Abstract
Introduction:
Neurogenic cough is a classification of chronic cough characterized by laryngeal hypersensitivity. Despite standard treatments, complete symptom resolution remains rare. Tetracaine lollipops, commonly used for oropharyngeal anesthesia, have not been studied for acute cough suppression. This study evaluates their efficacy in providing acute symptom relief in patients with neurogenic cough.
Material and Methods:
A retrospective, survey-based study was conducted with 23 patients diagnosed with neurogenic cough treated over a 1 year period with 0.5% tetracaine lollipops. Patients applied the lollipop to the posterior oropharynx for 20 to 30 seconds until numbing occurred. Cough relief was categorized as significant (≥70% reduction), moderate (40%-60%), mild (10%-30%), or no relief (0%).
Results:
Tetracaine lollipops reduced cough intensity by an average of 42%. Sixteen patients (69.6%) reported them effective, with 6 patients (26.1%) reporting significant relief, 6 patients (26.1%) reporting moderate relief, 9 patients (39.1%) reporting mild relief, and 2 patients (8.7%) reporting no relief. Cough suppression lasted 30 minutes to 2 hours in most cases (56.5%). A majority of the cohort (56.5%) expressed willingness to continue lollipop use, and 60.9% reported using them daily.
Conclusion:
Tetracaine lollipops are an effective short-term treatment option for acute neurogenic cough, providing improvement in most patients, with an average response reflecting moderate relief. They were well-adopted and frequently utilized daily. Further research is needed to refine their role in treatment.
Introduction
Neurogenic cough is a classification of chronic cough characterized by laryngeal hypersensitivity of the larynx and hypopharynx. Symptoms of neurogenic cough often progress through a self-perpetuating cough feedback loop, where recurrent coughing exacerbates neural irritation and further lowers the threshold for the cough reflex. Shapiro et al identified increased sensory nerve density in the airway mucosa of patients with neurogenic cough, while Irwin et al demonstrated heightened airway inflammation on histopathologic examination of the airway mucosa in patients with neurogenic cough.1,2 These findings suggest a pathophysiologic mechanism in which inflammation drives neural remodeling, resulting in heightened sensitivity and an exaggerated cough reflex. Effective management strategies of neurogenic cough focus on interrupting this cycle by reducing cough reflex sensitivity, subsequently minimizing cough frequency, and protecting neural pathways from further injury.
Treatment guidelines outlined by CHEST, the official journal of the American College of Chest Physicians, recommend neuromodulators and cough suppression therapy as first-line approaches for neurogenic cough in 2016.3,4 First-line neuromodulators include gabapentin, amitriptyline, and pregabalin.3,5 -7 Superior laryngeal nerve (SLN) blocks and laryngeal botulinum toxin A injections are often used to manage more refractory cases.7 -9 Despite significant advances in the management of neurogenic cough, complete symptom resolution remains rare, with most patients achieving only a tolerable level of relief.
Nebulized lidocaine is a widely-used anesthetic to suppress the cough reflex during endoscopic procedures. Existing meta-analyses have demonstrated its efficacy in alleviating cough symptoms, including chronic cough of unclear etiology or cough associated with conditions such as cancer, asthma, and chronic obstructive pulmonary disease (COPD). 10 Tetracaine lollipops are a widely available, compounded form of local anesthesia commonly used for oropharyngeal pain management after procedures or in the presence of mucosal lesions. They are generally used for short intervals of 20 to 30 seconds until a numbing effect is achieved and can be reused multiple times. Their application as gag reflex suppressants has been described in dental literature. 11 It is also commonly used for post-tonsillectomy pain control, especially in children due to the ease of use. 12 They are typically prepared by compounding pharmacies using a 0.5% tetracaine solution and flavored agents to improve palatability. To date, no studies have explored the use of tetracaine lollipops for cough suppression. This study aimed to investigate the efficacy of tetracaine lollipops as an adjuvant treatment for acute symptom relief in patients with neurogenic cough.
