Abstract
Objective:
This study aimed to identify key clinical predictors for obstructive sleep apnea (OSA) in pilots from routine aeromedical examination data and to assess the predictive value of the Psychomotor Vigilance Task (PVT).
Methods:
A retrospective 1:1 matched case–control study was conducted, including 37 pilots with polysomnography (PSG)-confirmed OSA and 37 matched non-OSA controls. Data from routine examinations, including anthropometric, biochemical, cardiovascular, and PVT parameters, were analyzed. The Least Absolute Shrinkage and Selection Operator (LASSO) regression was used to select significant predictors from a wide range of variables. These selected factors were then entered into a multivariable logistic regression model to determine their independent predictive value, calculated as odds ratios (ORs).
Results:
The OSA group had a significantly higher body mass index (BMI; 28.11 ± 2.72 vs 23.93 ± 1.89 kg/m2) and fasting plasma glucose (FPG; 5.29 ± 0.81 vs 4.82 ± 0.41 mmol/L) compared to controls (both P < .05). Conversely, no PVT metrics differed significantly between groups (P > .05). LASSO regression identified six key predictors: BMI, FPG, hyperuricemia, hyperlipidemia, systolic blood pressure (SBP), and high-density lipoprotein (HDL); no PVT parameters were selected by the model. Multivariable logistic regression confirmed BMI (OR = 3.43; 95% CI, 1.97-8.77; P < .001) and FPG (OR = 60.24; 95% CI, 3.45-5052.71; P < .05) as significant independent predictors of OSA.
Conclusion:
The core predictors for OSA in pilots are primarily indicators of metabolic syndrome, notably BMI and FPG. A framework based on the six physiological factors identified by LASSO regression provides a solid evidence-based foundation for developing an efficient and accurate OSA screening tool. In this study cohort, the PVT, as a measure of cognitive performance, demonstrated limited predictive value and is not recommended as a primary screening tool for OSA in pilots.
Introduction
Obstructive sleep apnea (OSA) is a disorder characterized by the recurrent collapse of the upper airway during sleep, leading to intermittent hypoxia and sleep fragmentation. OSA significantly impacts cardiovascular health, mental status, quality of life, and operational safety. 1 The resultant excessive daytime sleepiness and cognitive impairment pose a significant threat to aviation safety. Meta-analyses have confirmed that OSA significantly impairs psychomotor speed and executive function—cognitive domains critical for safe flight—thereby compromising a pilot’s ability to make complex decisions, particularly in abnormal or emergency situations.2,3 Furthermore, untreated OSA is a recognized risk factor for transportation safety, and the National Transportation Safety Board (NTSB) has emphasized that “flying fatigued is just as deadly as driving drunk,” highlighting the urgency of effectively identifying OSA in pilots.4 -6 Despite the direct threat OSA poses to aviation safety, current screening strategies for pilots face challenges. 7
Deficits in executive function can diminish a pilot’s capacity for complex decision-making and problem-solving, especially during abnormal or emergency scenarios. The link between untreated OSA and transportation accidents is well established. Research indicates that EDS in patients with untreated OSA is a major risk factor for motor vehicle accidents, particularly among males.4,5 This risk extends directly to aviation. Following a 2013 aviation accident, the U.S. National Transportation Safety Board (NTSB) issued a report emphasizing that “flying fatigued is just as deadly as driving fatigued.”7,8 Polysomnography (PSG), the diagnostic “gold standard,” is not suitable for large-scale, routine screening due to its high cost and complexity. On the other hand, commonly used screening tools, such as the Berlin Questionnaire (BQ) and the Stop-Bang questionnaire, while convenient, lack sufficient specificity. This can lead to a high rate of false positives, failing to meet the stringent accuracy requirements of aerospace medicine. 9 Therefore, developing a convenient, reliable, and precise OSA screening model based on data from routine pilot medical examinations is of significant clinical and practical importance. While risk factors such as obesity (measured by body mass index, BMI) and glucose metabolic abnormalities (measured by fasting plasma glucose, FPG) are well established, precisely identifying the core combination of predictors from a multitude of clinical variables is crucial for building an effective model. To address this, the present study utilizes the Least Absolute Shrinkage and Selection Operator (LASSO) regression analysis. This advanced machine learning method can automatically select the most critical predictive variables from high-dimensional data, overcoming the limitations of traditional methods. Additionally, given that OSA theoretically impairs vigilance, this study will also objectively evaluate the value of the psychomotor vigilance task (PVT) as a potential predictive tool.
