Abstract
This case report details a rare and severe presentation of refractory otomastoiditis caused by Mycobacterium abscessus in an immunocompetent pediatric patient who presented with a 1-month history of left ear pain, swelling, and fever unresponsive to antibiotics. Computed tomography imaging was suggestive of coalescent otomastoiditis, and she underwent urgent left mastoidectomy, subperiosteal abscess drainage, and myringotomy with tube insertion, followed by conventional antibiotic management. Despite these interventions, she remained symptomatic and cultures revealed M. abscessus 1 week later. Multidisciplinary management involved serial microdebridement and prolonged multidrug antimicrobial therapy with shared decision-making between otolaryngology, infectious diseases, and international experts. Pharmacological management was complicated by adverse effects including aminoglycoside-induced hearing loss, myelosuppression, and gastrointestinal intolerance requiring drug substitutions. Eighteen months after initial presentation, revision mastoidectomy, canaloplasty, mastoid obliteration, tympanoplasty, and ossiculoplasty were performed for the eradication of residual disease and reconstruction. At follow-up, there was no disease recurrence although left-sided moderate-to-severe mixed hearing loss persisted. This case highlights the importance of early recognition and coordinated medical-surgical interventions in atypical presentations of mastoiditis.
Case Presentation
An eight-year-old, otherwise healthy, girl presented to the emergency department in Southwestern Ontario, Canada, with a one-month history of left ear pain, swelling, and fever unresponsive to oral and topical antibiotic therapy. Her left ear was proptotic with postauricular erythema and fluctuance. Computed tomography imaging demonstrated complete opacification of the left mastoid and middle ear, bony erosion of the ear canal, mastoid cortex, and tegmen mastoideum, along with dural thickening