Abstract
Objective:
To investigate 6 month changes in hearing thresholds among neonates with hearing impairment and to explore potential factors associated with these changes.
Methods:
This retrospective study was conducted from June 2016 to June 2024 at a regional hospital. It included neonates with auditory brainstem response (ABR) thresholds exceeding 25 dBnHL in diagnostic audiology tests. Exclusions were made for those with auditory neuropathy spectrum disorder, craniofacial anomalies, non-type A tympanograms, and those with only one recorded hearing evaluation at corrected ages of 1, 3, and 6 months. ABR thresholds and birth medical records were analyzed, categorizing cases into “hearing changed” and “hearing unchanged” groups based on changes in ABR thresholds.
Results:
The study included 96 neonates (154 ears), during the 6 month follow-up, hearing thresholds improved in 45 ears and worsened in 2 ears. Significant associations were found between sex (P = .002), birth weight (P = .020), gestational age (P = .002), care unit (P = .006). Multivariate analysis indicated that sex was the only significant predictor of ABR threshold changes. Decision-tree analysis revealed that male neonates transferred to the neonatal intensive care unit (NICU) as well as male neonates cared for in the baby room or with an unknown care unit and a birth weight <2.6 kg, were more likely to demonstrate improvement in hearing thresholds in 6 months.
Conclusion:
Hearing thresholds in neonates with hearing impairment may improve over a 6 month follow-up period. These changes were associated with male sex, younger gestational age, lower birth weight, and postnatal transfer to NICU in univariate analyses; however, multivariate analysis identified sex as the only independent predictor. Given these findings, structured and ongoing audiological follow-up is crucial to facilitate early detection and timely intervention.
Introduction
Congenital hearing loss affects an estimated 1.86 per 1000 neonates at birth.1,2 Early detection and timely intervention are essential to minimize its impact on language, cognitive, and social development. The Joint Committee on Infant Hearing (JCIH) recommends the “1-3-6” principle: hearing screening by 1 month of age, audiological diagnosis by 3 months, and initiation of intervention by 6 months. 3 Neonates who do not pass the initial screening should undergo diagnostic evaluation by 3 months, and those with confirmed hearing loss should receive amplification and early intervention services within 1 month of diagnosis.
Multiple factors contribute to neonatal hearing loss, including genetic mutations (e.g., GJB2 and SLC26A4), 4 perinatal infections, 1 hypoxia or asphyxia at birth, hyperbilirubinemia, 5 prematurity, ototoxic drug exposure, and low birth weight (<1500 g).6,7 Additionally, neonates admitted to the neonatal intensive care unit (NICU) are at higher risk of developing hearing impairment. 8
Failure to detect and manage hearing loss early can result in significant developmental delays in auditory, speech, and language acquisition. Children with moderate to severe hearing loss often face challenges in academic achievement, communication, and socioemotional development. 9 The primary objective of early hearing detection and intervention programs is to enhance language and literacy outcomes for affected children.3,10 -15 Without early intervention, children with hearing impairment may experience persistent deficits in language, theory of mind, and cognitive skills compared with their normal-hearing peers.
Previous studies have reported that some neonates with congenital sensorineural hearing loss experience improvements in auditory thresholds within the first 6 to 12 months of life. Factors such as younger gestational age, lower birth weight, and NICU admission have been associated with early threshold fluctuations.8,16 -19 These findings highlight the importance of continuous hearing monitoring to guide timely management and intervention planning.
Prematurity, in particular, is associated with an increased risk of hearing loss and early threshold variability. 6 Accurate assessment of hearing in this population requires consideration of the neonate’s corrected age (CA), calculated by subtracting the number of weeks of prematurity from the chronological age. 20 Using CA allows developmental evaluations to align with expected milestones for gestational age, providing a more accurate interpretation of auditory function.
JCIH identifies auditory brainstem response (ABR) as the gold standard for assessing hearing thresholds under 6 months of age. 21 Accordingly, ABR was used at our institution to evaluate hearing thresholds at 1, 3, and 6 months of CA. These time points were selected to monitor potential deterioration or improvement in hearing thresholds during the first 6 months and to ensure timely and appropriate adjustment of hearing aids and early intervention strategies based on the infant’s current hearing threshold.
Despite these insights, most previous studies have focused on individual risk factors, and comprehensive analyses identifying predictors of early hearing threshold changes remain limited. Therefore, this study aimed to evaluate ABR threshold changes at 1, 3, and 6 months of CA and to identify clinical factors associated with early fluctuations using multivariate and decision-tree analyses.
