Abstract
Purpose:
Facial nerve stimulation (FNS) represents a significant complication of cochlear implantation (CI), yet its risk factors and optimal management strategies remain incompletely understood.
Method:
This study investigated 3750 CI recipients, identifying 265 cases (7.07%) of FNS, to evaluate its incidence and therapeutic outcomes using a novel stepped management protocol.
Results:
Analysis revealed a significant association between FNS and specific underlying etiologies (χ² = 27.10, P < .001), with large cavity malformation emerging as the predominant anatomical risk factor (Adjusted Residual = +3.12). Temporal distribution analysis demonstrated that 34.7% of FNS cases occurred within 1 week of device activation, while 26.0% manifested more than 1 year postimplantation, suggesting progressive etiologies such as electrode fibrosis. A four-tiered stepped management strategy was employed based on FNS severity: Grade 2 (mild, n = 62) achieved 100% resolution through programming adjustments, including pulse width reduction and bipolar stimulation; Grade 3 (moderate, n = 97) exhibited a 46.4% complete remission rate (45/97) with adjunctive steroid therapy; Grade 4 (severe, n = 87) showed improvement following selective electrode deactivation, with 55.2% downgraded to Grade 3 and 44.8% to Grade 2; and Grade 5 (refractory, n = 19) necessitated surgical revision.
Conclusion:
These findings underscore the importance of preoperative imaging in identifying high-risk patients, such as those with cochlear malformations, and demonstrate the efficacy of a stratified management approach in achieving symptom control. This study also establishes a robust, patient-centered framework for managing FNS in CI recipients, emphasizing adaptability to individual clinical profiles and optimizing therapeutic outcomes.
Level of Evidence for the Manuscript:
Level 3.
Keywords
Introduction
Cochlear implantation has become a commonly-performed procedure in children with severe-to-profound sensorineural hearing loss. Nevertheless, postoperative facial nerve stimulation (FNS) as a functional complication still has significant clinical controversy and research gaps regarding its incidence, risk factors, and management strategies. 1 The FNS can be unnoticeable to patients or can cause severe discomfort such as facial muscle twitch or spasm, which may stop the patients from using the implants. 2 The reported incidence of FNS varies widely (0.9%-15%), reflecting heterogeneity in study populations, diagnostic criteria, and intervention methods.3,4 While subjective reporting may underestimate occurrence, electrophysiological studies reveal rates as high as 31% to 78% in high-risk groups. This discrepancy underscores both diagnostic limitations and the need for more systematic analysis of FNS mechanisms and risk factors.5,6 The mechanism behind FNS symptoms is that the current emitted by the electrodes of the implanted cochlea spreads to the surrounding structures, stimulating the facial nerve, which is very close to the cochlea. Therefore, any factors that increase the stimulation current of the cochlear implant or decrease the impedance of the surrounding bony structures can promote the occurrence of FNS symptoms.7,8
The incidence of FNS varies significantly across existing studies, which may be related to inconsistent diagnostic criteria, differences in population characteristics, surgical techniques, and electrode design. Moreover, there is no consensus on the risk factors of FNS in current research, as different etiologies lead to FNS through distinct mechanisms. Additionally, existing intervention methods lack a stratified management framework. The objectives of this study were as follows: (1) to characterize the epidemiological profile of FNS in a Chinese cohort, by analyzing its incidence, temporal trends, and the spectrum of underlying etiologies present in affected patients; (2) to identify key risk factors for FNS through integrated analysis of anatomical abnormalities (eg, cochlear malformations) and relevant patient characteristics; and (3) to propose and evaluate a structured, progressive management strategy for FNS, assessing its initial clinical feasibility and outcomes in our retrospective cohort.
Material and Method
Participants
In total, 3750 CI recipients who had undergone switch-on and programing procedures by our team were included in this study, between January 1997 and January 2024. In total, 265 subjects were enrolled to observe the occurrence of FNS during the activation of cochlear implant devices. The cohort of 265 FNS participants was identified from the same overall study population of 3750 patients who received cochlear implants between 1997 and 2024. The primary inclusion criterion was simply being a recipient of a cochlear implant at our center during the study period. All such patients were considered for inclusion in this retrospective analysis. The exclusion criteria were minimal and focused on data integrity and consent, primarily including the following: (1) unwillingness to participate or inability to provide consent/assent; (2) incomplete medical records or loss to essential follow-up data required for the analysis; and (3) subjects who experienced additional health risks, which meant they were unable to continue participating in the study.
The onset of FNS after CI activation was recorded for each subject, along with the grade of FNS experienced. The definition of FNS is any reproducible facial movement that can be perceived by a third party or the subject themselves, occurring at any time during the activation of the cochlear implant device (when stimulation is given), with the stimulation intensity below the maximum comfort level and related to FNS symptoms. The grading scale for FNS ranged from 1 (no stimulation) to 5 (severe stimulation).
All patients provided written informed consent allowing the analysis of their data, and the study was approved by the Beijing friendship Hospital (approval no.113567209). The study complies with the Declaration of Helsinki.
The Grading System for FNS Post-CI
Based on our experience and relevant literature reports, 1 we classify the degree of FNS as follows, considering factors such as the presence of observable muscle twitching, the affected muscle regions, the presence of pain, and the impact on speech recognition ability:
Grade 1: No stimulation
Grade 2: Minor stimulation, characterized by the detection of slight facial muscle twitching through facial nerve electrophysiological monitoring or the patient’s self-awareness of minor twitching.
