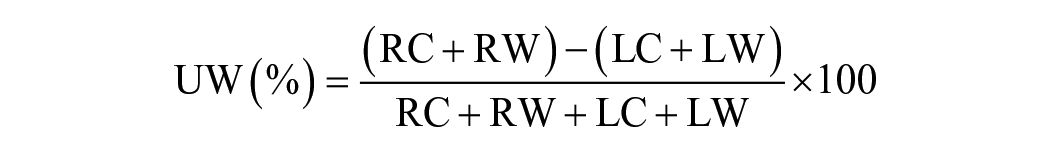Abstract
Background:
The monothermal caloric test (MCT) is a simplified alternative to the traditional bithermal caloric test for evaluating vestibular function. However, its diagnostic correlation with video head impulse test (vHIT) parameters, particularly in acute vestibular neuritis (VN), has not been thoroughly established.
Methods:
This retrospective study included 32 patients with acute VN who underwent bithermal caloric testing and vHIT within 5 days of symptom onset. Caloric responses were analyzed to determine unilateral weakness (UW) and monothermal caloric asymmetry (MCA) for warm and cold stimuli, with and without correction for spontaneous nystagmus (SN). The vHIT was performed using head impulse paradigm (HIMP) and suppression HIMP (SHIMP). Gain, gain asymmetry, and the presence of corrective saccades were evaluated. Correlation analyses were performed between caloric parameters and vHIT metrics.
Results:
Significant asymmetry was observed in the vHIT gain between the ipsilesional and contralesional ears in both the HIMP and the SHIMP (P < .001). Uncorrected warm MCA showed a stronger correlation with vHIT gain asymmetry (HIMP: r = 0.561, P < .001; SHIMP: r = 0.672, P < .001) than UW or SN-adjusted MCA values. Notably, SN correction reduced the correlation strength between MCT and vHIT results, suggesting SN as a clinically-relevant marker of vestibular asymmetry in the acute stage.
Conclusion:
Warm MCA without SN adjustment demonstrated the strongest correlation with high-frequency vestibulo-ocular reflex deficits measured using the vHIT in patients with acute VN. These findings suggest that the uncorrected MCT may be valuable and practical in assessing vestibular hypofunction, particularly when interpreted alongside the vHIT. The complementary use of the MCT and vHIT may enhance diagnostic accuracy and efficiency in acute vestibular evaluation.
Introduction
Caloric testing has traditionally been regarded as the gold standard for evaluating vestibular function by assessing the vestibulo-ocular reflex (VOR) of the horizontal semicircular canal, which is innervated by the superior vestibular nerve, during low-frequency stimulation.1 -4 It is particularly sensitive to mild vestibular asymmetries and remains useful in detecting subtle unilateral deficits. 5 With the introduction of the video head impulse test (vHIT), the clinical role of caloric testing has been reexamined, as the vHIT and caloric testing are believed to provide complementary information.6,7 Although caloric testing remains valuable, especially in chronic and subacute settings, clinicians are increasingly turning to the vHIT in acute settings because of its rapid execution and ability to assess high-frequency VOR function.6,8,9 Vestibular neuritis (VN) is one of the most common causes of acute vestibular syndrome, characterized by sudden-onset prolonged vertigo, unidirectional spontaneous nystagmus (SN), postural imbalance, and the absence of auditory symptoms or central neurological signs. It is generally attributed to inflammation of the vestibular nerve and typically results in unilateral dysfunction of the semicircular canals. This dysfunction can be evaluated using caloric testing and vHIT. 5 In addition to physical disability, VN can significantly impair quality of life and are often associated with psychiatric comorbidities such as anxiety or depression, underscoring the need for prompt and accurate diagnosis. 10
Despite the historical significance of caloric testing, it has several disadvantages. The test results can be influenced by multiple factors, including patient cooperation, middle ear status, and nature of the applied thermal stimuli. 11 In addition, the presence of SN may complicate the interpretation of caloric responses. 12 Furthermore, the test is time-consuming and frequently induces vertigo, making it an unpleasant experience for patients. To address these drawbacks, the monothermal caloric test (MCT) has emerged as a streamlined alternative.11 -15 The MCT can simplify vestibular assessment, reducing both the procedure duration and the patient discomfort. 12 Several studies have examined the efficacy and limitations of the MCT, with conflicting findings. 15 Although some studies have demonstrated adequate sensitivity and specificity in peripheral vestibular pathology,11,13,16 others contend that the MCT is suitable solely as a screening tool, questioning its diagnostic value.1,14,17 Most previous studies assessing the MCT have focused primarily on its correlation with traditional unilateral weakness (UW) parameters from bithermal caloric testing, leaving a significant gap regarding its correlation with vHIT parameters.1,7,15 Furthermore, these studies commonly included various vestibular disorders in their analyses, resulting in limited evidence specifically addressing the correlation between MCT and vHIT parameters in patients with VN. 7
Therefore, this study aimed to evaluate the clinical utility of the MCT in patients with VN by systematically comparing parameters derived from both caloric testing and the vHIT. Given that the MCT has not been thoroughly validated in an acute setting, particularly in VN, this study sought to clarify its potential role. By exploring the relationship between these 2 vestibular assessment modalities, we hope to provide deeper insights into the diagnostic value of the MCT in contemporary clinical practice.
