Abstract
Synovial sarcoma is a rare and aggressive soft tissue malignancy, with head and neck involvement comprising a small fraction of cases. Ethmoid sinus localization is exceptionally rare, with only a few cases documented in the literature. We present the case of a 34-year-old female who developed progressive right-sided nasal obstruction and epistaxis, with imaging revealing a locally invasive ethmoidal mass. Histopathological and immunohistochemical analysis confirmed a diagnosis of poorly differentiated synovial sarcoma. Incidentally, renal and intracranial findings suggested a possible underlying tuberous sclerosis complex. The patient was managed with a multimodal approach, including neoadjuvant chemotherapy (ifosfamide and doxorubicin), radiotherapy, and endoscopic surgical resection. Postoperative imaging demonstrated complete tumor clearance, and histology confirmed no residual malignancy. This case highlights the diagnostic and therapeutic challenges posed by ethmoidal synovial sarcoma and underscores the importance of individualized, multidisciplinary care, while also presenting an association with tuberous sclerosis that warrants further exploration.
Keywords
Introduction
Synovial sarcoma is a rare, malignant soft tissue tumor that originates from mesenchymal tissue, accounting for up to 10% of all soft tissue sarcomas. 1 Typically, it predominates in the extremities of young individuals, mostly in their third decade. 2 Less than 10% of synovial sarcoma cases involve the head and neck, with the paraspinal neck and hypopharynx being commonly affected. 3 Synovial sarcoma of the paranasal sinuses is a rare condition, with the ethmoid sinus being the rarest. To date, only 8 cases have been reported in the literature. Herein, we present a case of synovial sarcoma involving the ethmoid sinus.
Case Presentation
A 34-year-old female, a nonsmoker with an unremarkable past medical and surgical history, presented with a 1-month history of progressive right-sided nasal obstruction and intermittent epistaxis. She also reported a protruding mass from the right nostril (Figure 1), which subsequently detached spontaneously.
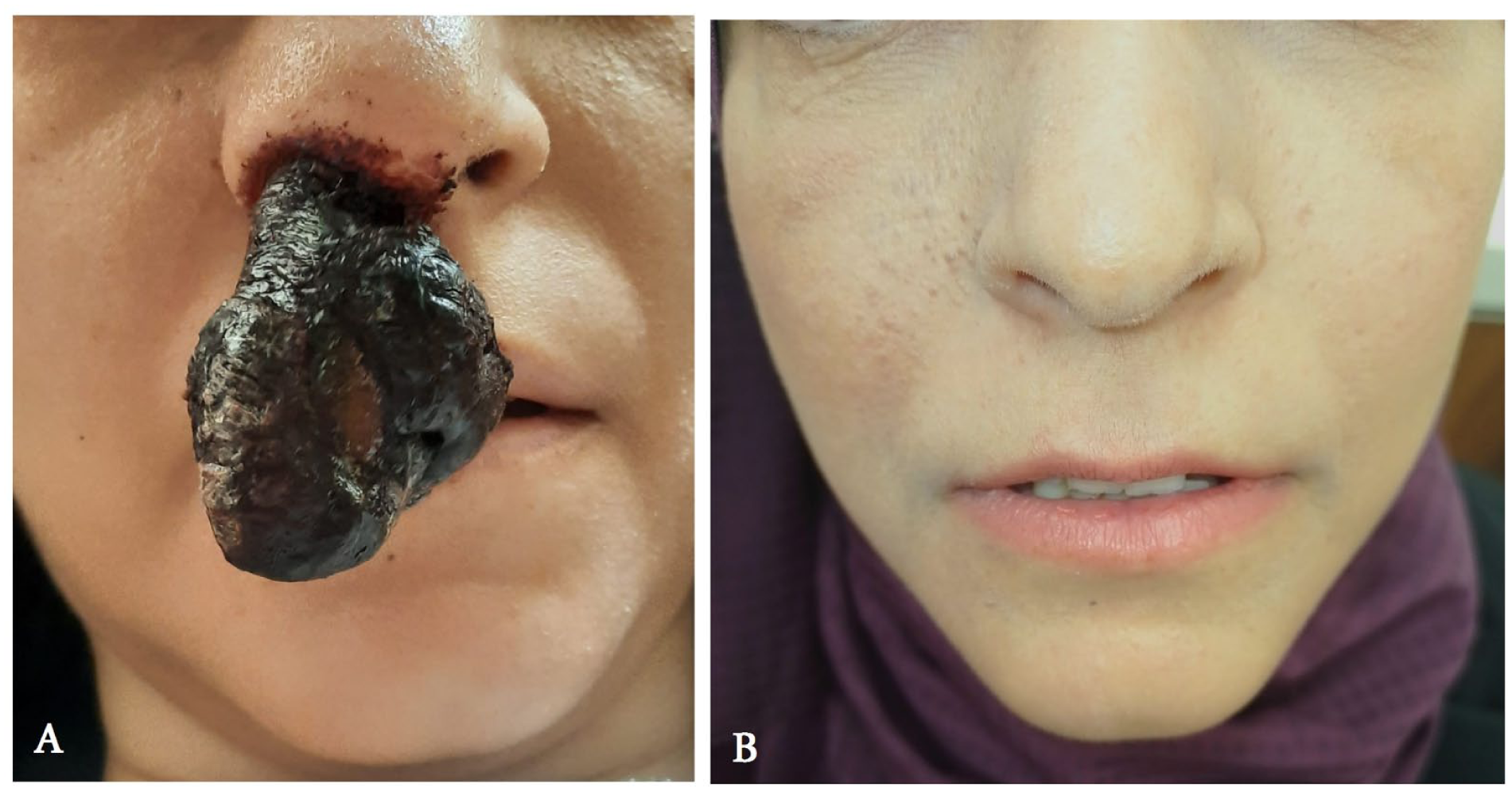
(A) A protruding mass from the right nostril. (B) Post-treatment appearance after disease resolution.
Histopathological examination of the detached lesion revealed a hypercellular tumor composed of round and spindle-shaped cells, with areas of necrosis, myxoid changes, and a characteristic staghorn vascular