Materials and Methods
Study Design
This study utilized a retrospective, survey-based design to collect data from 23 patients diagnosed with neurogenic cough and treated with tetracaine lollipops. This study is approved by the Institutional Review Board at the University of Arizona (IRB #STUDY00003289).
All patients prescribed tetracaine lollipops between April 2024 and May 2025 for symptoms associated with laryngeal hypersensitivity, including cough, chronic sore throat, vocal cord dysfunction, and throat clearing, were identified through electronic medical record review. All patients were treated by the senior author at a single institution beginning in April 2024. The indications expanded over the last year to other manifestations of laryngeal hypersensitivity as mentioned above. Patients were counseled on the out-of-pocket cost during the visit and were not prescribed the lollipops if they declined based on price. From a group of 123 patients prescribed tetracaine lollipops, 55 met our inclusion criterion of neurogenic cough being the primary complaint, while also having other symptoms of laryngeal hypersensitivity, including vocal cord dysfunction (VCD), chronic sore throat, and throat clearing. Neurogenic cough was based on a history of having triggers of cough that are not normal, including talking, laughing, singing, deep inhalation, and immediate position change from supine to sitting. They were invited to participate in a survey (Supplementary File) assessing patient reported outcomes in June 2025 via messaging through the electronic medical record’s secure patient portal. For patients without portal access, we contacted them by email. Twenty-three out of 55 patients completed the survey, yielding a respondent rate of 41.8%.
Data collection occurred through a combination of chart review for baseline demographic and clinical information and a research electronic data capture (REDCap) survey, which gathered patient-reported outcomes and experiences with tetracaine lollipops. Demographic and clinical information extracted from medical records included age, gender, body mass index (BMI), history of smoking, inciting event (if applicable), prior cough suppression therapy, and prior treatments with associated response, including neuromodulators, SLN blocks, and laryngeal botulinum toxin A injections. Response to prior treatment was assessed on a scale of 1 to 5: 1 = no response, 2 = mild improvement, 3 = moderate improvement, 4 = significant improvement, 5 = complete resolution. The lollipops were given as an adjunct to the existing step-up protocol that the author had instituted in her own practice, which typically includes initiation of neuromodulators ± cough suppression therapy, then progressing to SLN blocks, and laryngeal botulinum toxin A injection. 7 The senior author initiated all procedural therapies in these patients. The lollipops were introduced at various points in this protocol as the patients had already received multiple modalities in the treatment of neurogenic cough by the time the lollipops were incorporated into the practice.
Nebulized lidocaine has been described in the literature as a temporizing measure to address cough symptoms. 13 However, its use requires a nebulizer, which may require user education. Additionally, the liquid form of topical lidocaine at 4% is not accessible on an outpatient basis due to its potential cardiotoxicity. Tetracaine lollipops potentially offer a more practical and patient-friendly alternative, providing immediate, situational relief without the need for specialized equipment. Tetracaine lollipops prescribed were formulated with a 0.5% concentration of tetracaine, and prescriptions were filled through Acacia Pharmacy, with 2 branch locations in Tucson, Arizona. Patients were instructed to consume the lollipop for 20 seconds at a time, repeating every 2 hours as needed. The lollipop can be stored in a regular medicine canister and be reused until it is gone.
Patients were instructed to rate their cough relief into increments of 10 percentage points of improvement, ranging from 0% to 100%. For the purpose of our analysis, the improvements were then broadly divided into 4 categories, including significant (≥70% reduction), moderate (40%-60% reduction), mild relief (10%-30% reduction), or no relief (0% reduction).
Survey Questions
In the REDCap survey (Supplementary File), participants provided information on the duration of their cough symptoms in years, the length of time they had been using tetracaine lollipops, and prior treatments they had attempted to manage their symptoms. Treatment options included oral steroids, bronchodilator or corticosteroid inhalers, proton pump inhibitors, benzonatate (Tessalon Perles), gabapentin, pregabalin, amitriptyline, and an “other” category for additional therapies. Participants also evaluated the effectiveness of tetracaine lollipops, reporting their frequency of use, the reduction in cough intensity after use, and the duration of relief provided after a single use. Participants identified the specific situations in which they used the lollipops, such as before social encounters, leaving home, important meetings or events, work, exercise, nighttime, during symptom flare-ups, as part of their daily routine, an “other” option was provided for additional uses. The survey also evaluated potential adverse effects, including the presence and duration of any swallowing difficulties. Financial considerations were also assessed, including out-of-pocket expenses, cost per lollipop, insurance coverage, and co-payment amounts. Lastly, participants were asked about their plans to continue using tetracaine lollipops and, for those who discontinued, their reasons for doing so.