Materials and Methods
Patients
This retrospective study was approved by the Institutional Review Board (IRB) of Chosun University Hospital (approval number: 2025-05-032) and conducted in accordance with the ethical principles outlined in the 1975 Declaration of Helsinki. Given the retrospective nature of the study, the requirement for written informed consent was waived by the IRB. We reviewed the data of patients diagnosed with VN who underwent both vHITs and caloric testing on the same day between January 2022 and January 2025. The diagnosis of VN was based on the diagnostic criteria proposed by the Bárány Society, including acute prolonged vertigo, unidirectional horizontal-torsional SN, reduced horizontal VOR function, and the absence of auditory or central neurological signs. During the study period, no patients with isolated downbeat SN (suggestive of inferior-type VN) were identified, and only cases consistent with superior-type or total-type VN were included. 18 Patients were excluded if they had hearing loss based on pure-tone audiometry, middle ear disease confirmed by otoscopic examination, central vestibular, or neurological disorders identified on magnetic resonance image. Only patients who underwent both caloric testing and vHIT within 5 days of symptom onset were included in the analysis. Both tests were performed on the same day for all patients. The affected ear was identified based on clinical symptoms and the direction of the fast phase of the SN, in accordance with the Bárány Society diagnostic criteria. 18
Caloric Tests
Bithermal air caloric testing was performed using ICS AirCal irrigator (GN Otometrics, Taastrup, Denmark) delivering air at 24 °C (cold) and 50 °C (warm) for 60 s/irrigation. Eye movements were recorded using video-oculography (SLVNG, SLMED, Seoul, Korea) with 2 minutes recording times. A 5 minute interval was allowed between each caloric irrigation session. Eye movements were recorded using videonystagmography, and the maximum slow-phase velocity (SPV) was measured. UW was calculated using the Jongkees’ formula as follows 19 :
where RC is the SPV of right cold, RW represents right warm, LC represents left cold, and LW represents left warm. UW was considered abnormal when >25%, based on Jongkees’ formula.
Monothermal caloric asymmetry (MCA) 20 was calculated separately for warm and cold conditions as follows:
SN was recorded prior to caloric stimulation using video-oculography (SLVNG, SLMED) under fixation-removed conditions, with the patient in a seated position and eyes in primary gaze. SN adjustment was performed by adding or subtracting the baseline SN velocity to/from the caloric SPV, depending on whether the SN direction was the same as or opposite to the caloric-induced nystagmus. 13 Specifically, if the SN direction matched that of the caloric-induced response (eg, right-beating SN with right warm or left cold stimulation), the SN velocity was subtracted from the measured caloric SPV. Conversely, if the direction was opposite (eg, right-beating SN with right cold or left warm stimulation), the SN velocity was added to each measured caloric SPV. This adjustment aimed to minimize the confounding influence of SN on caloric responses. To differentiate between SN-corrected and uncorrected responses, we defined 2 MCA parameters: MCA without adjustment for SN (MCAraw), calculated using uncorrected caloric SPV values and MCAadj, calculated using SN-adjusted SPV values.