pattern. Mitotic activity was brisk. Immunohistochemical analysis showed strong positivity for CD99, CD56, BCL2, and TLE-1, with focal positivity for EMA, FLI-1, and Pan-CK. The tumor was negative for CD45, Desmin, Myogenin, Synaptophysin, Chromogranin, CD34, CD31, and SOX10. The Ki-67 proliferation index was ~70%. These findings were consistent with a diagnosis of poorly differentiated synovial sarcoma (Figure 2).
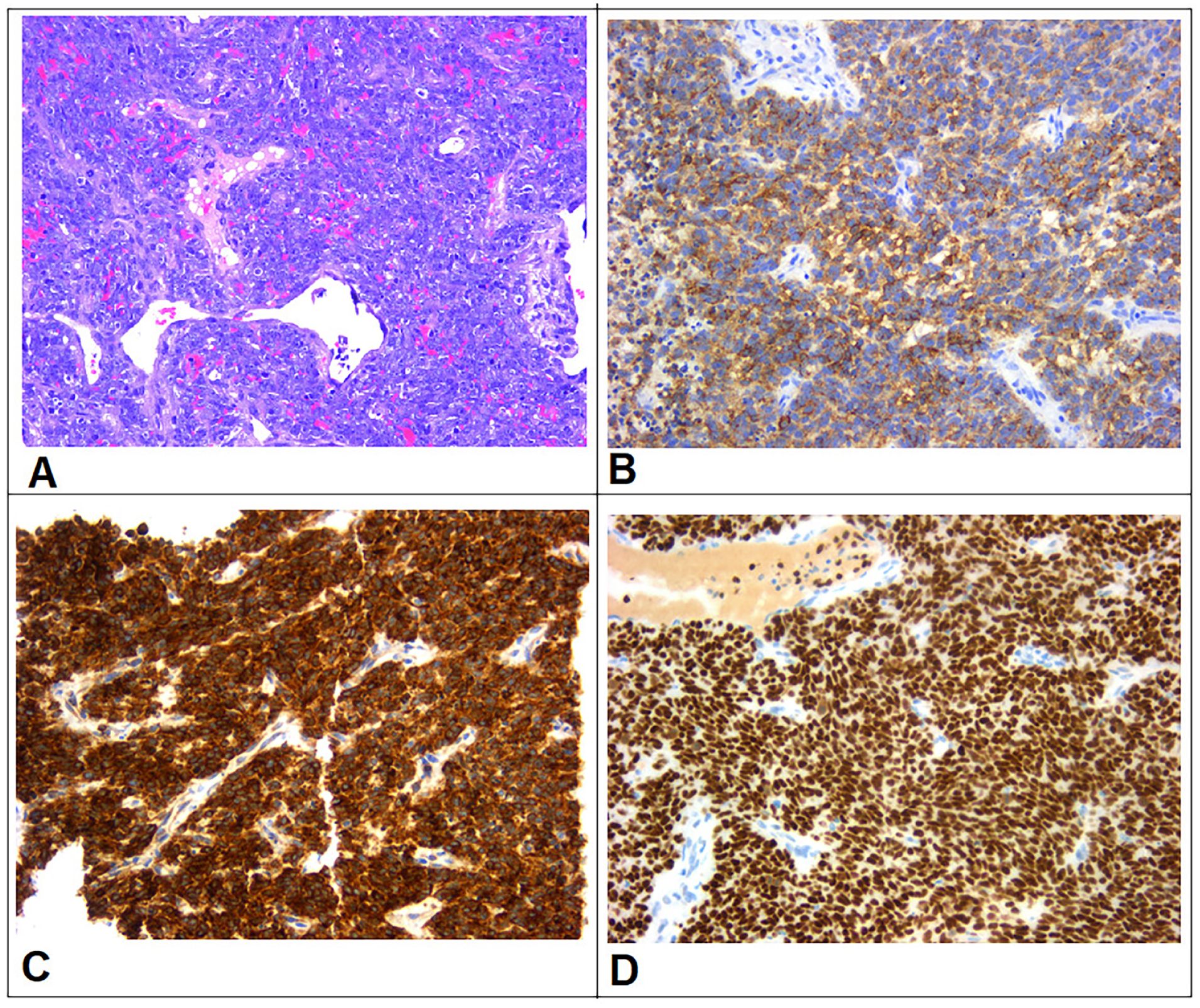
Histopathological examination. (A) H&E stain revealed a hypercellular tumor composed of round and spindle cells with overlapping nuclei and prominent nucleoli, accompanied by scant stroma, dilated vessels, and some mitotic figures. Immunohistochemistry stains showed that the neoplastic cells were diffusely positive for EMA (B), BCL2 (C), and TLE1 (D).
A brain computed tomography (CT) scan demonstrated a destructive mass lesion centered in the right ethmoid sinus, extending into the right nasal cavity, right orbit (compressing the medial rectus muscle), and the right side of the nasopharynx, measuring ~6.2 × 5.4 cm. The mass also extended externally beyond the nasal cavity. Subsequent MRI of the paranasal sinuses confirmed a locally aggressive mass involving the right ethmoid sinus, with