In summary, this study aims to (1) identify a core set of physiological and biochemical predictors for OSA from pilot routine medical examination data using LASSO regression; and (2) assess the objective predictive value of the PVT in screening for OSA in pilots.
Methods
Study Participants
This study enrolled 74 male pilots who presented at the Civil Aviation Shanghai Hospital in 2024. To control for demographic confounders, a 1:1 matched case-control design was employed. Controls were matched to OSA patients strictly based on age (±2 years) and sex. Notably, body mass index (BMI) deliberately was not used as a matching criterion to preserve its potential value as a metabolic predictor in the subsequent analysis. All procedures were performed in compliance with the Declaration of Helsinki and were approved by the Institutional Review Board of the Civil Aviation Shanghai Hospital (Ref: 2022-07). Written informed consent was obtained from every participant before any study-related activities commenced.
Polysomnography
All participants underwent overnight PSG for a minimum of 7 hours, recording EEG, EOG, ECG, chin EMG, oral and nasal airflow, SpO2, and thoracoabdominal respiratory effort. Recordings were manually reviewed by qualified technicians. Apnea was defined as ≥90% airflow reduction for ≥10 seconds; hypopnea as ≥50% airflow reduction for ≥10 seconds with a ≥3% desaturation or arousal. Arousal was a ≥3 seconds EEG shift. The apnea-hypopnea Index (AHI) was calculated as total apnea and hypopnea events per hour of sleep. OSA severity was classified by AHI: mild (5 to <15), moderate (15 to <30), and severe (≥30 events/hour).10,11
Anthropometric and Biochemical Analyses
Anthropometric assessments were performed with participants in light indoor clothing and without shoes. Body weight was recorded using an electronic scale, and height was measured as the maximal distance from the feet to the top of the head while the subject stood erect. Body mass index (BMI) was calculated as weight in kilograms divided by the square of height in meters (kg/m²). Participants were categorized according to the Chinese criteria: normal weight (BMI < 25.0 kg/m²), overweight (25.0 ≤ BMI < 30.0 kg/m²), and obese (BMI ≥ 30.0 kg/m²). Systolic (SBP) and diastolic (DBP) blood pressure were measured from the right upper arm in a seated position. For biochemical analysis, blood samples were drawn after a minimum 8-hour fast to determine the levels of fasting plasma glucose (FPG), total cholesterol (TC), triglycerides (TG), high-density lipoprotein cholesterol (HDL), low-density lipoprotein cholesterol (LDL-C), serum creatinine (SCr), and uric acid (UA).
Psychomotor Vigilance Task
In this study, all participating pilots underwent psychomotor vigilance task (PVT) testing according to standardized instructions following the completion of their diagnostic PSG. PVT is a simple visual reaction time task requiring participants to respond as quickly as possible to randomly appearing visual stimuli over a sustained period (typically 10 minutes for the standard version). It is highly sensitive to sleep deprivation and fatigue, capable of detecting subtle fluctuations in vigilance induced by insufficient sleep. 12 As the task itself is straightforward, PVT performance is minimally influenced by intelligence, learning, or practice effects, making it an ideal tool for longitudinally tracking individual vigilance changes. 13 The PVT provides a rich array of performance metrics. In this study, the key metrics of interest included (1) Lapses of Attention: Defined as reaction times (RT) significantly prolonged (typically ≥ 500 ms), representing failures in sustained attention. (2) Response Speed (Mean 1/RT): Calculated as the mean reciprocal of valid RTs, reflecting optimal performance levels. (3) Slowest 10% RTs: Focusing on the poorest responses, this metric is particularly sensitive to instabilities in vigilance. 4)False Starts: Defined as button presses without stimulus presentation or with RTs < 100 ms, indicating impulsivity or anticipatory responses. 14
Data Analysis
All statistical analyses were conducted using SPSS Statistics (Version 26.0, IBM Corp., Armonk, NY, USA). For comparisons of baseline characteristics between the OSA and control groups, continuous variables were analyzed using either the Welch’s two-sample t-test or the Wilcoxon rank-sum test, depending on the data distribution. Categorical variables were compared using Fisher’s exact test. To select the most predictive combination of features from the candidate variables, a LASSO (Least Absolute Shrinkage and Selection Operator) regression analysis was employed. LASSO regression was performed using R statistical software (v4.3.0) with the glmnet package. A total of 26 baseline variables were entered into the model. Continuous variables were standardized (z-scored) prior to analysis to ensure scale invariance, while categorical variables were dichotomized (dummy coded). The optimal penalty parameter (λ) was selected via 10-fold cross-validation. Subsequently, the key variables identified by LASSO were entered into a multivariable binary logistic regression model to calculate the odds ratios (ORs) and their 95% confidence intervals (95% CIs) for independent risk factors. All statistical tests were two-sided, and a P-value < .05 was considered statistically significant.
