Abstract
Airway involvement in epidermolysis bullosa (EB) is rare and most commonly associated with junctional EB. Laryngotracheal disease in epidermolysis bullosa simplex (EBS), particularly PLEC (Plectin gene)-related variants, is uncommon but may cause significant morbidity. We report a 7-year-old girl with autosomal recessive PLEC-related EBS who developed progressive supraglottic and glottic stenosis. She presented at 8 months old with severe respiratory distress requiring emergent tracheostomy. Serial endoscopies revealed persistent supraglottic granulation, an anterior glottic web, posterior glottic stenosis, and occasional bullae near the carina, with a patent subglottis and distal trachea. She underwent multiple direct laryngobronchoscopies for cold web release, dilation, mitomycin-C application, Kenacort injection, and excision of suprastomal granulation. Despite clinical stabilization, she remains tracheostomy-dependent with ongoing evaluation for potential decannulation. This case highlights the potential for significant airway disease in PLEC-related EBS, and underscores the importance of early recognition, gentle airway handling, and multidisciplinary management.
Introduction
Epidermolysis bullosa (EB) comprises a heterogeneous group of inherited blistering disorders characterized by marked epithelial fragility, where minimal friction results in blistering, erosions, and scarring. The estimated prevalence in the United States is approximately 11 per million people, with disease severity ranging from mild cutaneous involvement to life-limiting multisystem manifestations. 1
EB is classified according to the level of tissue cleavage into 4 major types: epidermolysis bullosa simplex (EBS), junctional EB (JEB), dystrophic EB (DEB), and Kindler EB. 2 EBS—the most common subtype—typically exhibits intraepidermal blistering and is most often inherited in an autosomal dominant pattern, though autosomal recessive forms associated with PLEC (Plectin gene) mutations are increasingly recognized. These variants may present with extracutaneous features, including muscular dystrophy, cardiomyopathy, or airway involvement. 3
Upper airway manifestations in EB are uncommon but potentially life-threatening. Reported abnormalities include laryngeal edema, supraglottic scarring, glottic webs, subglottic stenosis, and tracheal blistering, with JEB carrying the highest cumulative risk of progressive airway obstruction. 3 Nevertheless, airway disease has also been described in severe EBS, particularly in patients with PLEC-related phenotypes (previously classified as a subtype of JEB). 3 Airway involvement may lead to chronic stridor, dysphonia, recurrent respiratory distress, and the need for long-term tracheostomy. 4
Given the scarcity of literature on EBS with significant laryngotracheal disease, optimal airway management remains challenging and requires multidisciplinary, individualized strategies. Here, we report a rare case of a child with autosomal recessive PLEC-related EBS who developed progressive supraglottic and glottic stenosis requiring long-term tracheostomy. We additionally review the existing literature to highlight key considerations in the evaluation and management of airway involvement in EB.
Case Presentation
We report the case of a 7-year-old girl with genetically confirmed autosomal recessive PLEC-related EBS who has been tracheostomy-dependent since infancy due to progressive supraglottic and glottic stenosis. Patients’ legal guardian has provided informed consent to publish this case report.
Neonatal Course
She was born at full term via spontaneous vaginal delivery. Soon after birth, she developed respiratory distress requiring intubation for 3 days and was noted to have widespread skin blistering. A clinical diagnosis of EB was made at an outside facility. At 3 months of age, she was readmitted with respiratory distress associated with a viral infection, complicated by cardiorespiratory arrest requiring resuscitation, intubation, and mechanical ventilation for 5 days. Following extubation, she continued to experience persistent noisy breathing, exertional stridor, and a weak cry, though feeding was normal.
Hospital Course
At 8 months of age, she presented to our emergency department in severe respiratory distress with oxygen saturation of 60%, suprasternal retractions, and tachycardia. She was afebrile and hemodynamically stable, but her weight was below the 10th percentile. Dermatologic examination revealed tense bullae, erosions, and hemorrhagic crusts over the face and extremities, with diffuse subungual hyperkeratosis.