and a subperiosteal abscess, suggestive of coalescent otomastoiditis. She underwent urgent left mastoidectomy, subperiosteal abscess drainage, and myringotomy with tube insertion. The patient was treated with topical ciprofloxacin/dexamethasone and ceftriaxone. Magnetic resonance imaging (MRI) with gadolinium showed no intracranial extension (Figure 1), and she was discharged with 4-6 weeks of intravenous (IV) antibiotics.
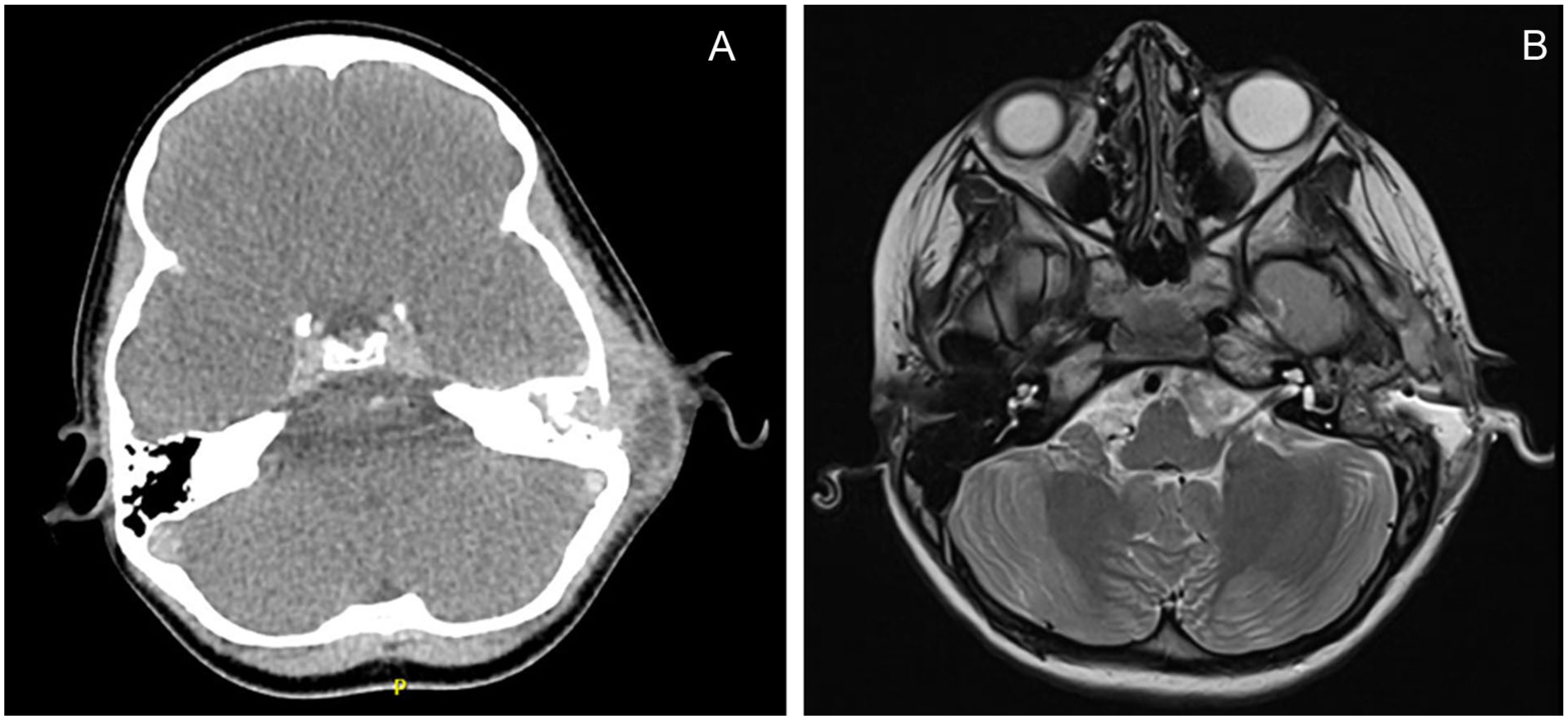
Initial preoperative and postoperative imaging. (a) Contrast-enhanced axial computed tomography of bilateral temporal bones showing complete opacification of left mastoid air cells and middle ear with erosion of the lateral wall of the mastoid process with a rim enhancing fluid collection laterally. (b) Magnetic resonance imaging with gadolinium following left mastoidectomy with enhancement of dura adjacent to mastoidectomy site and otherwise normal brain parenchymal signal.
The patient was readmitted 1 week later with fever, mastoid tenderness, and otorrhea, and the incision was drained and irrigated. Cultures from the initial surgery tested positive for Mycobacterium abscessus and histopathological analysis demonstrated features consistent with mycobacterial infection (Figure 2). Baseline hearing assessment demonstrated normal right-sided hearing and mild-to-moderate left-sided hearing loss. Differentiating conductive from sensorineural hearing loss was challenging since the patient could not tolerate bone conduction testing; it was presumed conductive based on the presentation. Empiric therapy was initiated with amikacin, meropenem, and clarithromycin (Figure 3). After identifying a functional erm(41) gene, clarithromycin was substituted with linezolid. Meropenem was switched to imipenem-cilastatin, and tigecycline was started alongside topical tigecycline and imipenem-cilastatin. Repeat audiology revealed worsening moderate low-frequency left-sided conductive hearing loss and middle ear dysfunction.
