Abstract
Objective:
To investigate the association between clinical sleep parameters and olfactory function (OF) in patients with obstructive sleep apnea (OSA) and to evaluate the therapeutic effect of 12-month continuous positive airway pressure (CPAP) on olfactory dysfunction (OD).
Methods:
A total of 146 participants were included: 55 healthy controls, 68 untreated OSA patients, and 23 CPAP-treated patients. OSA severity was classified according to the apnea–hypopnea index (AHI). OF was assessed using the Sniffin’ Sticks test. Clinical variables, sleep parameters, and olfactory scores were compared among groups. Subgroup analysis between CPAP-treated and untreated OSA patients used the Mann–Whitney U test. Multivariate logistic regression identified predictors of olfactory improvement.
Results:
Our findings demonstrated that OD was correlated with the severity of OSA. Olfactory scores were significantly negatively correlated with AHI (r = −.544, P < .001), but positively correlated with average SpO2% (r = .333, P = .006) and minimum SpO2% (r = .449, P < .001). After 12 months of CPAP treatment, the mean olfactory score reached 13.87 ± 1.96, significantly higher than 9.78 ± 4.217 in the untreated group, demonstrating substantial functional recovery.
Conclusion:
The results indicated a close relationship between OD and disease severity in patients with OSA and confirmed that long-term CPAP therapy can effectively improve OF. These findings provide new insights into the multisystem impact of OSA and suggest that olfactory assessment may serve as a complementary clinical tool for OSA evaluation.
Keywords
Introduction
Obstructive sleep apnea (OSA), characterized by recurrent obstruction of the upper airways during sleep, manifests itself primarily as a decrease in arterial blood SpO2%. The prevalence of OSA in the general population is approximately 30%, and the incidence rates in males and females are 24% and 9%, respectively.1,2 Untreated OSA can lead to a series of severe complications, including, but not limited to, hypertension, arrhythmias, myocardial infarction, cerebrovascular disease, impaired memory, and learning abilities, as well as various neurocognitive and mental health issues.3 -5 OSA has been established as a risk factor for mild cognitive impairment, and adults with severe OSA are more susceptible to cognitive dysfunction compared to those with mild-to-moderate OSA.
The olfactory function (OF), as a unique perceptual mechanism, is critical to daily social interactions and to avoid potential dangers in human life. 6 A recent large-scale population study revealed an overall incidence of olfactory dysfunction (OD) of 19.1%, olfactory hypoesthesia and anosmia are reported in 13.3% and 5.8% of patients, respectively. 7 Hummel et al found that the incidence of OD reached 20% among experimental subjects aged 36 to 55 years, and this phenomenon is even more prominent in older individuals. 8 Conventional therapeutic approaches for OD primarily include topical corticosteroids, antihistamines, and olfactory training.9,10 Existing research evidence shows that cognitive impairments, head injuries, upper respiratory infections, and nasal and sinus diseases can affect OF. A significant correlation has been demonstrated between OF and OSA. Hypoxia and reduced nasal airflow caused by OSA can induce OD.11,12
Recent studies have highlighted that the underlying mechanisms by which OSA affects OF can involve intermittent chronic hypoxia, which promotes oxidative stress and neuroinflammation in the olfactory bulb and associated neural pathways. Furthermore, repeated collapse of the upper airways and nasal obstruction can cause mucosal edema and epithelial damage, further exacerbating olfactory impairment (Figure 1).
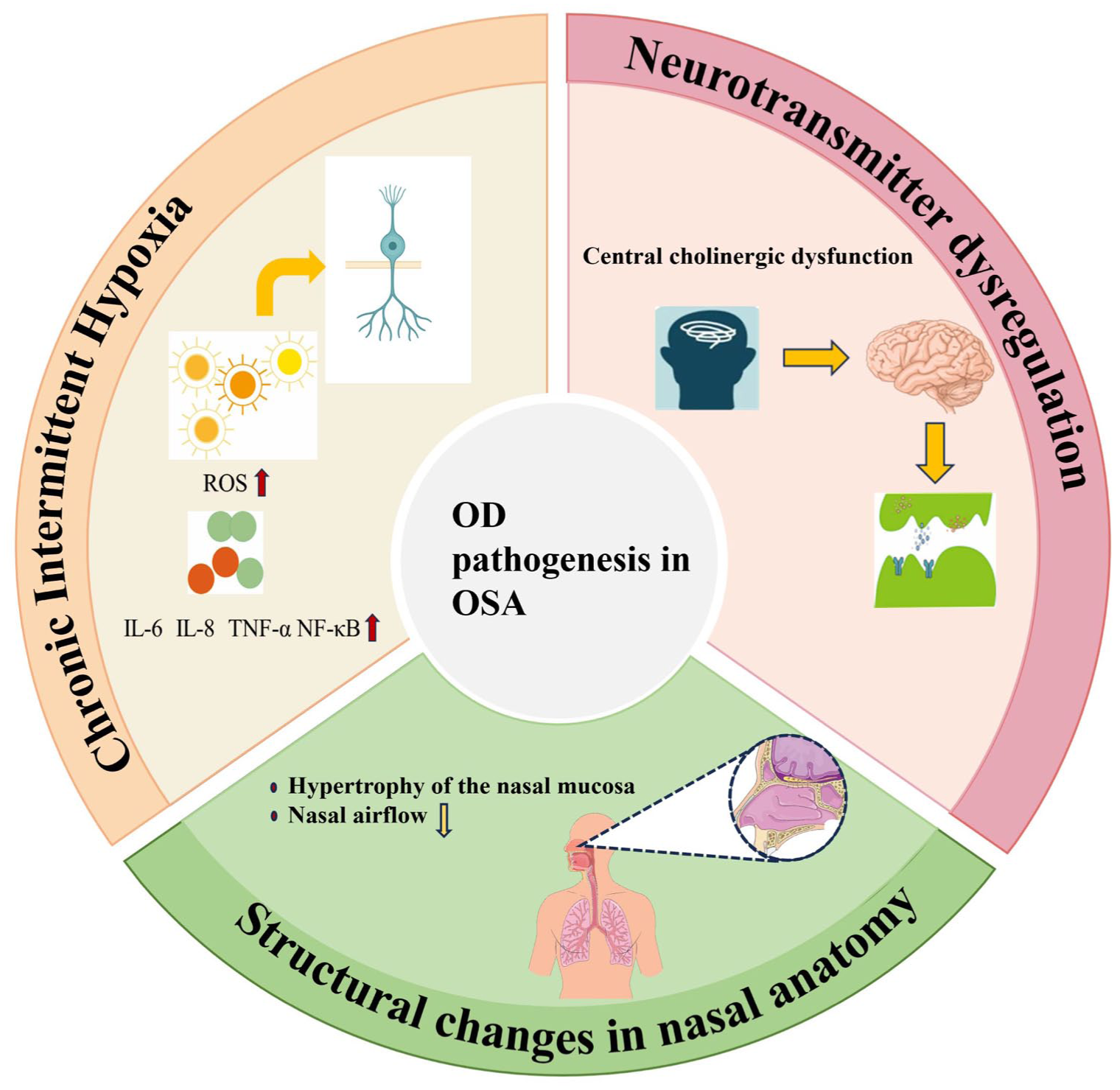
Schematic illustration of the mechanisms linking OSA to OD. Three major pathological pathways are highlighted: chronic intermittent hypoxia, which increases ROS and pro-inflammatory cytokines (IL-6, IL-8, TNF-α, NF-κB); neurotransmitter dysregulation, particularly central cholinergic dysfunction; and structural alterations of the nasal airway, including mucosal hypertrophy and reduced airflow. These mechanisms collectively impair olfactory function in OSA patients. OD, olfactory dysfunction; OSA, obstructive sleep apnea; ROS, reactive oxygen species.