Statistical Analysis
Statistical analysis was conducted via statistical product and service solutions (SPSS). Correlations between continuous variables were assessed using Pearson correlation coefficients. Correlations between ordinal variables and continuous variables were evaluated using Spearman’s rank correlation coefficients. Relationships involving ordinal and binomial variables were evaluated using Point-Biserial Correlation coefficients. Statistical significance was set at P < .05. Continuous variables are expressed in mean and standard deviation (SD). Categorical variables are expressed in ratio or percent.
Results
Demographics
A total of 23 patients with neurogenic cough treated with tetracaine lollipops completed our survey and were included in this study. The cohort’s mean age was 58.0 years (SD = 14.5), and 82.6% were female. Step-up therapy typically starts with neuromodulators (gabapentin, pregabalin, and amitriptyline) in addition to cough suppression therapy and then progresses to SLN blocks, and finally laryngeal botulinum toxin A injections. Twenty-one patients (91.3%) reported previous or current use of neuromodulators for cough symptom relief including gabapentin (91.3%), pregabalin (34.8%), and amitriptyline (30.4%). Additional treatments included cough suppression therapy in 8 patients (34.8%), SLN blocks in 9 patients (39.1%), and laryngeal botulinum toxin injections in 9 patients (39.1%). Mean follow-up time for this cohort was 6.35 months (range: 2.1-13.2 months). The average lapse between prescription and completion of the survey was 3.8 months (range: 0.2-10 months). Demographic data and prior treatments are illustrated in Table 1.
Cohort Demographics and Prior Treatments.
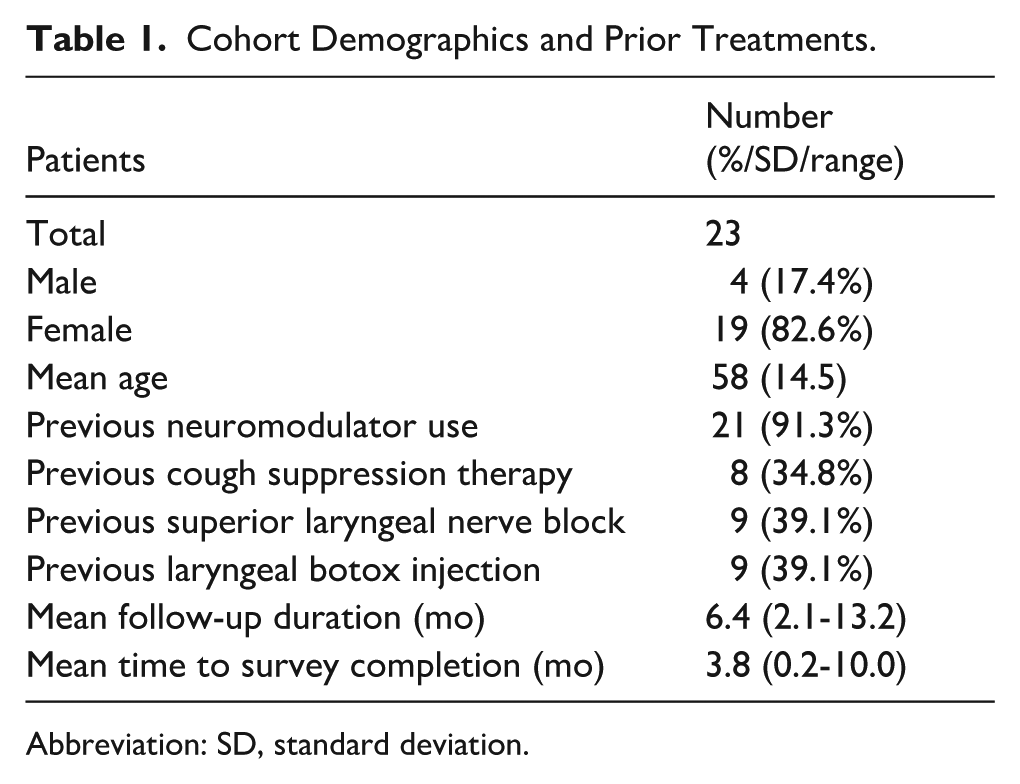
Abbreviation: SD, standard deviation.