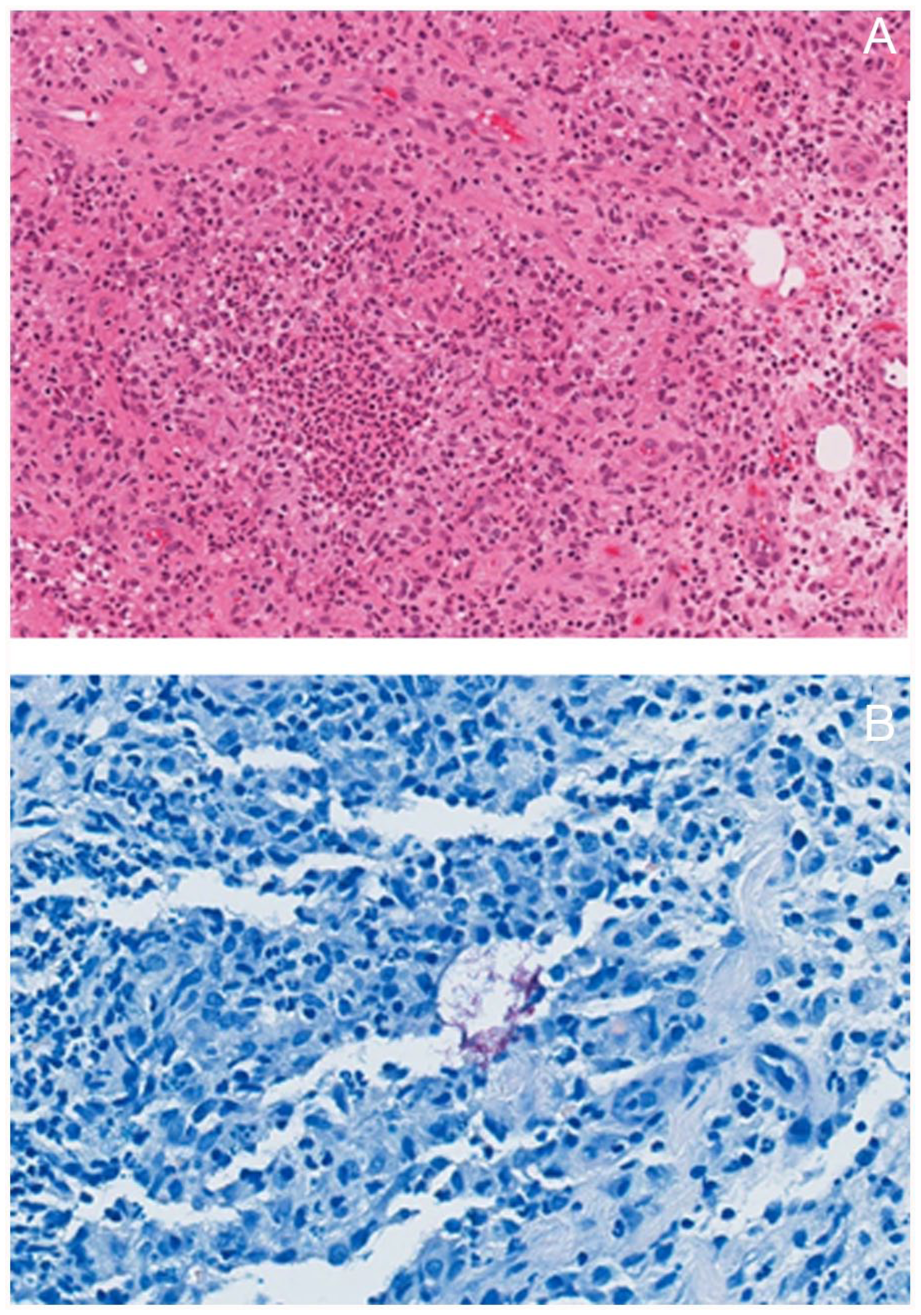
Histochemical staining of diagnostic samples from initial surgery. (a) Hematoxylin and eosin (20× magnification) showing granulation tissue with edematous, vascular-rich connective tissue with prominent mixed acute and chronic inflammation. (b) Ziehl-Neelsen (40× magnification) showing rare aggregates of red/pink rod-shaped microorganisms consistent with mycobacteria.