Results
Baseline Clinical Characteristics
Baseline demographic and clinical characteristics of the matched cohort are detailed in Table 1 and Supplemental Table S1. While age was well-balanced between groups (P > .05), BMI was significantly higher in the OSA group (P < .001), reflecting the deliberate decision to exclude BMI from matching criteria. Compared to healthy controls, pilots in the OSA group had significantly higher FPG (5.29 ± 0.81 vs 4.82 ± 0.41 mmol/L, P = .001), and prevalence of fatty liver. Regarding cardiovascular markers, the OSA group exhibited significantly lower HDL levels (1.12 ± 0.22 vs 1.30 ± 0.27 mmol/L, P = .003). Systolic blood pressure (SBP) and diastolic blood pressure (DBP) showed statistical differences; however, the values for both groups remained within the normal physiological range, suggesting limited clinical significance compared to metabolic indicators. No significant between-group differences were found for TC, TG, LDL, UA, or the prevalence of hyperuricemia and hyperlipidemia (all P > .05). In terms of PVT performance, although the mean RT, slowest 10% RTs, and number of lapses were numerically higher in the OSA group, these differences did not reach statistical significance (P = .178, P = .211, and P = .461, respectively), as shown in Table 2.
Baseline Characteristics After Matching.
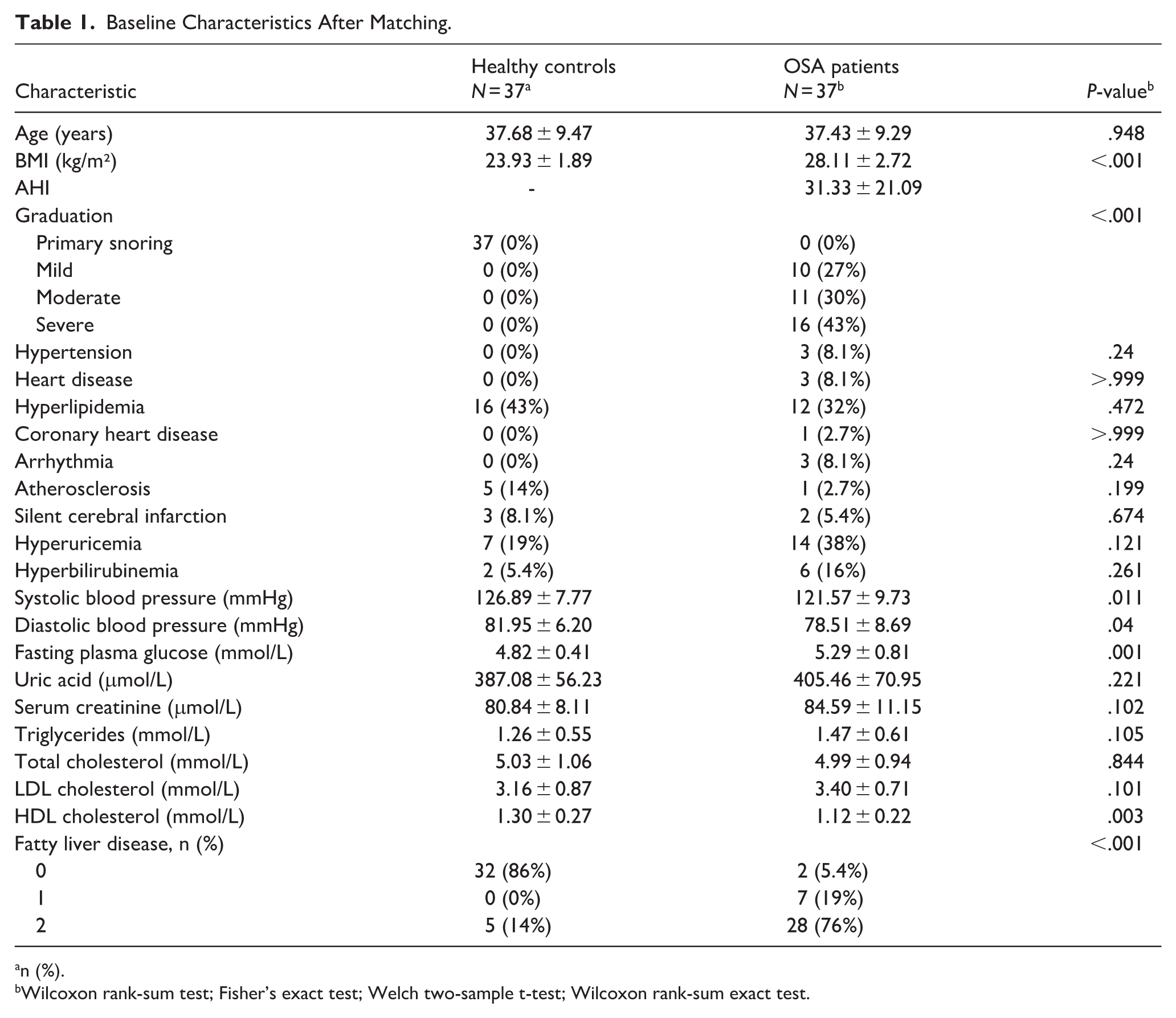
n (%).
Wilcoxon rank-sum test; Fisher’s exact test; Welch two-sample t-test; Wilcoxon rank-sum exact test.
Baseline Characteristics of PVT Parameters in the Two Study Groups.
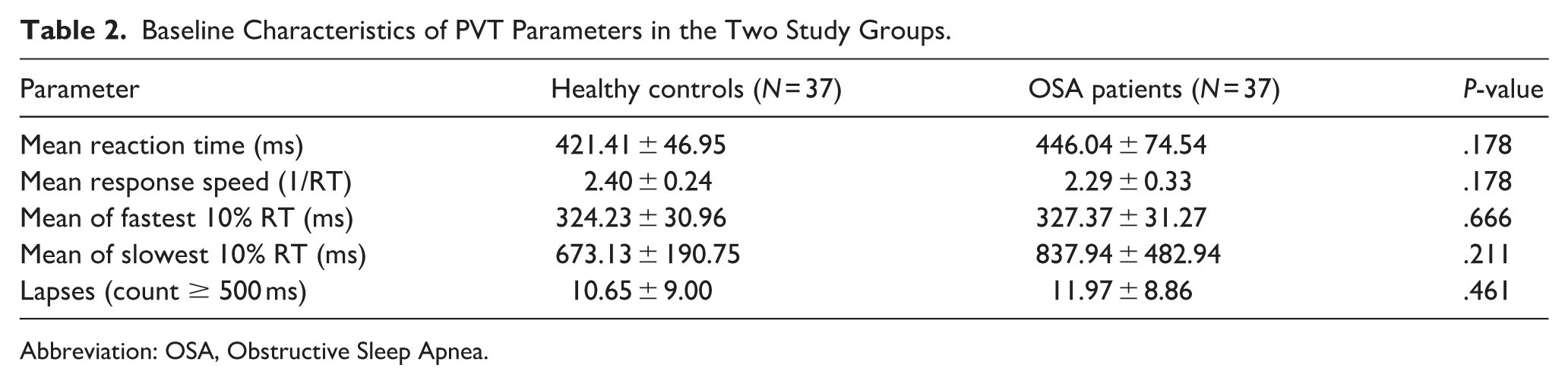
Abbreviation: OSA, Obstructive Sleep Apnea.
