BCC and were referred for WLE. Most referrals originated from dermatology. Patients were excluded if they were diagnosed with a non-basal cell skin cancer, or if they opted for Mohs surgery, observation, or palliative care instead of WLE.
Variables and Outcomes
The primary outcome was the presence or absence of residual cancer on final pathology following WLE. Collected variables included demographic data (age, sex, and race/ethnicity) and relevant medical history (diabetes mellitus, immunosuppressant use, transplant history, and prior radiation therapy). Procedural data included initial biopsy type (shave, punch, or curettage) and timing, histologic subtype, margin status, lesion size and location, and WLE findings.
Data Collection and Management
Diagnoses were identified using the International Classification of Diseases, 10th Revision (ICD-10). All patients seen in OHNS clinic with ICD-10 codes for diagnoses related to BCC or unspecified malignant neoplasm of the skin of the head, face, or neck were queried. All data were obtained through manual chart review of the electronic health record. Two reviewers performed all data abstraction. Any cases with incomplete or missing data were excluded from the final analysis.
Statistical Analysis
All statistical analyses were performed using SPSS version 25.0 (IBM Corporation). Descriptive data was presented as means, standard deviations, counts and frequencies. Categorical variables were compared using chi-square or Fisher’s exact tests, and continuous variables using 1-way ANOVA. Variables with significant bivariate associations were included in a multivariable logistic regression model. Odds ratios (OR) and its 95% confidence intervals (CI) were reported. A P-value of less than.05 was considered statistically significant.
Research Determination
The Research Determination Committee for the Kaiser Permanente Northern California region has reviewed this project and determined that it does not involve human subjects. This study was thus exempted from Institutional Review Board approval.
Results
Demographics