Flexible nasolaryngoscopy suggested glottic edema and limited vocal-fold mobility. Operative direct laryngobronchoscopy (DLB) demonstrated significant supraglottic granulation tissue, adhesions causing stenosis, and glottic edema. The subglottis and distal airway were patent. Due to impending airway failure, an emergent tracheostomy was performed using a size 3 neonatal tube. A multidisciplinary team initiated comprehensive management and reinforced non-adhesive skin care.
Systemic evaluation identified hepatomegaly, central hypotonia, global developmental delay, and failure to thrive. Cardiac evaluation excluded cardiomyopathy, and neuromuscular assessment ruled out muscular dystrophy. Laboratory tests, including hematologic, renal, and liver function panels, were normal. Pulmonary function testing later demonstrated fixed extrathoracic obstruction.
Genetics and Family History
The patient was born to first cousins, second-degree consanguineous parents with a strong family history of EB. Both families reported multiple affected relatives, though none had known airway disease. The patient had a younger sibling with EB who subsequently died at 1 month of age.
Genetic testing revealed a homozygous pathogenic PLEC variant (c.4552C>T p.(Gln1518*)) consistent with autosomal recessive EBS. She was also heterozygous for pathogenic variants in ACADM and MYO7A, indicating carrier status for medium-chain acyl-CoA dehydrogenase deficiency and Usher syndrome, respectively.
Subsequent Airway Course
Over the next several years, she experienced intermittent episodes of cyanosis attributed to tracheostomy-tube obstruction by bullae and crusts, each resolving with tube changes. She underwent multiple scheduled DLBs for routine tube exchanges and airway assessment. Intubation was consistently easy with a Cormack–Lehane grade I view, though obtaining intravenous access was always difficult and required silicone-based dressings to avoid skin trauma.