Grade 3: Mild stimulation, where observable muscle twitching occurs in one specific area of the face (eye, oral cavity, nasolabial fold, or forehead) that is noticeable to others, but without any other discomfort.
Grade 4: Moderate stimulation, defined by the occurrence of one or more than one of the following: (1) twitching in more than one area of the face; (2) accompanied by other discomfort such as pain; (3) intolerance to prolonged use of a cochlear implant; (4) impact on speech recognition ability; (5) reduced focus during communication.
Grade 5: Severe or higher stimulation, defined by the occurrence of one or more than one of the following: (1) twitching or spasmodic manifestations in multiple facial regions, accompanied by severe discomfort or pain, or other intolerable sensations; (2) intolerance to the use of CI; (3) severe impact on speech recognition ability.
The application of our new five-point FNS grading scale was fundamentally based on patient- and caregiver-reported outcomes. The final grade was assigned by a single clinical audiologist based on a synthesis of the following information:
(1) Primary Source: Patient Self-Report: For older children and adults, the description of the sensation (eg, tingling, twitching), its location, and its impact was directly reported by the patient.
(2) Corroborative Source: Caregiver Observation: For young children or individuals with limited communication skills, direct observation by caregivers or family members regarding visible facial movements (eg, eye twitching, mouth pulling) was essential and was used to corroborate or supplement the child's behavioral cues.
(3) Clinical Corroboration and Ruling Out Other Causes: The role of the clinical audiologist was critical in:
Verification: Confirming that the reported symptoms were consistent with FNS and not another phenomenon (eg, myogenic artifact, simple tactile sensation).
Stimulus-Evoked Confirmation: Systematically testing and observing the patient during programming sessions to confirm that the symptoms were reproducibly linked to specific electrical stimuli from the cochlear implant.
Final Grade Assignment: Integrating the subjective reports and objective observations to assign the final grade according to the defined, impact-based criteria.
Management Strategies of FNS
FNS management is stratified into four progressive tiers based on severity:
Level 1: For mild or transient stimulation (Grades 2-3), patient education and reassurance are prioritized to mitigate anxiety and ensure continued device use.
Level 2: In mild FNS, programming adjustments—including optimizing stimulation parameters (intensity, pulse width, gain) or employing triphasic pulses (device-dependent)—are implemented without compromising auditory perception or requiring adaptation periods.
Level 3: Moderate FNS necessitates modifying stimulation patterns (eg, bipolar mode to narrow field spread), altering coding strategies, or selective electrode deactivation. These interventions balance FNS reduction and auditory performance but may require auditory readaptation.
Level 4: Reserved for refractory cases, severe FNS (≥6-8 deactivated electrodes), or intolerance, surgical revision with alternative implant/electrode selection is considered to preserve speech recognition and usability.
This tiered approach ensures tailored management, minimizing FNS while optimizing auditory outcomes. Strategies escalate from behavioral counseling to technical reprogramming and ultimately surgical solutions, emphasizing individualized care without omitting critical therapeutic options. Surgical intervention was only pursued as a last resort when all available noninvasive options had been exhausted, and the clinical benefit of the cochlear implant was nullified by the severity of the FNS.
To clarify this methodology and enhance readability, we have incorporated a clear flowchart as shown in Figure 1. This diagram illustrates the direct link between the initial FNS grade and the corresponding first-line management interventions. The rationale for this grade-based approach is to implement the most appropriate and effective intervention at the onset, thereby maximizing efficiency and minimizing unnecessary procedures for the patient.