However, few literature reports focus on how OSA leads to OD and how to improve OF in individuals with OSA. Continuous positive airway pressure (CPAP) therapy is widely considered an effective treatment for OSA and has also been used to improve OD in OSA patients. 13 Additionally, mandibular advancement devices (MADs) have been shown to enlarge upper airway dimensions, particularly within the velopharyngeal region, thereby reducing airway collapsibility and apnea–hypopnea index (AHI). This provides an alternative therapeutic approach that may also influence upper airway physiology relevant to OF. 14
The purpose of this research was to identify the factors associated with OD in patients with OSA, examine the correlation between the severity of OSA and the degree of OD, and evaluate the efficacy of 12-month CPAP in improving OF.
Methods
Subjects
This research was approved by a local ethics committee. All participants provided their informed consent before beginning the research. A total of 283 participants from Shandong Provincial Hospital were enrolled in the current investigation. One hundred thirty-seven patients were excluded due to a history of upper respiratory tract infection within the past 3 weeks or obstructive nasal pathologies, including nasal septal deviation, inferior turbinate hypertrophy, nasal polyps, and allergic rhinitis. The final study population consisted of 146 participants, including 55 healthy controls, 68 untreated OSA patients, and 23 patients who had undergone 12 months of CPAP therapy (Figure 2). Individuals diagnosed with OSA on polysomnography (PSG) and who underwent CPAP treatment for 12 months were included in the current investigation. The links between olfactory scores and AHI, average SpO2%, and minimum SpO2% were investigated using data from these participants. CPAP treatment was provided in optimal therapeutic settings, and night-to-night adherence to CPAP was ensured by continuous supervision. CPAP devices were set to ensure nightly use ≥4 hours, the standard threshold for therapeutic compliance. Precise nightly usage was not systematically recorded, but all patients maintained the prescribed minimum. The patients then received CPAP for at least 12 months. Subsequently, the OF was evaluated as previously described. Ultimately, this study compared OF before and after CPAP.
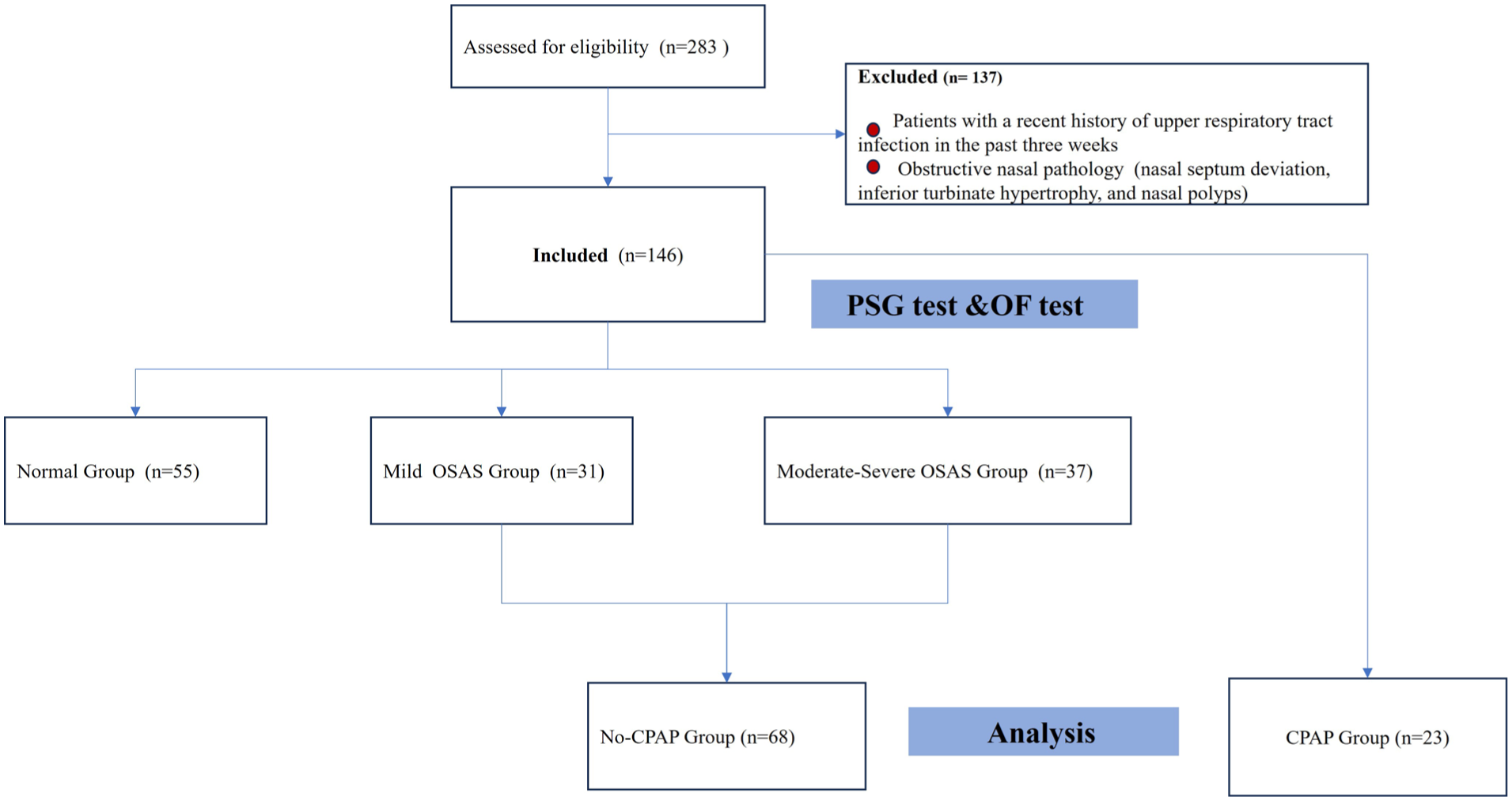
Flowchart of the study.