Materials and Methods
This study retrospectively reviewed the medical records of neonates referred to the Department of Otolaryngology at a regional hospital from June 2016 to June 2024. The study included patients with ABR thresholds above 25 dBnHL in the diagnostic audiology test. Neonates with auditory neuropathy spectrum disorder, craniofacial anomalies (such as microtia and cleft lip/palate), those with a 1000 Hz tympanogram (+) or a non-type A tympanogram at 226 Hz, and those with only a single recorded hearing evaluation before 6 months of CA were excluded. The variables collected for analysis included sex, birth weight, care unit, gestational age, laterality (unilateral or bilateral) and side of hearing loss, and initial degree of hearing loss. Audiological data included tympanometry, distortion product otoacoustic emission (DPOAE), and ABR results obtained from referred neonates at 1, 3, and 6 months of CA. Hearing threshold determination at each time point was based on ABR findings, while tympanometry and DPOAE were used as adjunctive assessments.
The study categorized 154 ears into 2 groups: the “Hearing Changed Group,” defined as ears with an ABR threshold difference of ≥20 dB or ears with an initial threshold >25 dBnHL that subsequently improved to a final threshold of 25 dBnHL, accompanied by passing DPOAE. The “Hearing Unchanged Group,” comprising all remaining ears. 22
DPOAE was conducted with 2 pure tone stimuli (F1/F2) presented simultaneously. The sound pressure level was calibrated based on the patient’s ear canal volume to approach 65/55 dB sound pressure level. Seven frequency sets between 1000 and 8000 Hz were tested with a frequency ratio of 1.22 (F2/F1). Distortion products were generated in response to the stimuli, and signals were accepted if the signal-to-noise ratio was greater than 8 dB, serving as the passing criterion for this study.
ABR was recorded with the negative electrode at the mastoid (M1/M2), the positive at the forehead (Fz), and the ground at the glabella (Fpz). During the procedure, children were tested in a natural sleep state, using inserted earphones (ER-3C) to deliver rarefaction clicks at a rate of 23.1 clicks per second, each lasting 0.1 ms. The bandpass filter was set between 100 and 2000 Hz, and waveforms were obtained by averaging over 1024 trials. If the hearing difference between ears exceeded 50 dB, masking noise was applied to the contralateral ear. The minimum ABR was measured at 25 dBnHL, with thresholds assessed in 10 dBnHL increments to ensure reproducibility by measuring each volume at least twice until the lowest volume at which Wave V could be identified was found.
Hearing thresholds were categorized as mild (26-40 dBnHL), moderate (41-70 dBnHL), severe (71-90 dBnHL), and profound (>90 dBnHL).8,23
Statistical analyses were conducted using R (version 4.4.1) with the packages readxl (1.4.3), data table (1.15.4), table1 (1.4.3), and car (3.1-2). Categorical variables were expressed as frequencies and percentages and compared using the chi-square test and Fisher’s exact test. Continuous variables were presented as means (standard deviations) and medians (range) and compared using the Wilcoxon rank-sum test. Univariate analyses were conducted, and variables with logistic regression coefficients P-values less than .1 were included in the multivariate model, with hearing change (yes/no) at 6 months as the outcome. In the decision-tree model, 80% of the samples were randomly assigned to the training set, while the remaining 20% constituted the test set for model validation. To ensure the stability and reliability of the model, a 5-fold cross-validation procedure was implemented within the training set. The final model performance was then assessed using the independent 20% test set, which remained unseen during the training phase.
All statistical tests were 2-tailed, with a significance level set at 0.05. For the decibel levels of hearing, paired t-tests were used to assess differences in hearing levels between different months for the same ear.
The study was approved by the Ethics Committee of Chia-Yi Christian Hospital (No: IRB2024093).
Results
A total of 128 neonates were included in this study, with a total of 203 ears. After excluding 4 cases with craniofacial anomalies, 2 cases with auditory neuropathy spectrum disorder, 7 cases with abnormal tympanograms, and 19 cases with inadequate follow-up, 96 neonates (154 ears) were enrolled in the study. The unchanged group had 107 ears, while the hearing change group had 47 ears, with 45 ears showing improvement and 2 ears showing deterioration. Data on initial degree of hearing loss, birth weight, sex, gestational age, side of hearing loss and care unit are summarized in Table 1. The distribution of ABR threshold changes in the hearing-impaired group is detailed in Table 2.