vHIT: Head Impulse Paradigm and Suppression Head Impulse Paradigm
The head impulse paradigm (HIMP) protocol was performed using the ICS Impulse (Otometrics) in a well-lit room with patients seated upright and fixated on a target 1 m away. Horizontal head impulses were delivered in unpredictable directions with peak velocities of 150 to 200 deg/second and amplitudes of 10° to 20°. The suppression HIMP (SHIMP) test was performed using the same equipment and basic procedure as the HIMP test, with a key modification to the fixation strategy. Instead of focusing on an earth-fixed visual target, participants were instructed to fixate on a laser dot projected onto the wall from a head-mounted laser source. This created a head-fixed target that moved synchronously with head rotations. 21 All vHITs were performed by a single experienced examiner trained in vestibular function testing. To compute the VOR gain, we calculated the area under the eye and area under the head velocity curves from the onset of head movement to the point where the head velocity crossed 0. VOR gain was defined as the ratio of these areas. To ensure data accuracy, the vHIT device was securely fitted to the head of each participant to minimize goggle slippage. Covert and overt saccades were visually identified and confirmed as pathological when they appeared in more than 20% of all trials, exhibited peak velocities exceeding 100 deg/second, and demonstrated similar amplitude and latency. 22 Abnormal VOR function was defined as a HIMP and SHIMP gain <0.8. 8 Asymmetry of gain was calculated separately for the HIMP and the SHIMP using Jonkee’s formula as follows:
All VOR gain values reported in this study reflect measurements of the horizontal semicircular canals, as both caloric testing and horizontal head impulse testing were limited to this plane.
Statistical Analysis
The normality of continuous variables was assessed using the Shapiro-Wilk test. Depending on the distribution, comparisons between ipsilesional and contralesional values were performed using the paired t-test or Mann-Whitney U test. Categorical variables were compared using the chi-squared test or Fisher’s exact test, as appropriate. Correlation analyses between the caloric and vHIT parameters were conducted using Pearson’s or Spearman’s correlation coefficients, depending on data distribution. All statistical analyses were conducted using IBM SPSS Statistics version 29 (IBM Corp, Armonk, NY, USA), and a P < .05 was considered statistically significant.
Results
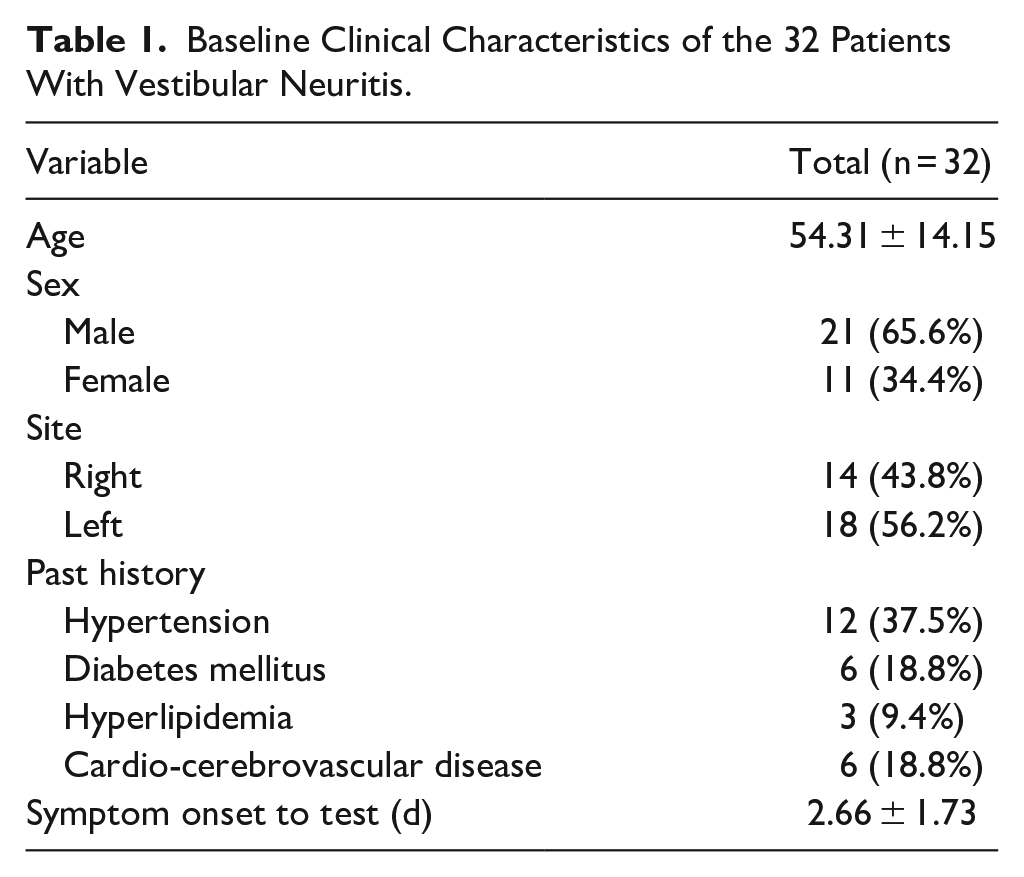
Thirty-two patients diagnosed with VN were included in this study. The mean age was 54.31 ± 14.15 years, with a predominance of male patients (21 men, 65.6%; 11 women, 34.4%). The right side was affected in 14 patients (43.8%), and the left in 18 patients (56.2%). Common comorbidities included hypertension in 12 patients (37.5%), diabetes mellitus in 6 patients (18.8%), hyperlipidemia in 3 patients (9.4%), and cardio-cerebrovascular disease in 6 patients (18.8%). The average time from symptom onset to vestibular testing was 2.66 ± 1.73 days (Table 1).
Baseline Clinical Characteristics of the 32 Patients With Vestibular Neuritis.
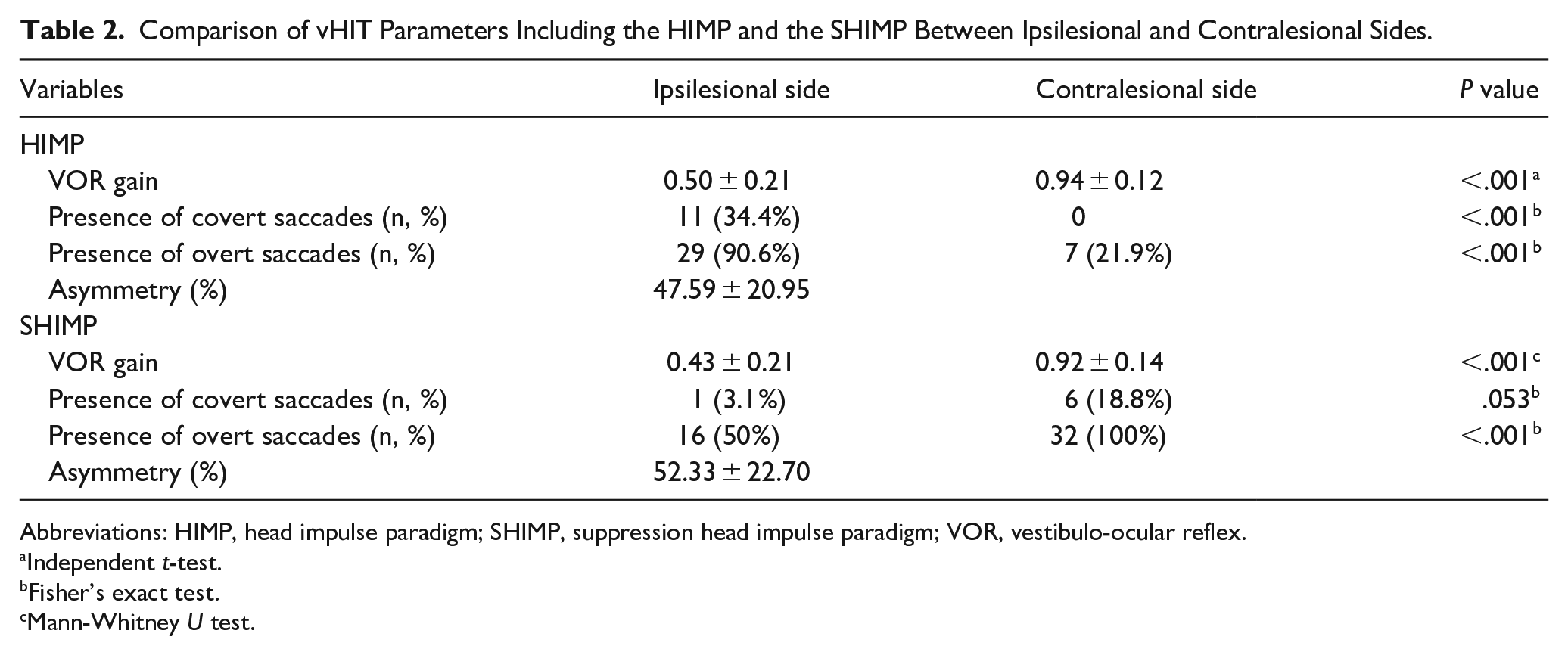
