Abstract
Background:
Administration of
Methods:
An animal model of hearing dysfunction was established by administration of
Results:
It was found that
Conclusion:
These findings suggested that
Introduction
Degeneration of the central auditory system is an important cause of age-related hearing loss. Animal model of
The auditory brainstem response (ABR) is an auditory-evoked potential extracted from ongoing electrical activity in the brain and recorded through electrodes placed on the scalp. The measured waves are a series of 6 to 7 vertex waves, which generated by the auditory branches of cranial nerve VIII and lower or upper parts of brainstem. The ABR test is often used to determine the hearing lost type and the degree as well as the auditory threshold estimation. 19,20
Huperzine A is a reversible and selective acetylcholinesterase (AChE) inhibitor,
21
and it has effects of antiaging and anti-inflammation in the neural and immune systems.
13,21,22
Thus, huperzine A has been used to inhibit immunosenescence and age-related disorders, such as Alzheimer disease.
21
-24
However, it has not been well-understood whether huperzine A can reduce
Materials and Methods
Animals and Drug Administration
Two-month-old male Sprague-Dawley rats (200-220 g) were maintained at room temperature (RT) under a 12-hour light/dark cycle with food and water ad libitum in The Laboratory Animal Center, Shanghai Medical College, Fudan University. After acclimatization to the laboratory conditions, the rats were randomly assigned to the following 3 groups (n = 12 per group): the
Auditory Brainstem Response Test
Basal threshold of ABR was tested in all rats prior to the use of drugs to confirm normal hearing function in all animals before the experiment. At the end of the experimental treatment, ABR test was conducted again. Briefly, the rats were anesthetized by intraperitoneal injection of pentobarbital with 80 mg/kg and received functional tests in the acoustic and electrical shielding room. During the test, body temperature was maintained with a constant heating pad. Stimulation signal and evoked potential recording were accomplished by TDT system III (RZ6; Tucker-Davis Technology, Washington, USA). The stimulus signal for ABR was 10 ms pure tone, with an ascending/descending time of 0.5 ms, and a repetition rate of 21.1 beats per second. Test frequency range (2-48) kHz was an octave range. At each frequency, the test starts at 90 dB SPL, with the minimum sound intensity of the III wave as the response threshold. Electrodes via hypodermic needle guide, parietal middle ear on both sides of the recording electrodes, reference electrode, and grounding electrode were applied. Bioelectrical signals were introduced by electrodes into RA16PA preamplifier, band pass filtering at 100 to 3000 Hz, and ABR addition of 1000 times.
Tissue Preparation
The rats were culled using an overdose of sodium pentobarbital and perfused through the heart using a 4% paraformaldehyde solution. The cochlea was postfixed in 4% paraformaldehyde overnight, decalcified with 10% EDTA for 2 weeks, and cryoprotected using a gradient sucrose solution (starting with 15%, followed by 30%). Serial sections were sliced at a thickness of 5 mm (Leica, Wetzlar, Germany).
Cellular Senescence Assay
Cellular senescence is believed to be associated with chronic hearing loss and, thus, senescence of cochlear cells was assessed. To accomplish this, a senescence-associated β-galactosidase (SA-β-Gal) staining kit (Beyotime Institute of Biotechnology, Suzhou, China) was used for SA-β-Gal staining following the manufacturer’s instruction. Briefly, the sections were fixed in a SA-β-Gal fixing solution for 15 minutes. After washing 3 times in phosphate-buffered saline (PBS), the sections were incubated overnight at 37°C with the SA-β-Gal working solution followed by photographing under a microscope.