Baseline Characteristics of Neonates With and Without Hearing Threshold Changes.
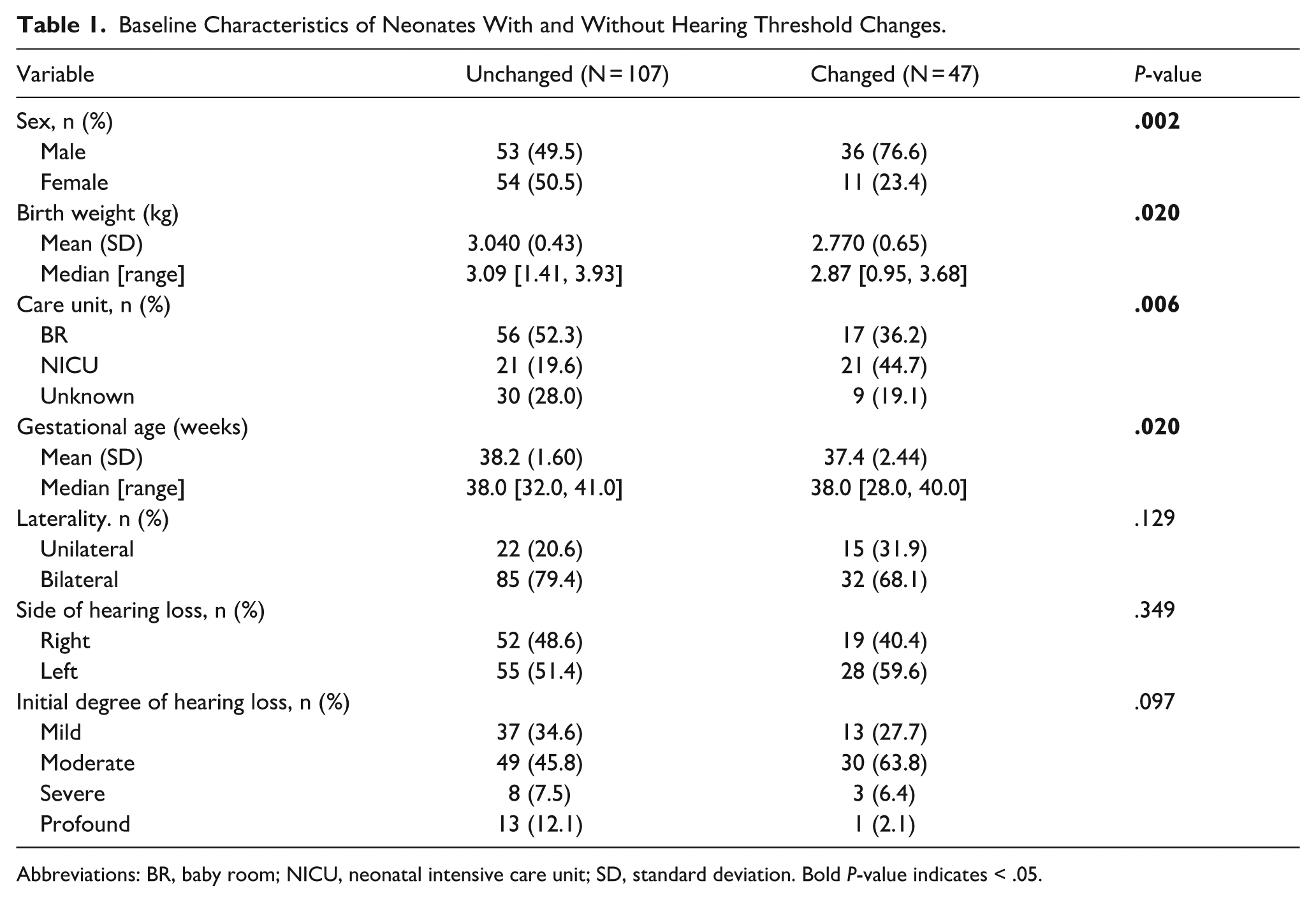
Abbreviations: BR, baby room; NICU, neonatal intensive care unit; SD, standard deviation. Bold P-value indicates < .05.
Distribution of ABR Threshold Changes in the Hearing Changed Group.
