Abstract
This prospective study investigated the potential benefits of deactivating the second most apical electrode to improve access to lower-frequency pitch and first formant information to help improve speech and music outcomes with a cochlear implant. Twenty-one adults (30 ears) with cochlear implants completed an A-B-A-B study to compare the participant's clinical map with all electrodes active (A) and their clinical map with the second most apical electrode deactivated (B). Test measures included pitch discrimination, speech understanding in noise, and subjective musical sound quality and enjoyment ratings. This study also investigated the impact of participant demographic and electrode placement factors on the degree of benefit derived from the experimental map (B). There was no significant difference between the two conditions on any measure at the group level. However, individual participants demonstrated improvements in pitch discrimination (33.3%), speech perception in noise (43.3%), musical sound quality (50.0%), and musical enjoyment (40.0%). Musical sound quality and enjoyment ratings were strongly correlated, and speech perception correlated with musical enjoyment but not sound quality. Electrodes outside scala tympani, smaller electrode-to-modiolus distances, and certain device manufacturers (Cochlear and MED-EL) predicted greater benefit from deactivating the second-most apical electrode. Certain adult cochlear implant users may benefit from selective apical electrode deactivation, depending on their demographic and electrode placement profile. Clinicians could consider deactivating the second most apical electrode with patients, who report poor musical sound quality or those who have disengaged from music since receiving their CI to assess potential benefits individually.
Keywords
Introduction
A cochlear implant (CI) is a medical device that helps individuals with moderate to profound sensorineural hearing loss (SNHL) hear (Buchman et al., 2020). CI listeners often perform well on tasks of speech understanding in quiet and rhythm perception because these signals primarily rely on temporal envelope cues, which are well preserved via CI signal processing (Holden et al., 2013; Jiam & Limb, 2019). In contrast, CI listeners perform more variably on tasks of speech understanding in noise and music perception (i.e., pitch, melody, timbre) because these signals rely more heavily on spectral cues, which are significantly degraded (Dornhoffer et al., 2021; Fowler et al., 2021). For example, in the speech domain, CI listeners may find it difficult to understand speech at signal-to-noise ratios (SNRs), such as +5 dB SNR, where listeners with normal hearing (NH) achieve 100% understanding without visual cues (Buss et al., 2015; Summers et al., 2013; Vermeire et al., 2016). In the music domain, there are well documented differences between individuals with CI and NH for pitch discrimination thresholds (CI: 3 semitones; NH: <1 semitone), isochronous melody recognition (CI: 25.1%; NH: 87.5%), and timbre recognition (CI: 45.3%; NH: 94.2%) (Drennan et al., 2015; Kang et al., 2009). Thus, developing programming strategies that improve access to and the quality of spectral information delivered through the CI is critical to improving outcomes.
There are both technical and biological constraints that degrade spectral cues through the CI. First, CI stimulation aims to restore access to spectral and temporal cues by converting incoming sound waves into tonotopically modulated biphasic electrical pulses of the auditory neural interface (Shannon et al., 1995). To achieve this, incoming sound is sectioned into different frequency bands using bandpass filtering. The temporal envelope is extracted for each individual band and is used to amplitude-modulate a biphasic electrical pulse train emitted from corresponding tonotopically organized electrodes. Importantly, spectral and temporal fine structure cues are largely discarded during this process. Second, CI electrode arrays contain 12–22 electrodes that stand in for thousands of sharply tuned auditory filters, thereby limiting spectral resolution. Third, CI recipients experience considerable spread of electrical excitation, commonly referred to as channel interaction (Jones et al., 2013; Won et al., 2014). Channel interaction occurs when electrical stimulation from one electrode activates neural populations intended to be stimulated by adjacent electrodes. This unwanted spread results from several factors, including (a) the electrode array being surrounded by highly conductive fluid, (b) electrodes often positioned at a distance from their target auditory neurons (requiring higher charge levels for stimulation), and (c) the use of monopolar stimulation, where current flows between an active intracochlear electrode and a distant extracochlear ground electrode, creating a broad electrical field. Consequently, intracochlear electrical stimulation activates large populations of peripheral auditory neurons, thereby limiting fine frequency resolution.
Recent work investigating mechanisms of channel interaction found that artificially increasing channel interaction across evenly distributed electrodes did not significantly impact speech-in-noise perception (Goehring et al., 2020, 2021). However, when channel interaction was clustered in the apical region, substantial degradation to speech understanding occurred, likely due to its importance for conveying first-formant information (Bosen & Chatterjee, 2016; Goehring et al., 2021). Berg et al. (2024) extended this work by examining how spectral blurring across different CI electrode regions affects speech recognition. Results showed that artificial channel interaction on all channels significantly decreased performance, but particular speech components were differentially affected by regional blurring. For example, vowel recognition showed greater sensitivity to apical and middle region channel interaction, whereas consonant recognition was more affected by basal region channel interaction.
CI channel independence varies by electrode positioning factors and the type of incoming signal. For example, CI users demonstrate asymptotic speech understanding with as few as 4–10 spatially selective channels, or active electrodes (Fishman et al., 1997; Friesen et al., 2001). Even in background noise, CI recipients with electrode arrays that are straight in design, positioned outside scala tympani, or with larger (>0.5 mm) electrode-to-modiolus distances are limited to 8–10 independent channels (Berg et al., 2020, 2021, 2022). For individuals with perimodiolar electrode arrays placed fully in scala tympani, recent evidence suggests that these CI recipients may have up to 16 independent channels for speech understanding in noise tasks (Berg et al., 2019b; Croghan et al., 2017; Gifford et al., 2022; Schvartz-Leyzac et al., 2017). On the other hand, music perception and sound quality are hindered even more by channel interaction as studies have shown music perception and sound quality minimally require 10–64 independent channels, depending on the number of musical lines and instruments (Berg et al., 2019a; Shannon, 2005; Smith et al., 2002). Though there are many factors at play, research has shown that the better musical sound quality ratings are obtained for CI recipients with greater electrode-to-modiolus distances, greater musical experience, and high levels of speech understanding (Berg et al., 2019a). Interestingly, the number of independent channels also varies by musical genre, with more rhythmic-based (i.e., temporal) genres like hip-hop or rap requiring just four independent channels and more pitch-based (i.e., spectral) genres like jazz requiring at least 12 (Berg et al., 2019a).