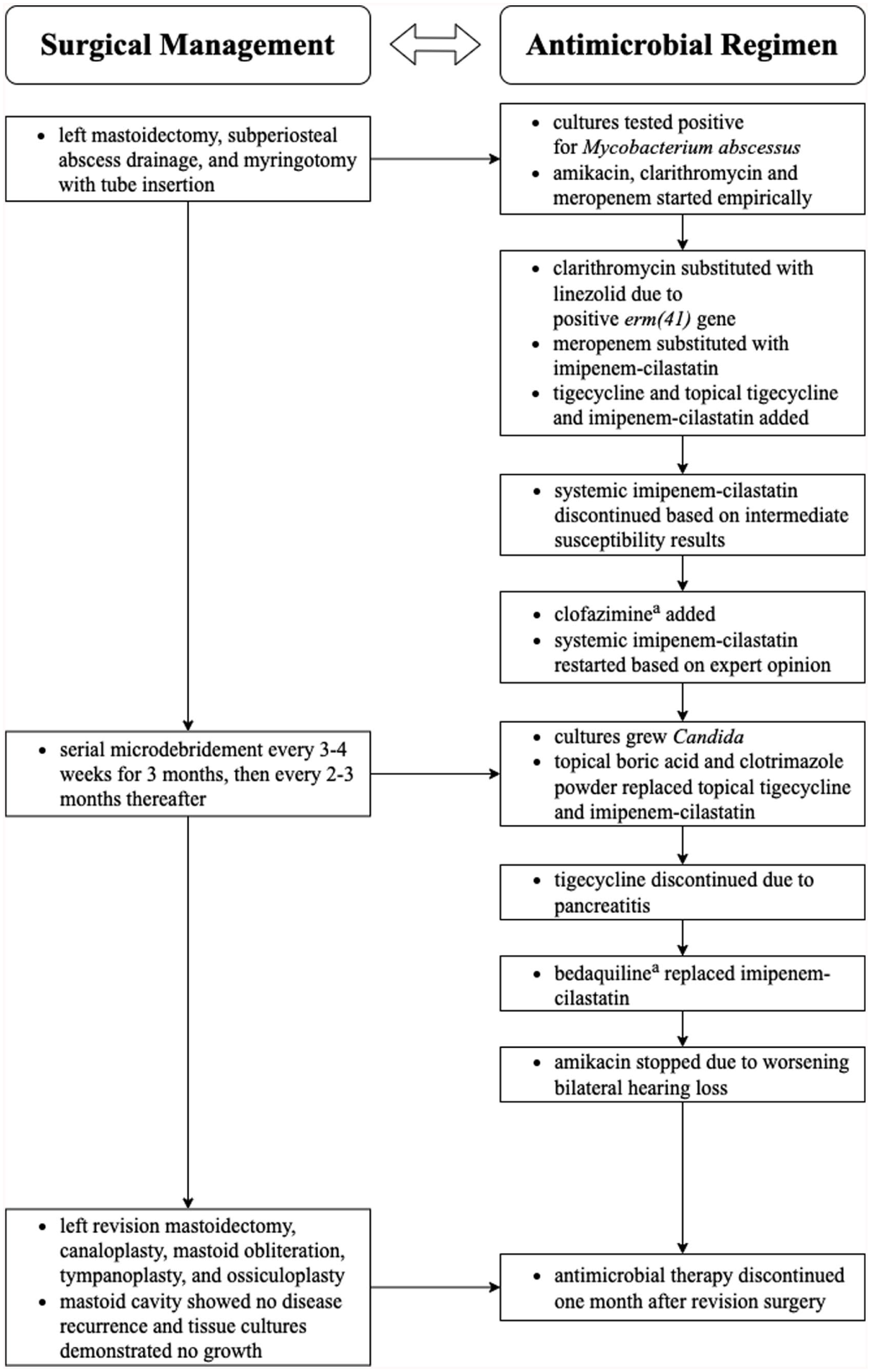
Overview of the combined surgical and antimicrobial management approach. This diagram outlines the general multimodal treatment strategy implemented in this case. Given the rarity and therapeutic complexity of pediatric Mycobacterium abscessus otomastoiditis infections, management plans should be individualized and informed by expert opinion.
Multidisciplinary meetings were ongoing (otolaryngology, infectious diseases, pediatric medicine) alongside consultation with international nontuberculous mycobacterium (NTM) experts. During the intensive phase (≥8 weeks), it was considered essential to maintain at least 4 systemic antimicrobial agents, followed by stepdown to 3 oral agents upon discharge. Adjunctively, the patient underwent serial microdebridement by otolaryngology every 3-4 weeks for 3 months, then every 2-3 months thereafter. During debridement visits, cultures grew Candida, resulting in the use of topical boric acid and clotrimazole powder, and discontinuation of topical tigecycline and imipenem-cilastatin.
