Abstract
Objective:
The purpose of this study was to evaluate and clarify the effects of menopause on voice by comparing postmenopausal female participants who did not receive hormone therapy (HT) to premenopausal female participants.
Methods:
The study included 50 postmenopausal women who had not undergone HT, as well as 50 premenopausal women. The acoustic voice analysis considered fundamental frequency (F0), jitter, shimmer, harmonic-to-noise ratio (HNR), and noise-to-harmonic ratio (NHR). Jitter is variations of F0, and shimmer is of peak amplitude. Experts completed the Grade, Roughness, Breathiness, Asthenia, Strain scale for perceptual assessment, while participants self-evaluated their voice using the Voice Handicap Index-10 (VHI-10).
Results:
Postmenopausal women had lower acoustic parameters (F0 [P = .01], G [P = .037], R [P = .011], and S [P = .031]) scores. Age had a link with G (r = 0.303, P = .002), R (r = 0.417, P < .001), A (r = 0.297, P = .003), and S (r = 0.370, P = .001) and the F0 (r = −0.455, P = .001). There was no significant difference in the VHI-10 (P = .526), jitter (P = .216), shimmer (P = .920), HNR (P = .261), and NHR (P = .301) values.
Conclusion:
Postmenopausal women’s voice have lower frequency, and perceptually-lower grade, roughness, and strain than premenopausal women; however, these changes do not affect their quality of life.
Introduction
Differences in lung magnitude, vocal folds, and laryngeal cartilage dimensions between males and females, as well as gender hormones such as estrogen, progesterone, and testosterone, all contribute to voice variances, particularly as they vary over time. Several studies have found that hormones regulate the larynx, with hormone receptors on the vocal cords contributing to gender differences in voice. Androgen, estrogen, and progesterone receptors have been identified in epithelial cells, gland cells, and fibroblasts in the lamina propria. 1
The vocal fold comprises 5 layers (deep to superficial layers): thyroarytenoid muscle, deep lamina propria, intermediate lamina propria, superficial lamina propria, and the squamous epithelium. The deep and intermediate lamina propria group forms the vocal ligament. 2
Estrogen contributes to a higher pitch and a softer tone in the female voice. Menopause can alter laryngeal tissues, leading to muscle and mucosal atrophy, enlargement of vocal folds, and loss of white and viscous mucosa. Abitbol et al and Caruso et al reported dystrophy and atrophy of the vocal fold epithelium and edema of the vocal folds in postmenopausal women. Furthermore, muscle atrophy of the thyroarytenoid and thickening of the overlying mucosa result in added weight of the mucosa, leading to a lower vibratory frequency. Both men and women also experience edema in the superficial lamina propria. This change results in a weaker and less flexible voice, which may reduce vocal power due to structural changes in the vocal cords. Furthermore, there may be a link between the distribution of the extracellular matrix related to age and sex in the vocal folds and hormone levels. 3
Menopause is described as the cessation of menstruation and the end of the reproductive cycle caused by a lack of ovarian follicular activity, which results in lower estrogen and progesterone levels. The human larynx is affected by hormonal swings; therefore, participant selection is critical to reducing the consequences of aging of the voice. 4
The decline in estrogen and progesterone levels after menopause results in vocal abnormalities known as “postmenopausal voice syndrome.” This syndrome can include diminished vocal power, loss of high notes, limited vocal range, voice fatigue, and flattening of the vocal timbre. Edema, thinning, atrophy, and mucus thickening are among the most common symptoms of the vocal cords. Acoustically, a lower fundamental frequency (F0) and higher perturbation values (F0, jitter, shimmer, harmonic-to-noise ratio [HNR], and noise-to-harmonic ratio [NHR]) may be observed.5,6
Blood samples were obtained from premenopausal and postmenopausal women to assess biochemical markers and discovered that estrogen and progesterone levels were much lower in the menopause group. There was a statistically-significant difference between premenopausal and postmenopausal women in practically every characteristic of speech, including F0, jitter, HNR, and signal-to-noise ratio. The quality of voice was measured by hoarseness, roughness, and breathiness, with the difference in breathiness only statistically significant in premenopausal and postmenopausal women. The overall score, which is the sum of these 3 factors and is inversely related to voice quality, was considerably lower in premenopausal women, implying that postmenopausal women had poorer voice quality. 7