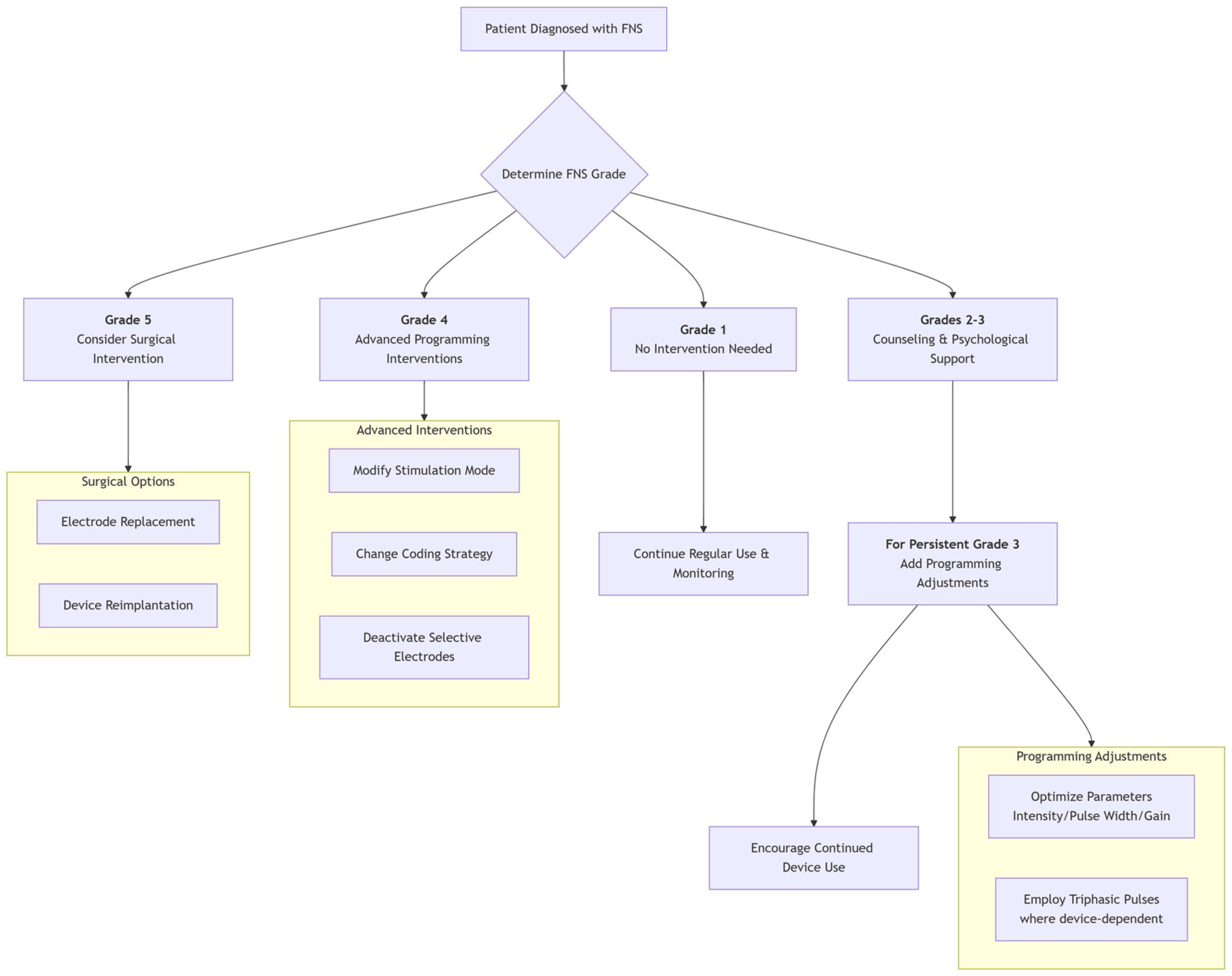
Progressive management strategy for FNS. FNS, facial nerve stimulation.
Success was primarily determined by a combination of the following: Patient Self-Report and Caregiver Observations regarding the resolution or significant reduction in FNS symptoms; Clinical Audiologist Assessment, where a single experienced clinician confirmed the absence of stimulus-evoked facial movements during programming. Crucially, successful management was defined as the elimination of FNS while maintaining stable and usable auditory perception for the patient, allowing for continued device use. This pragmatic approach prioritizes the patient's functional outcome.
Statistic Analysis
Subject characteristics were summarized using mean ± SD and range. Missing data were excluded. Analyses utilized SPSS v24 and R 4.1.2, with α = .05 (two-tailed). Categorical associations (gender, implantation side, device brand vs. FNS incidence) were assessed via chi-squared tests. Adjusted residuals are post hoc statistics used to identify which specific categories (etiologies, in this case) contribute most significantly to an overall chi-squared test result. They were calculated following a significant overall chi-squared goodness-of-fit test. The McNemar-Bowker test analyzed FNS classification improvements post-intervention. Temporal trends in FNS incidence (1 week, 1 year, >1 year post-activation) were examined using Cochran-Armitage trend tests.
Results
The Incidence of FNS
A total of 3750 cochlear implant recipients who had undergone switch-on and fitting procedures by our team were included in this study. In total, 265 subjects were enrolled to observe the occurrence of FNS during the activation of cochlear implant devices, with an overall incidence rate of 7.07%. The ages of the patients ranged from 1 to 63 years. Due to the right-skewed distribution of patient age (mean 14 ± 31.2 years), the supplementary report provided a median age of 6 years (IQR 2-18 years). Among the subjects, there were 149 males (56.2%, 149/265) and 116 females (43.8%, 116/265). Right-sided implants were performed in 199 cases (75.1%, 199/265), left-sided implants in 59 cases (22.3%, 59/265), and 7 cases (2.6%, 7/265) had FNS occur sequentially on one or both sides after bilateral implantation. Among all recipients, 5 cases required explantation and reimplantation, with 3 cases being re-implanted on the same side and 2 cases on the opposite side. There were 249 pre-lingually deaf patients and 16 post-lingually deaf patients. Patients were implanted with one of the three available cochlear implant brands (Advanced Bionics, Stäfa, Switzerland; Cochlear, Lane Cove, Australia; Med-El, Innsbruck, Austria; Oticon Medical, Vallauris, France) with perimodiolar (Cochlear Contour Advance®; Advanced Bionics HiFocus MidScala®), and straight electrodes (all brands). The Oticon Medical device was not included in the comparative analysis regarding brand incidence. The reason for its exclusion is that, during the data collection period for this study, the number of Oticon Medical implant recipients at our center was exceptionally low (constituting less than 0.1% of the total cohort). Out of the 265 subjects, 240 received Cochlear Corporation products, including 30 cases of CI24M, 67 cases of CI24RST, 18 cases of CI24RCS, 27 cases of CI24RCA, 77 cases of CI24RE-S, 14 cases of CI24RECA, and 7 cases of CI512. The implants from Me Del Company included 22 cases, with 7 cases of C40+, 5 cases of SONATAti100, 6 cases of PULSARci100, and 4 cases of Mi10xx Series. The products from the American company AB included 3 cases, with 2 cases of C2/Hifocus 1J and 1 case of 90K/Hifocus Helix.