A total of 321 patients were identified with the initial search terms. Patients were excluded based on the following criteria: diagnosis of squamous cell carcinoma (n = 43), diagnosis of melanoma in situ or invasive melanoma (n = 18), treatment with Mohs surgery (n = 9), and lack of WLE (n = 8). This left 243 patients with shave biopsy-confirmed BCC who underwent WLE in the final cohort. The mean age was 70.7 ± 13.6 years, with 26.7% over 80 years old. Most patients were male (60.1%, n = 146), and the cohort was predominantly White (93.0%, n = 226; Table 1).
Patients with Shave Biopsy-Confirmed Basal Cell Carcinoma: Demographic and Clinical Characteristics According to Residual Disease Status Based on Final Pathology Following Wide Local Excision.
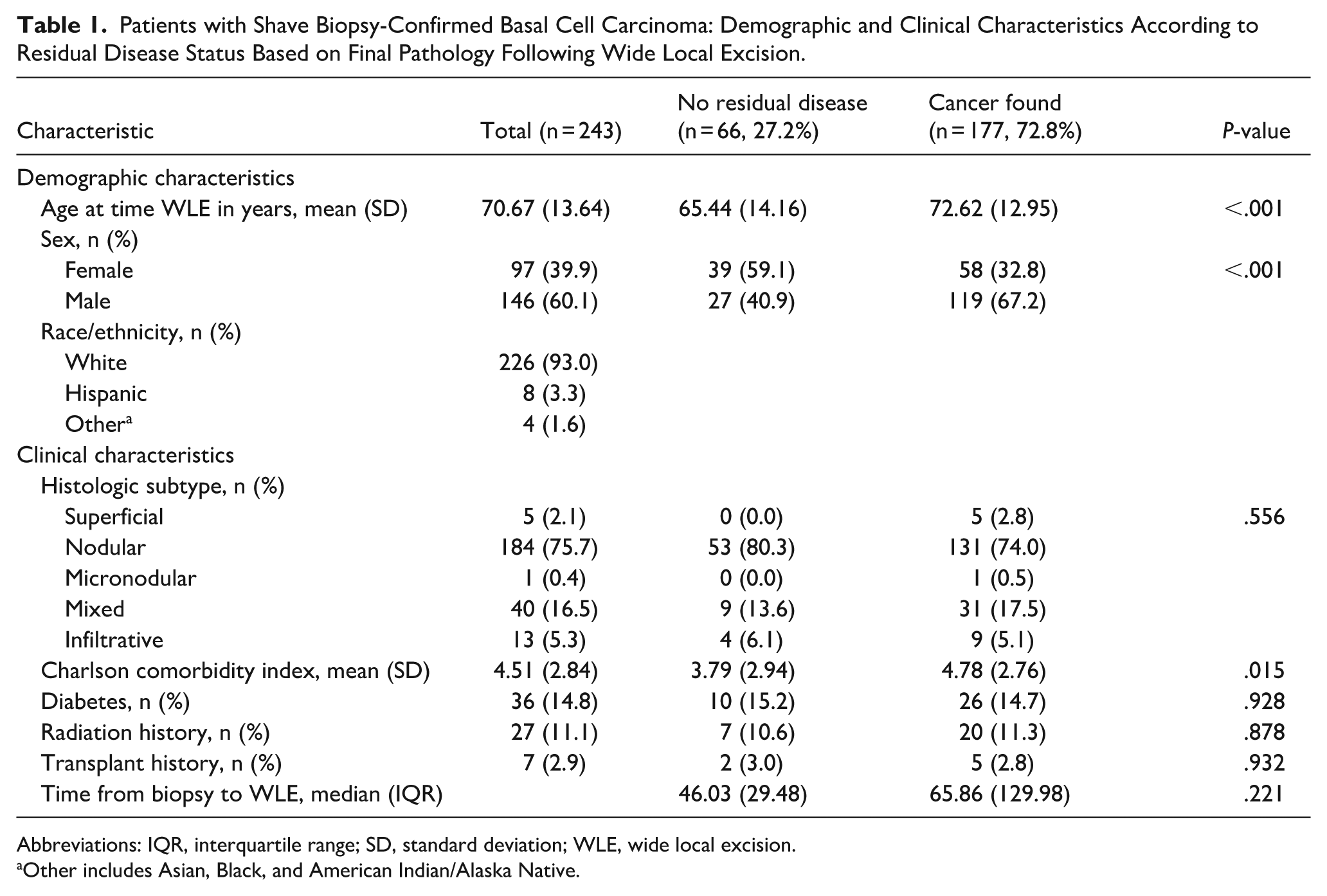
Abbreviations: IQR, interquartile range; SD, standard deviation; WLE, wide local excision.
Other includes Asian, Black, and American Indian/Alaska Native.
Overall Findings
Among all cases, 66 (27.2%) had no residual disease on final pathology following WLE (Table 1).
BCC Subtypes
The most common histologic subtype was nodular BCC (75.7%), followed by mixed (16.5%), infiltrative (5.3%), superficial (2.1%), and micronodular (0.4%). Table 1 provides the distribution of subtypes. No statistically significant differences in the rate of residual disease were observed across subtypes (P > .05 for all comparisons, Table 1).
Bivariate Analysis
Female patients were significantly more likely than male patients to have no residual disease (40.2% vs 18.5%, P < .001). No significant differences in residual disease rates were noted based on diabetes mellitus (P = .928), radiation therapy history (P = .878), or transplant history (P = .932; see Table 1).
Patients with no residual disease were younger than those with residual disease (65.4 ± 14.2 years vs 72.6 ± 12.9 years, P < .001) and had a lower Charlson comorbidity index (CCI; 3.8 ± 2.9 vs 4.8 ± 2.8, P = .015). Time from biopsy to WLE did not differ significantly between groups (46.0 ± 29.5 days vs 65.9 ± 130.0 days, P = .221; see Table 1).
Multivariate Analysis
Sex remained a significant predictor in multivariate logistic regression. Female patients had higher odds of having no residual disease (OR 3.398, 95% CI 1.80-6.42, P < .001), after adjusting for age, race/ethnicity, and CCI.