DLB findings remained notable for persistent supraglottic granulation and edema, immobile edematous vocal folds, and occasional bullae near the carina. The subglottis and trachea remained structurally patent.
Airway Interventions
By age 4, worsening airway symptoms and recurrent infections prompted repeat DLB, which revealed posterior glottic stenosis, an anterior glottic web, and reduced vocal-fold mobility (Figure 1a). A focal anterior tracheal ulcer likely caused by tube pressure was also noted. Her tracheostomy tube was upsized to a size 4 pediatric tube to bypass the ulcer. The glottic web was sharply released, and mitomycin-C was applied to reduce restenosis risk. A second-look DLB 2 weeks later showed expected postoperative edema and granulation with a slit-like glottic opening (Figure 1b). Clinically, she improved and tolerated daytime capping despite frequent tube obstruction by bullae and crusts.
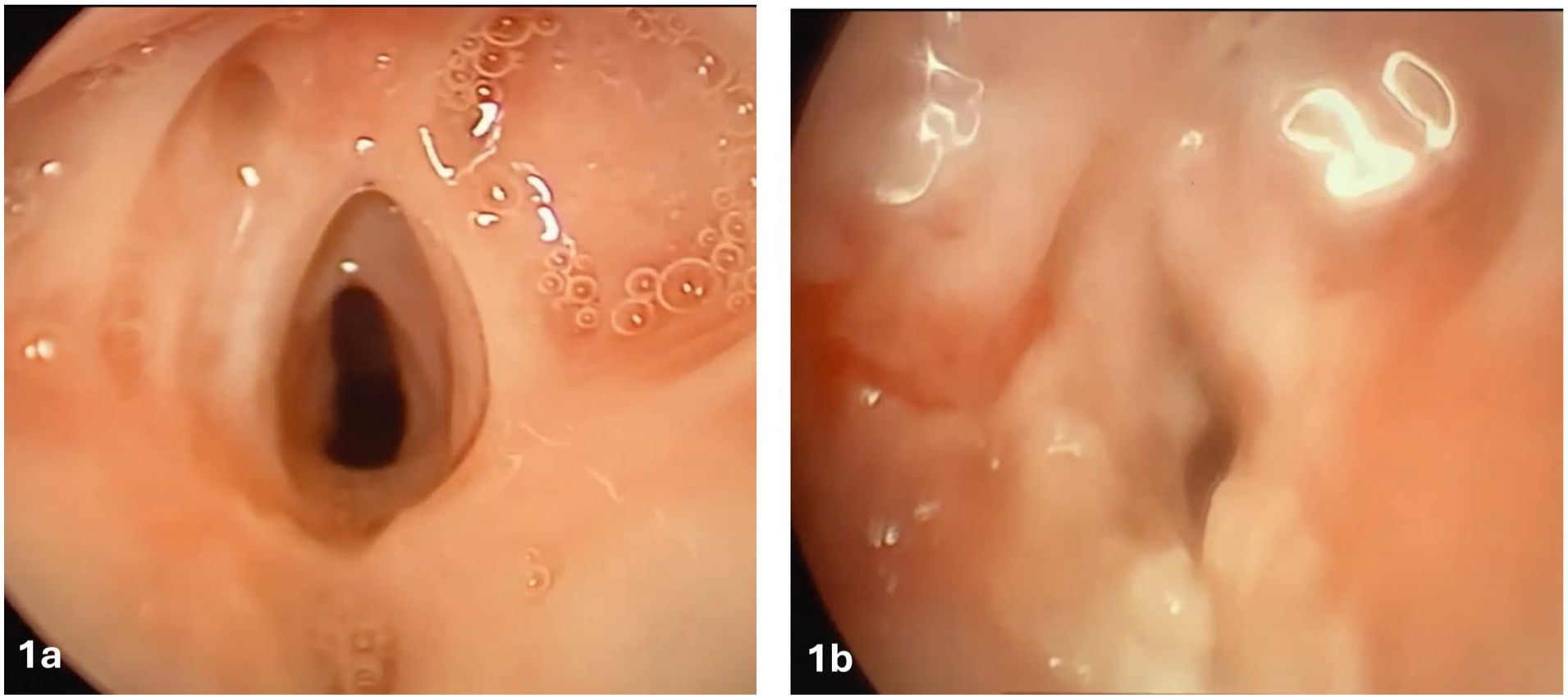
(a) Operative laryngoscopic view demonstrating anterior glottic web, posterior glottic stenosis, and supraglottic adhesions prior to intervention. (b) Second-look laryngoscopy 2 weeks after cold release and dilation showing marked edema, sloughing, and a slit-like glottic opening consistent with early postoperative changes.
At age 7, her most recent DLB demonstrated a persistent grade I anterior glottic web, grade I posterior glottic stenosis, suprastomal granulation, and sparse bullae near the carina and main bronchi (Figure 2). Suprastomal granulation was excised with a microdebrider and cold instruments, followed by intralesional Kenacort injection. Her tube was downsized to a size 3 neonatal tracheostomy tube. She has undergone 6 DLBs to date and is scheduled for another evaluation with consideration for decannulation.