The onset of FNS is as follows: 92 patients (34.7%) during the activation period (immediately upon activation and within 1 week), 104 patients (39.3%) within 1 year of activation, and 69 patients (26.0%) more than 1 year after activation.
Characteristic of CI Users
The causes and classification of FNS related to the patients include the following: 57 cases of large cavity malformation, 45 cases of Mondini malformation, 48 cases of cochlear nerve dysplasia or cochlear canal narrowing, 3 cases of temporal bone fractures, 18 cases of otosclerosis, 13 cases of meningitis and cochlear ossification, 33 cases of congenital hereditary deafness, and 48 cases due to other or unknown causes (Table 1).
Characteristics of CI Users.
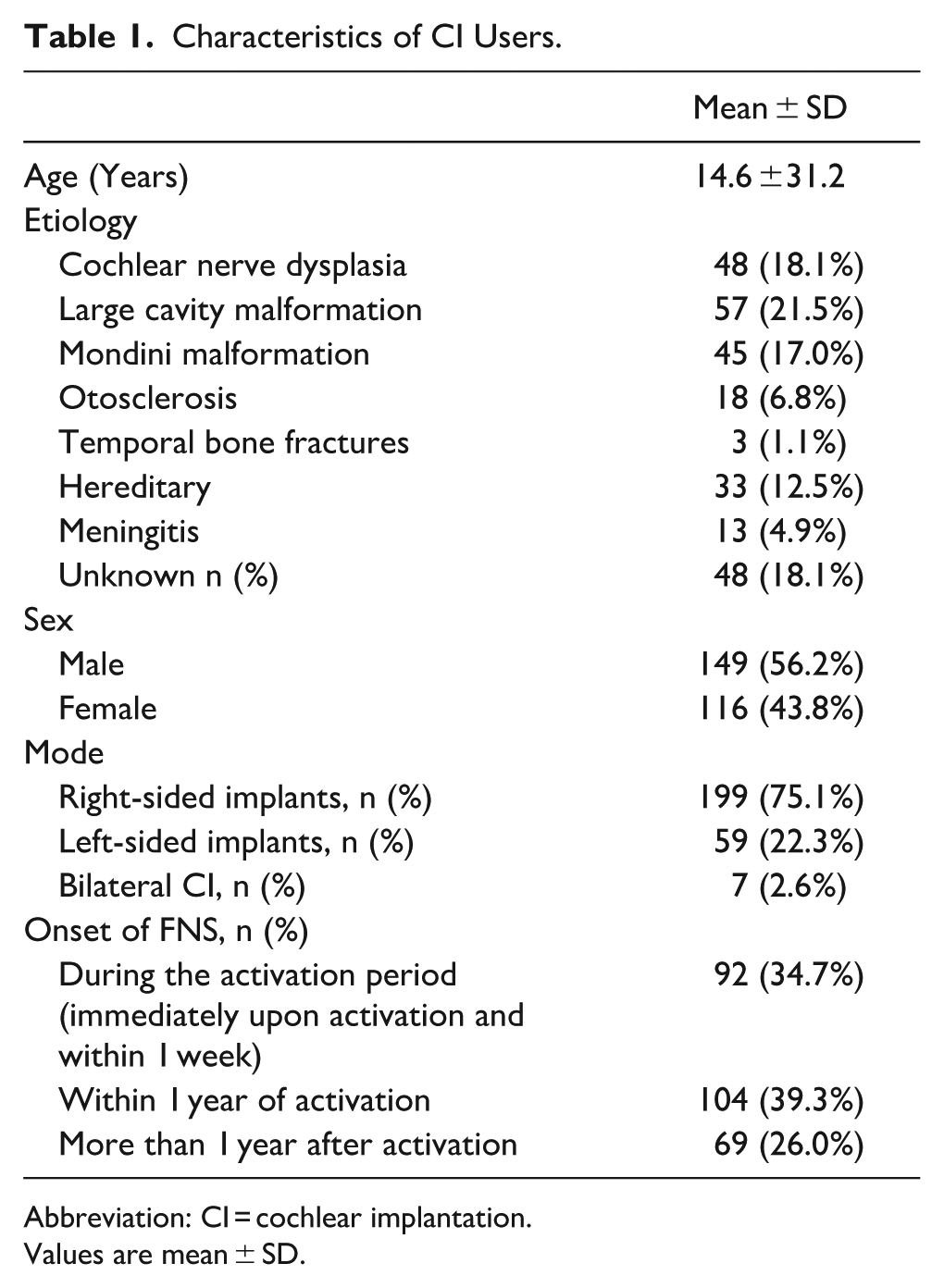
Abbreviation: CI = cochlear implantation.
Values are mean ± SD.