Survey Findings
In this cohort of 23 patients, tetracaine lollipops achieved an average reduction in cough intensity of 42%. Twenty-one patients (91.3%) recorded some measurable improvement in cough symptoms, and 16 (69.6%) rated the lollipops “effective.” Six patients (26.1%) achieved substantial relief (≥70% cough reduction), 6 patients (26.1%) achieved moderate relief (40%-60% cough reduction), 9 patients (39.1%) reported mild relief (10%-30% cough reduction), and 2 patients (8.7%) reported no relief (0% cough reduction). These results are illustrated in Figure 1.
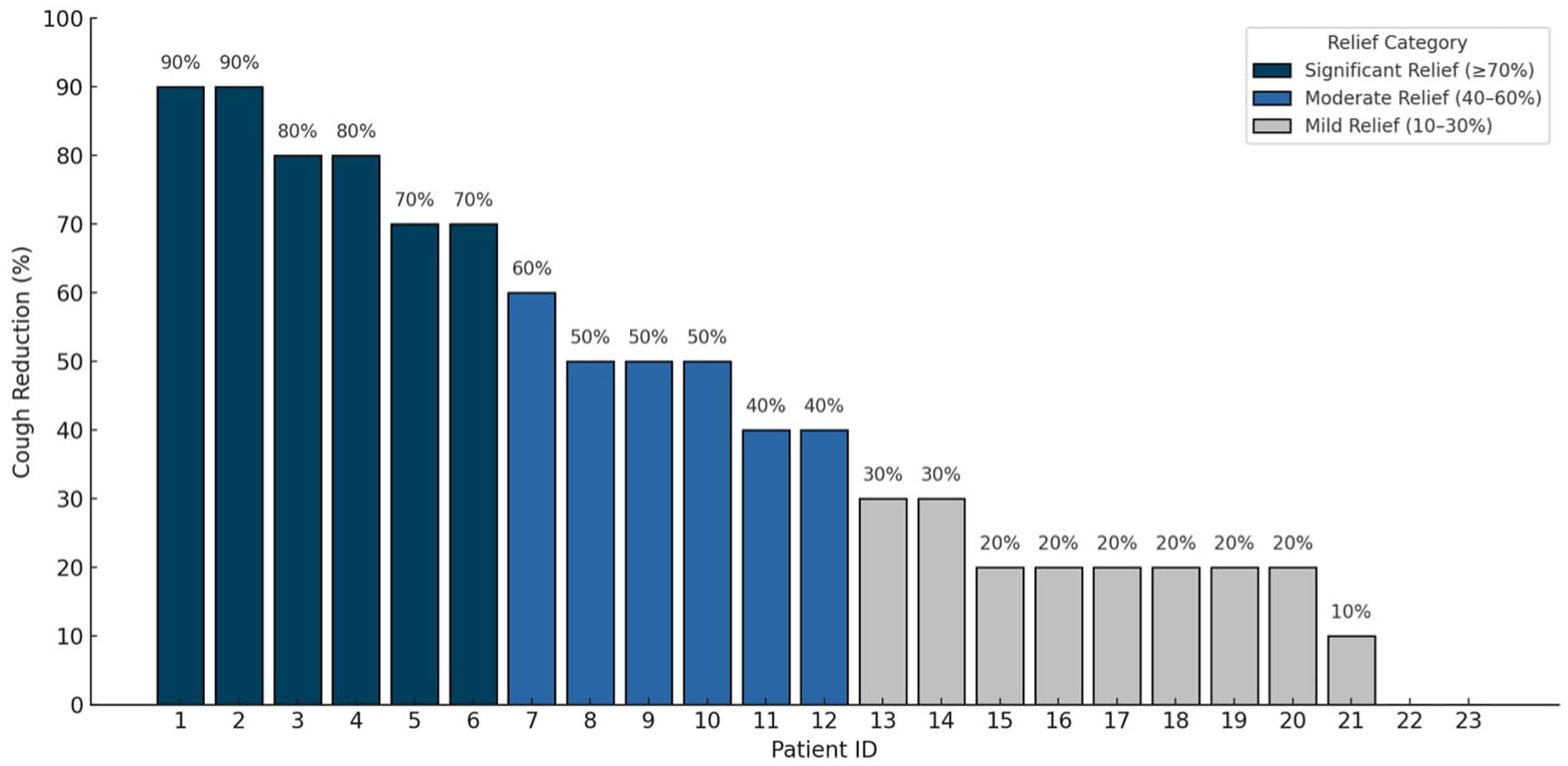
Patient-reported cough reduction in percent after tetracaine lollipop use.
After a single dose, relief duration was reported as <30 minutes by 4 patients (17.4%), 30 minutes to 1 hour by 9 patients (39.1%), 1 to 2 hours by 4 patients (17.4%), 2 to 3 hours by 4 patients (17.4%), and >5 hours by 2 patients (8.7%). Overall, 56.5% of patients experienced cough relief lasting between 30 minutes and 2 hours.
Thirteen patients (56.5%) chose to continue lollipop use while 10 (43.5%) decided to discontinue use. Among those who chose to discontinue, reasons included insufficient or transient relief (n = 6), side effects (tongue numbness [n = 1], dizziness [n = 1]), inconvenience of carrying the lollipops (n = 1), appropriate cough control with gabapentin alone (n = 1). Additionally, transient dysphagia after lollipop use was reported by 3 patients (13.0%), resolving within minutes. No other side effects were noted.