Discussion
In this cohort of BCC patients undergoing WLE, just over one-quarter of patients had no residual tumor on final pathology. This finding highlights the possibility of overtreatment of a cancer subtype that grows slowly and virtually never metastasizes. In many patients, after shave biopsy, there is no visible tumor, and WLE is performed on residual scar tissue. And in this study, we have confirmed that a large portion have no residual cancer at a microscopic level as well. In these cases, surgical intervention may be causing unnecessary morbidity, particularly when lesions are in cosmetically or functionally sensitive areas.
Our study identified several patient-level predictors of residual disease. Female patients had significantly higher odds of no residual cancer following WLE, even after adjusting for relevant covariates. This sex-based difference aligns with emerging research suggesting distinct biological or behavioral factors may influence tumor progression and immune response.6,7 Prior studies have shown that men may experience more aggressive disease courses in various skin cancers, potentially due to differences in health-seeking behavior, immune response, or environmental exposures.8,9 Further investigation is needed to elucidate the mechanisms underlying these sex-based disparities in residual tumor burden.
Age also emerged as a significant predictor; younger patients were more likely to have no residual disease. This could reflect differences in immune surveillance, tumor biology, or treatment approach.10 -15 Clinicians may have a lower threshold for excising lesions in younger individuals, particularly when cosmetic outcome or patient anxiety about malignancy influences decision-making. These findings invite further exploration into how provider and patient behavior preferences may influence treatment pathways.
Our data also show no significant association between residual disease and factors typically associated with increased oncologic risk, including diabetes, transplant history, or prior radiation.13-18 This may reflect several factors. First, our study may have been underpowered to detect such associations. Although relative differences in residual disease rates appeared notable across some comorbidity groups, the absolute number of patients with these risk factors—particularly transplant recipients and those with prior radiation exposure—was small. With a larger sample size, these differences might reach statistical significance. Second, it is possible that