evidence of bone destruction and avid contrast enhancement (Figure 3). Brain MRI and whole-body CT imaging revealed no evidence of distant metastases; however, imaging findings were suggestive of an underlying tuberous sclerosis complex (TSC). A few cortical subcortical tubers with characteristic radial bands, a calcified lesion in the right periventricular region, consistent with a subependymal hamartoma, measured ~1.1 cm. Multiple bilateral renal masses were identified. These were heterogeneous, containing both fat and soft tissue components, and were consistent with renal angiomyolipomas (Figure 4).
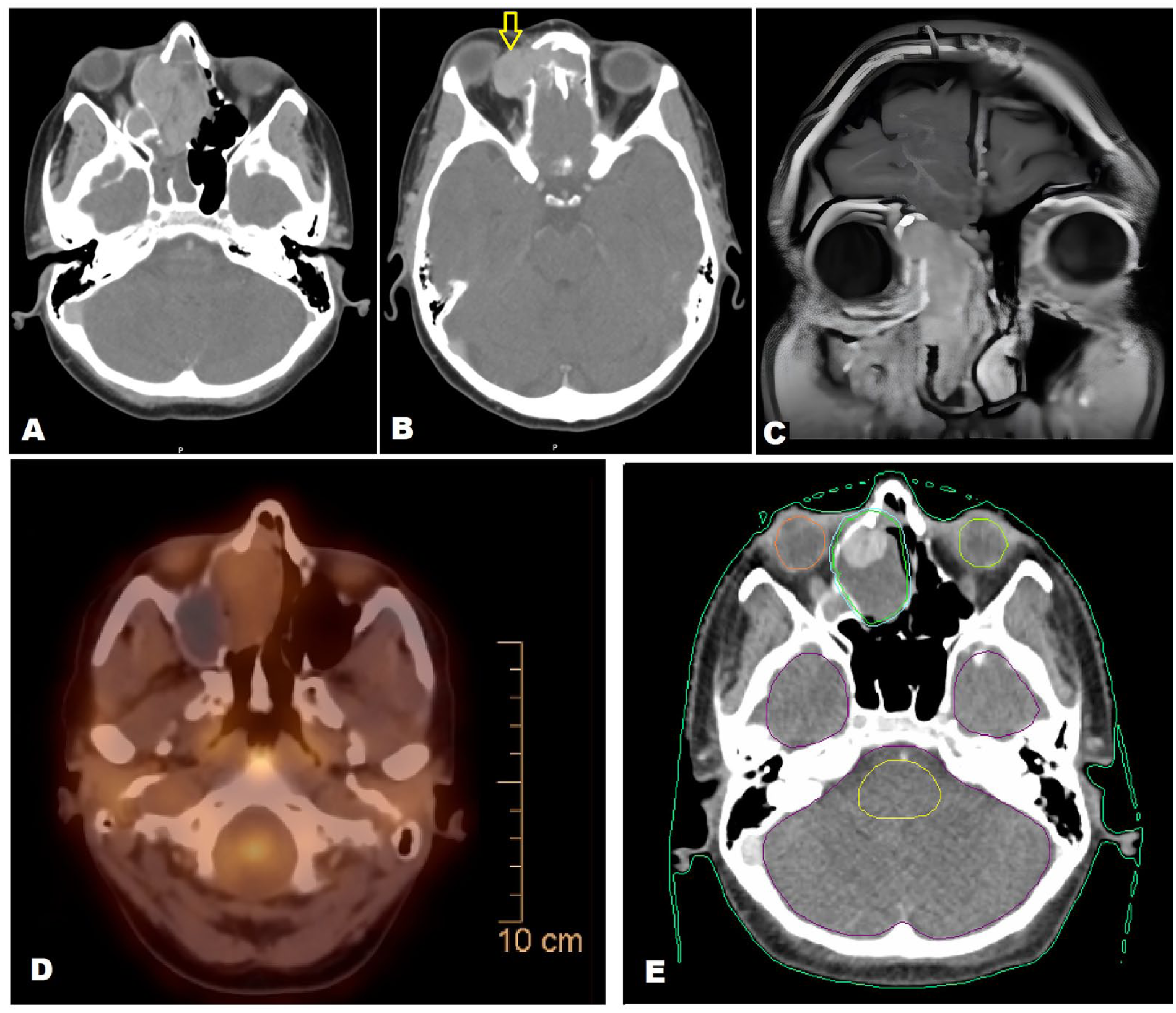
(A) Brain CT scan demonstrated a destructive mass lesion centered in the right ethmoid sinus, extending into the right nasal cavity and right orbit. (B) Intraocular extension of the tumor (arrow). (C) Coronal T1-weighted MRI with contrast of the paranasal sinuses showed a locally aggressive mass involving the right ethmoid sinus, with evidence of extension into the right orbit, bone destruction, and avid contrast enhancement. (D) PET-CT demonstrated moderate regression of the ethmoid mass. (E) Axial computed tomography CT scan for radiotherapy planning with organ-at-risk and target volume delineation.