In terms of use frequency, 7 patients (30.4%) used the lollipops multiple times per day, 7 (30.4%) used them daily, and 9 (39.1%) used them weekly. Duration of use ranged from 1 to 6 months in 15 patients (65.2%), 6 to 12 months in 6 patients (26.1%), and over 1 year in 2 patients (8.7%). Patients most often used lollipops during symptom flare-ups (69.6%), before important meetings or events (30.4%), before social encounters (26.1%), or at night (21.7%).
Twenty-two patients (95.7%) paid out of pocket at a mean cost of $22.9/lollipop (range $12-$50); 1 patient had insurance coverage through Aetna with a $15 co-pay.
Modality Score and Outcomes
In this cohort of 23 patients, prior treatment history ranged from none to all 4 standard neurogenic cough therapies. Neurogenic cough therapies included neuromodulators, cough suppression therapy, SLN blocks, and laryngeal botox injections. We assigned each patient a modality score from 0 to 4 based on the number of therapies already trialed, with 0 indicating no prior treatment and 4 indicating that all 4 therapies had been trialed.
Two patients with a modality score of 0 experienced a mean cough reduction of 75% and both continued lollipop use. Patients with a modality score of 1 (n = 3) had a mean cough reduction of 13% (range, 0%-40%), and 33% elected to continue use. Those with a modality score of 2 (n = 10) reported a mean cough reduction of 48% (range, 20%-90%), with 50% electing to continue use. Patients with a modality score of 3 (n = 6) achieved a mean cough reduction of 37% (range, 10%-80%), and 50% chose to continue use. Two patients with a modality score of 4 experienced a mean cough reduction of 35% (range, 20%-50%), and both elected to continue use. Overall, 18 patients (78%) had been on 2 or more modalities before being prescribed tetracaine lollipops, emphasizing its role as a late-stage adjunctive therapy.