The incidence of FNS in men was 56.2% (149/265), while in women it was 43.8% (116/265). The chi-square test showed no significant difference (χ² = 2.14, P = .143), indicating that gender is not a risk factor for FNS and there is no need for targeted adjustments in clinical interventions (Table 2). Regarding implant brands and FNS, there were no significant differences in FNS incidence among different brands (Cochlear 90.6%, Medel 8.3%, AB 1.1%) (χ² = 1.87, P = .392) and different sides (χ² = 2.65, P = .563). This suggests that the risk of FNS is not related to the implant brand and side.
Relationship Between the Occurrence of FNS and Various Variables.
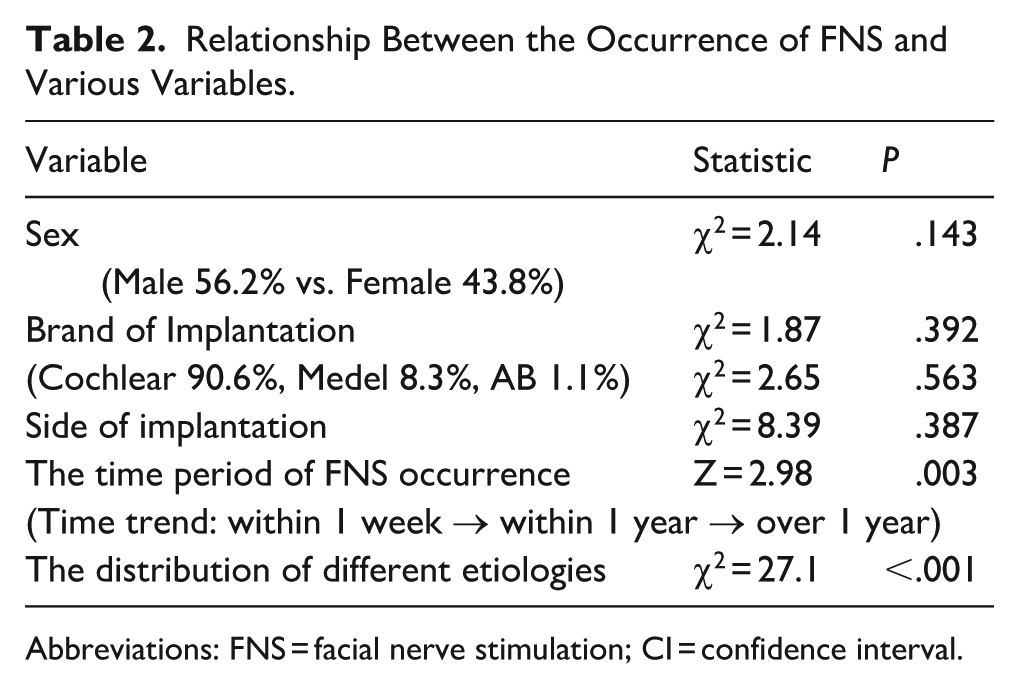
Abbreviations: FNS = facial nerve stimulation; CI = confidence interval.
The Cochran-Armitage trend test was used to examine the linear trend of ordered categorical variables (time trend: within 1 week → within 1 year → over 1 year), specifically the trend of FNS incidence over time (after device activation). The results showed that the incidence of FNS significantly decreased with prolonged activation time (34.7% → 26.0%, Z = 2.98, P = .003). This may be related to enhanced neural adaptability or the stabilization of electrode position due to fibrous encapsulation, supporting the strategy of extending the postoperative observation period.
The results showed that among patients with FNS, there is a significant difference in the distribution of different etiologies (χ² = 27.10, P < .001). The AR value for Large cavity malformation was 3.12, indicating that it was the main contributor to the distribution differences. The high incidence of Large cavity malformation (AR = +3.12) may be due to the increased risk of current spread caused by extensive electrode contact. In contrast, meningitis had a significantly low incidence (AR = −5.34), but it should be noted that the small sample size (13 cases) might affect the reliability of the results.
The Distribution of FNS Classification and Treatment Effects After Intervention
The overall response rate after intervention was 40.4%. After the intervention, 92.8% of patients showed a reduction in FNS grading (χ² = 58.3, P < .001, McNemar-Bowker test), but there was no improvement in patients classified as Grade 5 (refractory FNS), with 19 cases remaining unchanged. This suggests that the stepwise approach is effective for mild to moderate FNS, while severe cases require surgical intervention (Table 3).
Changes in FNS Classification Before and After Intervention.