Menopausal women taking hormone replacement treatment (HRT) had a higher natural pitch than those not on HRT.8,9 Existing data indicate that postmenopausal women who use HRT have a higher F0. Women with a normal body mass index appeared to benefit more from the medication. 8
Natural changes in the larynx and phonation structures can or may not cause changes in the elderly’s voice, such as lower volume, breathy voice, quivering voice, vocal fatigue, and shorter maximum phonation time (MPT). 10
Atrophy of the thyroarytenoid muscle, epithelium, and elastic fibers, which causes inadequate glottal closure, bowing of the vocal cord, and pronounced vocal process, is known as presbylarynx. Laryngeal cartilage undergoes calcification. Structural ankylosis, narrowing, and degeneration, including thinning and abnormalities of the articular surface and disorganization of the collagen matrix of the cricoarytenoid joint, concluded that smoothness and movement of the cords may be affected. An increase in type 1 and a decrease in type 2 fibers of the thyroarytenoid muscles and further a decrease in type 1 and an increase in type 2 fibers of the posterior cricoarytenoid muscle were detected, indicating laryngeal muscle atrophy. An increase in epithelial thickness, yellowing due to loosening of the underlying lamina propria connection, and decreased epithelial cell density were observed. Salivary glands in the larynx atrophy degenerate. 11
Voice abnormalities are also associated with higher levels of depression, state/health anxiety, self-reported trait anxiety, and neuroticism than in the placebo group, according to consistent data. On the contrary, there is a significant difference in the psychological qualities between them. 12
The literature lacks research on menopausal vocal issues, particularly objective examinations of voice quality in postmenopausal women. This research aimed to investigate the prevalence of voice problems, assess voice quality, and assess voice abnormalities in naturally-postmenopausal women because the general characteristics of postmenopausal voice are important to continue understanding the natural history of voice.
Materials and Methods
This study was approved by the institutional research ethics committee, the Scientific Research Ethics Committee of the Adana City Training and Research Hospital, because the study involved human participants (approval no. 64, date: July 25, 2024) and the study was carried out following the Declaration of Helsinki of 2013.
Participants
Between August 1, 2024, and October 1, 2024, 50 postmenopausal study groups and 50 premenopausal female control group participants who visited the Otorhinolaryngology Clinic at Adana City Training and Research Hospital were recruited. Participants between the ages of 45 and 60 were selected and invited to participate in the study. None of the patients refused to participate in the study. We looked at the literature and determined the appropriate sample size, 100 patients. All participants gave their informed written informed consent. Information on age and medical history was collected.
Voice Assessment
Participants underwent objective and subjective voice assessment techniques to determine vocal quality and function: acoustic voice analyses (F0, jitter [%], shimmer [%], HNR, and NHR), Grade, Roughness, Breathiness, Asthenia, Strain (GRBAS) scale assessment, the Voice Handicap Index-10 (VHI-10), and laryngeal assessment. In all voice analysis tests, higher scores are worse.
Objective Voice Assessment
Acoustic Analyses
For an objective assessment, acoustic analyses were used. Participants’ voices were recorded and analyzed using a voice recorder (Sony ICD-PX; Sony Corporation, Tokyo, Japan) and a portable computer (Samsung Intel Core i5, Giheung, South Korea). After 10 minutes of rest, participants were invited to sit 15 cm from the lip surface in the silent speech and language therapy room. The sustained speech sample /a/ was produced using habitual and constant vocalization for 10 seconds following a strong inspiration. This was done 3 times. The participants were then asked to read a standard Turkish text for 40 seconds while speaking in a relaxed manner. Acoustic analyses were performed using the Praat software (Version 4.4.13; Boersma and Weenink, Amsterdam, The Netherlands). F0, jitter (%), shimmer (%), HNR, and NHR were assessed in the acoustic voice analyses.
Subjective Voice Assessment
Perceptual Evaluation
Auditory perceptual assessment of voice characteristics was performed with the GRBAS scale. The GRBAS scale consists of four 4-point grading scales (0, normal; 1, slight; 2, moderate; 3, severe) for each parameter. For interobserver reliability, the samples were rated by 2 speech and language therapists. The concordance values of the 2 ratings of 2 speech and language therapists were 91%. The perceptual rating was performed blindly by 2 speech and language therapists.