Despite considerable variability in channel independence across CI recipients and signal inputs, CIs are currently programmed using a one-size-fits-all approach with respect to current delivery, frequency allocation, stimulation rate, and number of active electrodes. Strategic electrode selection is a promising approach for increasing channel independence by identifying electrodes causing overlapping stimulation (i.e., channel interaction), using perceptual or physiological metrics, and deactivating these electrodes in a patient's CI map. However, metrics for identifying the optimal subset of electrodes have varied across studies. Depending on the metric employed, different definitions of the optimal subset of electrodes for stimulation result, and not all studies have shown benefits. For instance, studies using tonotopicity or electrode discrimination have not shown speech benefits for strategic electrode selection at the group level (Debruyne et al., 2017; Henshall & McKay, 2001; Vickers et al., 2016). Conversely, studies using absolute thresholds for broad versus focused stimulation (Bierer & Litvak, 2016; Zhou, 2016), masked modulation detection thresholds (Garadat et al., 2013; Saleh et al., 2013; Zhou & Pfingst, 2012), or CT-based anatomical measurements of channel interaction (Labadie et al., 2016; Noble et al., 2013, 2014, 2016) have demonstrated significant benefits in auditory and speech perception compared to performance with all electrodes active.
Recent findings by Berg et al. (2025) provide compelling evidence that targeted deactivation of specific electrodes can also substantially improve music perception. Specifically, participants with deactivated apical electrodes showed the greatest benefits. For instance, among participants with at least one electrode deactivated among the three most apical, 38% exhibited improvements of more than one semitone in pitch discrimination thresholds at 262 Hz, compared to only 10% of participants without apical electrode deactivation. This pattern was consistent across multiple test frequencies (262, 330, and 392 Hz), with apical deactivation consistently providing greater improvements in pitch perception, underscoring this region's importance for music perception.
Electrode selection methods that incorporate electrode placement information (e.g., scalar location, electrode-to-modiolus distance, and angular insertion depth), such as CT-based anatomical measurements, may be advantageous over psychophysical methods alone (e.g., absolute thresholds, masked modulation thresholds), given the growing evidence that these factors significantly impact CI outcomes. For example, electrode arrays inserted entirely within scala tympani achieve superior audiologic outcomes (O’Connell et al., 2016; Skinner et al., 2002; Wanna et al., 2014, 2015). For straight arrays, deeper insertions up to approximately 650° are associated with improved speech recognition outcomes; however, insertions beyond this often lead to basilar membrane disruption and translocation out of scala tympani, resulting in poor structural preservation and speech recognition (Helbig et al., 2018; Morrel et al., 2020; O’Connell et al., 2017). In fact, the most significant positional predictors of outcomes for precurved arrays are full scala tympani insertion and modiolar distance, whereas angular insertion depth was found to be the most significant predictor for straight arrays (Chakravorti et al., 2019).
To address these challenges, each CI manufacturer has developed a unique signal processing approach. Advanced Bionics employs current steering, stimulating multiple electrodes concurrently to increase spectral resolution (Koch et al., 2007). In contrast, Cochlear uses ACE (an n-of-m strategy), processing sound through multiple frequency bands and selecting the most informative ones for stimulation (Holden et al., 2005). MED-EL takes a different approach with FS4, providing fine structure information on the apical four-electrode channels (Riss et al., 2014). Despite these innovations, optimal delivery of spectral cues remains challenging.
A recent study by Jiam et al. (2019) found significant benefits for pitch scaling using a CT-based electrode selection approach. Their algorithm deactivated electrodes if they were extracochlear, required frequency shifts greater than seven semitones, or had theoretical frequencies that exceeded the frequency boundaries of the cochlear implant (i.e., 70–8500 Hz) when electrode placement was compared to the modified Greenwood function (Stakhovskaya et al., 2007). However, this algorithm resulted in poorer performance on vowel recognition, a spectral-dependent task in the speech domain. Given these findings, and that music requires more independent channels, and is impacted by electrode positioning factors differently than speech, further investigation is necessary to determine the optimal subset of electrodes needed for music and speech stimuli to yield the best CI outcomes.
The apical region of the electrode array is important for music. For example, the entire left-half of a piano keyboard is approximately 400 Hz and below. To distinguish each key from one another, high levels of spectral resolution are required. However, CIs only have a couple of electrodes devoted to this region, as illustrated in Figure 1. In fact, the three most apical channel filers range from 238 to 587 Hz for Advanced Bionics, 188 to 563 Hz for Cochlear, and 70 to 469 Hz for MED-EL. This region is also the focus on the most commonly used clinical measure of music perception, the UW-CAMP, which tests pitch perception using base frequencies ranging from 263 to 396 Hz.

Illustration of the Range of the Piano Keyboard Covered by the Apical Region of the Electrode Array for the Three Cochlear Implant Manufacturers. Advanced Bionics (Navy), Cochlear (Yellow), and MED-EL (Red). The Apical Region of the Electrode Array Corresponds to the Pitch Range (263–396 Hz) of the UW-CAMP Pitch Perception Test.