Point-biserial correlation revealed a small, non-significant positive trend between modality score and willingness to continue (r = 0.03; P = .91), whereas Pearson correlation showed no association between modality score and percent cough reduction (r = −0.16; P = .45). These results suggest that patients with more extensive prior treatment histories may be more inclined to continue tetracaine lollipop therapy. The magnitude of acute cough relief remained independent of prior treatment burden.
Cough Reduction Correlations
Patient-reported magnitude of cough reduction was strongly associated with continued lollipop use (r = 0.727, P < .001). In addition, patients using tetracaine lollipops for more than 6 months reported significantly-greater cough reduction score than patients using them for <6 months (ρ = 0.519, P = .005).
No statistically-significant correlations were observed between cough reduction and BMI ≥30 (r = −0.119, P = .581), smoking history (r = −0.076, P = .726), gender (r = 0.238, P = .261), daily use of tetracaine lollipops (r = −0.144, P = .505), or duration of cough symptoms (r = −0.322, P = .150).
There was no statistically-significant correlation between neuromodulator response and percent improvement (Spearman’s ρ = −0.016, P = .943) nor between neuromodulator response and duration of relief (Spearman’s ρ = −0.048, P = .837). Similarly, SLN response was not significantly associated with percent improvement (Spearman’s ρ = −0.101, P = .781) or duration of relief (Spearman’s ρ = 0.050, P = .892). Botox response also showed no significant correlation with percent improvement (Spearman’s ρ = 0.317, P = .373) or length of relief (Spearman’s ρ = −0.197, P = .585).
Discussion
Neurogenic cough remains a challenging condition to manage, with complete symptom resolution rarely achieved. Many patients endure significant psychosocial burdens, including anxiety, social embarrassment, and social isolation. 14 First-line therapies for neurogenic cough include neuromodulators and cough suppression therapy.3,4 SLN blocks and laryngeal botox injections are often recommended for more refractory cases.7 -9 The current treatment regimen effectively addresses sustained symptom management but lacks adequate options for acute symptom relief.
Tetracaine and lidocaine are both widely-used local anesthetics, though they differ significantly in chemical properties, metabolism, potency, and duration of action. Tetracaine, an amino-ester anesthetic, has high lipid solubility and moderate protein binding, resulting in a prolonged duration of local action (up to 200 minutes). Once absorbed into systemic circulation, tetracaine is rapidly metabolized by plasma cholinesterases, resulting in a short systemic half-life (~20-30 minutes). This makes tetracaine highly effective at providing localized anesthesia while minimizing systemic exposure. 15 Patients who experienced symptom relief with tetracaine lollipops in our cohort most commonly (56.5%) experienced relief between 30 minutes to 2 hours. In contrast, lidocaine, an amino-amide anesthetic, has lower lipid solubility and protein binding, leading to a shorter duration of local action (~1-2 hours). Its systemic metabolism occurs in the liver via cytochrome P450 enzymes, giving it a longer systemic half-life (~1.5-2 hours) compared with tetracaine. 16 The primary safety concern with all local anesthetics is the risk of central nervous system and cardiac toxicity, with potential symptoms ranging from lightheadedness and blurred vision to severe adverse events such as seizures or cardiac arrest.15,16 For lidocaine, serum concentrations should not exceed 5 mg/L to avoid adverse events. The generally-accepted safe dosage range for lidocaine is between 100 and 200 mg/dose, though patients with hepatic disease require close monitoring due to impaired metabolism and elimination.16 -18 While specific guidelines for tetracaine are less well-established, it is generally recommended not to exceed 1.5 to 3 mg/kg/dose. Tetracaine has no formal contraindications.
Tetracaine lollipops are formulated using a 0.5% tetracaine solution, equivalent to 5 mg of tetracaine per milliliter. According to a formula published in the International Journal of Pharmaceutical Compounding, 2050 mg of tetracaine hydrochloride is distributed across 38 lollipops, with each lollipop containing ~54 mg of tetracaine hydrochloride. 11 This corresponds to about 48 mg of active tetracaine, as 1.13 mg of tetracaine hydrochloride yields 1 mg of active tetracaine. Tetracaine lollipops are applied to the oropharynx in short intervals of 20 to 30 seconds/use, releasing a small fraction of the total tetracaine content with each application. Even if the entire lollipop were to dissolve and be absorbed, the total active tetracaine dose of 48 mg remains well within the established safe dosage range in a healthy weight adult.