PSG Test
The PSG was used to record sleep and breathing throughout the night. Data analysis was carried out according to standard criteria. Apnea was defined as a ≥90% decrease in airflow from baseline before the event for ≥10 seconds. Hypopnea was defined as a decrease in airflow by ≥30% from the pre-event baseline for ≥10 seconds, together with a ≥3% oxygen desaturation from the pre-event baseline and/or an associated arousal.5,15 AHI was used to evaluate the severity of OSA. Participants consisted of newly diagnosed severe OSA (AHI >30/hour), moderate OSA (AHI >15-30/hour), mild OSA (AHI >5-15/hour), and normal groups (AHI <5/hour) according to PSG findings. 16
OF Test
OF was assessed using the Sniffin Sticks test (Chinese version, 16 odors), administered by the same investigator for consistency. During the test, the cap of the odorant-filled felt-tip pen was removed and the tip was positioned approximately 2 cm from the subject’s nostrils. Participants were instructed to smell the odor and identify it from the 4 options given. Each correctly identified odor was scored 1 point. The total score was used to assess the severity of OD. The severity was classified as follows: normal OF (14-16 points), mild OD (12-13 points), moderate OD (10-11 points), severe OD (7-9 points), and anosmia (0-6 points). 17
Statistical Analysis
The Kolmogorov–Smirnov test was utilized to probe the distribution of the data. Continuous data were reported as mean with standard deviation and categorical data as numbers. Differences between groups were analyzed using the Mann–Whitney U test or the Kruskal–Wallis test. Spearman’s correlation analysis was carried out to examine associations between olfactory scores and AHI, average oxygen saturation (average SpO2%), and minimum oxygen saturation (minimum SpO2%). Using multivariate regression analysis, we identified the factors influencing the improvement of OF through CPAP therapy in patients with OSA. The link between olfactory scores and sleep-related variables was established using a simple linear regression model. The P value <.05 was considered statistically significant. All statistical analyzes were performed using SPSS statistical software (SPSS version 25.0; IBM Corp., Armonk, NY, USA).
Results
Baseline Clinical Characteristics of the Normal, Mild, and Moderate-Severe OSA Groups
The main clinical characteristics, sleep characteristics, and OF test scores of the moderate—severe OSA, mild OSA, and normal groups are shown in Table 1. No significant differences were observed between the normal and OSA groups in terms of age, sex, smoking status, or alcohol consumption. Body mass index (BMI) and sleep-related parameters, including AHI, average SpO2%, and minimum SpO2%, differed significantly between the groups. In the moderate-to-severe OSA, mild OSA, and normal groups, the OF test scores showed a downward trend from the normal group toward the moderate-to-severe OSA group, which was statistically significant.
Demographic and Clinical Characteristics in Study Subjects With and Without OSA.
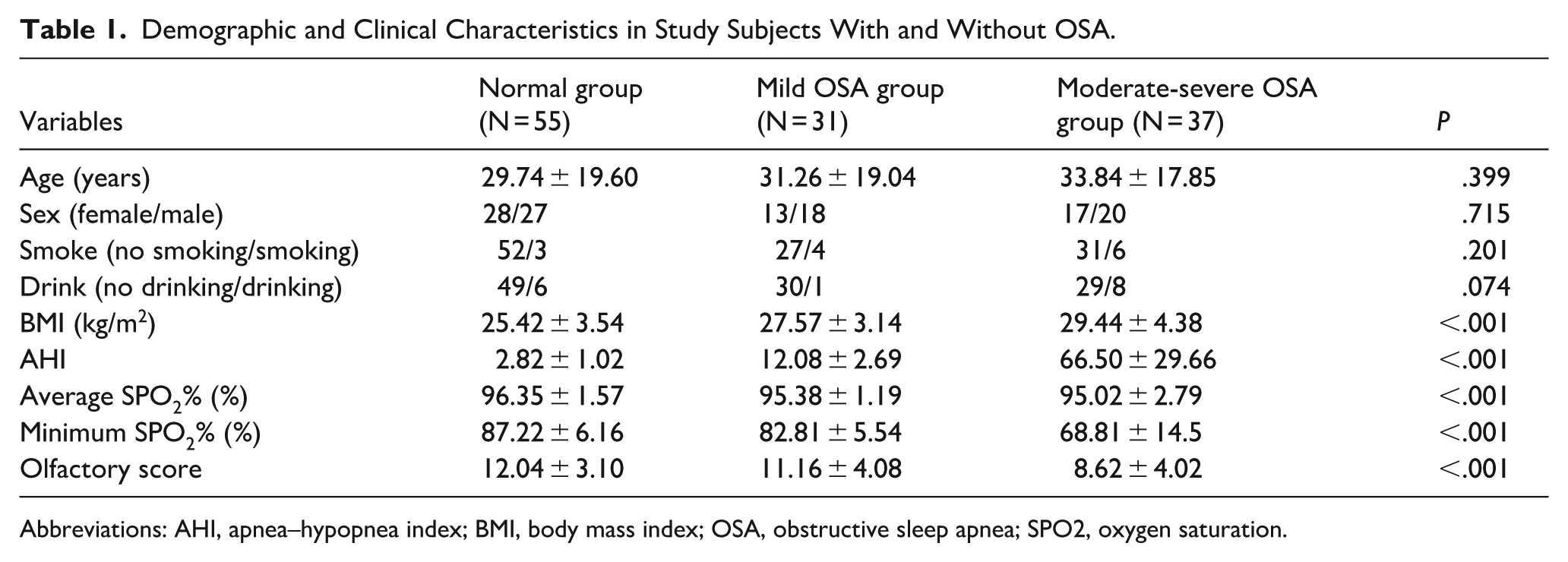
Abbreviations: AHI, apnea–hypopnea index; BMI, body mass index; OSA, obstructive sleep apnea; SPO2, oxygen saturation.
Differences in OF Between Normal and OSA Groups
The comparison of olfactory score differences between OSA and normal groups is shown in Figure 3. Olfactory scores were lower in the OSA group compared to the normal group. The difference between the OSA and normal groups was statistically significant.