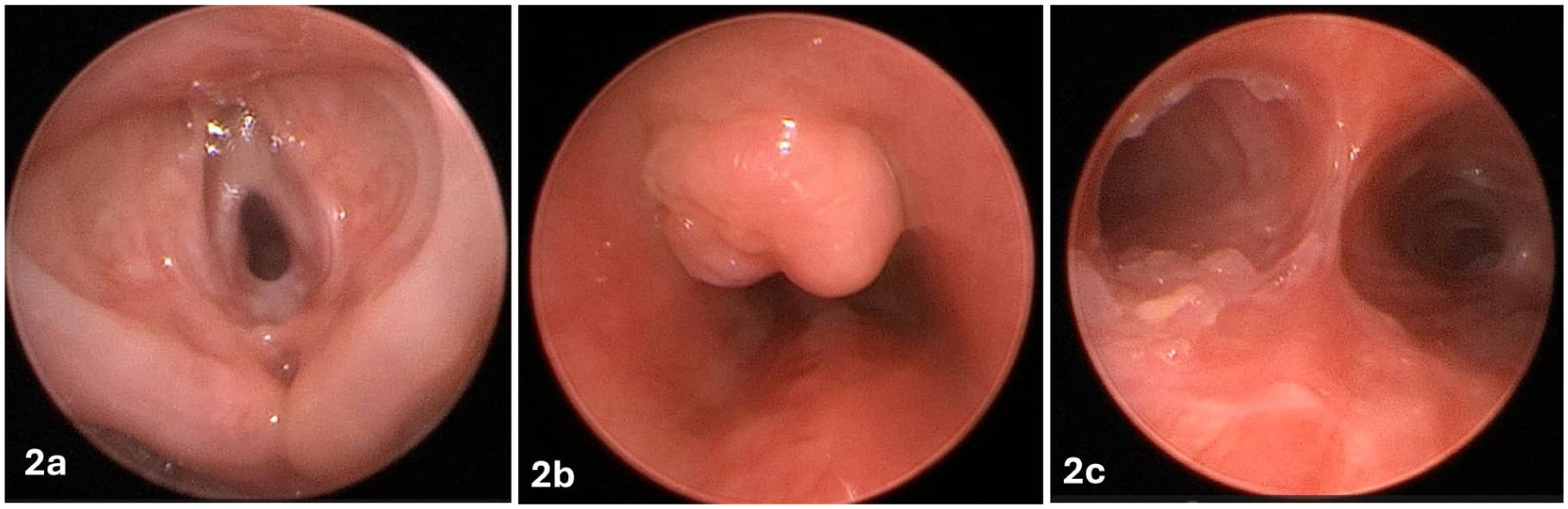
(a) Persistent grade I anterior glottic web and posterior glottic stenosis with supraglottic adhesions during airway evaluation at age 7. (b) Suprastomal granulation tissue prior to excision. (c) Bullae identified at the distal trachea, carina, and proximal bronchi.
Discussion
Airway involvement in EB is an uncommon but potentially life-threatening complication. Although JEB carries the highest predicted cumulative risk of laryngotracheal disease, significant airway pathology has also been described in severe forms of EBS, particularly in PLEC-related variants. 3 This case demonstrates progressive supraglottic and glottic involvement in autosomal recessive EBS, a presentation that remains rarely reported. A summary of previously published cases across EB subtypes is provided in Table 1, illustrating variability in age at presentation, affected airway location, management strategies, and outcomes.
Reported Cases of EB With Upper Airway Involvement.
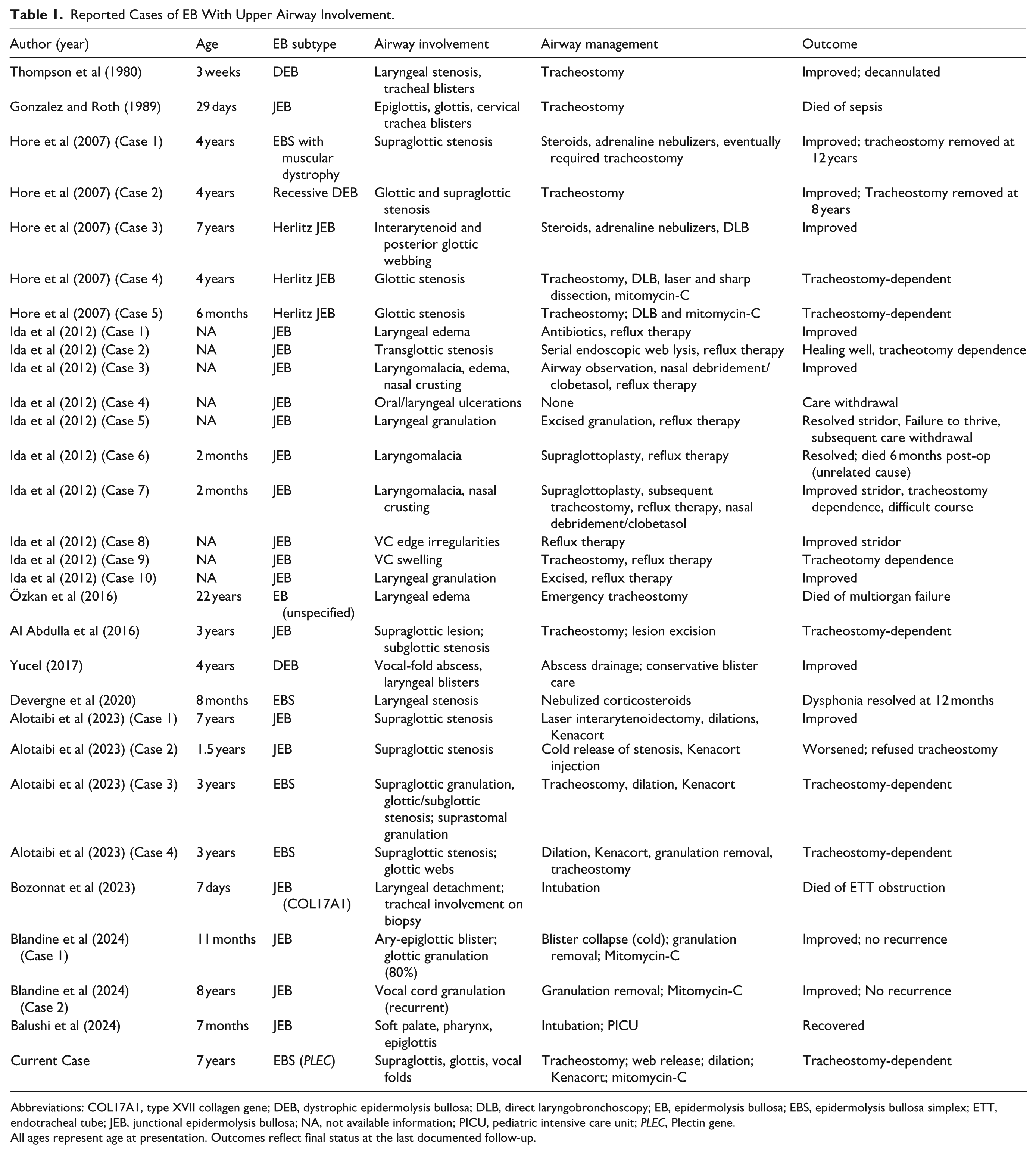
Abbreviations: COL17A1, type XVII collagen gene; DEB, dystrophic epidermolysis bullosa; DLB, direct laryngobronchoscopy; EB, epidermolysis bullosa; EBS, epidermolysis bullosa simplex; ETT, endotracheal tube; JEB, junctional epidermolysis bullosa; NA, not available information; PICU, pediatric intensive care unit; PLEC, Plectin gene.
All ages represent age at presentation. Outcomes reflect final status at the last documented follow-up.
Patterns of Upper Airway Involvement
Upper airway obstruction in EB may be acute or chronic. Reported abnormalities include supraglottic stenosis, glottic webs, posterior glottic stenosis, laryngeal edema, mucosal detachment, and vocal-fold immobility.5-16 Notably, most cases with significant airway disease occur in JEB or DEB. Only a small number of cases involve EBS, highlighting the rarity of meaningful laryngotracheal disease in this subtype and underscoring the importance of documenting similar presentations.
Tracheal involvement is exceedingly rare. As illustrated in Table 1, only a few cases have demonstrated blisters, mucosal sloughing, or severe tracheal obstruction.5,6,14 Compared to these reports, our patient exhibited persistent supraglottic and glottic pathology, while the subglottis and trachea remained largely spared except for focal granulation at the site of tracheostomy tube and occasional bullae at the carina.