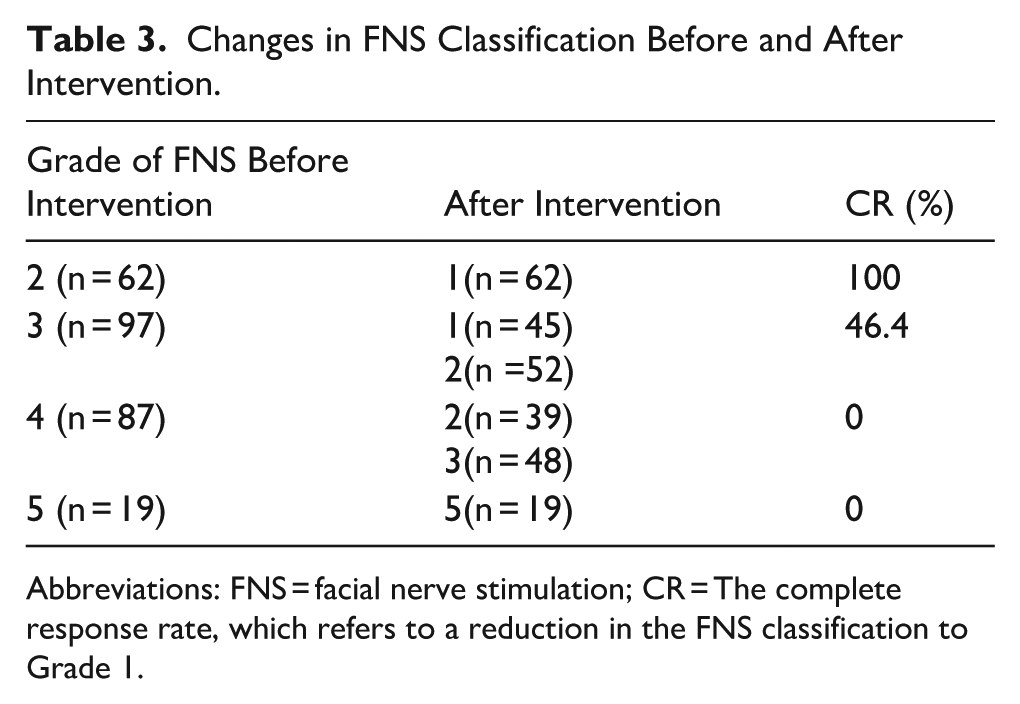
Abbreviations: FNS = facial nerve stimulation; CR = The complete response rate, which refers to a reduction in the FNS classification to Grade 1.
Discussion
Cochlear implantation is an effective treatment for patients with severe sensorineural hearing loss. However, FNS as a functional complication may significantly impact the quality of life and auditory rehabilitation outcomes of patients post-surgery. This study systematically investigates the epidemiological characteristics, mechanisms, and intervention effects of FNS based on a global cohort of 3,750 CI patients, focusing on 265 cases of FNS, in conjunction with clinical practices involving a stepwise management strategy. In our cohort, statistical analysis revealed no significant associations between FNS incidence and factors such as gender (χ² = 2.14, P = .143), side of implantation (χ² = 2.65, P = .563), or implant brand (χ² = 1.87, P = .392). These consistent nonsignificant findings robustly indicate that these variables are not primary risk factors for FNS. This reinforces the conclusion that the etiology of FNS is predominantly rooted in patient-specific anatomical and electrophysiological factors, rather than these baseline demographic or equipment-related variables. Consequently, clinical focus should remain on identifying high-risk anatomical profiles through preoperative imaging, rather than tailoring interventions based on gender, side, or device manufacturer.
The Prevalence of FNS
The prevalence of FNS significantly varies among patients undergoing CI, and the incidence in the present study was 7.07%(265/3750, The reported rate of FNS with multichannel cochlear implants varies from about 1% to 14.9%, with most recent studies reporting an overall incidence of approximately 7%. 9 Cochlear malformation, otosclerosis, and cochlear ossification have been associated with a higher incidence of FNS. 2 The prevalence of cochlear-facial nerve dehiscence (CFD) was 3.2% and 70% of cases with CFD experienced FNS. 10 Some studies have reported prevalence ranging from 1% to 58%. A meta-analysis reported that the prevalence of FNS among the CI populations was 5.29% (175/3306 patients). 11 FNS is also accompanied by specific conditions such as cochlear ossification, inner ear malformation, temporal bone fractures, meningitis, and otosclerosis. Other etiologies are similar to existing reports, such as abnormal electrode positioning due to cochlear ossification in patients with post-meningitis deafness, where the incidence of FNS can be as high as 31% to 78%.12,13
The Occurrence Time of FNS
The occurrence of FNS may happen immediately after CI activation, or it may occur later after a period of use, even appearing up to 10 years later. The onset of FNS is difficult to predict, as it typically increases over time or develops with new fitting maps. Late-onset FNS is not uncommon. Its pathogenesis may be due to the formation of adhesions following inflammatory processes within the temporal bone.14,15 Others suggest that it may be related to changes in current pathways, tissue impedance, or facial nerve sensitivity, while some believe this delay may be associated with increasing stimulation levels over time. 16 In our group of FNS patients, there were individuals with onset occurring at various time points, with a higher proportion (34.7%) experiencing symptoms during the initial activation. These patients often had cochlear structural deformities or auditory nerve developmental abnormalities, which may also be related to intraoperative mechanical injury or postoperative edema. The proportion of late-onset FNS occurring more than 1 year after activation also reached 26%, but the specific reasons may vary from person to person and could be related to fibrosis around the electrode or IL-6-mediated neuroimmune responses. 3
Possible Mechanisms Related to the Occurrence of FNS
The lack of a statistically-significant association between FNS incidence and demographic/surgical factors such as gender (χ² = 2.14, P = .143), side of implantation (χ² = 2.65, P = .563), or device brand (χ² = 1.87, P = .392) is a noteworthy finding. These results robustly indicate that these specific variables are not primary risk factors for FNS in our cohort. Consequently, they should not guide targeted screening or preemptive interventions. This reinforces the central conclusion that the etiology of FNS is predominantly rooted in patient-specific anatomical and electrophysiological factors, rather than these baseline demographic or equipment-related variables. FNS risk is multifactorial, linked to anatomical, electrical, and pathological factors. Predisposing conditions include otosclerosis, temporal bone fractures (pre-/postimplantation), osteoporosis, meningitis, hydrops, and congenital inner ear malformations (eg, cochlear hypoplasia, enlarged vestibular aqueduct). 17 Cochlear current leakage is a key contributor, particularly in cases with developmental cochlear anomalies or pathological bone conditions (eg, otosclerosis, post-meningitis ossification). These pathologies are believed to create aberrant, low-resistance pathways that facilitate current spread beyond the cochlea, stimulating the facial nerve. 18 Structural cochlear defects increase current diffusion, stimulating the labyrinthine facial nerve segment. 