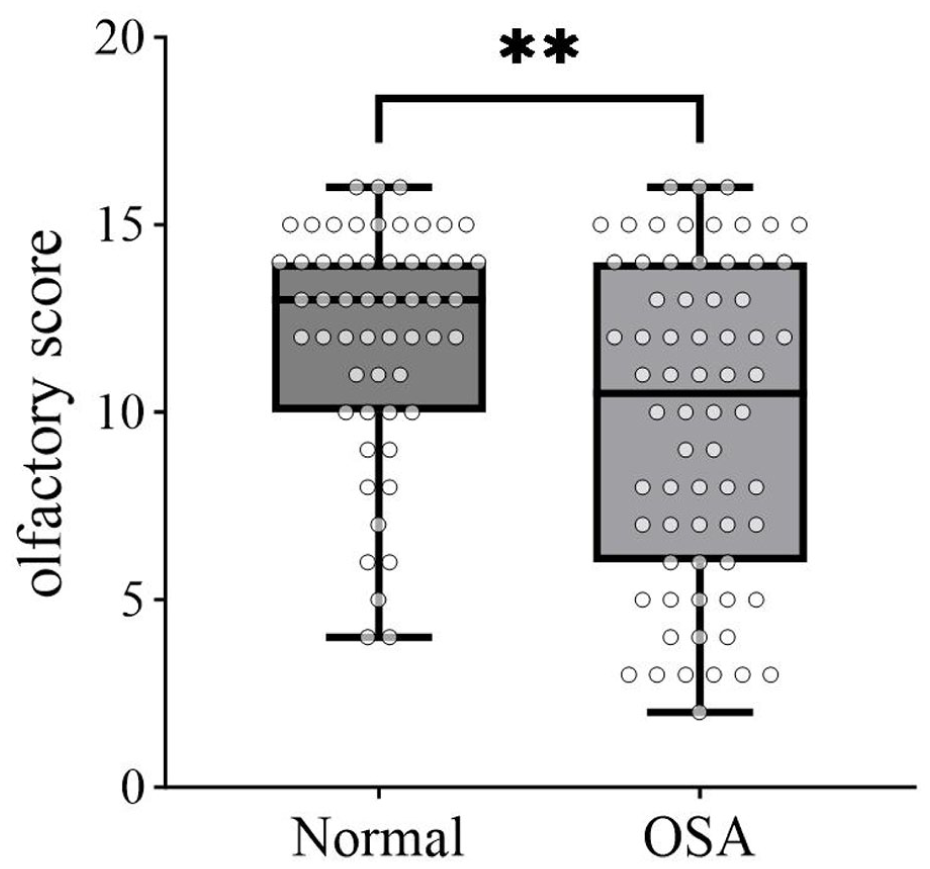
Box plot demonstrating the comparison of olfactory score differences between the normal group and the OSA group. OSA, obstructive sleep apnea.
Differences in OF Between the Mild OSA and Moderate-Severe OSA Groups
The comparison of olfactory score differences between mild OSA and moderate-severe OSA groups is demonstrated in Figure 4. OF test scores were significantly different in individuals with mild OSA and in the moderate-to-severe OSA groups.
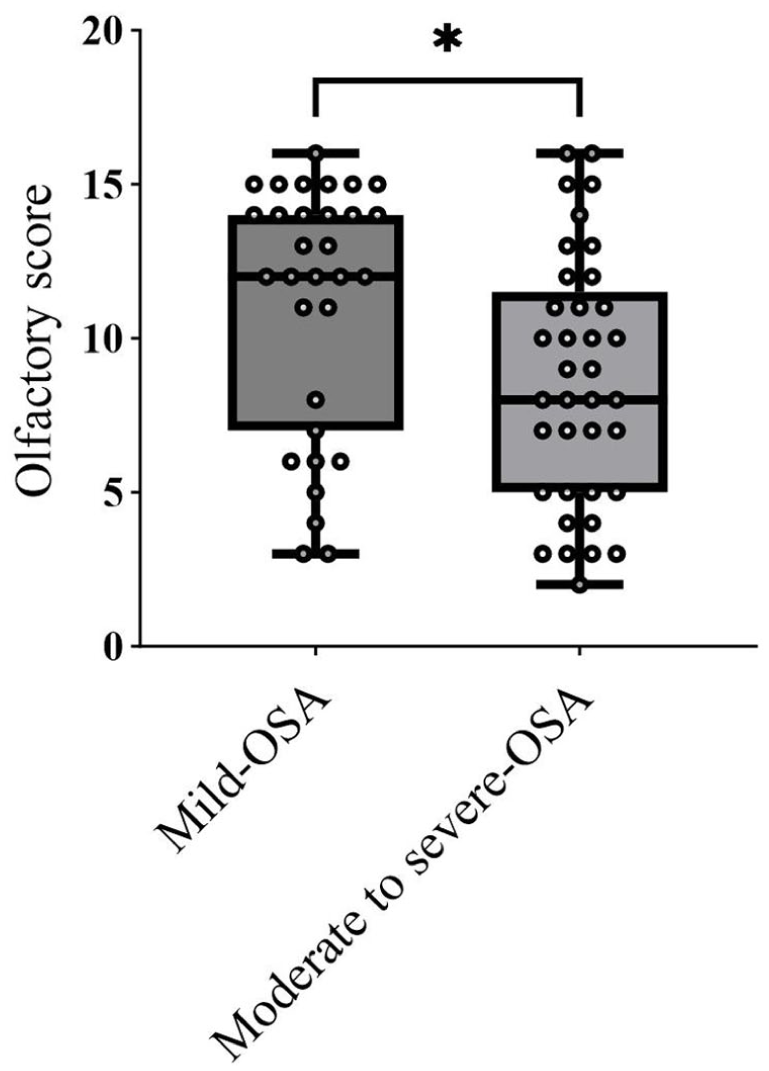
Box plot demonstrating the comparison of olfactory score differences between mild OSA group and the moderate-severe OSA group. OSA, obstructive sleep apnea.
Correlations Between Olfactory Scores and Sleep-Related Variables
The correlation of olfactory scores with sleep-related variables in the patient group is shown in Table 2. Spearman’s rank correlation coefficient was used to evaluate the relationships between olfactory scores and clinical sleep parameters. A negative correlation was detected between olfactory scores and AHI (
Correlations Between Clinical Data and Olfactory Scores in OSA Group.