Immunohistochemistry
The cochlea slices of the rats in each group were placed at RT for 30 minutes, fixed with 4% acetone for 10 minutes, and rinsed 3 times for 5 minutes each with PBS containing 1% (wt/vol) bovine serum albumin, 5% heat-inactivated goat serum, and 1% Triton X-100 in 0.1 mmol PBS. The cochlea slices were then incubated with 3% H2O2 for 5 minutes followed by rinsing 3 times for 5 minutes each. The primary antibodies, anti-neurofilament 200 antibody (1:100; Abcam, Cambridge, United Kingdom), anti-S-100 antibody (1:100, Abcam), and NF-κB (1:100, anti-p65-antibody) were allowed to react at 4°C overnight. After washing, the following secondary antibodies were allowed to bind for 4 hours at RT: goat anti-mouse immunoglobulin (Ig) G1-fluorescein isothiocyanate (FITC; 1:100; Santa Cruz Biotechnology, California), fluorescein-conjugated rabbit polyclonal secondary antibody to mouse IgG/IgM/IgA (1:100; Abcam), and FITC-conjugated goat anti-rabbit IgG (1:100, Kangchen Bio-tech, Shanghai, China). The slides were stained with 4′,6-diamidino-2-phenylindole (DAPI) for 15 minutes followed by photographing under a microscope. Number of positively stained neurofilament (red) was counted under the microscope by randomly selecting 5 high-power field (200× magnification). Average of the 5 fields between the 3 groups was compared.
Quantitative RT-PCR
Total RNA was extracted from the cochlear tissues using Trizol (Invitrogen, Carlsbad, California) followed by reverse transcription into complementary DNA using the reverse transcription system (Invitrogen). Real-time polymerase chain reaction (PCR) was then performed using a SYBR Premix reverse transcription-polymerase chain reaction (RT-PCR) kit (Invitrogen) following the manufacturer’s instructions. The primer sequences are listed in Table 1.
List of the Primers Used for the Quantitative Real-Time PCR.
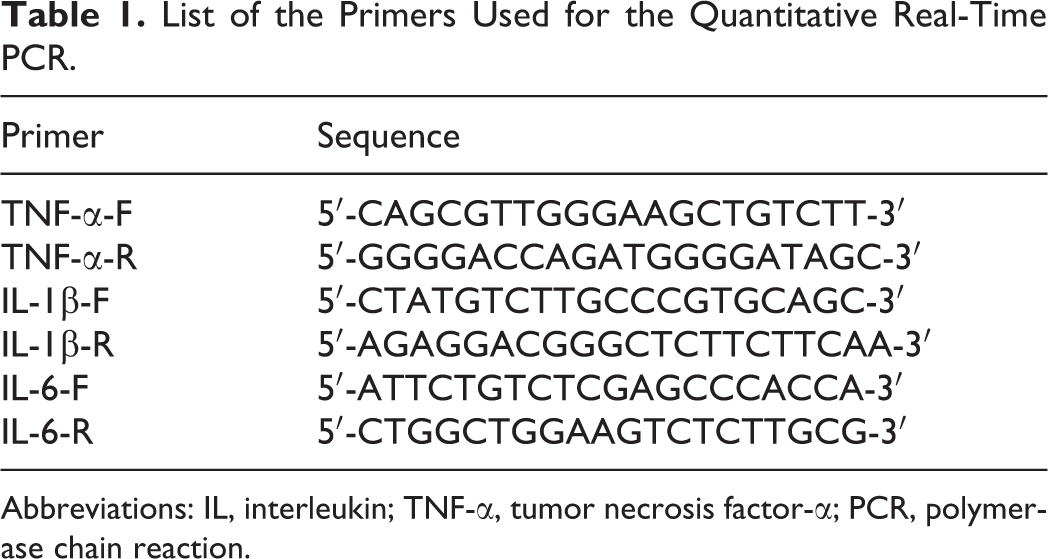
Abbreviations: IL, interleukin; TNF-α, tumor necrosis factor-α; PCR, polymerase chain reaction.
Image Acquisition and Statistics
The specific group identity was coded so that all microscopy and computer analyses were performed blinded to the treatment group. The data in all studies were expressed as the means ± standard error of the mean. A one-way analysis of variance was performed to evaluate the differences in the levels of protein and messenger RNA (mRNA). The difference was considered to be significant at P < .05.
Results
Effect on ABR Threshold
As shown in Table 2, there was no significant difference in basal frequencies of ABR between the groups. As shown in Table 3 and Figure 1, however, ABR threshold was significantly increased at 4 and 8 kHz sampling after administration of
Comparison of Basic ABR at Various Frequencies.