In this study, we chose to selectively deactivate the second most apical electrode, rather than the most apical, as a strategy to reduce channel interaction clustering in the apical region while preserving the benefits of deeper insertion. This investigation was conducted as a follow-up to our previous findings that showed participants with apical electrode deactivations demonstrated significant improvements in pitch perception (Berg et al., 2025). This approach targets apical channel interaction, which significantly impairs speech understanding (Bosen & Chatterjee, 2016; Goehring et al., 2021), while maintaining the potential advantages of deeper insertion provided by the most apical electrode (Chakravorti et al., 2019; Helbig et al., 2018; Morrel et al., 2020; O’Connell et al., 2017). Our study aimed to measure the impact of selective apical electrode deactivation on speech and music outcomes and determine the impact of demographic and electrode placement factors on the degree of benefit derived from the intervention. Additionally, we examined relationships between outcome measures to better understand how different speech and music outcomes are related. We hypothesized that selective apical electrode deactivation can improve both speech perception in noise and music perception/quality by reducing channel interaction in the apical region (Berg et al., 2025; Goehring et al., 2021). Our goal was to minimize apical channel interaction while maintaining as many active electrodes as possible, given that spectrally-weighted tasks such as speech understanding in background noise, pitch perception, musical sound quality and enjoyment ratings require a greater number of independent channels (Berg et al., 2019; Shannon, 2005; Smith et al., 2002). Participants with poorer electrode placement—as defined by translocation (i.e., having one or more intracochlear electrodes outside scala tympani)—were hypothesized to demonstrate greater benefits with selective apical electrode deactivation than participants with better electrode placement because of the greater amount of channel interaction they experience in their everyday map (Berg et al., 2020).
Materials and Methods
Participants
All participants provided written consent to participate in the study and allow results to published using the protocol approved by the local Institutional Review Board. Twenty-one postlingually deafened adult CI users (8 females; 14 right ears) with bilateral SNHL participated. Of the 21 unique participants, nine were bilateral CI recipients, each tested in both ears, and 12 were bimodal users, each tested in their CI ear alone. To recruit a more homogenous sample, participants were screened for musical experience using the Ollen Index of Musical Sophistication with no participant scoring above an amateur level (<500) (Ollen, 2006).
Each of the three FDA-approved CI manufacturers were equally represented with 10 Advanced Bionics (AB), 10 Cochlear, and 10 MED-EL devices. Our sample included 12 pre-curved and 18 straight arrays. Our inclusion criteria required participants to have at least 6 months of CI listening experience, at least 10 hr/day of CI processor wear time, no more than three electrodes deactivated in their clinical map, and the second most apical electrode (E2 for MED-EL and AB, E21 for Cochlear) could not have been deactivated prior to study participation.
Table 1 provides participant demographic information, including participant number by ear and by person in parentheses, age in years, biological sex, ear tested, CI experience in months, CI processor wear time in hours per day, electrode(s) deactivated in their clinical map and the reason for the deactivation, as well as CI-alone scores for CNC words, AzBio sentences in quiet, and AzBio sentences in noise (+5 dB SNR) as recorded from the most recent clinical appointment in the electronic medical record. Possible reasons for electrode deactivations included extracochlear (EC) or poor sound quality (SQ).
Participant Demographic Information.

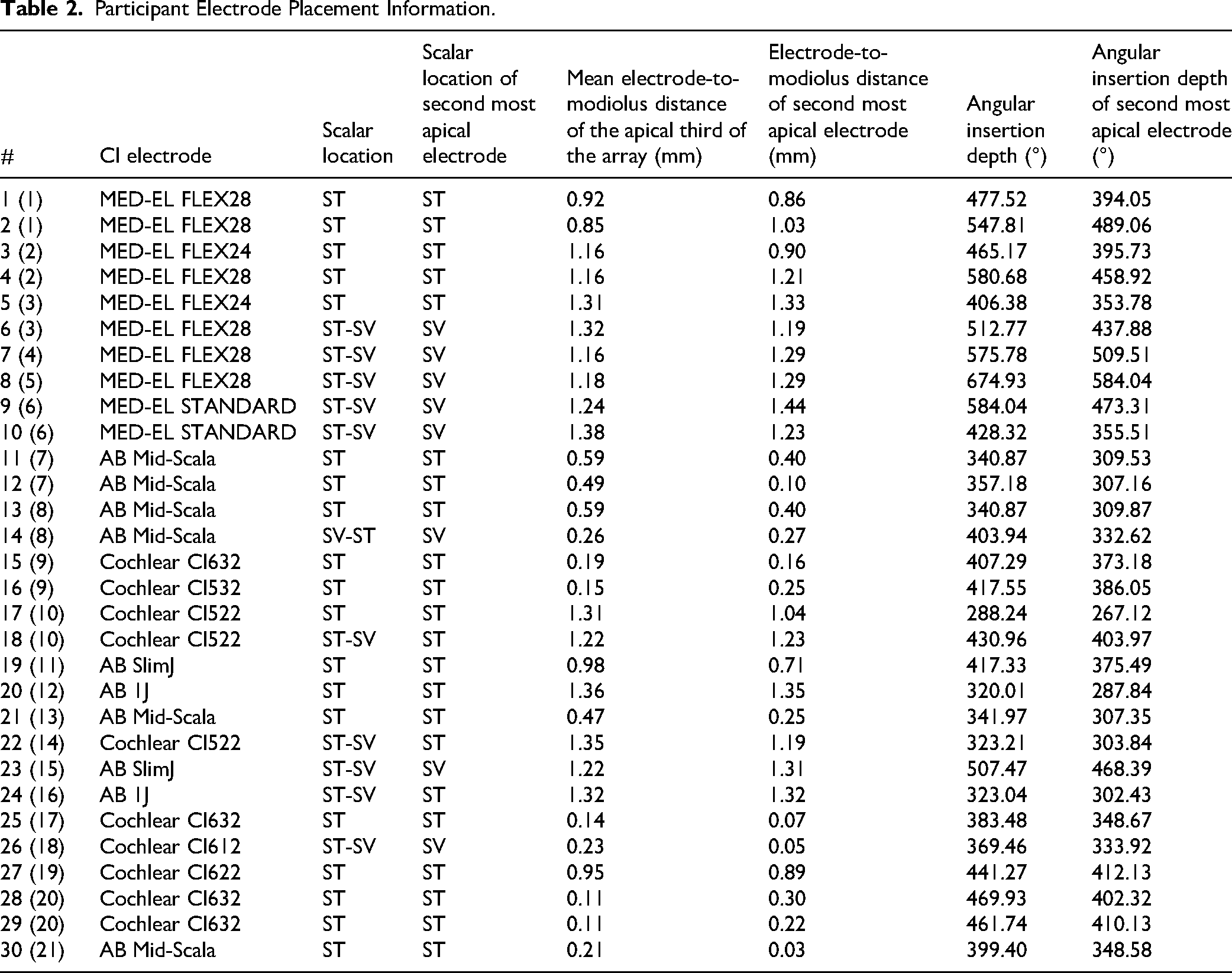
Electrode placement information was determined by CT imaging using validated CI position analysis algorithms (e.g., Noble et al., 2013). Table 2 provides electrode placement information, including overall scalar location, scalar location of the second most apical electrode (E2 for MED-EL and AB; E21 for Cochlear), mean electrode-to-modiolus distance of the apical third of the electrode array in mm, electrode-to-modiolus distance of the second most apical electrode, overall angular insertion depth in degrees, and angular insertion depth of the second most apical electrode for each participant. Possible scalar locations included scala tympani (ST) and scala vestibuli (SV).
Participant Electrode Placement Information.
Materials and Conditions