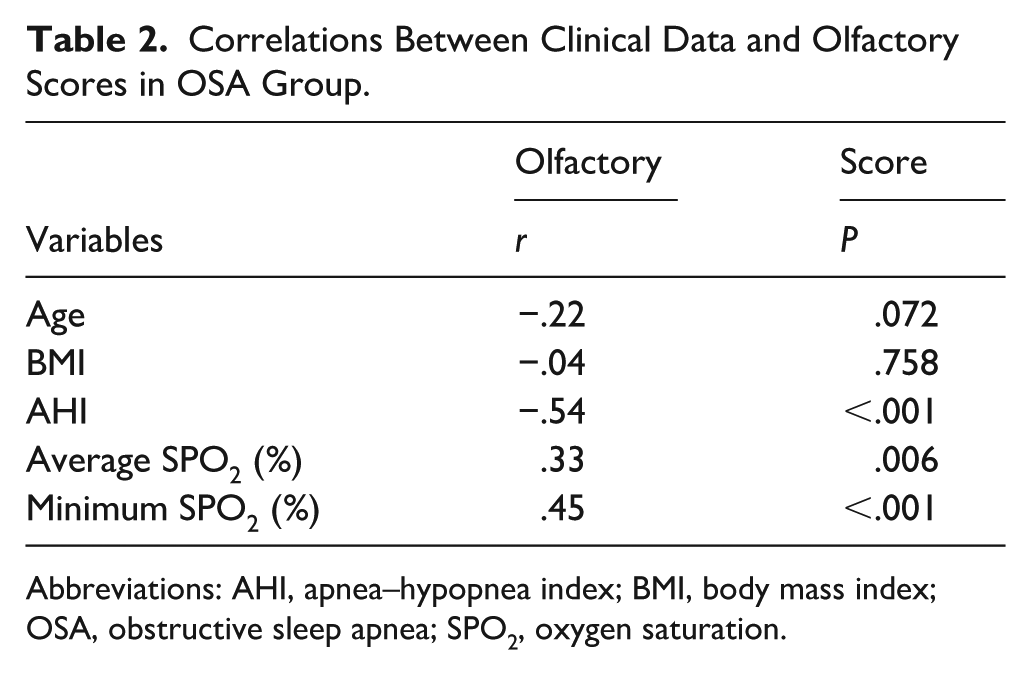
Abbreviations: AHI, apnea–hypopnea index; BMI, body mass index; OSA, obstructive sleep apnea; SPO2, oxygen saturation.
Univariate Linear Regression Analysis of Sleep Parameters and OF
To further examine these associations, univariate linear regression analyzes were performed with the olfactory score as the dependent variable and each sleep parameter as the independent variable. Analysis revealed a significant negative correlation between AHI and olfactory scores (r = −.54,
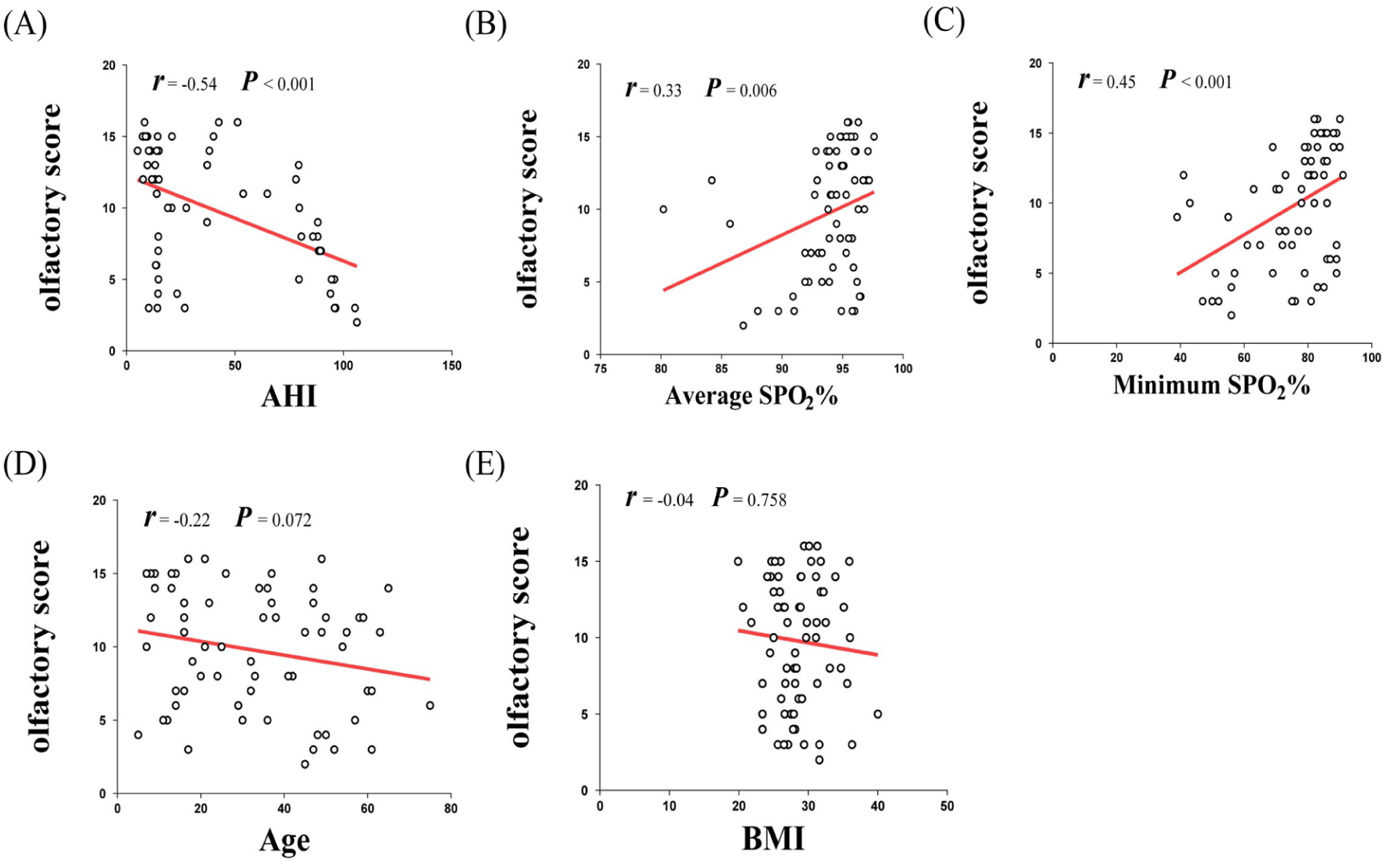
Scatter plots showing the associations between olfactory score and clinical parameters: (A) negative correlation with AHI (
Change in OF After 12 Months of CPAP Versus Non-CPAP Treatment
Table 3 presents the comparison of the main clinical characteristics, sleep characteristics, and OF test scores between the CPAP and non-CPAP treatment groups. After 12 months of CPAP treatment, the mean olfactory score was 13.87 ± 1.96 in patients treated with CPAP, which was significantly higher than 9.78 ± 4.217 in the untreated group. Characteristics such as smoking status, alcohol consumption, BMI, and sleep-related variables did not change significantly between CPAP and non-CPAP treatment groups after 12 months of treatment. Furthermore, a significant improvement in OF was observed after CPAP treatment (
Comparison of Baseline Variables Between CPAP Group and Non-CPAP Group.