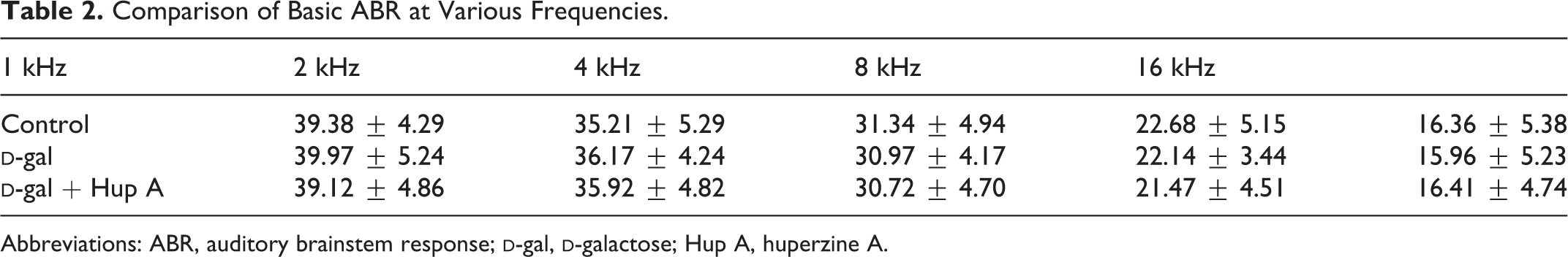
Abbreviations: ABR, auditory brainstem response;
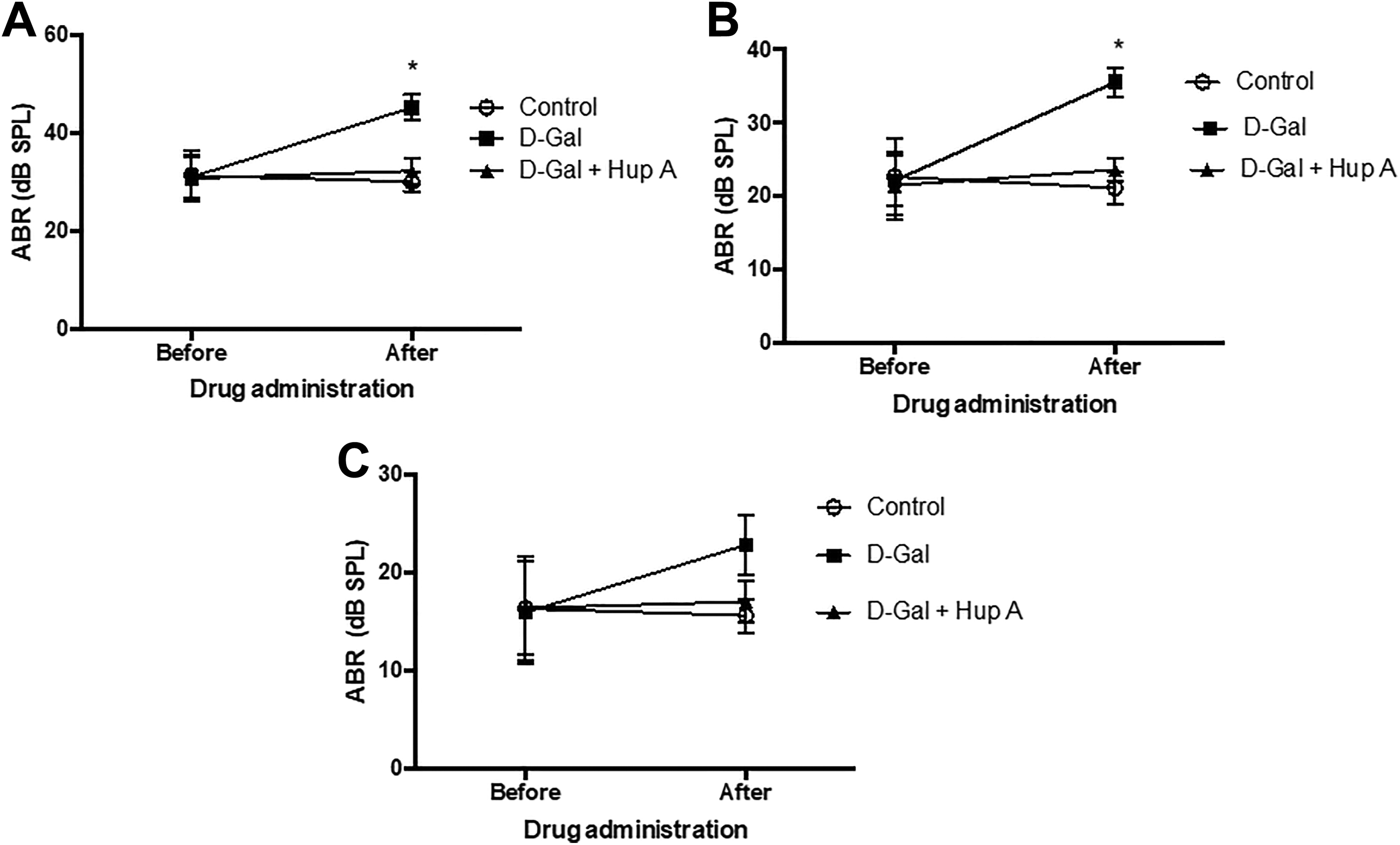
Auditory brainstem response (ABR) before and after administration of