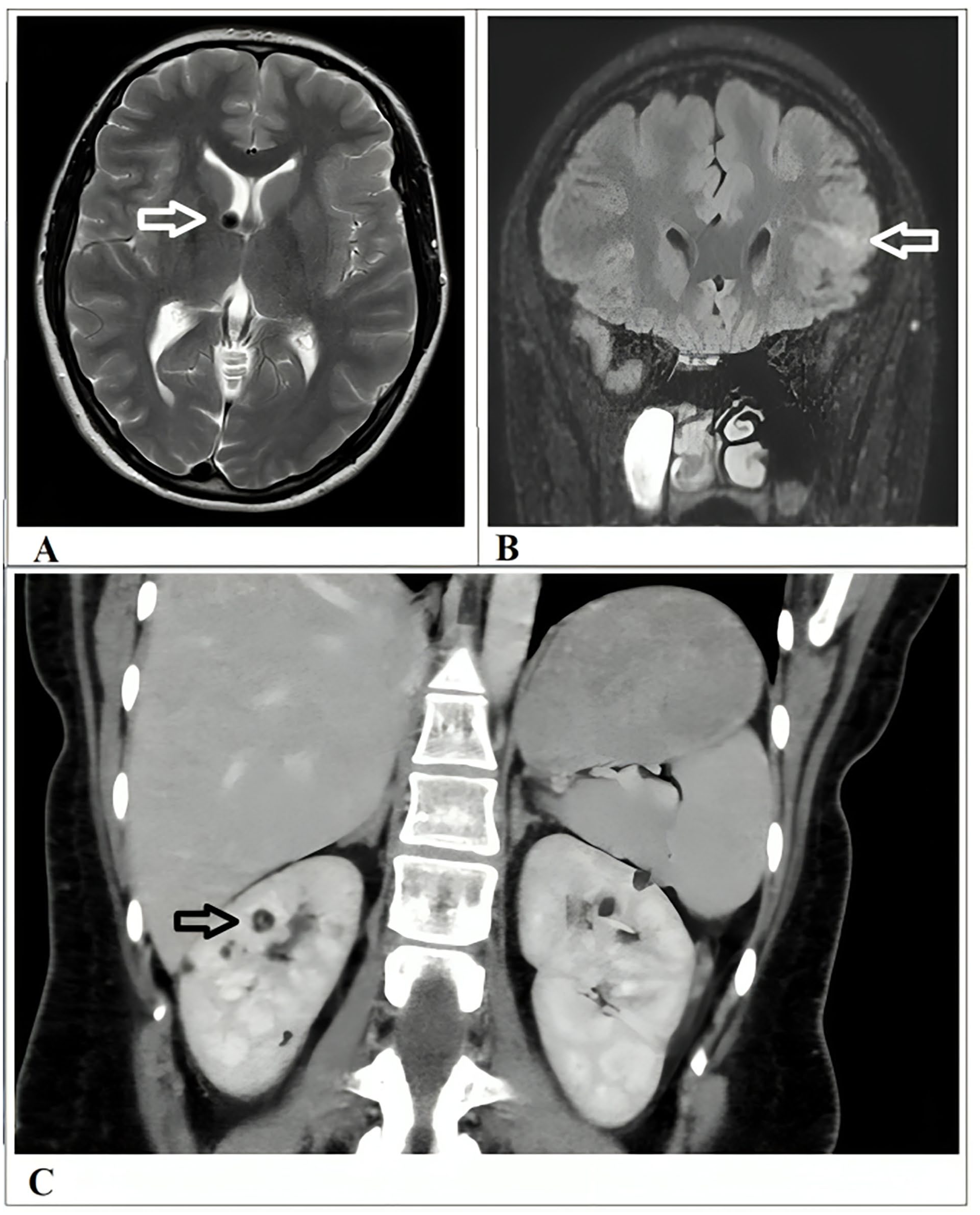
Radiological findings are suggestive of an underlying tuberous sclerosis complex. (A) A calcified lesion in the right periventricular region, consistent with a subependymal hamartoma, measured ~1.1 cm (arrow). (B) Few cortical subcortical tubers with characteristic radial bands (arrow). (C) Multiple bilateral renal masses were identified. These were heterogeneous, containing both fat and soft tissue components, and were consistent with renal angiomyolipomas (arrow).
A cardiac evaluation was within normal limits, with an ejection fraction of 55%. The patient received 6 cycles of chemotherapy following the IA protocol (ifosfamide and doxorubicin), which resulted in significant clinical and radiological improvement. Interim PET-CT demonstrated moderate regression of the ethmoid mass. A multidisciplinary head and neck tumor board reviewed the case. Due to the tumor’s proximity to critical structures, particularly the orbit, surgical resection with negative margins was deemed infeasible. The patient was subsequently treated with external beam radiation therapy, receiving 66 Gy in 33 fractions to the tumor site. The treatment was well tolerated (Figure 3).
Post-radiation MRI revealed a residual mass in the right maxillary sinus extending into the nasal cavity, with both cystic and solid components. The patient underwent functional endoscopic sinus surgery, with complete resection of the residual tumor, including both cystic and solid elements. Postoperative brain MRI confirmed total resection with no evidence of residual disease. Histopathological examination of the resected tissue demonstrated necrosis and chronic inflammation, with no evidence of residual malignancy. After 6 months follow-up PET scan was done showed no evidence of disease (Figure 5). The patient remains under regular follow-up in the radiation oncology clinic.
