Abstract
Chronic rhinosinusitis with nasal polyps (CRSwNP), particularly the type 2 endotype driven by IL-4/IL-13, remains challenging in a subset of patients refractory to surgery, allergen immunotherapy (AIT), and existing biologics. This case report describes a 29-year-old male with severe, multirefractory CRSwNP who achieved rapid and sustained remission with stapokibart (CM310), a novel anti-IL-4Rα monoclonal antibody, after failing 2 endoscopic sinus surgeries, omalizumab, and AIT. Following a regimen of 300 mg every 2 weeks, the patient exhibited significant olfactory improvement within 1 week and near-complete anosmia resolution by week 4. At 32 weeks, marked reductions in polyp burden, symptom scores (22-item Sinonasal Outcome Test (SNOT-22), Visual Analogue Scale (VAS), nasal congestion score (NCS), total symptom score (TSS)), and serum total IgE were documented. This case highlights stapokibart as a potent therapeutic option capable of inducing significant clinical response where conventional interventions have failed, supporting further investigation into IL-4Rα blockade for treatment-refractory CRSwNP.
Key Points
Stapokibart targets IL-4/IL-13-associated inflammatory mediators to reduce inflammatory burden in refractory CRSwNP.
Stapokibart achieved quick symptom relief with durable response in a severe uncontrolled CRSwNP patient.
Introduction
Chronic rhinosinusitis (CRS) affects 5% to 12% of the population and is characterized by persistent sinonasal inflammation leading to nasal obstruction, discharge, facial pain, and smell loss, resulting in substantial morbidity and impaired quality of life. 1 CRS is classified into CRS with nasal polyps (CRSwNP) and without (CRSsNP) based on endoscopic findings. The incidence of type 2 CRSwNP, which is eosinophilic-driven by cytokines including IL-4, IL-5, and IL-13, has increased in recent years. It is often associated with comorbidities such as asthma, allergic rhinitis (AR), and aspirin-exacerbated respiratory disease. Due to high recurrence rates after surgery, biologics have become promising alternatives, though targeted therapies remain underexplored.
We report a severe, treatment-refractory case of CRSwNP that did not respond to biologics, allergen immunotherapy (AIT), or endoscopic sinus surgery, but achieved rapid and sustained remission with stapokibart (CM310), a novel anti-IL-4Rα monoclonal antibody.
Case Presentation
A 29-year-old male with refractory CRSwNP and AR had a multiyear history of symptoms despite 2 sinus surgeries (2018 and 2020), omalizumab therapy, and unsuccessful AIT. He reported severe nasal obstruction, purulent discharge, and complete anosmia, greatly affecting his quality of life and work (Figure 1a). Pretreatment computed tomography (CT) showed bilateral opacification in all sinuses. Endoscopy confirmed recurrent polyps. Baseline scores were Visual Analogue Scale (VAS) 10, 22-item Sinonasal Outcome Test (SNOT-22) 56, nasal congestion score (NCS) 3, and total symptom score (TSS) 8. Sensitization to fungal and house dust mite allergens was detected.