Voice-Related Quality of Life
We used the Turkish version of the VHI-10 questionnaire, which was created by Kilic et al, for the subjective assessment of voice-related quality of life (VRQoL). 13 Ten items of the questionnaire assess the emotional, functional, and physical characteristics of the participant’s voice, and the scores range from 1 to 4.
Laryngeal Assessment
The larynx of each participant was examined by a single otolaryngologist with 70° rigid telescopes (Richard Wolf, Knittlingen, Germany), and participants with menopausal symptoms, previous vocal training, allergy history, acute infections, vocal nodules, polyps, papillomas, paralysis, gastroesophageal reflux, as well as those with severe chronic illness or malignancy, were excluded from the study. Smoking is a well-known risk factor for the development of Reinke’s edema and/or polypoid corditis, which can dramatically drop pitch. Given its association with voice and how this may mimic postmenopausal changes, smokers were excluded from the study. The participants confirmed menopause 12 consecutive months of amenorrhea after final menstruation by the gynecologist. Participants in the menstrual period were excluded. The premenopause group did not take hormone therapy (HT) of any kind, including oral contraceptive pills. Serum hormone levels in postmenopausal participants were consistent with menopausal values. The duration of menopause was between 4 and 12 months.
Statistical Analysis
All analyses were performed using IBM Statistical Package for Social Sciences statistics for Windows, version 26 (IBM Corporation, Armonk, NY, USA), and P < .05 was considered statistically significant. Continuous variables were summarized using the mean, standard deviation, median, and interquartile range, while categorical variables were summarized using frequency and percentage. The normality of the data distribution was assessed with the Shapiro-Wilk test. For parameters that followed a normal distribution, comparisons of means between 2 groups were performed using the independent samples t-test, while for non-normally-distributed parameters, the Mann-Whitney U test was applied. The chi-squared test was used to evaluate categorical variables. Numerous voice parameters (acoustic, perceptual, and self-reported) were tested with the Bonferroni correction. Correlation analyses were used to determine the direction and strength of the relationships between the study parameters.