Our study highlights the effectiveness and high patient acceptability of tetracaine lollipops for managing neurogenic cough. In our cohort, cough intensity was reduced by an average of 42%, with 14 out of 23 patients (60.9%) reporting a cough reduction of 30% or greater, and 6 patients (26.1%) experiencing substantial relief of cough symptoms (≥70% reduction). All patients who experienced at least a 30% improvement indicated a willingness to continue using tetracaine lollipops, with the exception of one individual who discontinued treatment after achieving sufficient symptom control with gabapentin alone. The lollipops were easily incorporated into patient routines, with 60.9% using them daily. They were most frequently used during symptom exacerbations (69.6%), prior to significant events (30.4%), before social interactions (26.1%), or at night (21.7%). For many patients, neurogenic cough contributes to social anxiety and withdrawal due to fear of public embarrassment. By offering even brief periods of reliable cough control, tetracaine lollipops enable patients to engage in social interactions with greater confidence, reducing isolation and improving quality of life.
In our cohort of 23 patients, 69.6% of patients reported that tetracaine lollipops effectively reduced their cough symptoms, with the average response indicating moderate symptom relief. This efficacy is comparable to established treatments for neurogenic cough. In our prior study of 56 patients with neurogenic cough managed using a step-up approach, symptom improvement was observed in 81.8% of those treated with neuromodulators (with or without cough suppression), 63.6% of those receiving SLN blocks, and 66.6% of those undergoing laryngeal botulinum toxin injections. 7 Similar response rates have been published in the literature.5,6 In a cohort of 28 patients with neurogenic cough treated with gabapentin, Lee and Woo observed symptom improvement in 68% of patients. 19 Simpson et al found that in a cohort of 18 patients with neurogenic cough treated with unilateral or bilateral SLN blocks, 83.3% experienced reduced symptom frequency and severity, with a 50% average reduction in Cough Severity Index scores after treatment. 8 Another study by Sasieta et al included 22 patients with neurogenic cough treated with laryngeal botulinum toxin injections and reported that 52% of the cohort experienced a >50% reduction in cough severity. 9
Patients who are elderly or unable to tolerate neuromodulators may benefit from tetracaine lollipops as a topical alternative for the treatment of neurogenic cough. Unlike neuromodulators, which require a titration period and can cause systemic side effects, tetracaine lollipops provides immediate, localized relief. Its topical mechanism minimizes systemic absorption, reducing the risk of sedation, dizziness, or cognitive impairment, which are particularly concerning in older adults. By offering a well-tolerated, easy-to-administer alternative, tetracaine lollipops can provide an opportunity for symptom relief in these patients.
Tetracaine lollipops in our study were prescribed from a single pharmacy with 2 branch locations, each offering different-sized lollipops (Figure 2). Patients in our practice have expressed preferences for one size over the other; the larger lollipops allow for more uses per lollipop, while the smaller lollipops provide more precise anesthesia and minimize the numbing of unintended structures. The cost of tetracaine lollipops ranged from $12 to $50, with an average price of ~$23/lollipop. For all but 1 patient in our cohort, the cost was not covered by insurance. One patient with Aetna insurance received partial coverage but still incurred a $15 co-pay. Despite these costs, no patients in the cohort reported discontinuing use due to financial concerns. This may be attributed to the ability to use a single lollipop multiple times before replacement. Although not formally queried in the survey, many patients have reported that a single lollipop can last up to 2 weeks even with regular use and much longer than that if used sporadically. The continued use among patients suggests that the perceived efficacy of tetracaine lollipops may justify the cost.

Tetracaine lollipop size comparison. A U.S. quarter (2.4 cm diameter) is included for scale. The left lollipop represents the larger size, while the right lollipop represents the smaller size.