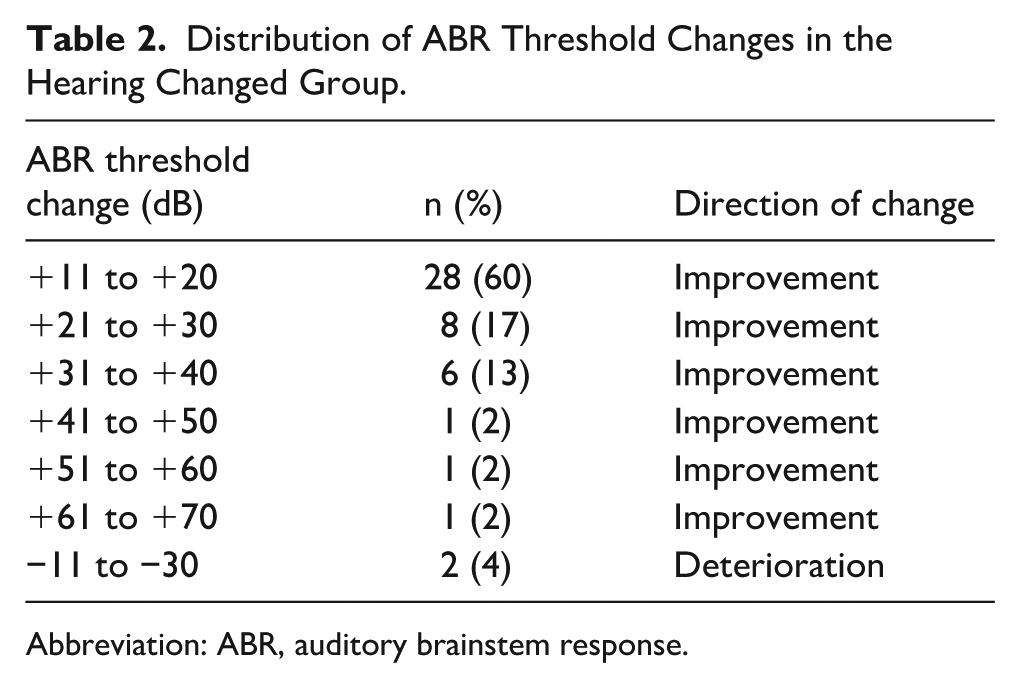
Abbreviation: ABR, auditory brainstem response.
Compared with the hearing unchanged group, the hearing changed group had a significantly lower mean birth weight (2.77 kg vs 3.04 kg, P = .02) and a lower mean gestational age (37.4 weeks vs 38.2 weeks, P = .02). Sex (P = .002) and care unit (P = .006) were also significantly associated with hearing changes. In contrast, no significant associations were observed between the 2 groups with respect to laterality (P = .129), side of hearing loss (P = .349), or initial degree of hearing loss (P = .097).
In univariate logistic regression analyses, birth weight (per 1 kg increase), sex, gestational age (per 1 week increase), and care unit (NICU) were identified as significant factors (P < .05) and were subsequently included in the multivariate model. In the final multivariate analysis, only sex remained a significant independent predictor of ABR threshold changes (adjusted odds ratio [aOR] 3.422; 95% confidence interval [CI] 1.543-7.592; P = .003). Although birth weight per kilogram increase demonstrated a trend toward a protective effect, it did not reach statistical significance (aOR 0.443; 95% CI 0.151-1.232; P = .125). Gestational age (aOR 1.053; 95% CI 0.793-1.389; P = .714) and care unit (NICU vs baby room [BR]: aOR 2.323; 95% CI 0.901-6.064; P = .081; unknown vs BR: aOR 0.887; 95% CI 0.329-2.285; P = .807) were also not significantly associated with ABR threshold changes (Table 3).
Univariate and Multivariable Logistic Regression Analysis Results.