Results
Characteristics of the Participants
The postmenopause group had an average age of 57.76 ± 8.12 years, while the control group had an average age of 57.92 ± 8.73 years, and the difference was not statistically significant (P = .580).
Comparison Between Premenopausal and Postmenopausal Women
Objective Voice Assessment
Acoustic Analyses
However, the mean value of F0 was found to be significantly lower in the postmenopause group than in the control group (P < .001). There were no significant differences in the jitter values between the postmenopause and control groups (P = .210). Similarly, the HNR values did not differ significantly between the groups (P = .261). Examination of the median and interquartile ranges for the shimmer values indicated that there was no statistically-significant difference between the postmenopause and control groups (P = .920). Similarly, no significant differences were found in the NHR values between the 2 groups (P = .301; Table 1).
Comparison Between Voice Parameters of the Postmenopause and Control Groups.
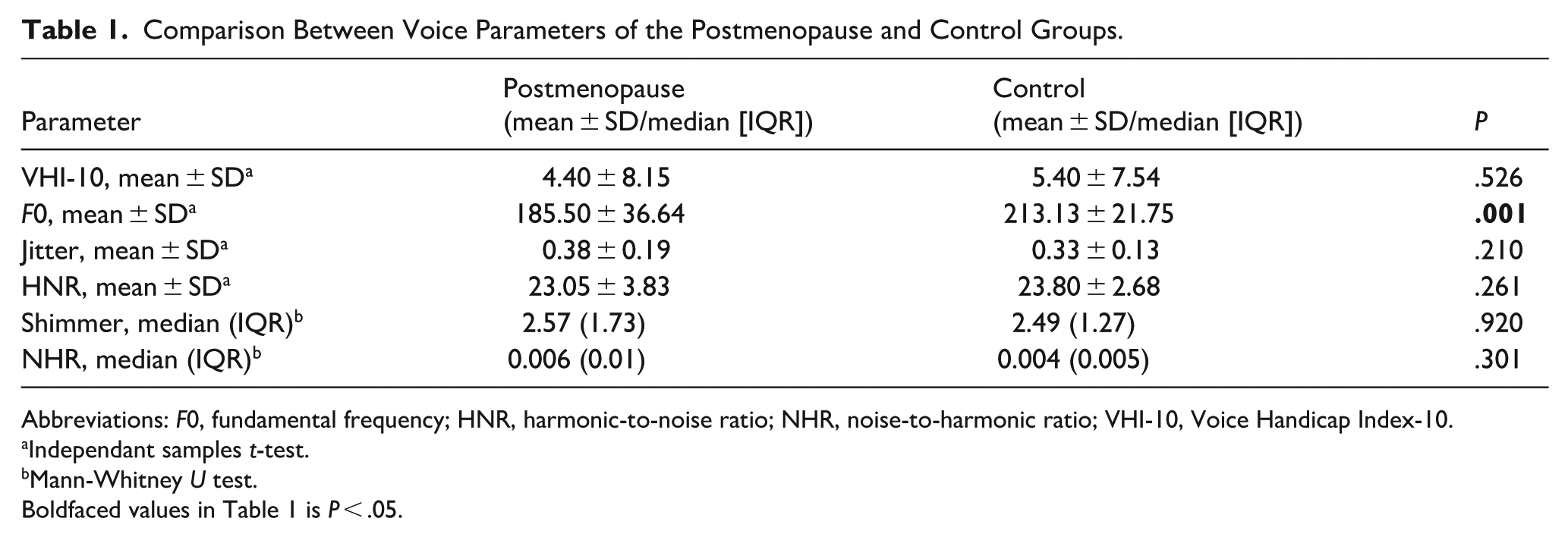
Abbreviations: F0, fundamental frequency; HNR, harmonic-to-noise ratio; NHR, noise-to-harmonic ratio; VHI-10, Voice Handicap Index-10.
Independant samples t-test.
Mann-Whitney U test.
Boldfaced values in Table 1 is P < .05.
Subjective Voice Assessment
Perceptual Evaluation
When GRBAS voice evaluation ratings were compared between the menopausal and control groups, the variable “current voice” showed no significant change (P = .803). We asked the patients how their voice was today and scored them as 0 normal, 1 mildly impaired, 2 moderately impaired, and 3 severely impaired. For the G score, 74% of postmenopausal participants received a score of 0, compared with 90% in the control group, and this difference was statistically significant (P = .037). The control group included fewer participants in category 0 (10%) than in the postmenopausal group (26%). For the R score (grid), 86% of the control group scored 0 compared with 64% in the postmenopausal group, which was statistically significant (P = .011). The scores of B did not show significant differences between the groups (P = .645). The A scores did not reveal significant differences between the groups (P = .070). Finally, 92% of the control group scored 0 on scale S, compared with 74% in the postmenopausal group, and this difference was statistically significant (P = .031; Table 2).