Three months after surgery, a clofazimine load was started for an intended two-week period but was limited by nausea and vomiting requiring dose adjustments. The patient also developed anemia and thrombocytopenia due to linezolid, requiring iron and dose adjustments, pancreatitis from tigecycline requiring discontinuation, and intermittent tinnitus from amikacin. Four months post-surgery, repeat MRI showed improvement in the mastoidectomy cavity and reduced dural thickening. After twelve weeks of inpatient antimicrobial therapy, the patient was discharged on bedaquiline, clofazimine, linezolid, and amikacin. Six months after surgery, amikacin was discontinued due to worsening bilateral high-frequency hearing loss.
Eighteen months after initial presentation, the patient underwent left revision mastoidectomy, canaloplasty, mastoid obliteration, tympanoplasty, and ossiculoplasty. The surgery aimed to remove residual disease, repair the perforated tympanic membrane, and address conductive hearing loss from ossicular erosion. Reassuringly, the mastoid cavity showed no disease recurrence and tissue cultures demonstrated no growth. One month later, antimicrobials were discontinued. Three months after revision surgery, the patient remained well with improved but ongoing left-sided moderate-to-severe mixed hearing loss (Figures 4 and 5).
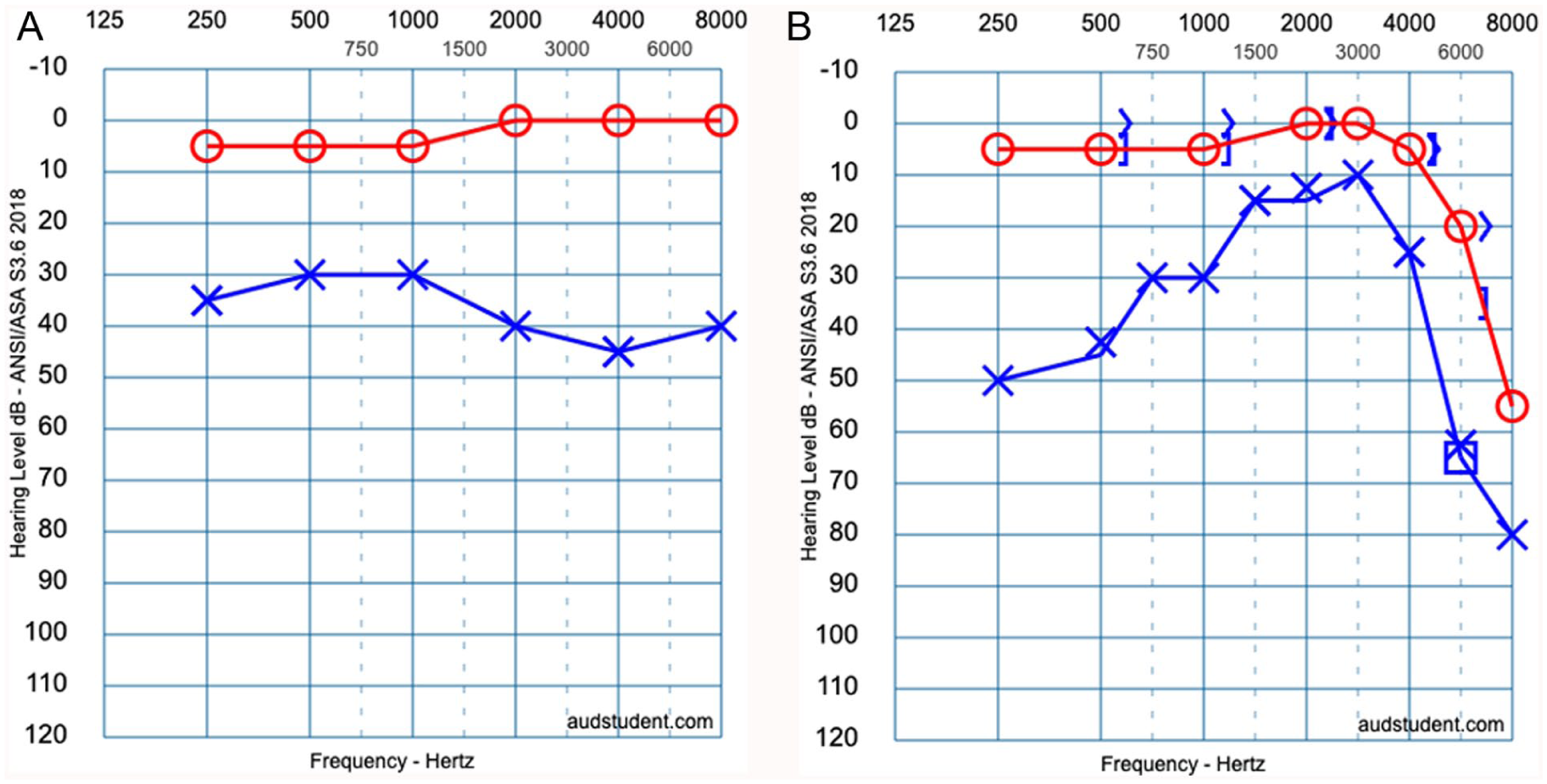
Pure tone audiometry and speech reception threshold measurements following initial surgery and revision surgery. (a) After the initial surgery, pure tone audiometry revealed normal right-sided hearing, and mild-to-moderate left-sided hearing loss across all frequencies. Pure tone average was 3 decibels for the right ear and 33 decibels for the left ear. Speech reception threshold was 5 decibels for both ears. Since bone conduction testing was not tolerated, conductive hearing loss was inferred from the clinical