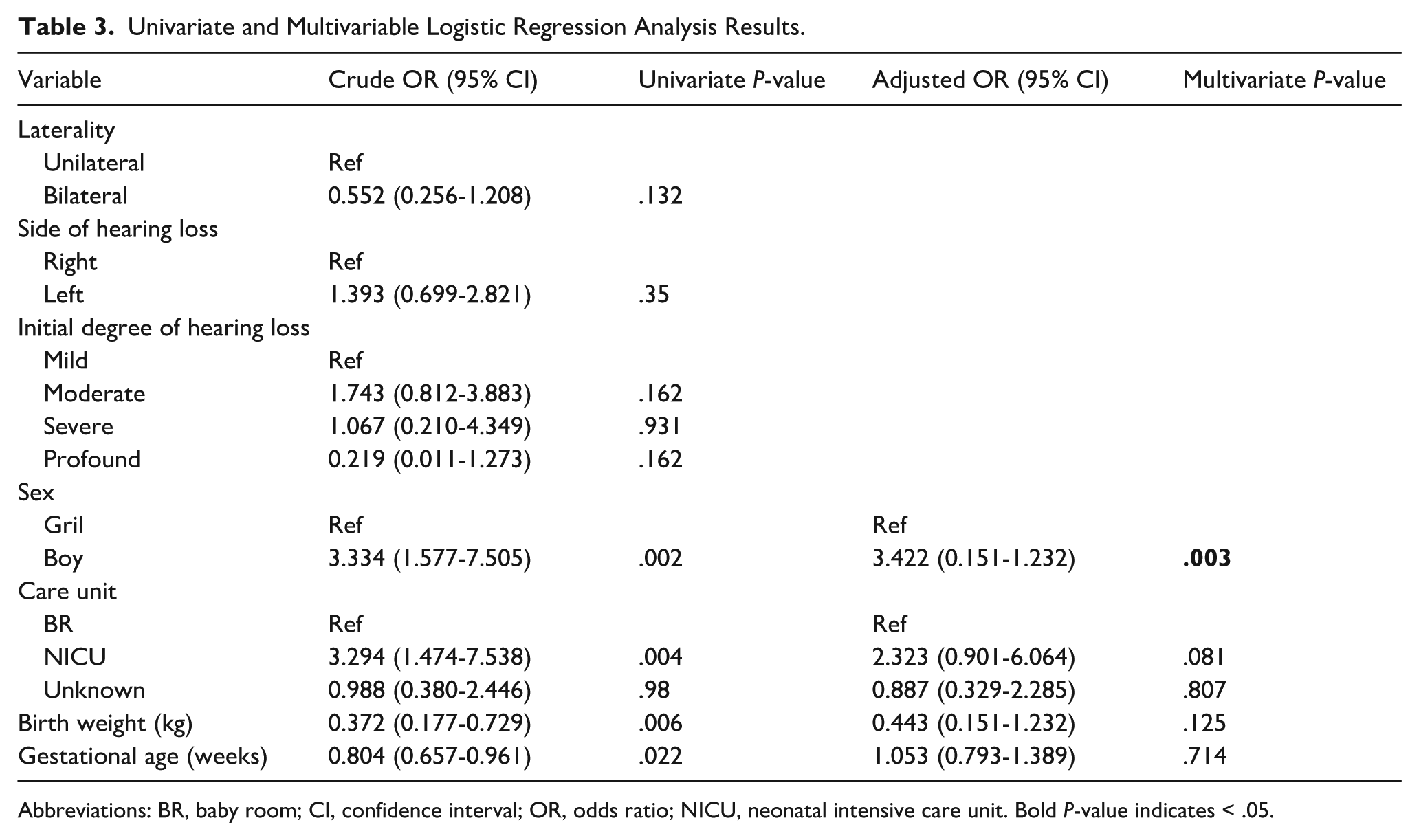
Abbreviations: BR, baby room; CI, confidence interval; OR, odds ratio; NICU, neonatal intensive care unit. Bold P-value indicates < .05.
In the paired t-test, significant differences in hearing levels were observed between the first and third months (mean difference [MD] 5.964, 95% CI 3.903-8.025, P < 0.001), the third and sixth months (MD 2.616, 95% CI 0.873-4.359, P = 0.003), and the first and sixth months (MD 7, 95% CI 4.463-9.536, P < .001). Among neonates with improved hearing thresholds, 8 ears showed improvement by 3 months, 37 ears by 6 months, and 2 ears demonstrated improvement during both the 1 to 3 and 3 to 6 month intervals. In contrast, among neonates with worsening hearing thresholds, deterioration was observed in 1 ear at 3 months and 1 ear at 6 months (Table 4).
Results of the Paired t-Test Analysis of Auditory Brainstem Response Thresholds in Newborns at 1, 3, and 6 Months of Corrected Age.