An A-B-A-B study design was used, comparing the participant's clinical map with the second most apical electrode (E2 for AB and MED-EL devices and E21 for Cochlear devices) active (A) and their clinical map with the second most apical electrode deactivated (B). The presentation order of starting with A versus B was randomized across participants to reduce bias for the clinical map and potential learning effects. AB participants used the Optima-S coding strategy with spanning disabled, Cochlear participants used the ACE or n-of-m strategy, and MED-EL participants used the FS4 strategy, which are the clinical default strategies for each manufacturer to represent a clinically applicable manipulation. All other mapping parameters were kept constant for each participant across the two study maps, except for the automatic re-allocation of frequency bands by the clinical software when the second most apical electrode was deactivated. This re-allocation ensured complete frequency coverage without spectral gaps by redistributing the deactivated electrode's frequency range to neighboring electrodes. Threshold levels were not adjusted from the participant's clinical map, however aided detection thresholds were verified to be within 20–30 dB hearing level (HL) for frequency-modulated pure tones (250–6000 Hz) in the sound field before the participant began the study (Firszt et al., 2004; Holden et al., 2007; Skinner et al., 1999). Initial upper stimulation levels were set behaviorally using a loudness scaling chart according to each manufacturer's clinical guidelines: “maximum comfort levels” for AB, “loud, but comfortable levels” for Cochlear, and “maximum comfort level” for MED-EL for both study maps. The two maps were then compared back and forth in live speech mode to ensure equivalent loudness perception, with global adjustments made to the upper stimulation levels as needed. While this method using live speech from the experimenter may introduce some variability compared to a constant sound source, it reflects commons clinical practice and allowed for real-time adjustments based on patient feedback. The test order of the conditions and the measures tested within each condition were randomized a priori. The participants used their own clinical processors throughout the study, but the same processor was used to test all conditions. All testing was completed using a loudspeaker at 0-degrees azimuth and 1-meter from the participant in a single walled sound booth. For bilateral CI users, the processor of the non-test ear was removed as none of the bilateral recipients had residual hearing in either ear. For bimodal users, the contralateral hearing aid was removed and the non-implanted ear was occluded with an E.A.R. plug and covered with a circumaural earmuff during testing as no participant had better than a moderate hearing loss in their non-implanted ear.
Test measures included the University of Washington Clinical Assessment of Music Perception (UW-CAMP) pitch discrimination sub-test (Kang et al., 2009), the Bamford-Kowal-Bench sentences in noise (BKB-SIN) test (Bench et al., 1979), and a test of musical sound quality and enjoyment subjective ratings (Berg et al., 2019), were tested once for each condition (twice per map). The UW-CAMP pitch discrimination sub-test uses a two-alternative forced choice, 1-up 1-down adaptive testing method (Kang et al., 2009). The stimuli included complex tones from a synthetic piano. Three base frequencies are tested, including 262 Hz (C4), 330 Hz (E4), and 392 Hz (G4), and a threshold in semitones is provided for each base frequency. The UW-CAMP pitch discrimination sub-test was presented at a calibrated level of 70 dB sound pressure level (SPL). One list-pair of the BKB-SIN test was completed per condition and the average score in dB signal-to-noise ratio (SNR) across the two lists was used for analyses. BKB-SIN sentences were presented at a calibrated level of 60 dB SPL in 4-talker babble. The sentences were presented at prerecorded SNRs that decrease in 3-dB steps. Musical sound quality and enjoyment were assessed for each condition using a randomly selected subset of ten 30-s song clips out of a group of 20 possible songs, all from various genres and styles, listed in Table 3. All song clips were presented at a calibrated level of 70 dB SPL and the participant was asked to make a subjective rating of sound quality and enjoyment immediately after they listened to each clip. The participant typed in two ratings after each clip, one for subjective sound quality and one for how much they enjoyed the song. A 10-point rating scale was used for both subjective sound quality (0 = very poor; 10 = very good) and enjoyment (0 = no enjoyment; 10 = high enjoyment) presented on a touch-screen computer. The average rating across the 10 clips for musical sound quality and enjoyment for each participant were used for analyses.