The vHIT analysis showed marked asymmetry between the ipsilesional and contralesional sides in both the HIMP and SHIMP protocols. The mean ipsilesional VOR gain was significantly lower than that in the contralesional side for both paradigms (HIMP: 0.50 ± 0.21 vs 0.94 ± 0.12; SHIMP: 0.42 ± 0.24 vs 0.92 ± 0.14; P < .001 for both). Figure 1 provides a visual representation of the variability in horizontal VOR gains on the ipsilesional and contralesional sides for both the HIMP and SHIMP protocols using box-and-whisker plots (Figure 1). Covert saccades were observed in 11 patients (34.4%) in the ipsilesional HIMP condition, while overt saccades were observed in 29 patients (90.6%). SHIMP results showed overt saccades in 16 patients (50%) ipsilesionally, whereas 100% of the patients demonstrated overt saccades on the contralesional side (Table 2).
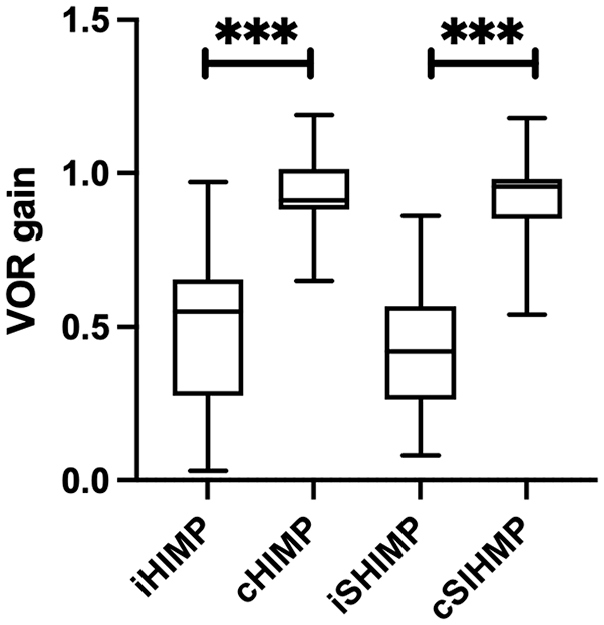
Distribution of horizontal VOR gain values measured by video head impulse test in the ipsilesional (i) and contralesional (c) ears. Data are presented separately for the HIMP and the SHIMP. Box-and-whisker plots indicate the median (horizontal line), interquartile range (box), and full data range (whiskers: minimum-maximum). VOR, vestibulo-ocular reflex; HIMP, head impulse paradigm; SHIMP, suppression head impulse paradigm. ***P < .001.
Comparison of vHIT Parameters Including the HIMP and the SHIMP Between Ipsilesional and Contralesional Sides.
Abbreviations: HIMP, head impulse paradigm; SHIMP, suppression head impulse paradigm; VOR, vestibulo-ocular reflex.
Independent t-test.
Fisher’s exact test.