Comparison of G, R, B, A, S Scores and Current Voice Assessment in Postmenopause and Control Groups.
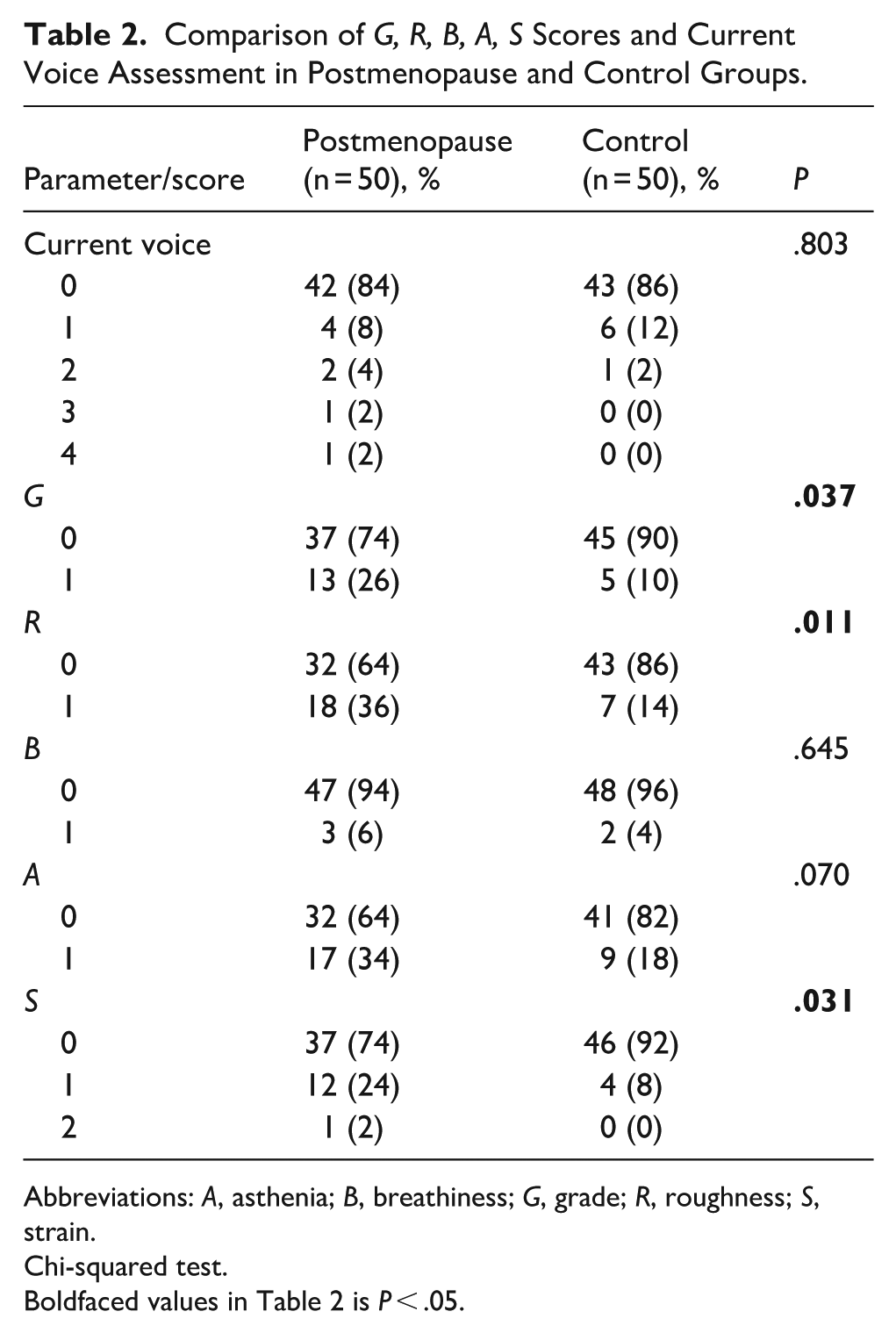
Abbreviations: A, asthenia; B, breathiness; G, grade; R, roughness; S, strain.
Chi-squared test.
Boldfaced values in Table 2 is P < .05.
Voice-Related Quality of Life
In terms of VHI-10 scores, there were no statistically-significant differences between the average scores of the postmenopause and control groups (P = .526).
Laryngeal Assessment
Observation by laryngoscopy continues to be a subjective assessment technique that lacks the sensitivity to detect small changes in the mucosa. In our study, no differences were found between premenopausal and postmenopausal women.
Age and Voice Parameters
Age was found to have a moderate positive association with voice metrics such as G (r = 0.303, P = .002), R (r = 0.417, P < .001), A (r = 0.297, P = .003), and S (r = 0.370, P < .001). Furthermore, there was a moderate negative connection between age and the F0 parameter (r = −0.455, P < .001). There were no significant correlations between age and current voice (r = 0.153, P = .128), B (r = 0.118, P = .242), VHI-10 (r = 0.033, P = .742), jitter (r = 0.107, P = .291), shimmer (r = 0.068, P > .05), NHR (r = 0.173, P = .085), or HNR (r = −0.076, P = .451; Table 3).
Correlation Analysis to Determine the Relationship Between Voice Parameters and Age.

Abbreviations: A, asthenia; B, breathiness; F0, fundamental frequency; G, grade; HNR, harmonic-to-noise ratio; NHR, noise-to-harmonic ratio; R, roughness; S, strain; VHI-10, Voice Handicap Index-10.
Spearman correlation analysis.
Boldfaced values in Table 3 is P < .05.
Discussion
There are not enough studies on menopause-related voice changes. Our study aimed to fill this gap by evaluating and comparing the vocal characteristics of postmenopausal women with those of premenopausal women. In this study, we found that in postmenopausal women, the acoustic parameters (F0 of the /a/ vowel, G, R, and S scores) on the GRBAS scale were significantly lower than in premenopausal women (P < .001).