Songs Included in the Musical Sound Quality and Enjoyment Task.

Data Analysis
All statistical analyses were performed using R version 4.0.0 (R Core Team, 2021). Average scores across the two trials using the same study map on the UW-CAMP pitch discrimination subtest, the BKB-SIN test, and the musical sound quality and enjoyment subjective ratings were used for all analyses. Age, electrode-to-modiolus distance of the second most apical electrode, and angular insertion depth of the second most apical electrode were mean-centered and the musical sound quality and musical enjoyment ratings were converted to z-scores prior to analyses. To evaluate multicollinearity among potential predictors, variance inflation factor (VIF) scores were calculated for age, biological sex, electrode type (straight versus precurved), CI manufacturer (AB, Cochlear, and MED-EL), scalar location, electrode-to-modiolus distance of the second most apical electrode, and angular insertion depth of the second most apical electrode. All VIF scores were <5, except electrode type, which was consequently excluded from the regression models.
To assess the impact of deactivating the second most-apical electrode (experimental condition), linear mixed-effects models were implemented using the lme4 package in R. For each outcome measure, a null model with only participant as a random effect was compared to a model with condition (control vs. experimental) as a fixed effect and participant as a random effect. Model comparison was performed using likelihood ratio tests, and the intraclass correlation coefficient (ICC) was calculated to estimate the proportion of variance explained by individual differences.
To identify factors associated with individual benefit from the intervention, individual benefit scores were calculated by subtracting the control condition score from the experimental condition score for each outcome measure. These benefit scores were then used as dependent variables in backward stepwise regression models. Initial models included age, biological sex, CI manufacturer, scalar location, electrode-to-modiolus distance of the second most apical electrode, and angular insertion depth of the second most apical electrode as predictors. Model selection was performed using the Akaike Information Criterion (AIC). Variables were removed sequentially until no further improvement in AIC greater than 2 points could be achieved. Final models were evaluated for goodness-of-fit using R-squared values and residual diagnostics.
To examine relationships between outcome measures, Pearson correlations with 95% confidence intervals were calculated using baseline condition scores. Correlation strength was interpreted as weak (.01 ≤ r < .3), moderate (.3 ≤ r < 5), strong (.5 ≤ r < .7), or very strong (r ≥ .7).
Results
Comparison of Selective Apical Electrode Deactivation With Baseline Performance
Pitch Perception
Linear mixed-effects analyses comparing the null model to a condition-only model for the UW-CAMP pitch discrimination subtest average scores (Figure 2A) showed no significant effect of the experimental condition (χ²(1) = 0.03, p = .859). The experimental condition resulted in a non-significant mean difference of −0.03 semitones (SE = 0.19, 95% CI: −0.45 to 0.38) compared to baseline. Based on the 95% confidence intervals, 10 participants (33.3%) showed improvement with the experimental condition, 11 (36.7%) showed no reliable difference between conditions, and 9 (30.0%) performed better with the baseline condition.

Comparison of Performance with the Baseline and Experimental Conditions Across the Outcome Measures. Box Plots Show Median (Horizontal Line), Interquartile Range (Box), and Range (Whiskers) with Individual Participant Data Overlaid as Connected Points. (A) UW–CAMP Pitch Discrimination Thresholds Averaged Across Three Base Frequencies (262, 330, and 392 Hz), where Lower Values Indicate Better Performance. (B) BKB–SIN Speech–in–Noise Thresholds, where Lower Values Indicate Better Performance. (C) Musical Sound Quality Subjective Ratings on a 0–10 Scale, where Higher Values Indicate Better Perceived Quality. (D) Musical Enjoyment Subjective Ratings on a 0–10 Scale, where Higher Values Indicate Greater Enjoyment. Gray Shading Indicates the Interquartile Range for the Baseline Condition. Arrows Indicate the Direction of Better Performance for Each Measure.
For the 262 Hz base frequency, there was no significant effect of the experimental condition (χ²(1) = 0.24, p = .624). The experimental condition resulted in a non-significant mean difference of 0.12 semitones (SE = 0.24, 95% CI: −0.41 to 0.64) compared to baseline. Based on the 95% confidence intervals, 8 participants (26.7%) showed improvement with the experimental condition, 12 (40.0%) showed no reliable difference between conditions, and 10 (33.3%) performed better with the baseline condition.
For the 330 Hz base frequency, there was no significant effect of the experimental condition (χ²(1) = 0.37, p = .543). The experimental condition resulted in a non-significant mean difference of −0.22 semitones (SE = 0.36, 95% CI: −1.04 to 0.59) compared to baseline. Based on the 95% confidence intervals, 11 participants (36.7%) showed improvement with the experimental condition, 13 (43.3%) showed no reliable difference between conditions, and 6 (20.0%) performed better with the baseline condition.
For the 392 Hz base frequency, there was no significant effect of the experimental condition (χ²(1) = .001, p = .971). The experimental condition resulted in a nonsignificant mean difference of 0.01 semitones (SE = 0.25, 95% CI: −0.54 to 0.56) compared to baseline. Based on the 95% confidence intervals, 11 participants (36.7%) showed improvement with the experimental condition, 10 (33.3%) showed no reliable difference between conditions, and 9 (30.0%) performed better with the baseline condition.