Mann-Whitney U test.
Caloric testing revealed significant differences between ipsilesional and contralesional responses. Without adjustment for SN, warm stimulation on the ipsilesional side showed negative SPV values (−0.69 ± 10.26 deg/second), likely due to baseline SN in the opposite direction attenuating the caloric-induced response. Contralesional warm SPV also decreased slightly after adjustment (35.16 ± 21.63-23.53 ± 24.14 deg/second). The mean UW was 56.27% ± 19.27%. Warm MCA was high before SN adjustment (147.23% ± 118.09%), but dropped to 15.31% ± 98.01% after SN adjustment (Table 3).
Quantitative Results of Caloric Testing Including SPV, UW, and MCA.
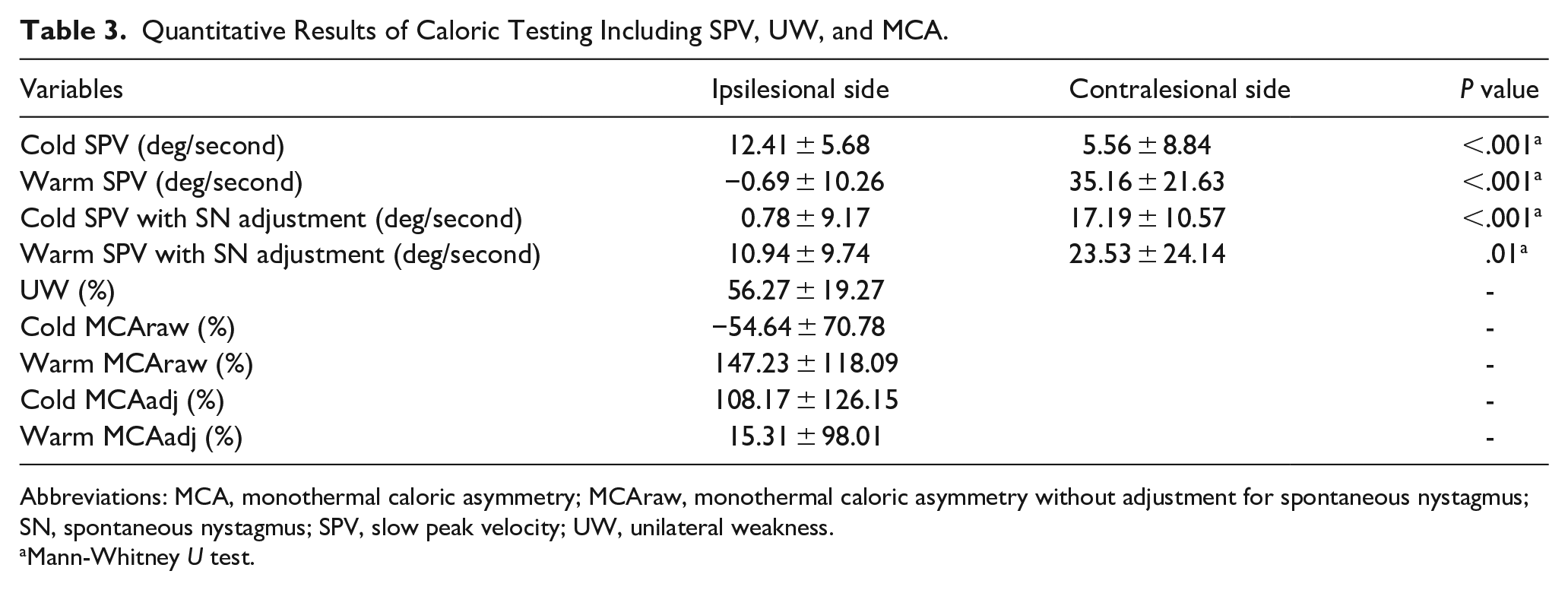
Abbreviations: MCA, monothermal caloric asymmetry; MCAraw, monothermal caloric asymmetry without adjustment for spontaneous nystagmus; SN, spontaneous nystagmus; SPV, slow peak velocity; UW, unilateral weakness.
Mann-Whitney U test.