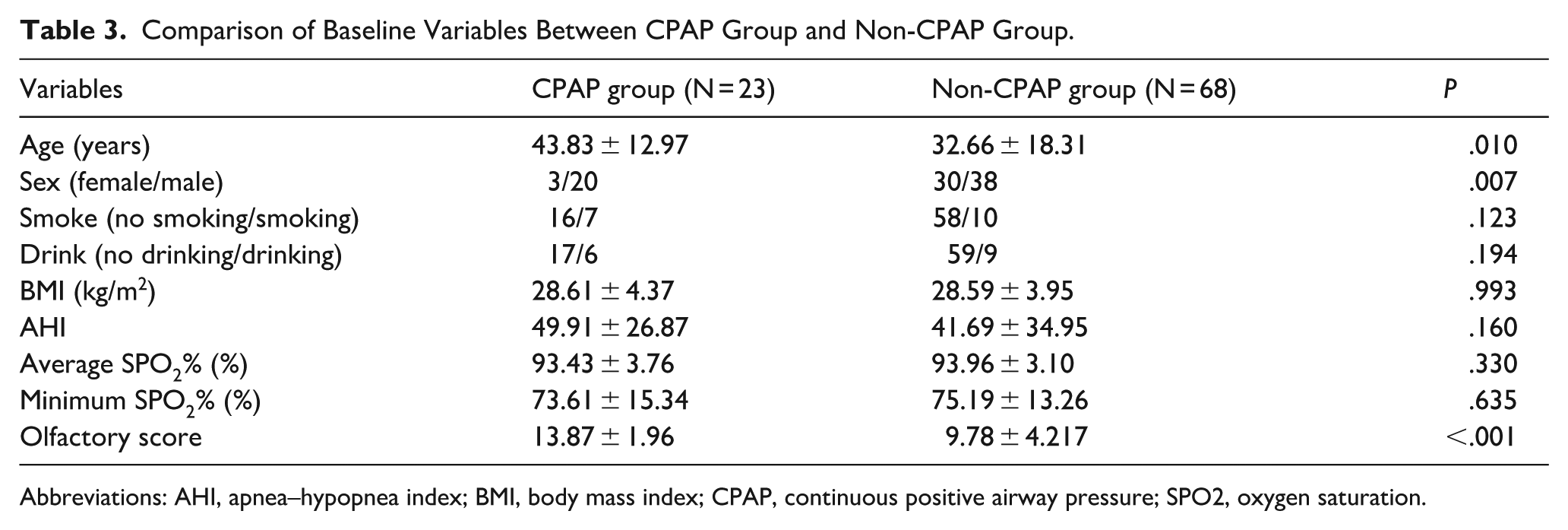
Abbreviations: AHI, apnea–hypopnea index; BMI, body mass index; CPAP, continuous positive airway pressure; SPO2, oxygen saturation.
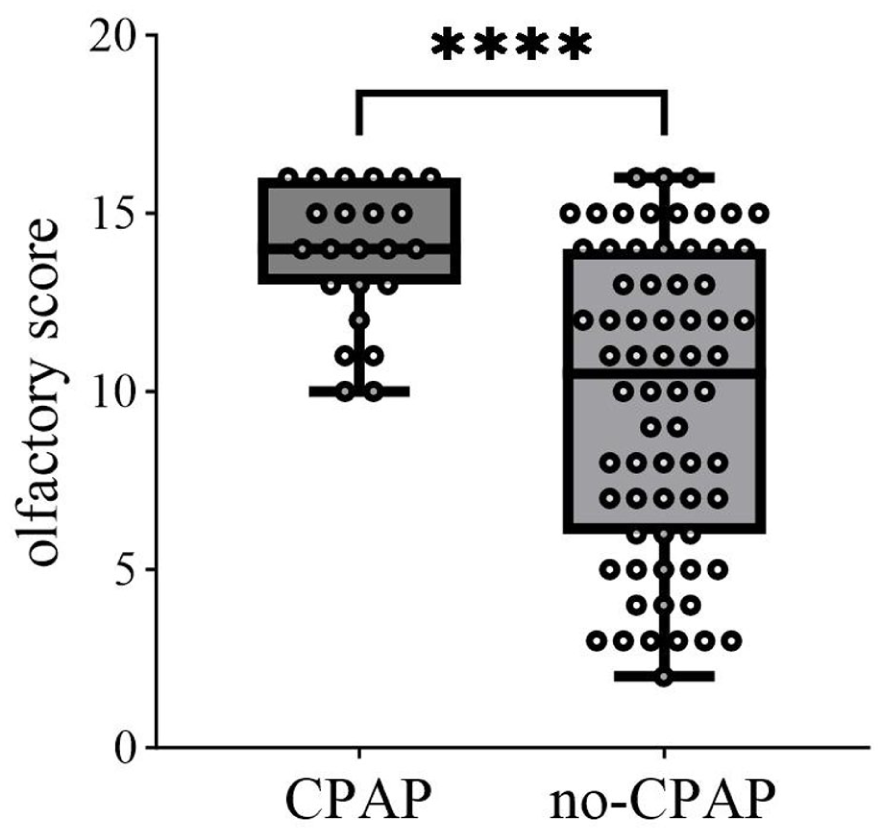
Box plot demonstrating the comparison of olfactory score differences between the CPAP group and the no-CPAP group. CPAP, continuous positive airway pressure.