findings. (b) After revision surgery, pure tone audiometry revealed moderate high-frequency right-sided sensorineural hearing loss, and mild-to-moderate left-sided conductive hearing loss at lower frequencies and severe mixed hearing loss at higher frequencies. Pure tone average was 3 decibels for the right ear and 30 decibels for the left ear. Speech reception threshold was 10 decibels for the right ear and 20 decibels for the left ear. Figure graphics were generated using an online tool (audsim.com).
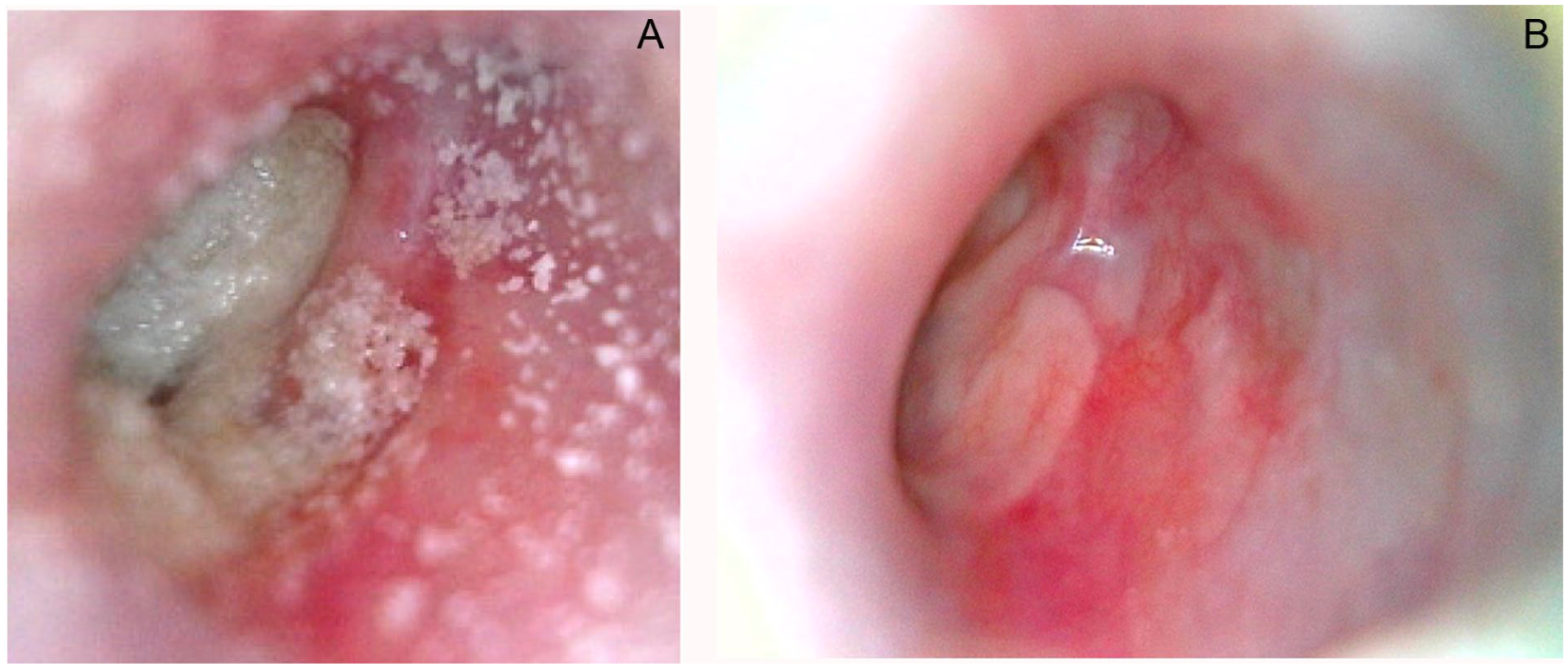
Transcanal examination of the left ear before and after the revision surgery. (a) Before the revision surgery, tympanic membrane is covered by debris. (b) After the revision surgery, tympanic membrane is intact, and there is no residual disease, infection, or fistula.
Discussion
This case study highlights an atypical and severe presentation of acute coalescent otomastoiditis caused by M. abscessus in an immunocompetent pediatric patient. While M. abscessus is increasingly recognized as an emerging pathogen in pediatric infectious disease, its presentation in otomastoiditis remains rare and diagnostically challenging, as it mimics more common bacterial etiologies of mastoiditis. This case underscores the importance of considering atypical pathogens in treatment-refractory mastoiditis and illustrates key principles in diagnostic reasoning, antimicrobial stewardship, and surgical decision-making in M. abscessus otomastoiditis.
M. abscessus is a virulent NTM that frequently originates from contaminated water sources and health care settings due to inadequate sterilization of medical equipment. 1 There are three known subspecies, M. abscessus subsp. abscessus, M. abscessus subsp. bolletii, and M. abscessus subsp. massiliense. Resistance to macrolides, often considered the backbone of therapy for NTM infections, can result from the presence of an inducible erm(41) gene in M. abscessus subsp. abscessus or a functional erm(41) gene in subsp. bolletii. In contrast, M. abscessus subsp. massiliense lacks an inducible erm(41) gene and is generally associated with superior treatment outcomes. 