the biology of residual BCC after shave biopsy is influenced more heavily by procedural and anatomic variables (e.g., biopsy depth and tumor location) than by systemic host factors. Additionally, the time between biopsy and surgery did not significantly differ between residual and non-residual cases, suggesting that surgical delay alone does not account for the absence of residual disease in a substantial proportion of patients.
Taken together, these results emphasize the potential to better stratify patients by risk of residual disease after biopsy-confirmed BCC. We propose a management algorithm (Figure 1) that integrates these demographic factors to guide preoperative counseling and shared decision-making. As prior literature has suggested, the identification of patients who may benefit from watchful waiting—rather than immediate excision—should be guided by data, particularly in older or frailer individuals for whom surgical risks may outweigh clinical benefit.
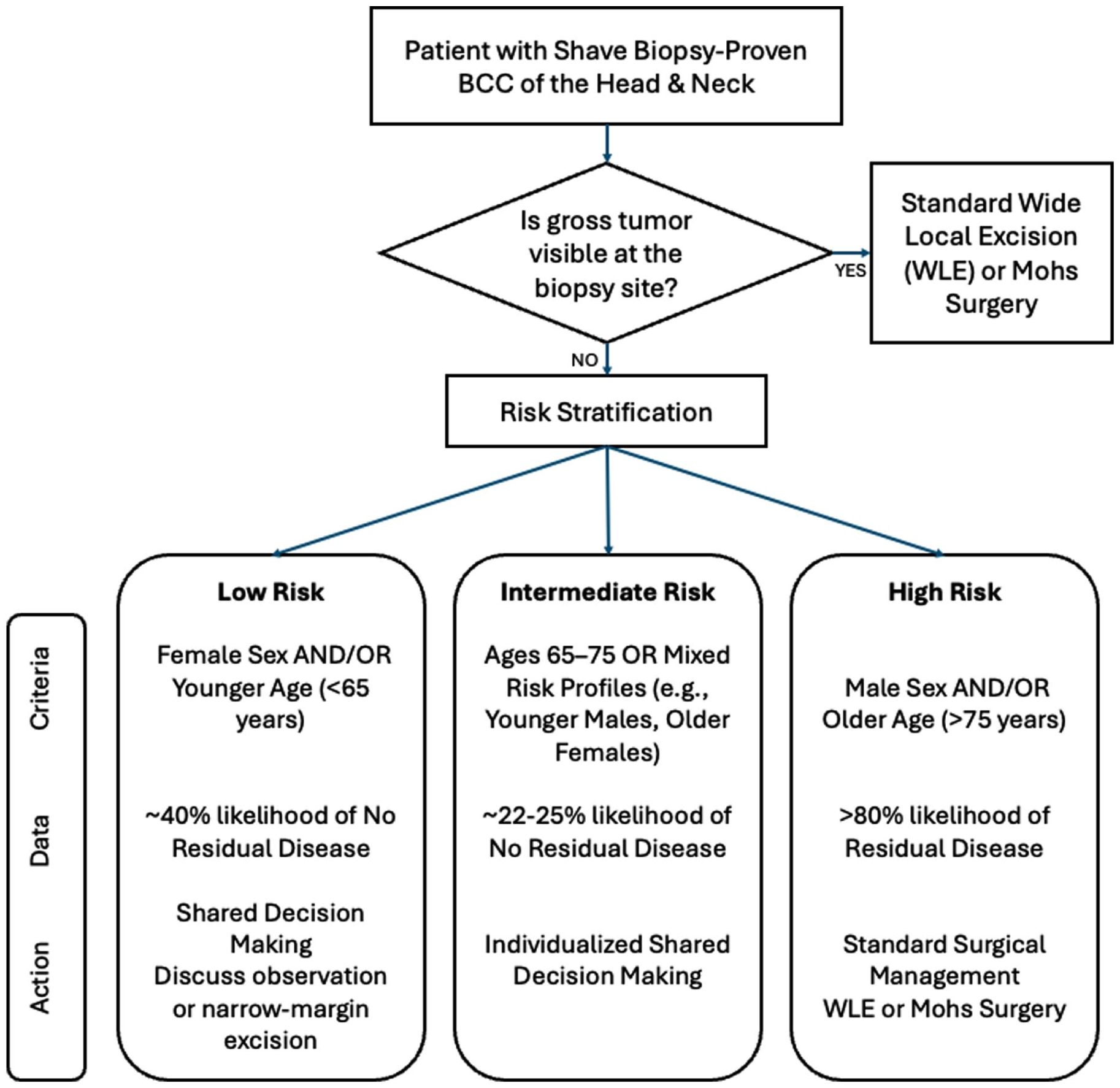
Proposed management algorithm for head and neck biopsy-proven basal cell carcinoma. A decision-making framework incorporating the study’s findings on predictors of residual disease. Patients with no visible tumor who are female and younger have a significantly higher probability of having no residual disease at the time of excision. In these patients, particularly if frail or in cosmetically sensitive areas, a discussion regarding the risks and benefits of immediate aggressive surgery versus conservative management or narrow-margin excision is warranted.
Our findings align with a growing body of literature reporting comparable rates of no residual disease following biopsy. Alcalay et al found that 25% of patients had no residual tumor after biopsy, even when initial pathology indicated incomplete excision. 19 In a separate analysis, the same group observed tumor disappearance in 22% of patients with nodular BCC following shave biopsy prior to Mohs surgery. 20 In a more recent cohort reported by Gurunluoglu et al, the rate of no residual disease after excision of biopsy-proven BCC was 15%. 5 Our observed rate of 27.2% is consistent with or slightly higher than these prior findings, likely reflecting differences in biopsy technique or lesion selection. These comparisons reinforce the notion that a substantial proportion of BCCs may be completely excised at the time of biopsy—particularly with deep shave approaches—and support the use of clinical predictors to guide selective management.