Feature Selection by LASSO Regression
All baseline physiological and biochemical variables were included in the LASSO regression model. Through 10-fold cross-validation, the algorithm selected 6 features with the highest predictive value for OSA: BMI, FPG, hyperuricemia, hyperlipidemia, SBP, and HDL. Notably, during the optimization process, the LASSO algorithm shrunk the regression coefficients of all PVT-related metrics to zero, indicating that these cognitive indicators were not key predictors within this dataset Multicollinearity analysis confirmed that the variables in the final model were independent. All variance inflation factors (VIF) were well below the threshold of 5 (eg, FPG VIF = 2.20, BMI VIF = 2.83), as shown in Supplemental Table S2.
Independent Risk Factors for OSA
The 6 variables selected by LASSO were subsequently entered into a multivariable logistic regression model to identify independent risk factors while adjusting for confounding effects. The predictive performance of the final model was excellent, as illustrated by the ROC curve in Figure 1 (AUC = 0.961, 95% CI: 0.917-0.993). The results (Figure 2) revealed that after mutual adjustment, BMI (OR = 3.43, 95% CI: 1.97-8.77, P < .001) and FPG (OR = 60.24, 95% CI: 3.45-5052.71, P < .05) were strong independent risk factors for OSA in pilots. Although the 95% CI for FPG was wide, suggesting that the precision of the effect estimate may be limited by sample size, its statistical significance and the sheer magnitude of the OR strongly indicate that elevated FPG is associated with a dramatically increased risk of OSA. The other variables in the model—HDL, SBP, hyperuricemia, and hyperlipidemia—did not demonstrate independent statistical significance (all P > .05).