2 Therefore, accurate subspecies identification is crucial, as it can significantly impact therapeutic decision-making based on susceptibility. In children, M. abscessus infections typically present as lymphadenitis, pulmonary disease, postoperative wound infections, or catheter-related infections. M. abscessus infections are uncommon in immunocompetent children, but reports are increasing due to heightened awareness, longer survival in chronically ill children, and improved laboratory identification.3,4
In the case presented, the patient’s clinical presentation of fever, postauricular swelling, and mastoid tenderness initially raised concerns for bacterial coalescent mastoiditis. However, the absence of clinical improvement following surgical intervention and conventional antibiotic therapy warranted reevaluation. Diagnostic delays are common. One case series of seven patients with chronic tympanostomy tube otorrhea with confirmed M. abscessus infection reported a mean delay of 81 days, from initial presentation to diagnosis. 5 For our patient, the time to diagnosis was just over 1 month. A high index of suspicion is essential in recalcitrant cases, along with acid-fast bacilli (AFB) cultures and molecular susceptibility testing.
M. abscessus is well-known for its multidrug resistance. Treatment requires prolonged multidrug therapy along with repeated surgical debridement of infected tissue. 2 Treatment is guided by susceptibility testing, subspecies typing, and clinical tolerance. Currently, there are no established management guidelines for pediatric M. abscessus infections, and clinical practice is largely guided by expert opinion.