Although postmenopausal women have good quality voices, their F0 values, grade, roughness, and strain scores are lower. A decrease in the F0, G, and R scores can be attributed to edema of the vocal folds, while a decrease in the S score can be the result of atrophy of the thyroarytenoid muscle, leading to a softer and weaker voice in postmenopausal women. Our understanding may be correct, but more studies are needed, particularly focusing on laryngeal cytological aspects in postmenopausal women using vocal epithelium smears and examining the presence of gender hormone receptors in the female larynx. There may also be confounding factors, such as age. We excluded acute infections, allergy, vocal nodules, polyps, papillomas, paralysis, and gastroesophageal reflux, as well as individuals with severe chronic diseases or malignancies by laryngoscopy, but we did not observe a difference between the 2 groups.
In the study by Shankar et al, it was found that postmenopausal female participants had lower voice quality, with significant differences in F0, pitch, and perturbation. 7 However, no significant differences were found in the disturbance of voice intensity, nor was there a significant difference between estrogen and progesterone levels and voice quality. In our study, we found that postmenopausal women have a lower F0 but no difference in perturbation parameters.
In the study by D’haeseleer et al, premenopausal women were found to have a smaller frequency and intensity range and a lower F0. 14 By comparing premenopausal and postmenopausal women, they found that postmenopausal women who did not receive HT had a lower fundamental speaking frequency and were able to produce sounds at lower frequencies. In analysis of covariance (ANCOVA), postmenopausal women exhibited significantly-lower mean phonation quotient and F0 during reading, but no significant difference in voice quality. The homogeneity of the slope assumption was tested before ANCOVA. The reliance of ANCOVA can be discussed; therefore, we used the Bonferroni correction for further analysis. In our study, we did not include women who received HT; we selected naturally-menopausal women. Similarly, we found a lower F0 and no significant differences in voice quality.
In a meta-analysis by La et al investigating the effects of menopause on vocal measurements, 8 articles were evaluated, focusing on 6 voice metrics: mean F0 in speech and sustained vowel /a/ frequency perturbation measures, jitter, shimmer, NHR, and MPT. 15 Compared with those in premenopausal women, the speaking and sustained vowel /a/ frequencies in postmenopausal women were 0.94 and 1.18 semitones lower, respectively. Despite the significance, these reductions remained within a range that did not fall below the threshold for perceivable differences. There were no significant differences in jitter, shimmer, NHR, or MPT. In our study, the mean F0 was lower, but no significant differences in jitter, shimmer, and NHR were observed. Evaluating acoustic measurements that reflect a single aspect of voice production can obscure the effects of hormonal changes during menopause, as various interacting factors affect voice production, and acoustic measurements alone are limited in predicting the association of steroid hormones with the state of vocal folds.
Although cell degeneration of the vocal folds is observed during menopause, there is considerable variability between individuals. This suggests that hormonal changes in the female larynx during menopause are highly individual. Furthermore, it is not clear how changes in the vocal cord mucosa are related to the acoustic properties of the voice.
Mendes-Laureano et al divided women into groups based on whether they were premenopausal or postmenopausal and whether they used HT. 16 They compared the sustained vowels (/i/ and /e/) and the HNR between the groups. No significant differences were found in the F0 or HNR values, nor changes were observed in the vocal cords on video laryngoscopy (VLS). We excluded HT participants from our study and found no differences in HNR values nor any changes in vocal folds on indirect laryngoscopy in postmenopausal women.
Meurer et al compared premenopausal and postmenopausal periods and found no significant differences in voice, F0, intensity, formant, or verbal diadochokinesis postmenopause. 17 However, we found low F0, grade, roughness, and strain of voice in our postmenopausal participants.
Lindholm et al found a reduction in the average F0 and the sound pressure level (SPL) for sustained sounds in postmenopausal women compared with those in premenopausal women. 18 Uniformly, we find a reduction in average F0 in postmenopausal women. To support this, Linville and D’haeseleer et al also demonstrated a decrease in speaking fundamental frequency (SFF) postmenopause.19,20 D’haeseleer et al noted that postmenopausal women using HT had an SFF of ~13 Hz, while those not using HT had an SFF of around 14 Hz. 20 A non-significant decrease in SFF was observed in postmenopausal women with a higher BMI, possibly due to lower estrogen levels when BMI is high.