Speech Perception in Noise
Linear mixed-effects model analysis comparing the null model to a condition-only model for BKB-SIN scores (Figure 2B) showed no significant effect of the experimental condition (χ²(1) = 0.40, p = .529). The experimental condition resulted in a non-significant mean difference of −0.25 dB SNR (SE = 0.40, 95% CI: −1.15 to 0.65) compared to baseline. Based on the 95% confidence intervals, 13 participants (43.3%) showed improvement with the experimental condition, 9 (30.0%) showed no reliable difference between conditions, and 8 (26.7%) performed better with the baseline condition.
Musical Sound Quality Subjective Ratings
Linear mixed-effects model analysis comparing the null model to a condition-only model for subjective musical sound quality ratings ( ) showed no significant effect of the experimental condition at the group level (χ²(1) = 3.33, p = .068). The experimental condition resulted in a non-significant mean increase of 0.26 points (SE = 0.14, 95% CI: −0.05 to 0.57) in ratings compared to baseline. Based on the 95% confidence intervals, 15 participants (50.0%) reported improved musical sound quality with the experimental condition, 11 (36.7%) showed no reliable difference between conditions, and 4 (13.3%) reported better sound quality with the baseline condition.
Musical Enjoyment Subjective Ratings
Linear mixed-effects model analysis comparing the null model to a condition-only model for musical enjoyment ratings (Figure 2D), no significant effect of the experimental condition was observed at the group level (χ²(1) = 0.97, p = .325). The experimental condition resulted in a non-significant mean increase of 0.13 points (SE = 0.13, 95% CI: −0.16 to 0.42) in ratings compared to baseline. Based on the 95% confidence intervals, 12 participants (40.0%) reported improved musical enjoyment with the experimental condition, 12 (40.0%) showed no reliable difference between conditions, and 6 (20.0%) reported better enjoyment with the baseline condition.
Impact of Demographic and Electrode Placement Factors on Intervention Benefit
Pitch Perception
Backward selection for UW-CAMP benefit scores resulted in a final model with no significant predictors (AIC = 89.10, BIC = 91.90). The null model was the most parsimonious, indicating that none of the examined factors reliably predicted individual benefit for pitch perception.
Speech in Noise
Similar to pitch perception, the backward selection process for BKB-SIN benefit scores resulted in a null model (AIC = 135.75, BIC = 138.55), with no significant predictors identified among the demographic or electrode placement factors.
Musical Sound Quality
The backward selection procedure for musical sound quality benefit identified two significant predictors: electrode-to-modiolus distance of the second-most apical electrode and scalar location. The final model is shown in Table 4. Participants with lower electrode-to-modiolus distance demonstrated greater benefit (β = −0.84, SE = 0.25, p = .002). Additionally, participants with electrodes positioned outside the scala tympani showed significantly greater benefit (β = 1.19, SE = 0.23, p < .001) compared to those with electrodes within the scala tympani. The final model (AIC = 54.81, BIC = 61.81) accounted for 48% of the variance in musical sound quality benefit (adjusted R² = .48, F(3, 26) = 9.88, p < .001).
Predictors of Intervention Benefit for Musical Sound Quality.

Musical Enjoyment
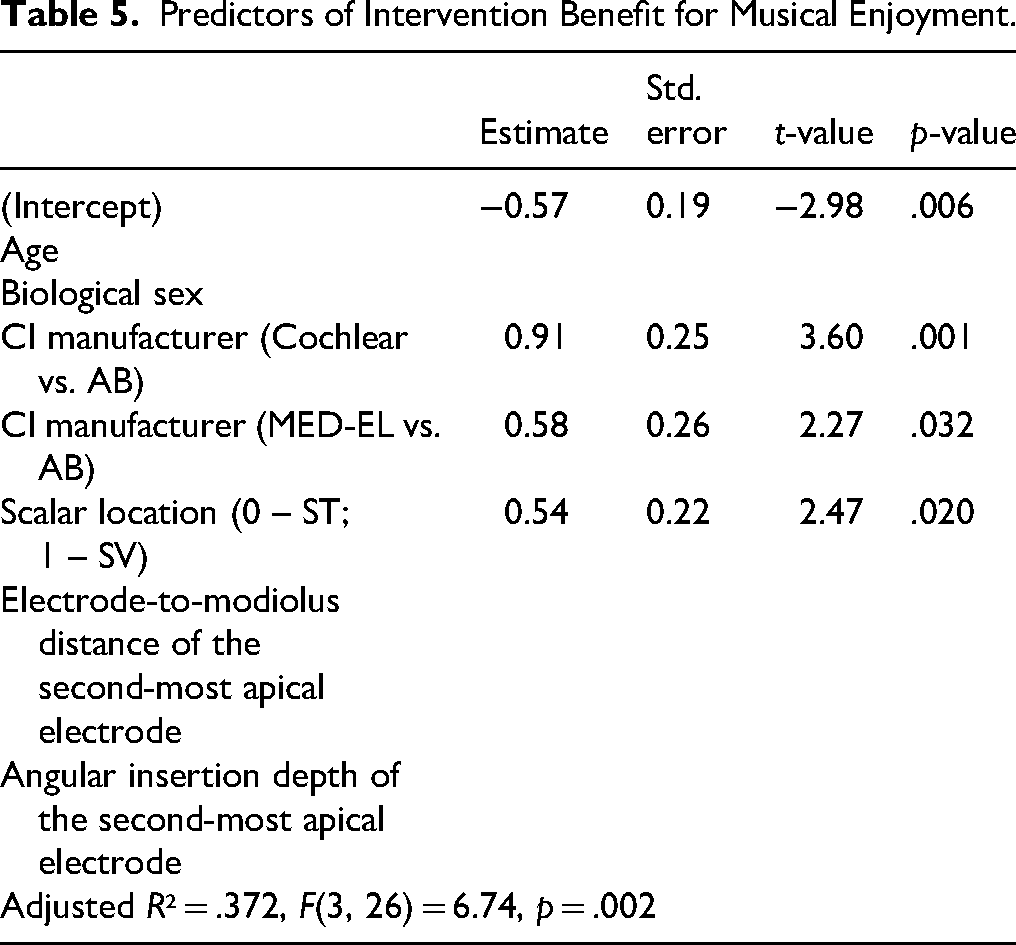
For musical enjoyment benefit, the backward selection identified CI manufacturer and scalar location as significant predictors. The final model is shown in Table 5. Participants with electrodes outside the scala tympani showed significantly greater benefit (β = 0.54, SE = 0.22, p = .020) compared to those with electrodes within the scala tympani. Additionally, CI manufacturer was a significant predictor, with Cochlear recipients showing greater benefit (β = 0.91, SE = 0.25, p = .001) compared to Advanced Bionics recipients, and MED-EL recipients also showing greater benefit (β = 0.58, SE = 0.26, p = .032) compared to Advanced Bionics recipients. The final model (AIC = 56.83, BIC = 63.83) explained 37% of the variance in musical enjoyment benefit (adjusted R² = .37, F(3, 26) = 6.74, p = .002).