Amikacin, meropenem, and clarithromycin were empirically initiated for our patient. After detecting a functional erm(41) gene, clarithromycin was discontinued and replaced with linezolid. Over the course of treatment, several agents (imipenem-cilastatin, tigecycline, clofazimine, and bedaquiline) were sequentially added, reflecting a dynamic, individualized approach aligned with current practices for NTM management.2,3 As the intensive phase of treatment resolved, a key objective was to facilitate discharge planning by transitioning to oral agents where possible, based on susceptibilities. Thus, clofazimine and bedaquiline were the therapeutic options chosen. However, access was limited by the requirement to obtain both medications through Health Canada’s Special Access Program, which introduced logistical delays.
Clofazimine is increasingly used for its in vitro activity. In a case series of 27 children with M. abscessus odontogenic infections treated with a clofazimine-containing drug regimen, all patients demonstrated improvement in their jaw osteomyelitis with no significant cardiotoxicity. 6 With limited evidence for clofazimine in pediatric NTM management, dosing was individualized with expert consultation. Bedaquiline is a diarylquinoline approved for multidrug-resistant tuberculosis, but it has been proposed to provide therapeutic benefits in refractory NTM cases due to its in vitro activity.7,8 In a case series of 10 adult patients with treatment-refractory NTM infections, adding bedaquiline to their drug regimen was associated with moderate clinical benefit and acceptable short-term safety. 9 However, given the scarcity of pediatric safety data, this drug should be used cautiously.
Adverse effects secondary to therapy significantly impacted care. The patient experienced ototoxicity from prolonged amikacin exposure, myelosuppression from linezolid, gastrointestinal intolerance, and hepatotoxicity from clofazimine and tigecycline, as well as pancreatitis attributed to tigecycline. This is consistent with previously reported pediatric M. abscessus infections, where prolonged therapy is often complicated by serious adverse effects.2,3 To mitigate the adverse reactions that our patient experienced, pharmacotherapeutic adjustments were made, with continued monitoring for hearing, QTc prolongation, and liver, kidney, and pancreatic function.
Ultimately, surgical management remains a cornerstone in M. abscessus otomastoiditis, both for source control and diagnostic sampling. In our case, the patient required two surgeries: an initial mastoidectomy followed by a revision that included ossiculoplasty and cavity obliteration. Between these surgeries, the patient underwent serial debridement of the ear canal. The eighteen-month gap between the first and second surgeries allowed for reconstruction to be delayed until the infection was adequately controlled. Studies have shown that surgical debridement with systemic antimicrobials improves clinical outcomes in NTM otomastoiditis. A recent case report described the successful use of systemic and topical antimicrobials alongside mastoid surgery in a similar pediatric case. 10
Despite successful resolution of the infection, our patient experienced mixed hearing loss, likely due to damage to the ossicles, fibrosis of the middle ear, and toxicity from aminoglycosides. After the revision surgery, the pure tone average was 3 decibels for the right ear and 30 decibels for the left ear compared to 3 decibels for the right ear and 33 decibels for the left ear after the initial surgery. This underscores the potential for permanent hearing impairment due to M. abscessus infection and emphasizes the importance of early and ongoing audiologic monitoring during aminoglycoside therapy.
In conclusion, clinicians should consider NTM, particularly M. abscessus, in any child presenting with mastoiditis or otorrhea unresponsive to conventional management. When NTM is suspected, it is essential to request AFB cultures and pursue subspecies identification to inform treatment decisions. Early involvement of a multidisciplinary team, comprised of infectious diseases, otolaryngology, pharmacy, and audiology, is critical for managing complex therapeutic decisions and monitoring. Given the potential for significant drug toxicities, antimicrobial regimens should be tailored to susceptibility patterns and patient tolerance. Lastly, surgical intervention plays a vital role in both diagnosis and source control, with reconstruction deferred until the infection has fully resolved.
Footnotes
Acknowledgements
Special thanks to Dr. M. Elise Graham for her invaluable contributions to project conceptualization and data acquisition.
Ethical Considerations
Our institution does not require formal ethics approval for reporting individual cases.
Consent to Participate
Participant and legally authorized representatives provided informed written and verbal consent to participate in the study.
Consent for Publication
Participant and legally authorized representatives provided informed written and verbal consent to conduct the study, publish the study on an open access basis, and have their photographs or images used. All information has been anonymized where possible.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statements
The data that support the findings of this study are available from the corresponding author upon request.