Multivariate Analysis of Factors Associated with the Improvement of Olfactory Function
Multivariate logistic regression analysis was performed to identify baseline predictors of olfactory improvement after CPAP therapy. The results demonstrated that higher AHI (OR = 0.947, 95% CI: 0.902-0.995, P = .032), older age (OR = 1.056, 95% CI: 1.015-1.098, P = .007) and male sex (OR = 0.126, 95% CI: 0.022-0.720,
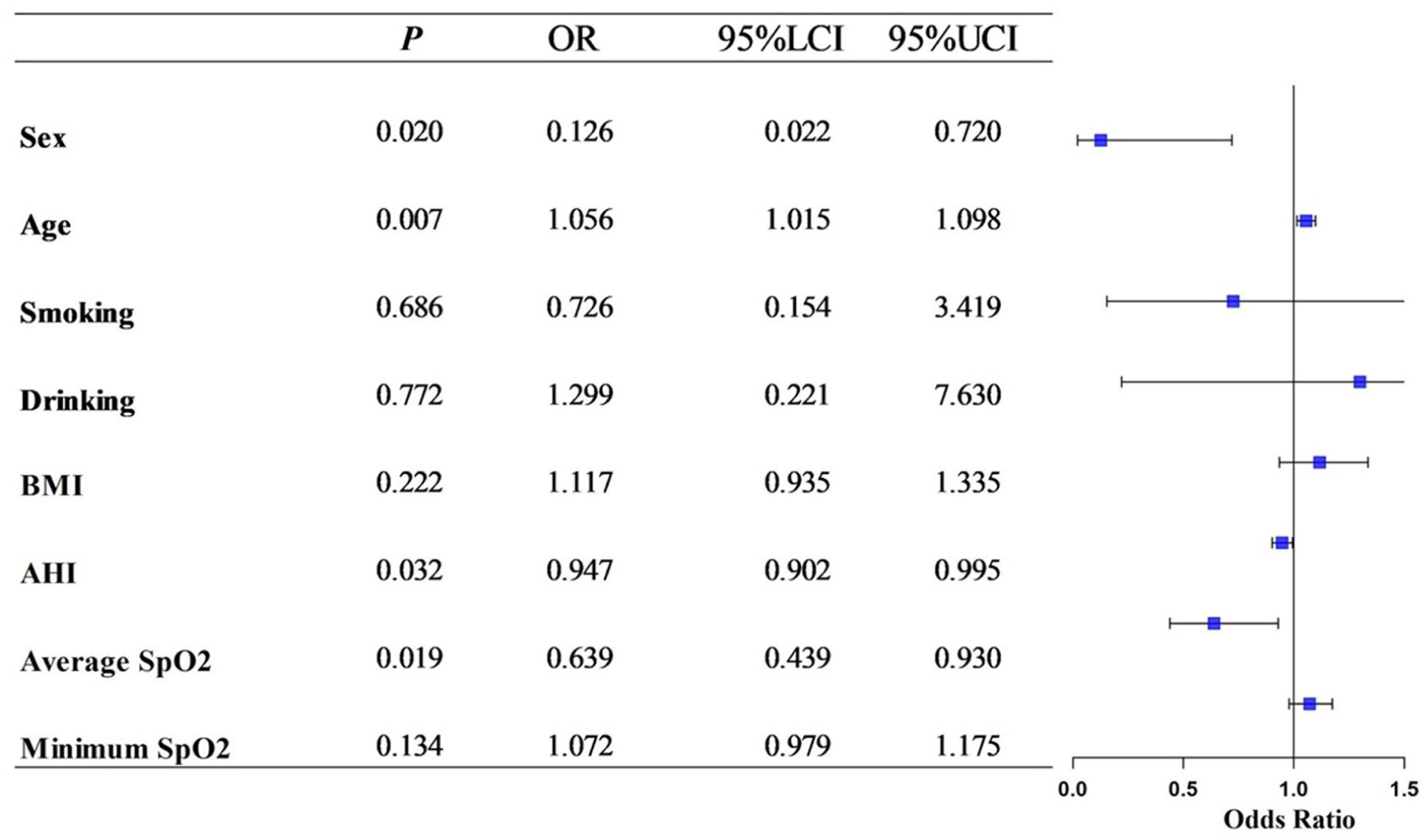
Forest plot of factors associated with olfactory improvement following CPAP therapy. OR with 95% CI were derived from multivariate logistic regression analysis. Variables included in the model were AHI, average SpO2%, minimum SpO2%, age, sex, BMI, smoking status, and alcohol consumption. AHI, older age, and male sex were significantly associated with reduced likelihood of olfactory improvement, whereas higher average SPO2% was a protective factor (
Discussion
OSA is a common sleep-related breathing disorder characterized by recurrent episodes of collapse of the upper airways during sleep, leading to apnea and hypopnea, which in turn results in intermittent hypoxia and sleep fragmentation.18 -20 OSA is particularly prevalent among middle-aged and older individuals and is strongly associated with a variety of systemic conditions, including hypertension, cardiovascular disease, metabolic syndrome, and cognitive impairment. 21
The pathogenesis of OSA is multifactorial, involving anatomical abnormalities of the upper airways, impaired neuromuscular control, and dysregulation of central respiratory control.22,23 As research into the systemic effects of OSA continues to advance, accumulating evidence suggests that OSA can also adversely affect sensory systems, particularly olfaction, which has recently garnered increasing attention.18,24
Olfaction is a critical sensory modality that plays a significant role in appetite regulation, emotional perception, and overall quality of life. 25 OD can be the result of a wide range of causes, including viral infections, nasal diseases, neurological disorders, and systemic illnesses. 6 In patients with OSA, OD may be associated with various mechanisms. Structural nasal abnormalities commonly associated with OSA, such as septal deviation, turbinate hypertrophy, and nasal polyps, can alter the dynamics of intranasal airflow, thus limiting the transport of odorant molecules to the olfactory epithelium. 26 In addition to these peripheral factors, chronic intermittent hypoxia (CIH), a hallmark feature of OSA, causes repeated cycles of hypoxia and reoxygenation. 27 This process increases the production of reactive oxygen species, leading to oxidative stress and cellular injury. 28 At the same time, CIH activates inflammatory signaling pathways, resulting in elevated levels of cytokines such as IL‑6, IL‑8, TNF‑α, and NF‑κB, which further damage olfactory sensory neurons.29,30 Furthermore, fragmentation of sleep and intermittent hypoxia can alter the central cholinergic system, producing abnormal activity in cholinergic neurons within olfactory pathways (Figure 1). 31 These combined effects contribute to impaired olfactory processing, supporting the view that hypoxia-related mechanisms play a key role in the pathogenesis of OD in OSA.
This study aims primarily to investigate whether OSA is associated with the onset of OD and whether 12 months of CPAP ameliorates OF in individuals with OSA. Current research reveals that the difference in olfactory scores between the mild OSA, moderate-to-severe OSA, and normal groups was statistically significant. Our findings are consistent with those reported in another recent report, which has demonstrated significant statistical differences in olfactory scores between control groups and patients with OSA, with lower olfactory scores observed in individuals with severe OSA. 32
In this study, correlation analysis indicated that olfactory scores were significantly associated with key indicators of OSA severity. Specifically, olfactory scores were negatively related to AHI and positively related to both average SpO2% and minimum SpO2% (Figure 5A-C). These findings imply that impaired OF may be related to the degree of nocturnal hypoxia. No significant correlations were found between olfactory scores and age or BMI (Figure 5D, E), indicating that these factors may not play a major role in OD in this population.
However, due to limitations such as limited sample sizes, measurement heterogeneity, and potential confounders, the possibility of underlying associations cannot be completely excluded. Therefore, a linear regression analysis was performed to determine the independent effects of these variables on OF.
As shown in Figure 5, a negative correlation was found between olfactory scores and AHI in this study. Furthermore, olfactory scores were positively linked to both average and minimum SpO2%. Similarly, previous research has also demonstrated significant negative correlations between AHI and olfactory scores. 33 Although BMI did not have significant effects on OF in the present study, a previous study observed a weak correlation between olfactory scores and BMI. 34 This indicates that OD can lead people to make unhealthy food choices, increasing their risk of obesity.