Comparison of ABR at Various Frequencies After Treatment.
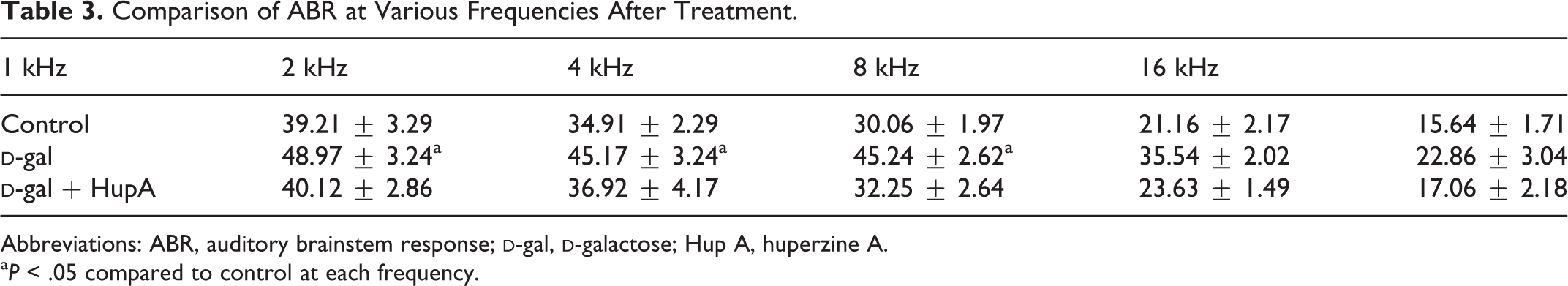
Abbreviations: ABR, auditory brainstem response;
aP < .05 compared to control at each frequency.