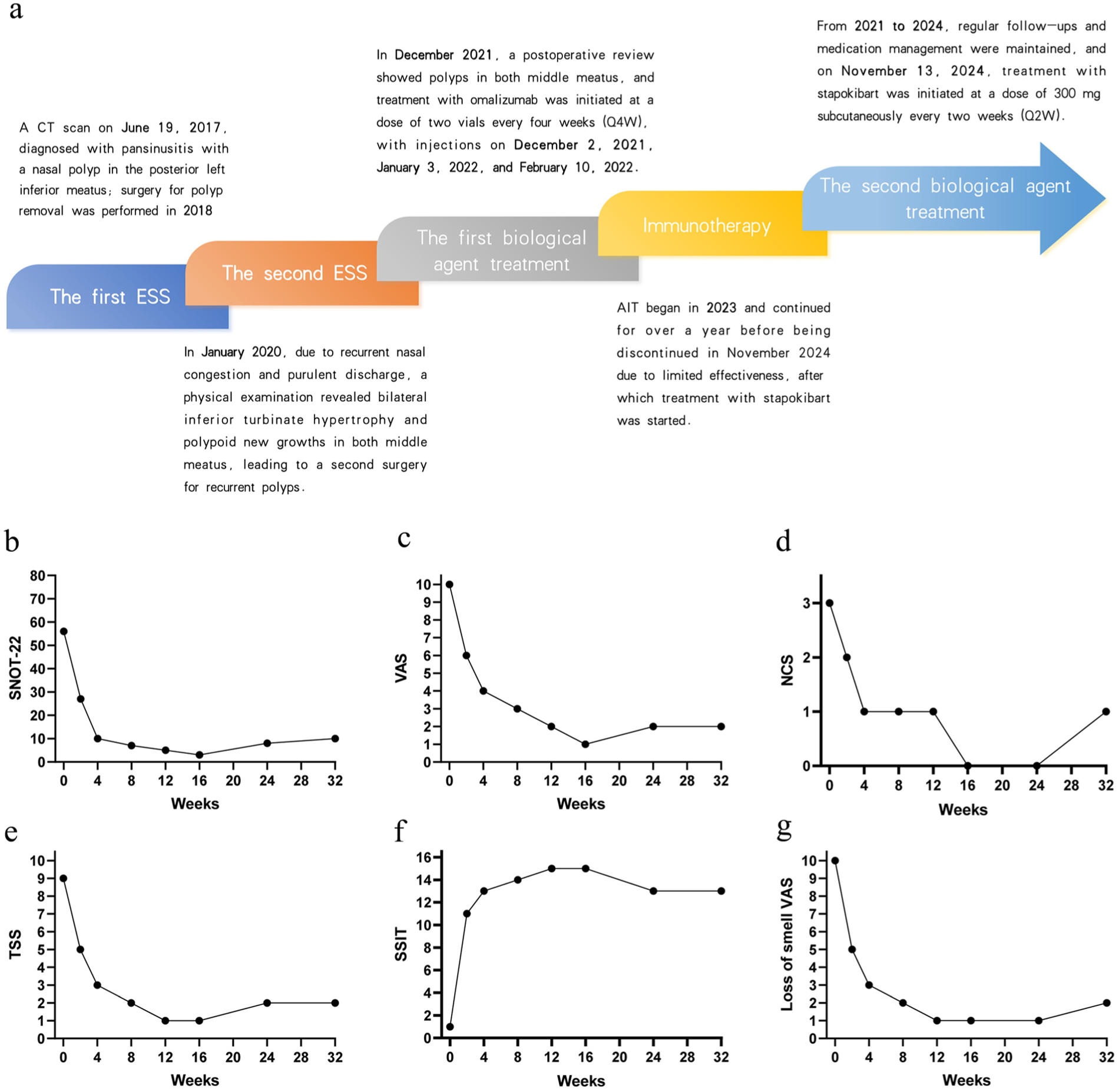
(a) Treatment timeline and therapeutic interventions for the patient. (b-g) Symptom scores before and after the administration of stapokibart comparative analysis of (b) SNOT-22, (c) VAS, (d) NCS, (e) TSS, (f) SSIT, and (g) loss of Smell VAS scores. SNOT-22, 22-item Sinonasal Outcome Test; VAS, Visual Analogue Scale; NCS, nasal congestion score; TSS, total symptom score; SSIT, sino-nasal symptom interview test.
Stapokibart was started at 300 mg loading dose, then 300 mg every 2 weeks for 4 months, alongside intranasal corticosteroids. Clinical monitoring included regular nasal endoscopies (Figure 2a), sinus CT imaging (Figure 2b), and serial symptom assessments using standardized metrics (VAS, SNOT-22, NCS, TSS). Olfaction improved within 1 week. At week 2, endoscopy showed >50% polyp reduction and partial smell return (3/5 identification). By week 4, anosmia nearly resolved (5/5). During follow-up at week 32, symptom scores (Figure 1b) improved markedly (SNOT-22: 10; VAS: 2; NCS: 1; TSS: 2; sino-nasal symptom interview test (SSIT): 13). Total IgE fell from 499 to 306 KU/L. Olfactory function remained stable in follow-up, with no obstruction recurrence. However, during a dermatology revisit for eczema flare-up, nasal polyps were identified in the nasal passages (Figure 2b). A consolidation dose of stapokibart was subsequently administered.