Predictors of Intervention Benefit for Musical Enjoyment.
Relationships Between Outcome Measures
To examine relationships between outcome measures, we calculated Pearson correlations across all participants using baseline condition scores. Musical sound quality and musical enjoyment ratings were strongly correlated (r = .69, 95% CI: [0.43, 0.84], p < .001). Pitch discrimination was not significantly related to musical sound quality (r = −.15, 95% CI: [−0.49, 0.22], p = .42) or musical enjoyment (r = −.29, 95% CI: [−0.59, 0.08], p = .12). BKB-SIN performance was not significantly correlated with musical sound quality (r = −.14, 95% CI: [−0.48, 0.23], p = .46) but was moderately correlated with musical enjoyment (r = −.54, 95% CI: [−0.75, −0.22], p = .002), where better speech in noise (lower thresholds) was associated with higher enjoyment ratings.
Discussion
To improve CI outcomes, it is essential to develop programming strategies that enhance the accessibility and quality of spectral information provided by the CI. Currently, CIs are programmed using a one-size-fits-all approach, which may not be optimal for this goal. Prior research has established that speech and music have different requirements for channel independence and are affected differently by electrode positioning factors (Berg et al., 2019a, 2019b, 2020, 2021, 2022; Croghan et al., 2017; Fishman et al., 1997; Friesen et al., 2001; Gifford et al., 2022; Schvartz-Leyzac et al., 2017; Shannon, 2005; Smith et al., 2002). Apical channel interaction has been identified as a significant cause of performance decrements, suggesting the need to focus on this region in programming strategies (Bosen & Chatterjee, 2016; Goehring et al., 2021). Image-guided electrode selection has shown promise in promoting channel independence and improving spectral resolution (Jiam et al., 2019; Labadie et al., 2016; Noble et al., 2013, 2014, 2016; Saleh et al., 2013). However, the optimal subset of electrodes required for speech and music perception remains unknown. Therefore, the current study used a prospective, A-B-A-B design to (a) assess the impact of selective apical electrode deactivation on speech and music outcomes and (b) determine how demographic and electrode positioning factors influence the benefits of this intervention.
Contrary to our hypothesis, we did not observe significant group-level benefits from selective apical electrode deactivation for any outcome measure we tested. This finding contrasts with our previous work, which showed that participants with apical electrode deactivations demonstrated improvements in pitch perception (Berg et al., 2025). However, the current study deactivated only one electrode in the experimental map for all participants regardless of their individual electrode placement characteristics. Previous studies showing benefits using image-guided electrode selection typically deactivated more than one electrode with some deactivating as many as 14 electrodes, and tailored the approach to each patient's specific electrode placement (Labadie et al., 2016; Noble et al., 2013, 2014, 2016; Saleh et al., 2013). Even studies focusing on music perception that yielded significant pitch perception benefits deactivated up to four electrodes, though these deactivations were not necessarily concentrated in the apical region (Jiam et al., 2019). Given these findings, we theorize that more than one electrode deactivation is required to obtain noticeable benefits for most CI recipients, and the deactivation pattern should be tailored to each individual.
Despite the absence of significant group-level effects, our individual participant data revealed notable patterns that support this personalization theory. For pitch discrimination, 33% of participants showed improvement with the experimental condition. For speech perception in noise, 43% demonstrated improvement. Most notably, 50% of participants reported improved musical sound quality with electrode deactivation, and 40% reported enhanced musical enjoyment. These findings suggest that while not universally beneficial, selective apical electrode deactivation can provide meaningful improvements for a substantial subset of CI users.
In an attempt to identify characteristics of those who might benefit most from this intervention, we completed linear mixed-effects models to identify predictors of individual benefit for the outcome measures. For musical sound quality, participants with electrodes positioned outside the scala tympani and those with lower electrode-to-modiolus distance at the second-most apical electrode demonstrated greater benefit. For musical enjoyment, both scalar location and CI manufacturer were significant predictors, with participants having electrodes outside scala tympani and those using Cochlear or MED-EL devices showing greater benefit compared to Advanced Bionics recipients. These findings suggest that electrode placement characteristics and device type may influence the effectiveness of selective apical electrode deactivation. This underscores the need for a personalized approach to CI programming, rather than the current one-size-fits-all strategy.
Since our small sample size did not permit running comprehensive post-hoc statistical analyses, we completed visual inspection of our data, which revealed interesting patterns across different device types. For musical enjoyment and sound quality, participants with electrodes positioned outside scala tympani generally showed greater improvements with the experimental condition. When examining device-specific patterns, we observed that Cochlear recipients with lower electrode-to-modiolus distances appeared to derive greater benefit from selective apical electrode deactivation. For MED-EL recipients, we noted that those with longer electrode arrays (28 and 31.5 mm) tended to show more improvement than those with shorter arrays (24 mm).