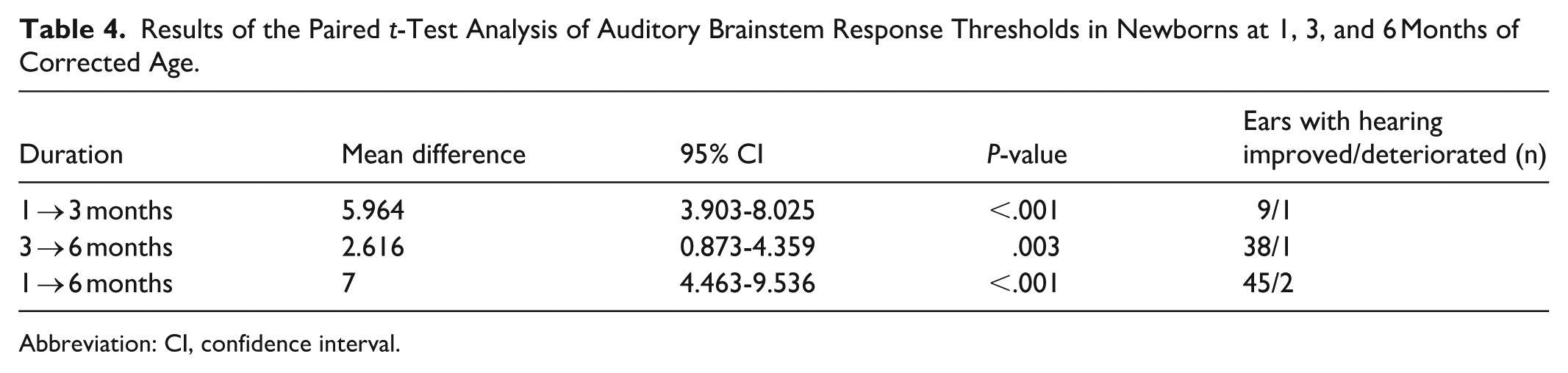
Abbreviation: CI, confidence interval.
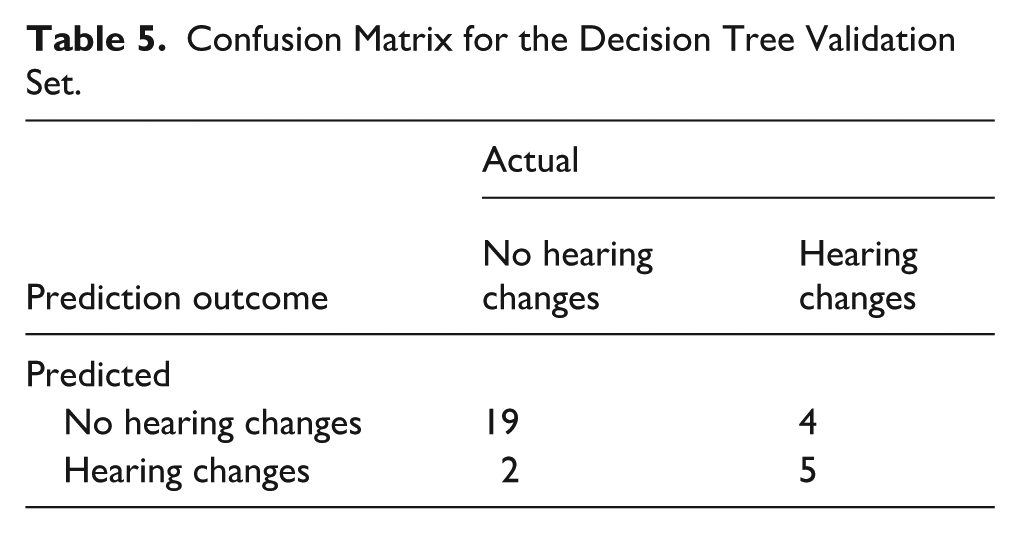
Decision-tree analysis was performed to identify predictors of hearing threshold changes over 6 months. Among male neonates, care unit further influenced outcomes: those admitted to the NICU had a higher proportion of hearing changes (67%) compared with those not admitted (12%). Among males not admitted to the NICU, neonates with a birth weight less than 2.6 kg were more likely to experience hearing changes (71%), whereas those with a birth weight of 2.6 kg or above predominantly showed unchanged hearing thresholds. Overall, the model suggests that male neonates admitted to the NICU, as well as male neonates cared for in the BR or with an unknown care unit and a birth weight <2.6 kg, were more likely to demonstrate improvement in hearing thresholds (Figure 1). The model was validated using the validation set, achieving an accuracy of 0.8 (Table 5).