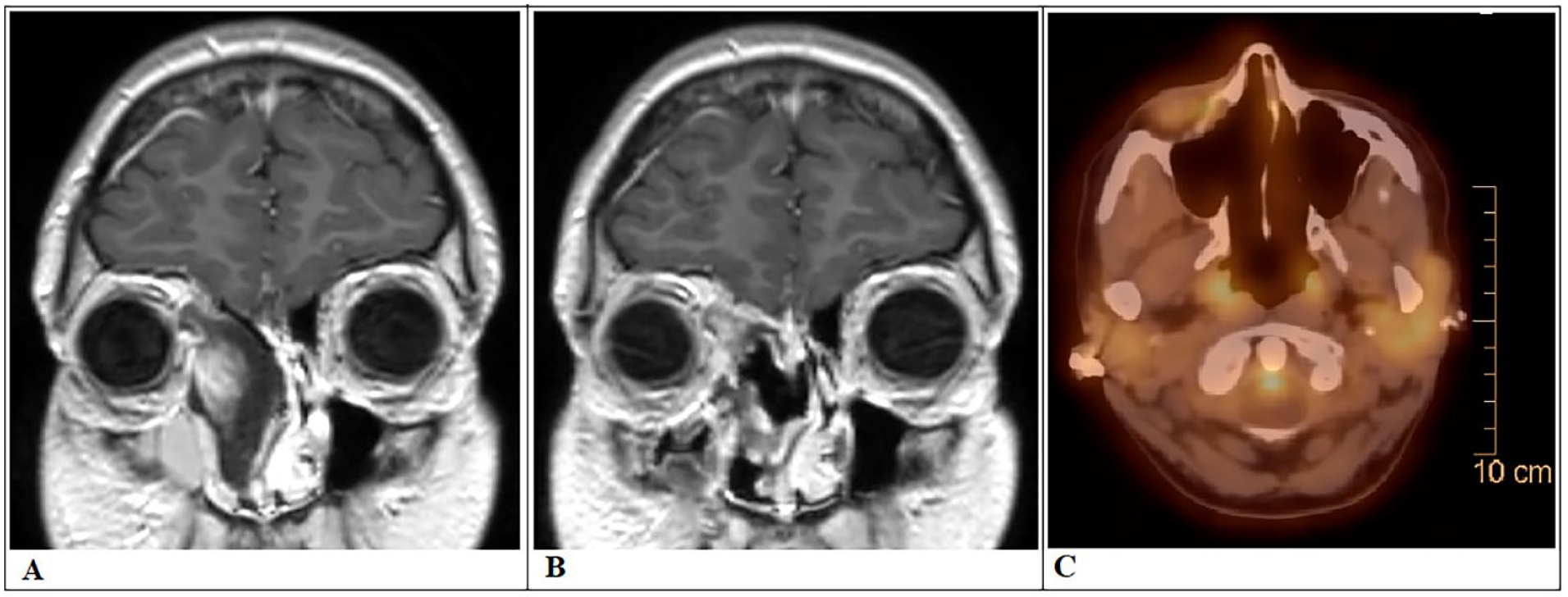
Post-treatment imaging. (A) Coronal brain MRI revealed a residual cyst with an eccentric enhancing nodule in the right ethmoidal sinus extending into the nasal cavity. (B) Post-FESS, coronal brain MRI confirmed total resection with no evidence of residual disease. (C) Six-month PET scan follow-up showed no evidence of disease. FESS, functional endoscopic sinus surgery.
Discussion
Synovial sarcomas are high-grade malignant tumors that arise from primitive mesenchymal cells, which are unrelated to the synovial tissues. 4 It is considered a rare tumor, accounting for 8% to 10% of soft tissue sarcomas, and ~5% to 10% of synovial sarcomas can be found in the head and neck region. 5 It primarily originates in the periarticular regions of the extremities between the third and fifth decades of life, with a male predominance. 6 Paranasal synovial sarcoma usually remains asymptomatic until the tumor size increases, resulting in compressive symptoms in adjacent structures. In previous cases of ethmoidal sinus sarcomas, patients typically presented with epistaxis, nasal pain, nasal obstruction, headache, eye pain, and proptosis. There is 1 case that involved spreading to the cavernous sinus, which led to facial nerve palsy and dysarthria, suggesting recurrent attacks of stroke. 7 However, our case presented with typical symptoms of nasal obstruction and epistaxis, along with a protruding mass from the right nostril and a sign of tuberous sclerosis.