Effect on the Cochlear Tissue
To evaluate the effect of
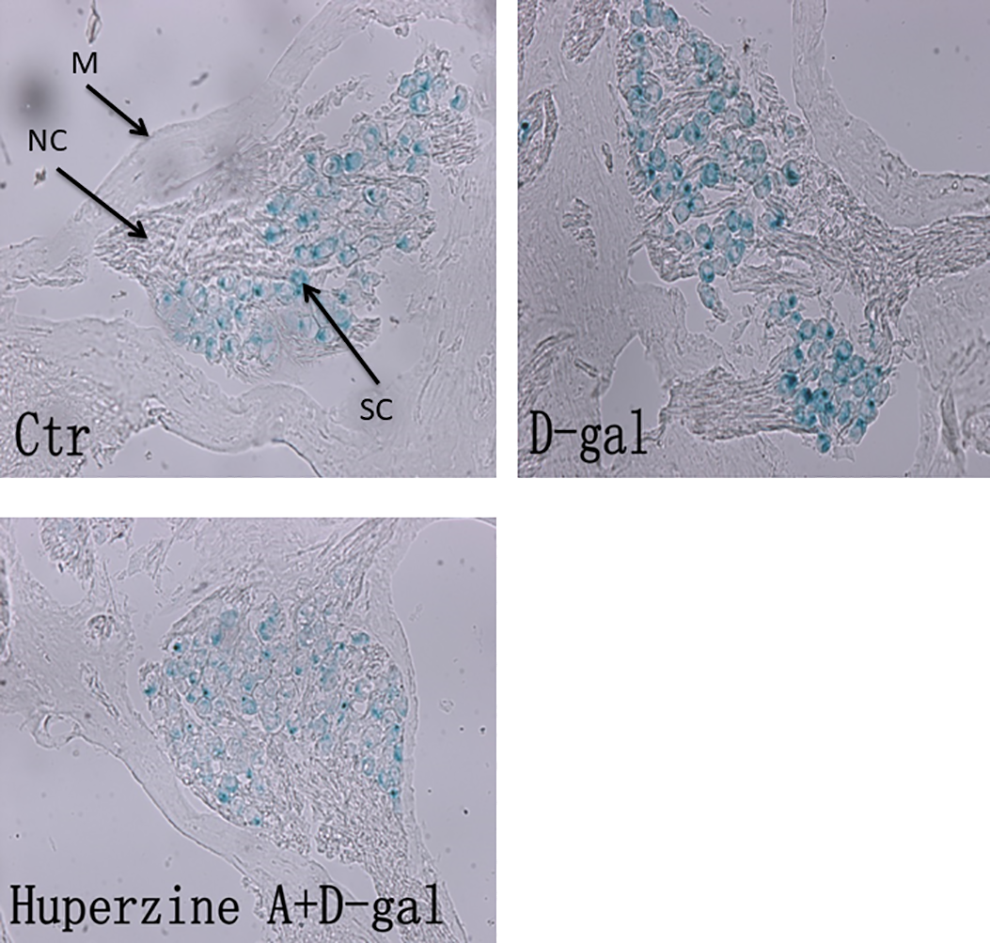
Senescence-associated β-galactosidase (SA-β-Gal) staining in the cochlear tissues. Cochlear tissues were obtained, and SA-β-Gal staining was performed as described in the Materials and Methods section. Ctr, control group;
Next, the effect of
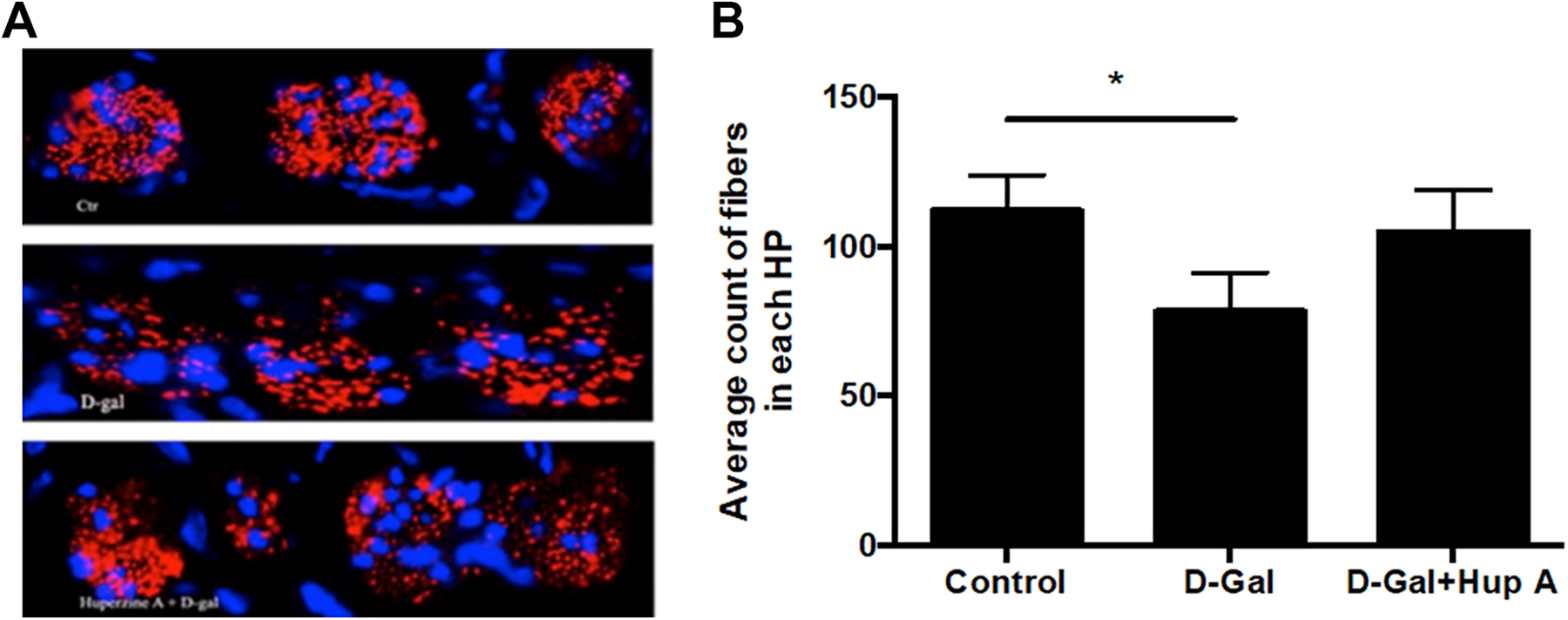
Immunostaining of nerve fibers. Immunofluorescence staining for nerve fibers with anti-NF200 antibody (red) and nuclei staining with 4′,6-diamidino-2-phenylindole (blue) was performed as described in the Materials and Methods section. A, Representative image data. Top, control group; middle, animals treated with
Effect on the Pro-Inflammatory Cytokine Synthesis
Gene expression of pro-inflammatory cytokines, IL-1β, IL-6, and TNF-α was quantified by real-time RT-PCR. As shown in Figure 4, mRNA expression of pro-inflammatory cytokines, IL-1β, IL-6, and TNF-α was significantly upregulated in the animals injected with
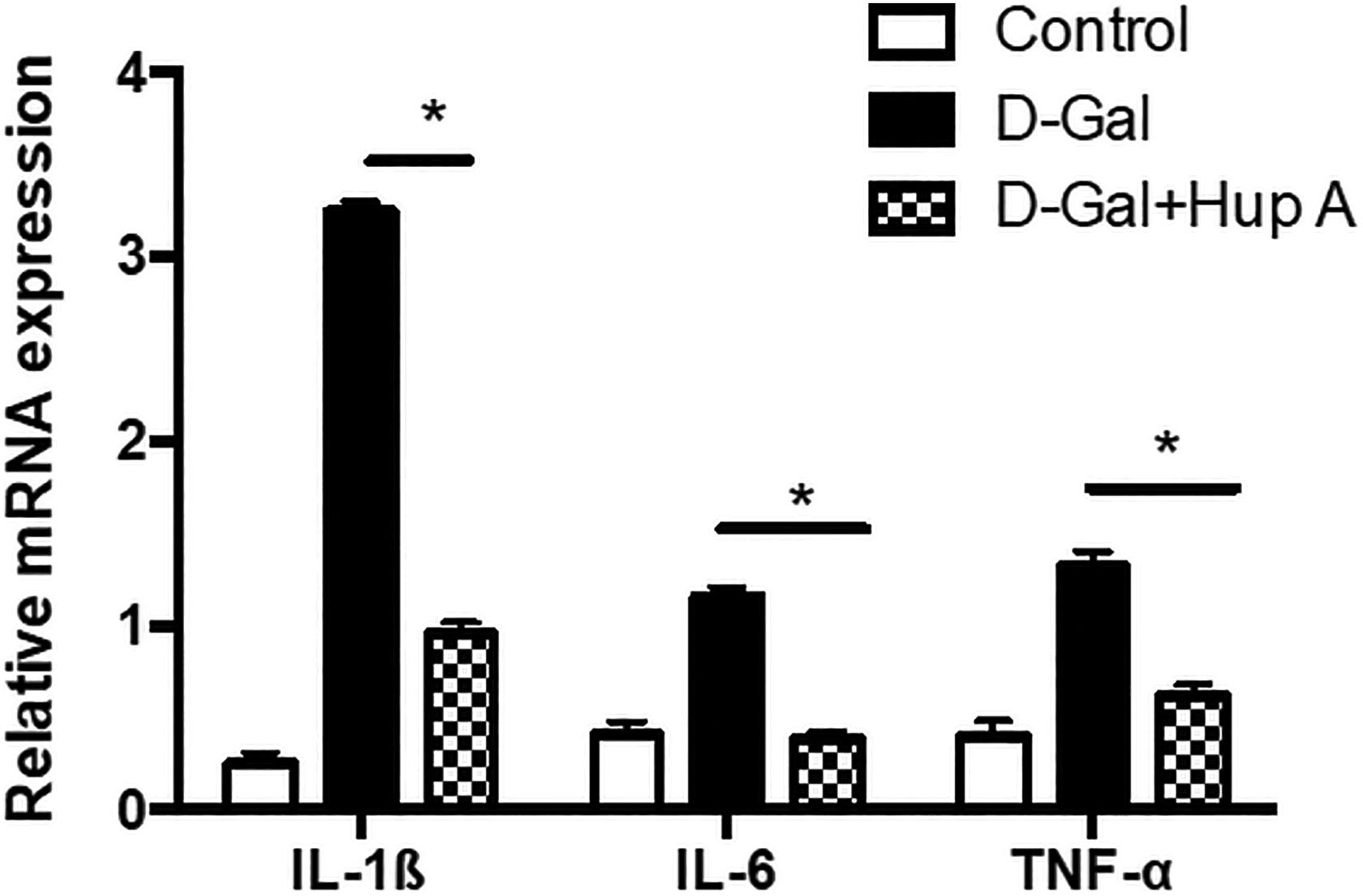