For Cochlear recipients with lower electrode-to-modiolus distances, we hypothesize that perhaps this small intervention of deactivating a single electrode is effective enough to result in a meaningful reduction in channel interaction because they tend to have less inherent channel interaction than other array types (Berg et al., 2019). For MED-EL recipients, we hypothesize that perhaps this small intervention of deactivating a single electrode is effective enough to result in a meaningful reduction in channel interaction because recipients with longer electrodes (28 and 31.5 mm) tended to benefit more than shorter arrays (24 mm). Perhaps this benefit is afforded because these longer arrays have greater interelectrode spacing between contacts, so they inherently might have less channel interaction than arrays with closer spacing.
This pattern suggests that interelectrode spacing may play an important role in determining who benefits from this intervention. Longer arrays typically have greater spacing between contacts, which could influence how deactivating a single electrode affects overall channel interaction. Interestingly, angular insertion depth was not a significant predictor in our models, further supporting the hypothesis that interelectrode spacing may be more important than insertion depth alone. It is important to note that our models assume uniform neural survival across the electrode array, which may not reflect the biological reality. Future work should investigate how neural survival patterns and potential “dead regions” in the cochlea might influence which patients benefit from selective electrode deactivation.
Our correlation analyses revealed both expected and unexpected relationships between outcome measures. Musical sound quality and enjoyment ratings were strongly correlated (r = .69), consistent with previous work demonstrating that subjective music measures capture related constructs in CI users (Gfeller et al., 2008; Looi et al., 2012). Pitch discrimination was not correlated with subjective musical sound quality or enjoyment ratings, highlighting the need to consider music perception and appreciation as two distinct outcomes for CI users.
Perhaps most surprisingly, however, was BKB-SIN performance, which showed a significant moderate correlation with musical enjoyment (r = −.54) but not with musical sound quality. This selective relationship contrasts with previous studies reporting minimal associations between speech and music perception (Looi & She, 2010; Won et al., 2010). Interestingly, while we recently found that speech-in-noise correlated with general sound quality ratings (Berg et al., 2025), the current findings suggest speech perception abilities may selectively influence musical enjoyment without affecting perceived sound quality. This distinction could reflect shared cognitive demands between speech-in-noise processing and music listening (e.g., auditory scene analysis, selective attention) or general listening confidence that affects enjoyment but not sound quality.
An important limitation of the current study was the possible bias for map A, which was the clinical map used by participants in their daily lives. Despite randomizing the condition order and blinding participants to the map being tested, familiarity with the clinical map may have contributed to the non-significant group-level results. Future research should include an extended acclimatization period to determine if the benefits of selective apical electrode deactivation become more pronounced with adaptation.
Additionally, our study focused exclusively on deactivating the second most apical electrode, based on previous findings suggesting benefits from apical deactivation. However, this uniform approach may not be optimal for all participants. Future studies should consider combining selective apical deactivation with CT-guided approaches that account for individual electrode placement characteristics and assessments of auditory neural survival to determine the optimal subset of electrodes for each recipient.
Our findings also have important clinical implications. While not all CI users will benefit from selective apical electrode deactivation, those with specific electrode placement profiles may experience meaningful improvements in musical sound quality and enjoyment ratings. In clinical settings, this intervention could be implemented as a simple programming modification for selected patients, particularly those reporting poor musical sound quality or those who have disengaged from music since receiving their CI. During a standard clinical mapping appointment, a clinician can create a second program with the second-most apical electrode deactivated to save to the patient's processor. This allows the patient to take the experimental program home and compare it with their standard program, assessing potential improvements in musical sound quality during their everyday listening experiences without requiring additional specialized equipment or extended appointment times.
Conclusions
This study evaluated selective apical electrode deactivation as a strategy for reducing channel interaction and improving CI outcomes. While results did not show benefits at the group level, our findings suggest that certain subgroups of CI recipients may benefit from this approach. Specifically, patients with smaller electrode-to-modiolus distances, electrode arrays positioned outside scala tympani, and those using Cochlear or MED-EL brand devices may be the most promising candidates for this intervention. These findings emphasize the importance of personalized CI programming strategies over one-size-fits-all approaches. From a clinical perspective, deactivating the second most apical electrode represents a simple intervention that can be easily implemented in routine programming sessions for select patients. For patients reporting poor musical sound quality or those who have disengaged from music since receiving their CI, clinicians could consider a trial deactivation to allow the patient to test the program in their everyday environments. This approach moves away from a uniform programming strategy, instead offering a more individualized method of CI optimization. However, further research is needed to develop precise criteria for identifying optimal candidates for this intervention.
Footnotes
Ethical Considerations
Participants were consented using the protocol approved by the Vanderbilt University Institutional Review Board (#171526).
Consent to Participate
All participants provided written consent prior to study participation.
Consent for Publication
The authors have obtained written consent from all participants to publish this study.
Author Contributions
KAB: conceptualization, methodology, investigation, data analysis, visualization, writing—original draft, funding acquisition; JHN: software, resources, writing—reviewing and editing, funding acquisition; BMD: software, resources, writing—reviewing and editing, funding acquisition; RHG: conceptualization, methodology, resources, writing—reviewing and editing, funding acquisition, supervision.
Funding
We would like to acknowledge our funding sources that made this research possible, including F32 DC020347 (PI: Berg), R01 DC014037 (PI: Noble), R01 DC014462 (PI: Dawant) & R01 DC009404 (PI: Gifford).
National Institute on Deafness and Other Communication Disorders (grant number F32 DC020347, R01 DC009404, R01 DC014037, R01 DC014462).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