Symptoms and prevalence of menopause-related changes in voice vary. In surveys, 77% of elite professional voice users, such as singers, reported loss of high notes, hoarseness, and reduced vocal flexibility during menopause. 8 However, the prevalence of the survey may appear high due to low response rates. Among nonsingers, symptoms included reduced pitch range, decreased voice volume, vocal fatigue, difficulty in voice control, reduced intensity, and voice breaks. Dysphonia was observed in 17% of menopausal women, while 46% experienced voice changes, and 33% reported vocal discomfort. In VLS, increased mucosal viscosity, swelling, Reinke’s edema, and edema were observed along the free edge of the vocal cords, while these changes were absent in postmenopausal women without voice complaints. 8 Our study did not include elite professional voice users. We did not observe any changes in indirect laryngoscopy in postmenopausal women.
A reduction in F0 between 10.1 and 13.4 Hz was observed in reading and sustained /a/ sounds in menopause. This decrease was 2 standard deviations below the mean for both sinus tones and sounds in the 160 to 200 Hz range. This drop affects women differently, whether they are elite singers or professional voice users. There was no significant difference in the frequency perturbation (jitter, shimmer, HNR) or MPT values. 8 Comparably, we did not observe a significant difference in frequency perturbation values.
Hamdan et al found a higher habitual pitch and jitter in women using HT, but did not observe differences in other parameters. 9 They also reported greater vocal fatigue and throat dryness after menopause. But we found no difference in jitter or other parameters.
Basilio et al observed a decrease in the total voice symptoms score, a higher functional VHI score, and a lower socioemotional VRQoL score. 21 In our study, we used the VHI-10 for the perceptual evaluation of voice quality and found a significant difference in voice and quality of life in postmenopausal women.
Ferraz et al found lower average F0, maximum and minimum F0 values, higher variability of F0, and standard deviation. 22 For voiceless sounds, a lower MPT was observed for postmenopausal women, with no differences in the values of the glottal-to-noise excitation ratio between the groups. Postmenopausal women were found to have voices that were more pleasant, but lower in pitch and rougher in quality. There were no differences in total and detailed voice query language scores. Perceptually, mild roughness, strain, and instability were observed in both groups. Additionally, the lower average F0, perceptual grade, roughness, and strain of the postmenopausal group were significantly lower in our study.
In the study by Sovani and Mukunan, postmenopausal women were found to have lower values of F0 and SFF during reading and spontaneous speech, with higher values of NHR and voice turbulence index. 23 Lower VHI scores were observed in menopause. In our study, postmenopausal women were found to have lower F0 but no difference in NHR or VHI scores.
Raj et al reported lower values of F0, fundamental resonance, and maximum phonation duration and higher S/Z ratios, with no differences found in other parameters. 24 We observed a low F0, but no differences in other parameters.
Menopause brings hormonal changes as well as age-related consequences. With aging, laryngeal muscles shrink, cartilage stiffens, collagen fibers decrease, and vocal cords become stiffer. 25 The effects of aging and hormonal changes in menopause are mixed.
However, voice is both an acoustic and a psychosocial construct. Certain age intervals through which people go affect the mental, emotional, and social health of the individual. Therefore, it is vital to find and treat the factors that lead to changes in vocal behavior.
Menopause is an intricate and varied condition that affects women in a variety of ways. Women suffer a variety of physical symptoms that can have a substantial influence on their quality of life. Menopause has a substantial psychological influence, including mood swings and anxiety. Women may feel isolated and disconnected from their social networks as a result of losing their sense of identity or social role. 12
Women in menopause have a variety of symptoms as a result of hormonal changes, which can have a substantial influence on their daily lives, particularly their occupational lives. 12
The incorporation of acoustic characteristics that define voice qualities is greatly complicated and multifaceted, and the variances found in the literature can be resolved through flaws in the methodologies applied until now, and that may be driven by various variables that include various techniques deployed to calculate, shifts in recording processes, or instrumentation.
In conclusion, F0, grade, roughness, and the strain of voice quality were found to be significantly lower in postmenopausal women. Aging may affect the voice of women. Aged women’s voices may have lower frequencies, resulting in a weaker voice. However, vocal alterations are not to be associated with the quality of life of postmenopausal women.
Footnotes
Acknowledgements
The author thanks all participants who participated in the study.
Ethical Considerations
This study was approved by the institute research ethics committee, the Scientific Research Ethics Committee of the Adana City Training and Research Hospital, because the study involved human participants (approval no. 64, date: July 25, 2024), and the study was carried out according to the Declaration of Helsinki 2013.
Consent to Participate
Written informed consent was obtained from all participants.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data supporting this study are not publicly available due to ethical and legal restrictions.