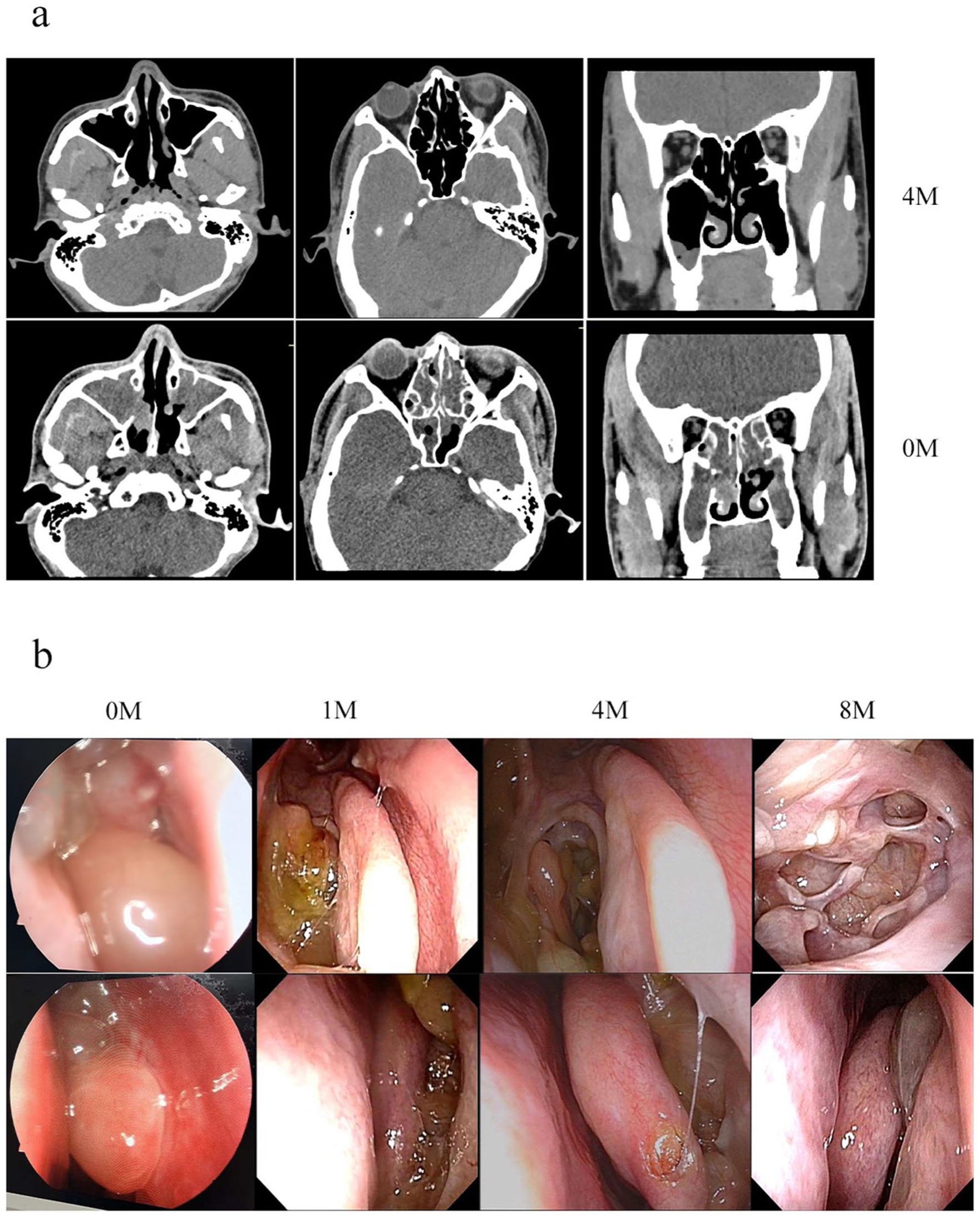
(a) Pretreatment (0M) CT scan of the paranasal sinuses revealed soft tissue opacities filling the bilateral maxillary, frontal, ethmoid, and sphenoid sinuses, with enlargement of the bilateral maxillary sinus ostia and similar abnormal densities occupying the middle and superior meati. Follow-up CT scan after 4 months of treatment demonstrated near-complete resolution of the soft tissue opacities in all 4 bilateral sinuses and improvement of the sinus mucosa. (b) Comparison of endoscopic findings before and after stapokibart therapy. (0M) Prior to treatment, nasal polyps were observed. At the 1-week follow-up after initiation of stapokibart, endoscopic examination showed reduction in polyp size. By week 4, there was significant disappearance of polyps within the meatus. At week 8, nearly complete resolution of polypoid lesions in the nasal cavity was achieved. CT, computed tomography.