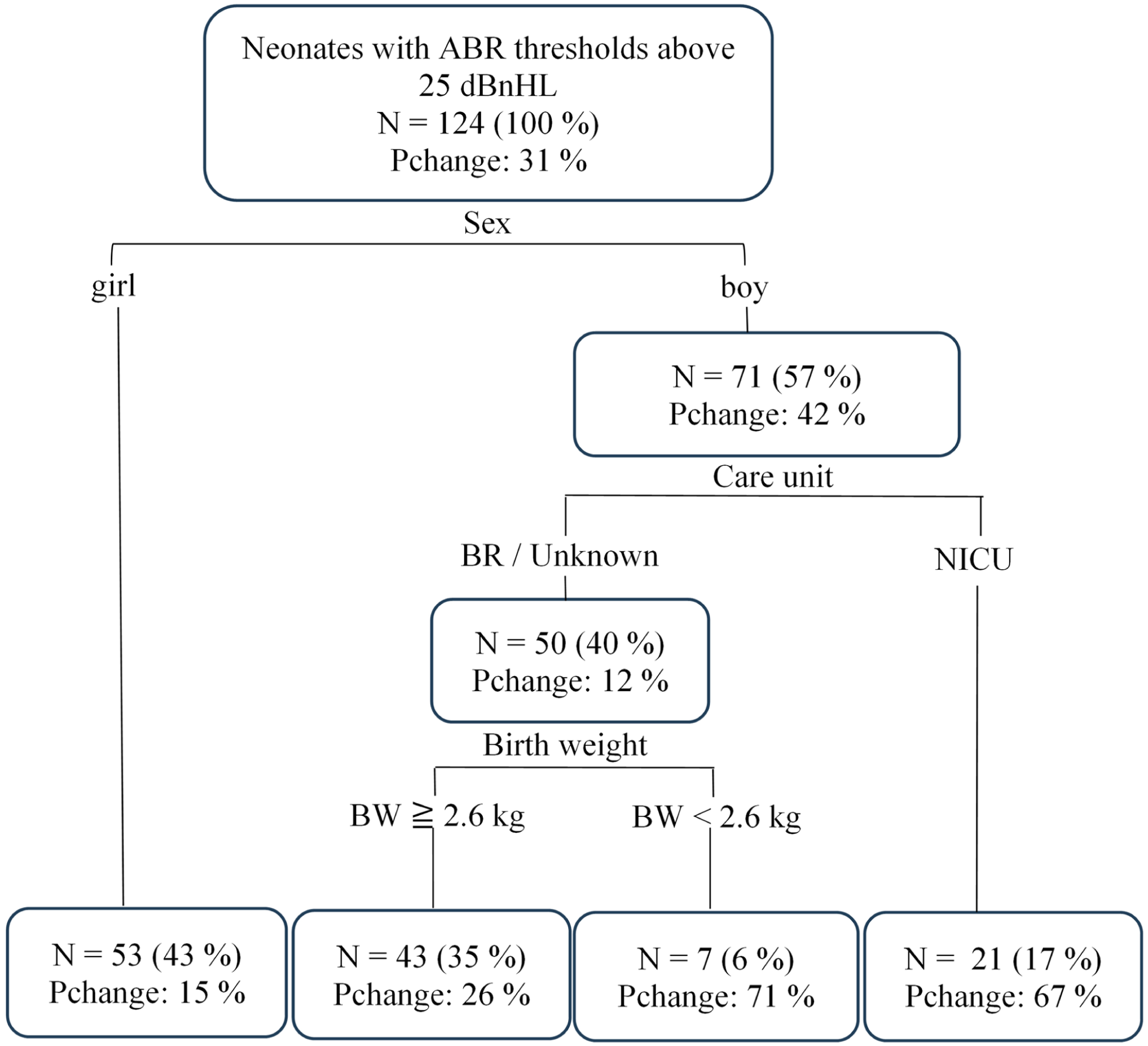
Decision-tree model for the training set predicting hearing changes in neonates within 6 months of corrected age. Node values, from top to bottom, represent the proportion of observed data at each node relative to the total sample, and the proportion of hearing changes. ABR, auditory brainstem response; BR, baby room; BW, birth weight; NICU, neonatal intensive care unit; Pchange, proportion of changed hearing threshold.
Confusion Matrix for the Decision Tree Validation Set.
Discussion
This study investigated patterns of ABR threshold changes before 6 months of CA. Our findings indicate that transfer to the NICU, male sex, lower birth weight, and reduced gestational age were associated with threshold improvements during early infancy. However, multivariate analysis identified sex as the only significant predictor of hearing changes. Decision-tree analysis revealed that male neonates transferred to the NICU and male neonates cared for in the BR or with an unknown care unit and a birth weight <2.6 kg were more likely to demonstrate improvement in hearing thresholds.