19 Adjusted residuals are post hoc statistics used to identify which specific categories (etiologies, in this case) contribute most significantly to an overall chi-squared test result. They were calculated following a significant overall chi-squared goodness-of-fit test (χ² = 27.10, P < .001), indicating that the observed distribution of etiologies in the FNS group differed from the expected distribution. Notably, large cavity malformations are overrepresented (AR = +3.12), while post-meningitis ossification is underrepresented among FNS cases, suggesting etiology-specific anatomical influences on current spread. 20 The AR value quantifies and confirms that large cavity malformation is a predominant anatomical risk factor for FNS in our cohort. This makes pathophysiological sense: These malformations (eg, common cavity) feature a large open bony lumen that fails to contain the electrical current. This drastically increases the risk of current spread to the nearby labyrinthine segment of the facial nerve, consistent with the “current leakage” hypothesis. This finding directly informs clinical practice. It underscores the critical importance of preoperative CT imaging to identify patients with such malformations. For these high-risk individuals, surgeons can then proactively consider strategies such as selecting more flexible or shorter electrode arrays, planning a specific insertion trajectory to maximize distance from the facial nerve, and informing the audiologist to cautiously initiate stimulation at lower parameters postoperatively.
Otosclerosis may elevate FNS risk due to abnormal current density from cochlear bony shell thickening. 21 Similarly, temporal bone fractures may disrupt insulation, enabling aberrant stimulation. 22 Excessive stimulation intensity also drives FNS, often necessitated by cochlear nerve hypoplasia, narrow cochlear canals, prolonged auditory deprivation, or electrode failure, which require higher currents to evoke hearing, raising “leakage” risks. 22 In our cohort, patients with cochlear nerve hypoplasia or narrow canals exhibited an 18.1% FNS incidence.
Anatomic proximity between electrodes and the facial nerve further modulates risk. The upper basal turn of the cochlea (UBTC) lies closest to the labyrinthine facial nerve segment. Studies report thinner UBTC-facial nerve bony partitions in FNS patients. 23 While electrode count/position within arrays shows no FNS correlation, 24 reduced bony resistance (eg, otosclerotic spongiosis), or bone loss-induced cavity formation may shorten electrode-nerve distances, facilitating current diversion. 25
Preoperative high-resolution CT with 3D reconstruction and intraoperative navigation can identify high-risk anatomy (eg, UBTC-facial nerve bone thickness/density), guiding electrode selection (eg, flexible arrays) or initial stimulation parameter reduction to mitigate FNS.22,27 This preoperative assessment aids in ear-of-implantation decisions and personalized surgical planning. 26
The Rationale of New Five-Point Scale and Strategies to Manage FNS
Our decision to employ a new five-point scale in this study was primarily driven by considerations of clinical practicality and patient assessment accuracy, based on the following rationale:
(1) Clinical Limitations of the Existing Standard: The intermediate grades (2-5) in the Kelsall system strictly depend on the patient's ability to accurately report the number of affected facial anatomical regions (eg, eye, mouth, nasolabial fold, forehead). However, in clinical practice, especially for patients with mild or intermittent FNS, it is often difficult for them to precisely identify and report the exact number of regions involved. This ambiguity can lead to inaccurate grading, which may subsequently affect the clinician's objective assessment of the severity and impede optimal management decisions.
(2) Core Concept of the New Standard: A Shift Toward “Functional Impact”: Our new grading system aims to address this issue. Its core principle is to shift the focus from merely counting the “number of regions” to evaluating the “actual impact on the patient’s function and quality of life.” Our Grade 2 (Minor Stimulation) encompasses all mild, intermittent symptoms that do not affect device use, without requiring the patient to perform the difficult task of regional counting. Our Grade 4 is a critical composite grade. It consolidates various clinically-significant negative outcomes, such as “involvement of multiple regions,” “accompanying pain,” “impact on speech recognition,” or “inability to use the device for extended periods.” This grade signifies that FNS has progressed from a symptom to a functional problem that poses a substantial impediment to implant use.
We believe this impact-oriented grading (eg, distinguishing “inability to use for extended periods” from “complete inability to use”) provides more intuitive guidance for escalating clinical management strategies.
(3) Driven by Clinical Feasibility: We believe this new standard, which focuses more on functional consequences, reduces the variability introduced by patients' subjective and imprecise descriptions and improves inter-rater consistency. Consequently, it demonstrates greater feasibility and reproducibility in routine clinical follow-up.