A CT scan, while useful in detecting malignancy, is not pathognomonic for synovial sarcoma, as its characteristics frequently overlap with other paranasal sinus masses. Therefore, histopathology is essential for a definitive diagnosis. Synovial sarcoma is categorized into 2 main cell types: epithelial and spindle cells. It can be classified into various subtypes—monophasic fibrous (the most common), monophasic epithelial, biphasic, and poorly differentiated—based on the relative proportions of the 2 cellular components and the degree of differentiation. 4 In this case, histopathological examination revealed hypercellularity with round and spindled cells. In addition, immunohistochemical staining was positive for CD99, CD56, BCL2, and TLE-1, with focal positivity for EMA, FLI-1, and Pan-CK, thereby confirming the diagnosis of monophasic synovial sarcoma. 8
A summary of the previously reported 8 cases of ethmoid sinus synovial sarcoma, including clinical features and outcomes, is provided in Table 1.9 -11 Although various treatment approaches for synovial sarcoma have been described, the current standard modality includes surgical excision, typically followed by adjuvant chemotherapy and radiation therapy to improve outcomes and reduce recurrence. The main challenge during paranasal sinus surgery is obtaining a negative margin while preserving the integrity of adjacent vital structures. 12 As a result, our case was managed with 6 cycles of neoadjuvant chemotherapy following the IA protocol (ifosfamide and doxorubicin), followed by radiotherapy and subsequent surgical resection.
Summary of Cases of Synovial Sarcoma of the Ethmoid Sinus.
The chemosensitivity of paranasal synovial sarcoma is not fully understood due to the rarity of cases. Nevertheless, chemotherapy has been shown to reduce tumor size, lower the likelihood of distant metastasis, and improve survival time, thereby facilitating surgical resection. 3
Radiotherapy has demonstrated efficacy in managing cancer by improving survival rates and local disease control. Andrä et al 12 retrospectively reviewed 26 patients with head and neck sarcomas who received radiotherapy. Surgical resection was performed in 81% of patients, with only 38% achieving negative margins, while the remainder had positive margins and gross residual disease in 23% and 19% of cases, respectively. All patients received 66 Gy of radiation, and half of them were treated with chemotherapy. Five-year local control and overall survival rates were 86% and 82%, respectively.12 -14
According to the International Tuberous Sclerosis Complex Consensus Conference diagnostic criteria, a definite diagnosis of TSC can be established in the presence of 2 or more major clinical features, even in the absence of genetic testing. In this case, the patient exhibited a subependymal hamartoma and bilateral renal angiomyolipomas, both of which are classified as major diagnostic criteria. In addition, the presence of cortical tubers further supports the diagnosis. Therefore, based on imaging findings alone, this patient meets the clinical threshold for a definitive diagnosis of TSC. 15
This case highlights the diagnostic challenges of ethmoidal synovial sarcoma, particularly when it arises in anatomically complex and surgically constrained regions, such as the paranasal sinuses. The coexistence of synovial sarcoma with radiologically confirmed features of TSC raises the hypothesis of a potential pathogenic link between TSC-associated genetic pathways and mesenchymal tumorigenesis. Given the rarity of both the tumor location and its association with TSC, this case adds a novel dimension to the current understanding of sinonasal sarcomas and warrants further molecular investigation.
Conclusion
Synovial sarcoma of the ethmoid sinus presents significant challenges due to its rarity, aggressive nature, and complex anatomical location. This case highlights a rare coexistence of the tumor with features of TSC, indicating a possible association between the 2 conditions. In addition, it emphasizes the importance of a multimodal, individualized management approach to achieve effective treatment while preserving adjacent structures.
Footnotes
Acknowledgements
The authors are grateful to the doctors in the radiology and pathology departments at Augusta Victoria Hospital for their assistance in collecting patient data.
Ethical Considerations
Ethical approval to report this case was obtained from the Research Ethics Committee at Al-Quds University.
Consent to Participate
The patients gave written informed consent for the publication of this study. The editor-in-chief of this journal can review a copy of the written consent upon request.
Author Contributions
Elias Edward Lahham: lead author; primary manuscript writing and case synthesis. Rahmeh Deek: writing assistance; editorial support and manuscript revision. Mohammad Abuzir: writing assistance, editorial support, and manuscript revision. Ibrahem Eid: radiation oncology insights; contributed to data analysis and interpretation. Marwan Qubaja: pathology expertise; slide review and data analysis. Mahmoud Ramahi: radiological follow-up; case imaging review and interpretation.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data used to support the findings of this study are included in the article.