The rapid and sustained response, including exceptional olfactory recovery, highlights stapokibart’s potential in refractory CRSwNP.
Discussion
The management of CRSwNP remains clinically challenging. First-line therapy typically includes intranasal corticosteroids and saline irrigation, with short courses of systemic corticosteroids reserved for severe exacerbations. Endoscopic sinus surgery is indicated for patients’ refractory to medical management, but long-term outcomes are often unsatisfactory, with high rates of recurrence, persistent olfactory dysfunction, and need for revision surgery.
Biologics targeting type 2 inflammation have emerged as promising therapeutic options.
Stapokibart (CM310) is a humanized monoclonal antibody that binds the interleukin-4 receptor alpha subunit (IL-4Rα), thereby blocking both IL-4 and IL-13 signaling pathways. It was approved in China for the treatment of CRSwNP in December 2024 and for seasonal AR in February 2025. 2 Clinical studies demonstrated that stapokibart significantly improves nasal and ocular symptoms, with a favorable safety profile.3 -5
Mechanistically, stapokibart differs from dupilumab, which also targets IL-4Rα. While dupilumab binds to the receptor and blocks both IL-4 and IL-13 signaling, stapokibart interacts with a distinct conformational epitope on IL-4Rα, located closer to IL-4’s natural binding site (M39A, S95A, L135A). 6 Notably, this unique epitope enables stapokibart to bind IL-4Rα across multiple species—including human, cynomolgus monkey, and rat—which facilitates direct preclinical evaluation in immunologically intact animal models, unlike dupilumab that primarily binds only to human and primate receptors. This cross-species reactivity may enhance the translational relevance of its preclinical profile. 6 In vitro studies show that stapokibart effectively prevents IL-4 and IL-13/IL-13Rα1 binding to IL-4Rα, with neutralizing activity comparable to or exceeding that of dupilumab. 7
In the present case, stapokibart induced a rapid and sustained clinical response, with particularly striking restoration of olfactory function in a patient with complete anosmia. Treatment also improved sinonasal symptoms, reduced polyp burden, and decreased biomarker levels. These findings suggest that stapokibart represents a novel and potent therapeutic option for patients with severe, refractory CRSwNP. Nevertheless, larger controlled studies are warranted to further define its efficacy, optimal dosing, and long-term safety profile.
Conclusion
Current evidence, though still limited, supports the use of biologics as a safe and effective intervention for CRSwNP refractory to conventional medical and surgical treatment. In this case, stapokibart led to significant improvement in sinonasal symptoms, restoration of olfactory function, and reduction in polyp burden. However, questions regarding the durability of response and optimal maintenance strategies remain. Definitive evaluation of both efficacy and safety will require multicenter, randomized controlled trials with extended follow-up periods.
Footnotes
Ethical Considerations
Ethical approval to report this case was obtained from the Ethics Committee of Union Hospital, Tongji Medical College, Huazhong University of Science and Technology (UHCT-IEC-SOP-016-03-01). The patient included in the study have signed informed consent, and the treatment methods and testing methods adopted in this study are known safe methods in clinical practice; the general information and clinical data are only used for study analysis, not for other purposes. Informed written consent was obtained from the patient for publication of this report and any accompanying images.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: work was financially supported by the Natural Science Foundation of Hubei Province (2024AFB615, ZLQ) and the National Natural Science Foundation of China (82201301, CH).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The corresponding author will provide access to relevant data upon reasonable request.