Mechanisms of Airway Injury
The fragile mucosa characteristic of EB renders the airway susceptible to blistering, trauma, and scarring. Repeated intubations, suctioning, coughing, and recurrent infections may accelerate the formation of webs, adhesions, and stenosis. 1 In this case, early-life intubations and multiple infectious episodes likely contributed to progressive supraglottic and glottic narrowing. Chronic inflammation and intermittent tracheostomy tube obstruction by bullae added complexity to airway management. Upon reviewing the literature, only 2 cases had a documented history of prior intubation,7,13 underscoring the fragility of the affected airway mucosa in such cases where even minor injuries should be avoided.
Challenges in Anesthetic and Airway Management
Airway instrumentation in EB requires meticulous care to minimize friction and tissue trauma. Recommended strategies include avoiding adhesive materials, using well-lubricated smaller tubes, minimizing repeated instrumentation, and securing intravenous access with silicone-based dressings.4,7 Despite structural airway disease, our patient consistently demonstrated an uncomplicated Cormack–Lehane grade I view, though IV access remained difficult.
Surgical management often includes cold web release, dilation, intralesional steroids, and topical antifibrotics such as mitomycin-C. While these interventions may improve airway patency, they also carry the risk of perpetuating the injury–scar cycle. Individualized decision making is therefore essential, balancing potential improvements with the risk of further mucosal trauma.
Tracheostomy Dependence and Long-Term Course
Table 1 demonstrates that many EB patients with supraglottic or glottic involvement ultimately require tracheostomy and often remain tracheostomy-dependent. This aligns with the prolonged airway course observed in our patient. Tracheostomy care in EB is uniquely challenging due to mucosal fragility, increased risk of granulation, and potential for obstruction by bullae or crusts. Frequent airway assessments are necessary to ensure tube patency and to identify suprastomal complications early. Although decannulation may be considered once supraglottic healing stabilizes, success depends on the degree of residual glottic mobility and stenosis.
Systemic Manifestations and Multidisciplinary Care
Our patient exhibited hypotonia, developmental delay, failure to thrive, and hepatomegaly, which may reflect broader systemic involvement of PLEC-related EBS or nutritional compromise. EB-associated feeding difficulties, increased metabolic demands, and chronic inflammation contribute to poor growth trajectories.3,8,12 Coordinated care involving dermatology, nutrition, pulmonology, cardiology, genetics, anesthesia, and ENT is therefore essential.
Genetic Counseling
Given the autosomal recessive inheritance of the pathogenic PLEC variant, genetic counseling was provided to the family. A 25% recurrence risk in future pregnancies was discussed, along with reproductive options such as preimplantation genetic diagnosis, chorionic villus sampling, and amniocentesis. Although the patient was also a carrier for ACADM and MYO7A mutations, these are not known to be associated with EB or airway disease.
Clinical Significance
This case expands the limited literature on airway involvement in EBS and demonstrates that PLEC-related EBS can present with progressive, clinically significant supraglottic and glottic disease. Compared with most published EBS airway cases—which typically present in infancy—our patient’s presentation at 7 years of age highlights the potential for ongoing progression. Early recognition, cautious airway handling, and repeated endoscopic surveillance are critical to avoid life-threatening complications and optimize long-term outcomes.
Conclusion
Laryngotracheal involvement in EBS is rare but can lead to progressive, life-threatening airway obstruction. This case highlights the potential severity of airway disease in PLEC-related EBS and underscores the importance of early recognition, cautious airway manipulation, and close endoscopic surveillance. Management often requires staged interventions, thoughtful tracheostomy care, and coordinated multidisciplinary support. Awareness of this presentation is essential, as timely diagnosis and individualized management can stabilize the airway, reduce complications, and improve long-term outcomes in affected children.
Footnotes
Consent for Publication
Informed consent is signed by patient’ father (legal guardian) for publication of this case report. Patients’ privacy ensured.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Further data are available upon request.