Several studies have reported that early ABR thresholds in neonates can improve completely or partially over time. Kang et al observed changes in hearing thresholds among neonates with congenital sensorineural hearing loss during the first year of life, emphasizing the importance of follow-up evaluations. 17 Hof et al found that 64% of preterm infants demonstrated threshold improvements—some returning to normal levels—suggesting auditory pathway maturation before 80 weeks of gestation. 19 Similarly, Bovo reported significant improvements in hearing thresholds among full-term infants by 6 months, while most preterm infants improved after 70 weeks, with 1 case showing improvement after 85 weeks. 16 Psarommatis et al noted that 42% of infants with initial thresholds ≥80 dBnHL exhibited normalization or improvement during follow-up assessments at 4 to 6 months. 18 Consistent with these findings, our study demonstrated statistically significant differences in hearing thresholds at CAs of 1, 3, and 6 months. Although the mean improvement of 7 dB observed between 1 and 6 months fell within the test–retest variability range, an overall trend toward improved hearing thresholds was evident.
The influence of sex on early ABR threshold changes remains inconclusive in the literature. In the present study, a significant difference between sexes was identified, suggesting that males may be more susceptible to early hearing fluctuations. Previous studies have primarily reported sex-related differences in ABR waveform characteristics, such as latency and amplitude, which reflect auditory pathway maturation rather than hearing sensitivity. Borenstein-Levin et al reported a strong association between male sex and prolonged interpeak latencies in both preterm and full-term neonates, indicating delayed auditory brainstem maturation. 24 Similarly, Li et al demonstrated that males exhibited longer wave III and V latencies, extended interpeak intervals, and smaller amplitudes than females across gestational groups. 25 These findings suggest that males, regardless of gestational age, exhibit slower ABR maturation, potentially reflecting delayed myelination. Further studies are warranted to elucidate the biological mechanisms underlying sex-related differences in early auditory development.
This study also explored whether specific characteristics in neonates predispose them to early hearing changes. Hof et al reported that younger gestational age and lower birth weight were associated with improved hearing outcomes, 19 whereas Frezza et al found that greater gestational age predicted better recovery. 8 In contrast, Aldè et al found no significant association between hearing improvement and either gestational age or birth weight. 26 In our study, smaller gestational age and lower birth weight were associated with hearing threshold changes in univariate analysis, but neither remained significant in the multivariate model.
NICU admission has been identified as a potential risk factor for hearing loss, but may also relate to subsequent hearing improvement. Frezza et al reported that 47% of NICU patients experienced hearing recovery, with shorter NICU stays predicting favorable outcomes within the first year. 8 In our study, 50% of neonates transferred to the NICU exhibited threshold changes, a significantly higher proportion than among those who remained in the nursery. However, NICU transfer was not an independent predictor in multivariate analysis, suggesting that other unmeasured clinical factors may contribute to these changes.
Limitations
Its retrospective, single-center design may introduce selection bias and limit the generalizability of the findings. Important high-risk factors, such as hypoxia, hyperbilirubinemia, and exposure to ototoxic medications, were not analyzed and may have influenced the interpretation of hearing outcomes. In addition, bone-conduction ABR was not performed; therefore, despite otoscopic examination by an otolaryngologist and tympanometry to screen for conductive pathology, we could not fully distinguish sensorineural from conductive components of hearing threshold changes. Separate categorical analyses for hearing deterioration were not feasible due to the limited number of cases. Future multicenter, prospective studies incorporating broader clinical and genetic data are warranted to validate these findings and further clarify predictors of early auditory changes.
Conclusion
Hearing thresholds in neonates with hearing impairment may improve over a 6 month follow-up period. These changes are associated with male sex, younger gestational age, lower birth weight, and postnatal transfer to the NICU, although multivariate analysis identified sex as the only independent predictor. Given these findings, structured and ongoing audiological follow-up is essential for early detection, timely intervention, and optimal auditory and developmental outcomes.
Footnotes
Acknowledgements
We express our gratitude to Lo Chuang, Yi-Ju Chen, and Pei-Hsuan Wu at the Chia-Yi Christian Hospital audiometry room for their invaluable help.
Ethical Considerations
This study was approved by the Institutional Review Board (IRB) of the Ditmanson Medical Foundation Chia-Yi Christian Hospital (IRB no. 2024093). The data were collected retrospectively, and the patients were anonymized before data analysis.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Ditmanson Medical Foundation Chia-Yi Christian Hospital Research Program (R113-048).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