A single tetracaine lollipop can last 2 weeks when used according to the instructions. The base ingredient of tetracaine is bitter, and therefore, flavoring agents must be added. The usual flavors are fruity. The sweetener can be stevia, and saccharine sodium. The senior author surveyed 2 compounding pharmacies, 1 in Tucson, AZ, and 1 in Little Spring, CO. The lollipops are made a batch at a time, in a quantity of about 30 lollipops. They are made from a lollipop base, and the active ingredient, tetracaine, and stabilizing agents are added. They require 24 to 48 hours of hardening time and technically easy to make. Many compounding pharmacies also offer mail ordering of the lollipops at United States postal service (USPS) rates after receiving a prescription from the prescriber’s office. Tetracaine lollipops’ mechanism of action is most closely akin to cough drops. Tetracaine works via sodium channel blockade, which targets nerve reflex sensitivity characteristic of neurogenic cough. Menthol, the main ingredient in many over-the-counter cough drops, activates transient receptor potential melastatin-8 (TRPM8) channels, inducing a cooling sensation and desensitizing nociceptive nerve fibers in the upper airway.20 -22 There is some evidence that it exhibits weak sodium channel-blocking activity, which could contribute to local anesthetic properties. Studies on menthol have shown evidence to provide short-term relief for mild, acute coughs. 22 Its effect is limited by tachyphylaxis, where repeated activation of TRPM8 channels desensitizes these receptors over time. By the time patients are referred to otolaryngology, they have typically-exhausted over the counter antitussives, including cough drops, antihistamines, and therapies for asthma and gastroesophageal reflux disease (GERD) with limited success.23 -25 Comparative studies evaluating placebo, menthol cough drops, and tetracaine lollipops across varying concentrations are needed to determine their relative efficacy and roles in neurogenic cough management.
This study is limited by its small sample size, retrospective survey-based design, absence of a control group, and potential recall bias. Additionally, patient-reported outcome measures (PROMs) were not collected, which limits the ability to objectively quantify symptom improvement. Future studies should include PROMs and objective cough reduction metrics, incorporate a control group, and utilize standardized tetracaine lollipop administration protocols to better characterize the efficacy and clinical utility of this treatment adjunct in managing neurogenic cough.
Conclusion
This study demonstrates that tetracaine lollipops are a safe and effective short-term treatment option for neurogenic cough, providing symptom improvement in most patients, with moderate relief on average. Most patients experienced relief lasting 30 minutes to 2 hours. The lollipops were well-adopted, with over 60% of patients using them daily or multiple times per day. Although each lollipop costs ~$23, the ability to reuse a single lollipop multiple times helps reduce overall expense. Further research is warranted to better define the efficacy of tetracaine lollipops and to identify patient characteristics associated with favorable response.
Supplemental Material
sj-pdf-1-ear-10.1177_01455613251395927 – Supplemental material for Investigating Tetracaine Lollipops as an Adjuvant Therapy Option for Neurogenic Cough
Supplemental material, sj-pdf-1-ear-10.1177_01455613251395927 for Investigating Tetracaine Lollipops as an Adjuvant Therapy Option for Neurogenic Cough by Nader Wehbi, Troy Weinstein, Austin Lever and Helena T. Yip in Ear, Nose & Throat Journal
Footnotes
Author Note
This study was presented as a poster at the Combined Otolaryngology Spring Meetings (COSM) 2025, May 14-18, New Orleans, LA, USA.
Ethical Considerations
This study was conducted at the University of Arizona College of Medicine – Tucson and was approved by the Institutional Review Board of the University of Arizona (IRB #STUDY00003289).
Author Contributions
Nader Wehbi: data curation; formal analysis; investigation; writing—original draft; writing—review and editing. Troy Weinstein: data curation; formal analysis; investigation; writing—review and editing. Austin Lever: methodology; supervision; writing—review and editing. Helena T. Yip: conceptualization; data curation; methodology; project administration; supervision; validation; writing—review and editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author on reasonable request. The data are not publicly available due to privacy and institutional restrictions.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