Expression of pro-inflammatory cytokine messenger RNA (mRNA). Total RNA was extracted from cochlear tissues of the animals treated with or without
Since Schwann cells secrete a variety of active substances including cytokines, and NF-κB signaling regulates pro-inflammatory cytokine production, NF-κB activation in the Schwann cells was evaluated by double staining of S-100 (biomarker of Schwann cell) and p65. As shown in Figure 5, compared with the control group, intensity of Schwann cell staining (red) and NF-κB staining (green) was stronger in the cochlea of
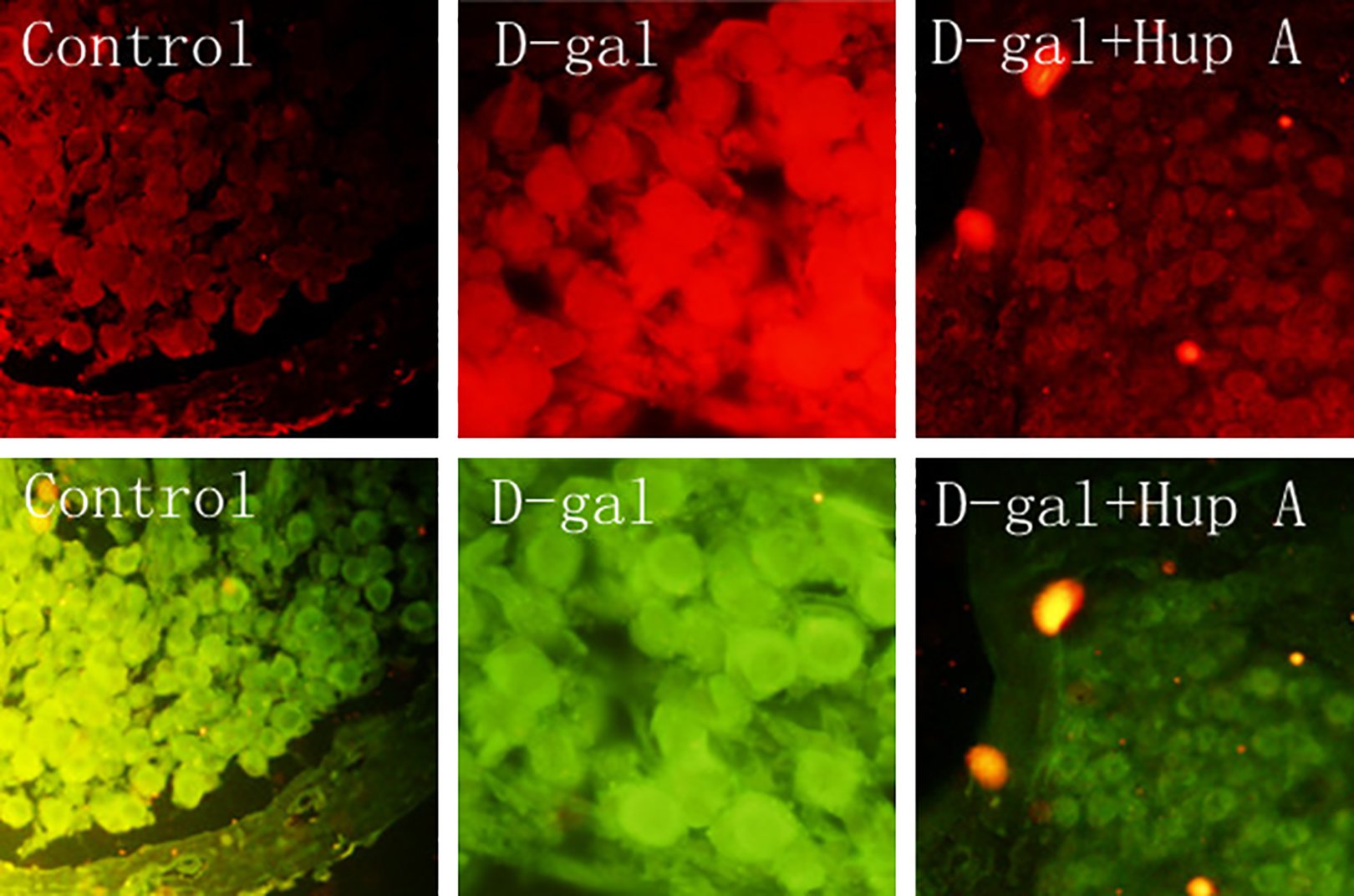
Schwann cells and NF-κB immunostaining. Schwann cells were identified by immunostaining with anti-S-100 antibody (red), and NF-κB was identified by anti-p65 antibody (green) as described in the Materials and Methods section. Data presented were one representative of 3 separate experiments. Control, animals were not treated;
Discussion
In this study, the effect of huperzine A on
Chronic administration of
Huperzine A is a reversible and selective AChE inhibitor.
21