Cognitive deficits have been reported in those with OSA, including memory, attention, and sensory functions as well as olfaction.35,36 OD has been shown to be linked to central cholinergic dysfunction and decreased cognitive function. 37 A previous investigation suggests that intermittent hypoxia caused by OSA could be a major contributor in OD progression.38,39 In our study, OD was found to be associated with AHI, average SpO2%, and minimum SpO2%. A recent study also revealed that nocturnal hypoxemia can cause changes in cholinergic neurotransmission, and central cholinergic dysfunction can also lead to OD. 40 Furthermore, cholinergic neurotransmission in OSA may be altered in regions that are more vulnerable to intermittent hypoxia at night. 41 A previous study investigated the link between cholinergic dysfunction and OD. Their results indicated that cholinergic dysfunction was a strong predictor of hyposmia in individuals with OSA, and olfactory deterioration and cholinergic dysfunction could together determine the risk of impaired cognitive functions in individuals with OSA.41,42
Multiple clinical studies have shown that CPAP can improve respiratory and cognitive performance in OSA.43,44
In line with our findings, previous research has demonstrated a clear relationship between OSA severity and OD. A meta-analysis reported that patients with OSA exhibit impaired OF, which can be significantly improved with CPAP therapy. Specifically, CPAP treatment over 3 months resulted in significant enhancements in odor threshold, discrimination, and identification among moderate-to-severe OSA patients.45,46 Furthermore, these improvements were correlated with reductions in AHI, suggesting that intermittent hypoxia alleviation plays an important role in restoring olfactory performance. 46 However, CPAP may also cause nasal dryness, mucosal irritation, and nasal congestion which could modestly limit its positive effects on olfactory recovery in some patients.47,48 Although previous studies have shown that CPAP has a limited impact on nasal patency or nasal symptoms, these findings do not contradict our results. The improvement of OF observed in our study is more likely attributed to the correction of OSA-related hypoxemia, reduction of systemic inflammation, and improvement in sleep fragmentation than changes in nasal airflow or intranasal mucosal condition. 49
Furthermore, the negative associations we found between advanced age and male sex with olfactory improvement are consistent with established data on sensory physiology. Women generally outperform men in odor detection and identification tasks, while OF progressively decreases with increasing age in sexes.46,50 These demographic effects likely underlie the differential response to CPAP observed in our cohort.
In the multivariate logistic regression model, we identified several independent predictors associated with improved OF after CPAP therapy. Specifically, AHI was significantly associated with a lower probability of olfactory recovery, suggesting that greater severity of the disease may attenuate the therapeutic effect. 46 Similarly, older age and male sex were associated with a poorer response to treatment, while a higher average SpO2% appeared to be a protective factor. These findings indicate that the efficacy of CPAP in restoring OF is influenced by both baseline hypoxemia and individual patient characteristics.
The inverse relationship between AHI and treatment efficacy can be attributed to cumulative neural damage caused by intermittent chronic hypoxia and inflammation in patients with OSA.45,51 Beyond this, CPAP can improve OD in individuals with OSA through various interactions.45,52 The cholinergic neurotransmitter system participates in several cognitive functions and is vulnerable to cerebral hypoxemia. CPAP may relieve cognitive dysfunction in patients with OSA by normalizing the synthesis of neurotransmitters. 43 Elevated oxygen saturation has been shown to be beneficial for neurocognitive function, including olfaction.13,32 Our study found that 12-month CPAP treatment significantly elevated olfactory scores in individuals with OSA. Therefore, it can reasonably be inferred that CPAP can at least partially reverse OD in the OSA population. Moreover, structural airway improvements achieved with MAD therapy—particularly enhanced velopharyngeal airway space—have been associated with reduced severity of obstruction, suggesting that therapies targeting airway patency may indirectly support olfactory recovery. 53
The inverse relationship between AHI and treatment efficacy may also be related to age-related degenerative changes in the olfactory epithelium and hormonal differences between sexes, which may further contribute to the observed variability in treatment outcomes.
Several limitations in the current research should be acknowledged. First, the volume measurement of the olfactory bulb is lacking. Second, long-term follow-up is limited. Third, there is no control group for patients with untreated OSA due to ethical considerations. Moreover, baseline differences in age and gender between the CPAP and non-CPAP groups may introduce potential confounding effects. Although a multivariate logistic regression analysis was applied to adjust for these variables, the possibility of residual confounding cannot be fully excluded. Additionally, while CPAP adherence was monitored to ensure ≥4 hours per night, exact usage data was not collected, which may limit the precision of the observed olfactory improvements. Nevertheless, the consistent minimum usage supports the reliability of our findings. However, our study still offers valuable information on the relationship between OSA and OD, highlighting the potential efficacy of CPAP therapy in reversing OD. Nevertheless, the lack of objective neuroimaging, such as MRI-based olfactory bulb volumetry, and comprehensive neurocognitive assessment represents a limitation. These evaluations will be a focus of our future studies to further elucidate the neural mechanisms underlying OSA-related OD. By exploring the mechanisms that link sleep apnea-related hypoxia and OD, our findings may pave the way for further investigations into the cognitive and sensory consequences of OSA. These findings offer a basis for formulating more comprehensive therapeutic strategies aimed at improving both respiratory and sensory functions in this patient population. Furthermore, early detection and intervention for OD in patients with OSA are crucial in clinical practice.
Conclusions
This study underscores the relationship between OSA and OD, demonstrating that OD is a common issue in patients with OSA. Importantly, 12-month CPAP therapy can improve OD in these patients, suggesting that it is a potential therapeutic approach for sensory impairments related to OSA. Olfactory testing may also serve as a useful, noninvasive adjunct in the clinical assessment or screening of OSA patients. Future research should investigate the long-term effectiveness of CPAP and the underlying mechanisms behind its efficacy in improving OF.
Footnotes
Ethical Considerations
The Ethics Review Committee for Human Subject Biomedical Research, Shandong Provincial Hospital of Shandong First Medical University, approved our interviews (approval: SWYX:NO. 2025-347) on June 16, 2025 Respondents gave their written consent for review and signature before starting interviews.
Author Contributions
X.L. and J.L. were responsible for the conceptualization and design of this study. H.Y. conducted the experiments and collected the data. H.Y. and Z.W. analyzed and interpreted the data. H.Y. drafted the manuscript. X.L. supervised and managed the study. All authors approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Natural Science Foundation of China (82271152 and 82071013).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data supporting the findings of this study are available from the corresponding author on a reasonable request.