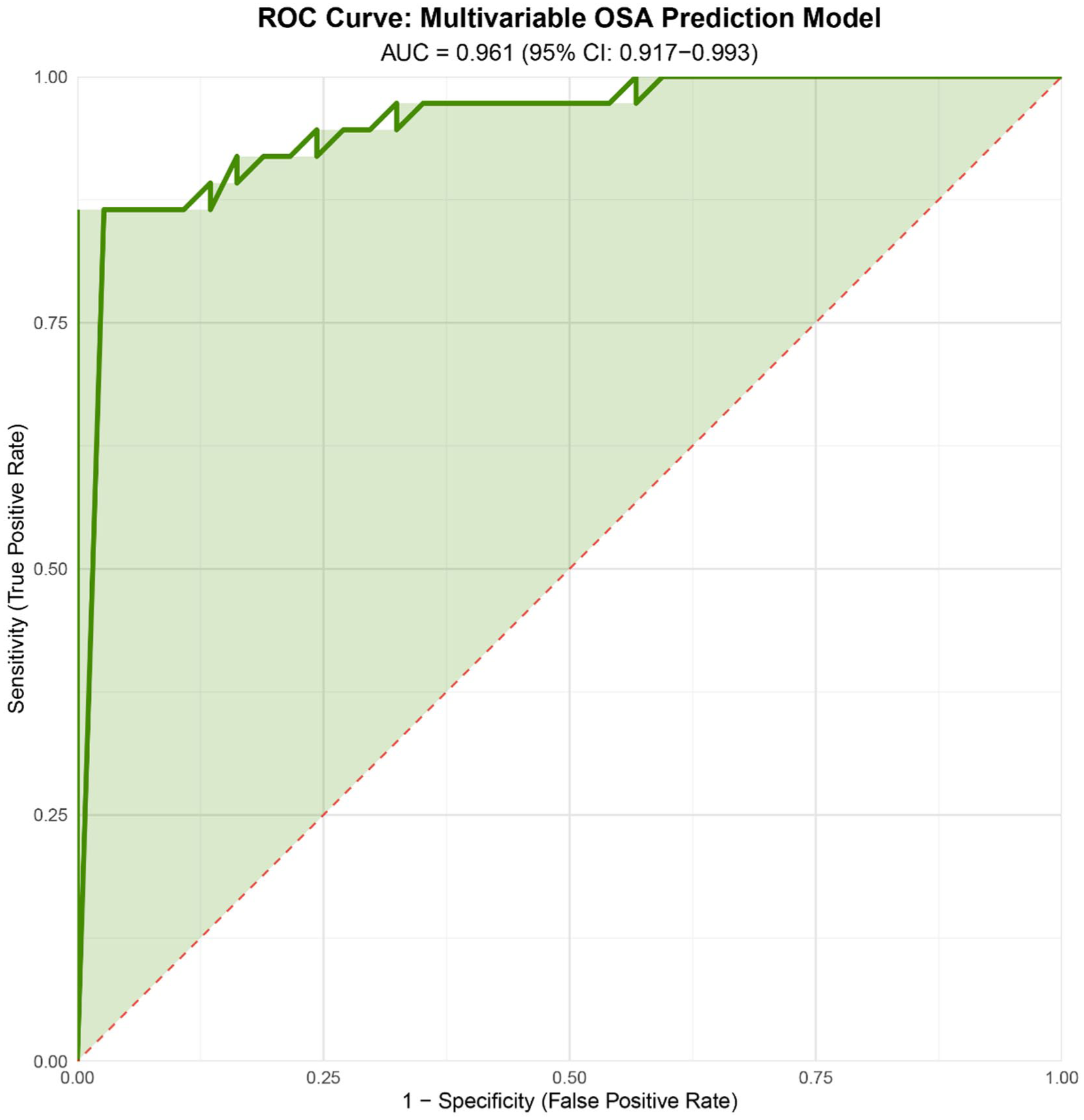
ROC curve of the final multivariable prediction model. The model, incorporating BMI and FPG, achieved an AUC of 0.961 (95% CI: 0.917-0.993). This demonstrates the excellent diagnostic accuracy of the metabolic-based model in distinguishing OSA patients from healthy controls. The red dashed line represents the line of no discrimination (AUC = 0.50). ROC, receiver operating characteristic; BMI, body Mass Index; FPG, fasting plasma glucose; AUC, area under the curve; OSA, obstructive sleep apnea.
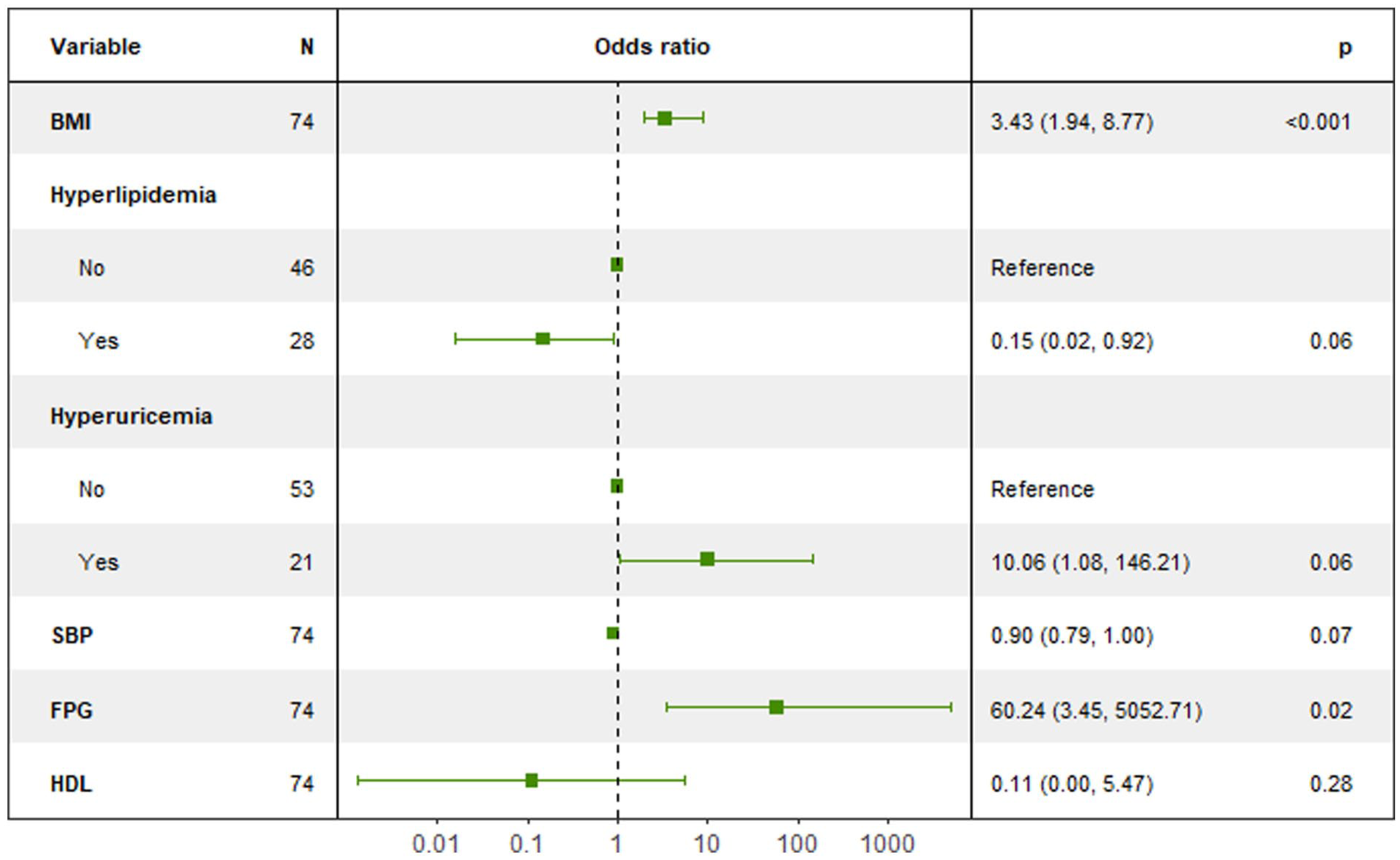
Forest plot of multivariate logistic regression analysis for risk factors of OSA in pilots. The squares represent the point estimates of the odds ratios (OR), and the horizontal lines indicate their 95% confidence intervals (CI). The vertical dashed line at an OR of 1 is the line of no effect. A predictor was considered statistically significant and independent if its 95% CI did not cross this line. OSA, obstructive sleep apnea.
Discussion
This study employed a rigorously matched case–control design and a LASSO machine-learning algorithm to identify core predictors of OSA within a specific population of pilots. The principal finding is that body mass index (BMI) and FPG are the most robust and significant independent predictors of OSA. Conversely, PVT, an objective metric of sustained attention, demonstrated negligible predictive value for screening in this context.
Our multivariable logistic regression model confirmed the strong independent predictive effects of BMI and FPG. Notably, FPG exhibited a remarkably high odds ratio (OR = 60.24). While this magnitude suggests a mathematical phenomenon known as “quasi-complete separation”—attributable to the distinct metabolic profiles between the case and control groups in this sample size—the directionality and statistical significance of the association are robust. This indicates that in a highly screened occupational group such as pilots, even minor dysregulation of glucose homeostasis serves as a potent “red flag” for OSA. Similarly, BMI (OR = 3.43) was identified as a critical risk factor, consistent with established literature regarding the anatomical burden of adiposity on the upper airway.
The link between these metabolic markers and OSA is likely bidirectional and self-perpetuating. Anatomically, central obesity promotes fat deposition in the parapharyngeal pads and tongue base, directly increasing upper airway collapsibility. 15 Metabolically, the intermittent hypoxia and sleep fragmentation characteristic of OSA activate the sympathetic nervous system and systemic inflammation, 16 which can induce insulin resistance and glucose dysregulation. 17 Unlike general populations, active-duty pilots are subject to strict medical standards; therefore, the emergence of these metabolic deviations, however slight, appears to be highly specific for underlying sleep pathology in this cohort.