(4) Regarding Validation: We acknowledge that as a newly-proposed standard, its broader validity and reliability require further verification in future large-scale studies. However, the retrospective data from our present study preliminarily indicate that this scale can effectively distinguish FNS cases of differing severity and shows a good correlation with the corresponding levels of management strategies we employed.
In summary, our proposed new scale is not intended to replace the Kelsall system but rather to offer a complementary framework that is more functionally oriented and clinically operable. We believe it holds significant value for optimizing the personalized management of FNS and improving clinician-patient communication.
Accordingly, the handling of FNS in this study is based on the degree and impact of FNS, with the aim of controlling and alleviating FNS using appropriate methods to minimize impact on the implant recipients. For those with mild and moderate cases, it is essential to provide sufficient explanations to alleviate fears, which is a critical aspect that can foster understanding and cooperation from both the recipients and their parents. The specific principles must be developed to ensure minimal impact on the subjects. Therefore, the first measures taken involve adjusting stimulation parameters, including changes to stimulation intensity and pulse width, adjustments in gain, or selecting certain products that use three-phase pulses instead of bipolar pulses. 27 Braun and others studied the application of three-phase pulse stimulation in patients with cochlear implants and found that it can effectively alleviate FNS and improve the user experience. 28 These interventions have minimal effect on the recipients’ auditory perception. If these measures do not resolve the issue, the next step is to identify and deactivate the specific electrode causing FNS. If necessary, changes to the electrode stimulation pattern or the speech coding scheme may be made, striving to address the problem using a programming mode. Of course, if the number of deactivated electrodes exceeds a certain limit (eg, 6-8) and significantly impacts speech recognition ability, or if the patient refuses to use the cochlear implant, or if the FNS reaches Grade 4, it may be necessary to consider selecting a more appropriate electrode array or even replacing the implant to ensure the recipient benefits from cochlear implantation. 29 Similar handling methods are seen in related articles, but a clear step-by-step processing strategy has not been documented. In this study, a stair-step treatment strategy based on the degree of FNS was applied to all patients, achieving excellent results with an overall efficacy rate of 92.8%. Other research has reported strategies to reduce FNS as well. Kim et al 30 found that using intraoperative neurophysiological monitoring during cochlear implantation can effectively reduce the incidence of FNS and assist surgeons in optimizing electrode placement. Zhou et al. proposed a deep learning-based signal processing strategy to decrease the frequency of FNS occurrences during cochlear implantation. 31
Strengths and Limitations
This study also has the following limitations: The retrospective design may lead to selection bias, there is a lack of control data from non-FNS patients, and the number of long-term follow-up patients (>5 years) is insufficient to assess the risk of late-onset FNS. It should also be noted that electrode design characteristics, such as the distinction between pre-curved and straight arrays, constitute another potential variable warranting targeted investigation in future research to further elucidate the multifaceted risk profile of FNS. Our analysis identified large cavity malformation as a predominant etiology among patients who presented with FNS, suggesting its clinical importance in this specific cohort. However, the lack of a direct control group of non-FNS implant recipients prevents us from concluding that this malformation is a statistically-independent risk factor. Therefore, we interpreted this finding as highlighting a clinically-significant anatomical profile that warrants heightened awareness and preoperative planning, with its definitive predictive value to be established in future controlled studies. The practical application of this grading system might be challenging because of subjective interobserver variability in reporting grades.
Conclusions
FNS, a frequent CI complication, arises from multifactorial etiologies and may cause discomfort or implant disuse. While onset can occur immediately, delayed presentation is more common, necessitating meticulous preoperative planning (eg, device selection) and counseling to mitigate risks. Management combines programming adjustments (eg, parameter optimization, electrode deactivation) and, in refractory cases, surgical revision. Proactive patient/parent education on FNS mechanisms and impacts is critical to alleviate anxiety and ensure adherence. Systematic analysis of causative factors—including anatomical anomalies, stimulation intensity, or current leakage—enables targeted interventions to preserve auditory outcomes. A tiered approach prioritizing cost-effective strategies (eg, reprogramming) over invasive measures optimizes clinical efficacy while minimizing burden.
Footnotes
Acknowledgements
We discussed the results and implications and commented on the manuscript, at all stages. We acknowledge and thank the following: The Beijing Friendship Hospital Cochlear Implant Team and Clinical Auditory Diagnostic Center, particularly Xia Yuqi and Chen Zhe for their assistance with data collection; Chen Yuxin for the testing equipment and stimuli calibration; and our patients for their time and participation in this study. We would like to designate Chaogang Wei and Yuhe Liu as the corresponding authors for this manuscript.
Ethical Considerations
All procedures were conducted in accordance with the Declaration of Helsinki and approved by the the Beijing friendship Hospital (approval no.113567209).
Consent to Participate
Written informed consent was obtained from all participants prior to inclusion in the study. The study protocol was approved by the Beijing friendship Hospital (approval no.113567209).
Author Contributions
Chaogang Wei has contributed significantly to the experimental design and data analysis, while Yuhe Liu has provided substantial input on the manuscript writing and revisions.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received grants from the following funding sources: National Natural Science Foundation of China (NSFC)—82471185; Chinese Institutes for Medical Research, Beijing (Grant No. CX24PY13); Capital’s Funds for Health Improvement and Research-No. 2022-1-2023.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