Huperzine A has been widely used to improve cognitive and memory deficits in patients with benign senescence forgetfulness, Alzheimer disease, and vascular dementia via non-cholinergic mechanisms, including attenuating oxidative stress, inhibiting apoptosis, and interfering with amyloid precursor protein metabolism.
27
-30
In addition, huperzine A may protect against diverse neurodegenerative diseases by blocking N-methyl-
While the mechanisms of
Several mechanisms may be associated with age-related chronic hearing loss. In this regard, telemase-associated cellular senescence, abnormal differentiation of the cochlear cells, and membrane hypothesis have been considered to be associated with chronic hearing loss. Therefore, in this study, cellular senescence of cochlear tissues was examined. Consistent with the aforementioned concept, this study demonstrated that
Huperzine A is an inhibitor of AChE and often used for the treatment of age-related diseases including Alzheimer disease and vascular dementia.
33,34
However, a systemic review and meta-analysis of 20 randomized clinical trials with1823 participants indicated that huperzine A appeared to have beneficial effects on improvement of cognitive function, daily living activity, and global clinical assessment in the patients with Alzheimer, the authors suggested that the findings should be interpreted with caution due to the poor methodological quality of the randomized clinical trials.
34
Here, we reported that huperzine A could significantly antagonize
There are several limitations in this study. First, only 1 dose of
Conclusion
Taken together, this study demonstrated that administration of
Footnotes
Authors Contributions
CL and SS contributed to the conception and design of the study, acquisition, and analysis of data. CL wrote the manuscript. All authors reviewed and approved the final version of the manuscript. All necessary information about this study was presented in the manuscript. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Science and Technology Commission Fund of Changning District, Shanghai (Grant No. CNKW2016Y05).