Such clinical factors with growing evidence of predictive value for higher-risk BCC are tumor location and histological subtype. For example, 2 studies21,22 show that the vast majority of BCC cases are located on the face (90% and 54.7%, respectively), with the highest rates of residual tumor—determined either via clinically visible tumor or histological testing—being found on the nose or cheek and characterized by an infiltrative or morpheic/sclerosing histology.21,22 These histological trends in particular are consistent with the pathophysiological growth patterns of these subtypes, which are more aggressive, invade more deeply, have ill-defined borders, and have poorer margin clearance, often due to subclinical spread that is difficult to detect. 23 Thus, this identification of key subgroups that contribute the most to the rate of residual or recurrent cancer potentially provides a key approach to risk-stratifying BCCs to better target aggressive excisional approaches to patients who would benefit most. Similar algorithmic approaches have recently been proposed for site-specific BCC management, such as those of the scalp, and our findings suggest a broader application for head and neck lesions. 24
Limitations
This study has several limitations. First, the retrospective design introduces risks of selection and documentation bias. All data were collected from 2 medical centers within a single health system, which may limit generalizability. While the cohort was diverse in age and gender, the racial distribution was predominantly White, limiting applicability to more diverse populations. Lesion-specific data such as depth, ulceration, and biopsy margin status were not consistently available and therefore not analyzed, despite their potential relevance to residual disease.
A key limitation lies in the pathological assessment technique. WLE specimens are processed using the bread-loafing method, which samples tissue at 2- to 4-mm intervals and may miss residual tumor between sections.1,2 This differs from the en-face technique used in Mohs micrographic surgery, which evaluates nearly 100% of surgical margins. 15 Thus, a “no residual disease” result from bread-loafing may not confirm a truly tumor-free specimen. Nonetheless, if tumor is undetectable on standard pathology, this raises a valid question of whether excision is really necessary, particularly in frail and elderly patients.
Lastly, shave biopsy was the predominant diagnostic method, which introduces variability in the depth and extent of tissue sampled. Shave biopsies are operator-dependent and can range from superficial shaves to deep saucerizations. In cases where a large or deep shave is performed, the entire lesion may be removed during the initial biopsy. Conversely, a superficial biopsy is more likely to lead to residual disease. This heterogeneity in biopsy technique likely contributes to the observed rate of no residual disease and may limit the generalizability of our results to institutions with different diagnostic practices.
Additionally, while local recurrence is the primary clinical outcome of interest, many studies, including ours, lack long-term follow-up data. As a result, the relationship between lack of residual disease on WLE and local recurrence remains inadequately defined. Recurrence data were not assessed in this study, limiting our ability to directly correlate WLE outcomes with long-term patient outcomes.
Conclusion
An appreciable proportion of patients with shave biopsy-confirmed BCC had no residual tumor upon WLE. Female sex and younger age were significant predictors of no residual disease. These findings suggest that individualized risk stratification may reduce overtreatment and its associated morbidity, especially in patients unlikely to benefit from surgery due to age, comorbidities, or personal preference. Further research should focus on predictive models to guide shared decision-making and reduce overtreatment.
Footnotes
Ethical Considerations
The Research Determination Committee for Kaiser Permanente Northern California reviewed this project and determined that it does not involve human subjects research and is therefore exempt from Institutional Review Board oversight. Committee documentation regarding approved exemption of this project is available on request. All data were obtained from existing electronic medical records and analyzed in de-identified form, thus protecting patient data in accordance with guidelines set forth by the Declaration of Helsinki.
Consent to Participate
Because this was a retrospective study using only de-identified data, and the Kaiser Permanente Northern California Research Determination Committee determined that the project does not constitute human subjects research, requirements for obtaining patient informed consent were waived.
Author Contributions
Bryan D. Le: Design, conduct, analysis, and presentation of the research.
Sacha Moufarrej: Conduct.
Lisa Chionis: Conduct.
Miranda Weintraub: Conduct and analysis.
Benjamin D. Malkin: Design, conduct, and analysis.
Kevin H. Wang: Design, conduct, and analysis.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and analyzed during the current study are not publicly available due to institutional data-use restrictions and patient privacy, but de-identified data may be available from the corresponding author on reasonable request and with permission from Kaiser Permanente Northern California.