A key contribution of this study is the objective assessment of the PVT's role. Contrary to the hypothesis that OSA-induced sleep fragmentation would impair vigilance, PVT metrics failed to effectively differentiate OSA patients from controls. This was objectively confirmed by the LASSO regression analysis, which automatically excluded all PVT-related metrics (including mean reaction time and lapses) from the final model during the regularization process. We propose two explanations for this lack of sensitivity. First, pilots are a highly selected professional group with significant cognitive reserve, potentially masking performance deficits. Second, PVT performance is susceptible to unmeasured confounders such as circadian rhythm phases and cumulative fatigue, which were not strictly standardized in this retrospective setting. However, we do not dismiss the utility of PVT entirely. The exclusion of PVT in our model may partly be attributed to the limited sample size (n = 74), which restricts the statistical power to detect subtle cognitive deficits (type II error). While metabolic markers showed a dominant signal in this cohort, vigilance deficits remain theoretically linked to sleep fragmentation. Future studies with larger, multicenter cohorts might still validate PVT’s predictive potential, especially under strictly controlled experimental conditions.
To contextualize our findings, we compared the predictive performance of our LASSO-derived metabolic model against traditional screening questionnaires reported in the literature. Previous studies assessing tools such as STOP-BANG or NoSAS in aviation or similar occupational cohorts typically report area under the curve (AUC) values ranging from 0.60 to 0.75. 9 These subjective tools often suffer from limited specificity and the “healthy worker effect,” where pilots may underreport symptoms (eg, loud snoring or tiredness) due to fear of grounding. In stark contrast, our model, relying entirely on objective biological markers (BMI and FPG), achieved an AUC of 0.961. This suggests that in professional cohorts where subjective reporting may be unreliable, objective metabolic profiling provides a significantly more accurate method for OSA risk stratification.
These findings have direct implications for aviation medical protocols. Current screening often relies on self-reported symptoms or generic physical findings. Our data suggest a paradigm shift toward precision screening. Given that PVT and subjective questionnaires may lack sensitivity in this context, Aviation Medical Examiners (AMEs) should prioritize the “metabolic phenotype” (BMI and FPG) during routine class I examinations. We recommend that pilots exceeding specific metabolic thresholds be automatically flagged for polysomnography, regardless of their self-reported sleepiness. This objective, data-driven approach would minimize reliance on subjective reporting and enhance the early identification of high-risk individuals, thereby improving flight safety.
Limitations
This study has several limitations. First, the sample size (n = 74), while sufficient to identify strong predictors, limited the stability of coefficient estimates for the FPG variable. Second, the retrospective design precluded the inference of causality and the strict control of PVT testing conditions. Finally, the absence of an independent external validation cohort means the generalizability of our model requires further confirmation in larger, multicenter pilot populations.
Conclusion
In conclusion, this study identifies indicators of metabolic syndrome as the core predictors for Obstructive Sleep Apnea (OSA) in pilots. While an initial six-factor model was derived from LASSO regression, subsequent multivariable logistic regression revealed that only body mass index (BMI) and fasting plasma glucose (FPG) were robust, independent risk factors. The other candidate variables—hyperuricemia, hyperlipidemia, systolic blood pressure, and HDL—did not retain statistical significance in the final adjusted model. Furthermore, the Psychomotor Vigilance Task (PVT) demonstrated limited predictive utility and is not recommended as a primary screening metric for OSA in this cohort. These findings provide a targeted, evidence-based framework for developing an efficient and objective OSA risk assessment tool for aviation personnel.
Supplemental Material
sj-pdf-1-ear-10.1177_01455613251413546 – Supplemental material for Metabolic and Anthropometric Factors Outperform Psychomotor Vigilance Task in Predicting Obstructive Sleep Apnea in Pilots: A LASSO-Based Analysis
Supplemental material, sj-pdf-1-ear-10.1177_01455613251413546 for Metabolic and Anthropometric Factors Outperform Psychomotor Vigilance Task in Predicting Obstructive Sleep Apnea in Pilots: A LASSO-Based Analysis by Yan Shao, Ziying Song, Li Fu, Lin Liu, Yunan Chen, Lanhui Zhou and Jingjing Huang in Ear, Nose & Throat Journal
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Civil Aviation Safety Capacity Building Project (Grant No. 268).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