To assess the clinical utility of the MCT, correlation analyses were performed between caloric asymmetry parameters and vHIT metrics. HIMP and SHIMP asymmetries were moderately correlated with caloric UW, with both associations reaching statistical significance (r = 0.451, P = .01; r = 0.428, P = .015, respectively; Table 4 and Figure 2). Warm MCAraw demonstrated stronger correlations with vHIT asymmetry (HIMP asymmetry: r = 0.561, P < .001; SHIMP asymmetry: r = 0.672, P < .001; Figure 2), indicating a closer association between monothermal caloric responses and high-frequency VOR function measured by vHIT. In contrast, when MCA values were adjusted for SN, the correlations with vHIT parameters became weak and statistically nonsignificant. Other caloric asymmetry parameters, including cold MCAraw and adjusted MCA values, did not show significant correlations with HIMP asymmetry. Additionally, warm MCAadj showed no meaningful association with either HIMP or SHIMP asymmetry (Table 4).
Correlation Between vHIT and Caloric Parameters.
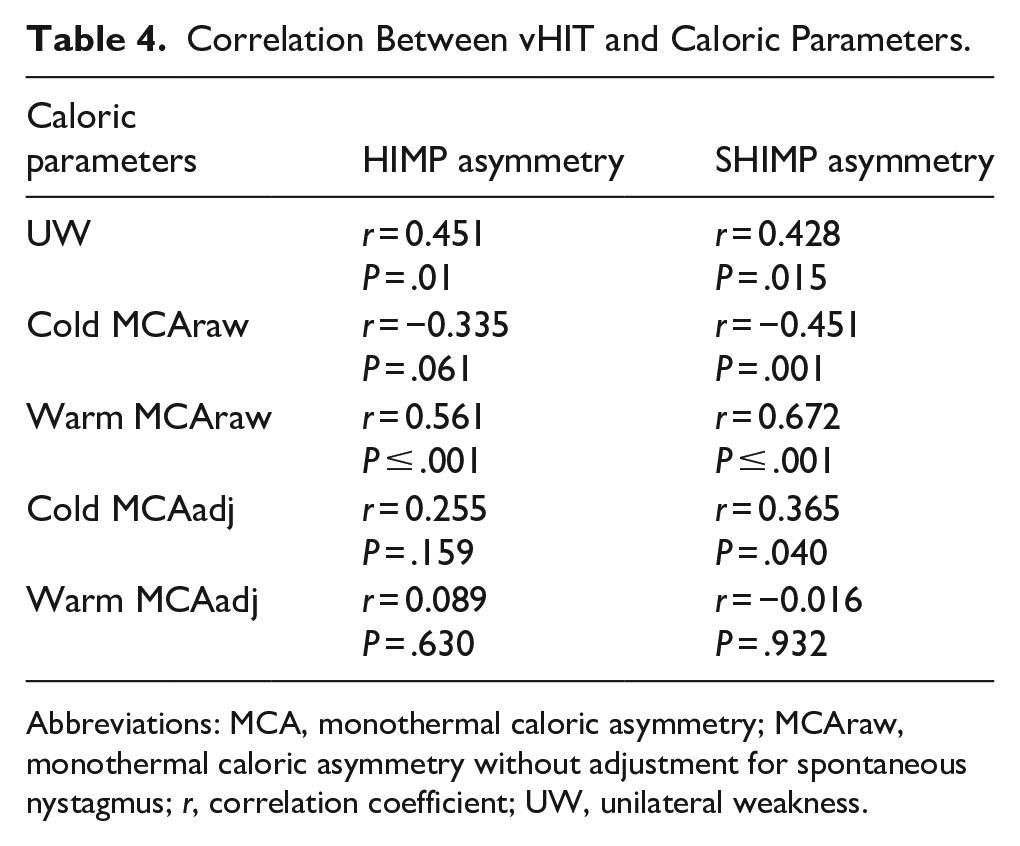
Abbreviations: MCA, monothermal caloric asymmetry; MCAraw, monothermal caloric asymmetry without adjustment for spontaneous nystagmus; r, correlation coefficient; UW, unilateral weakness.
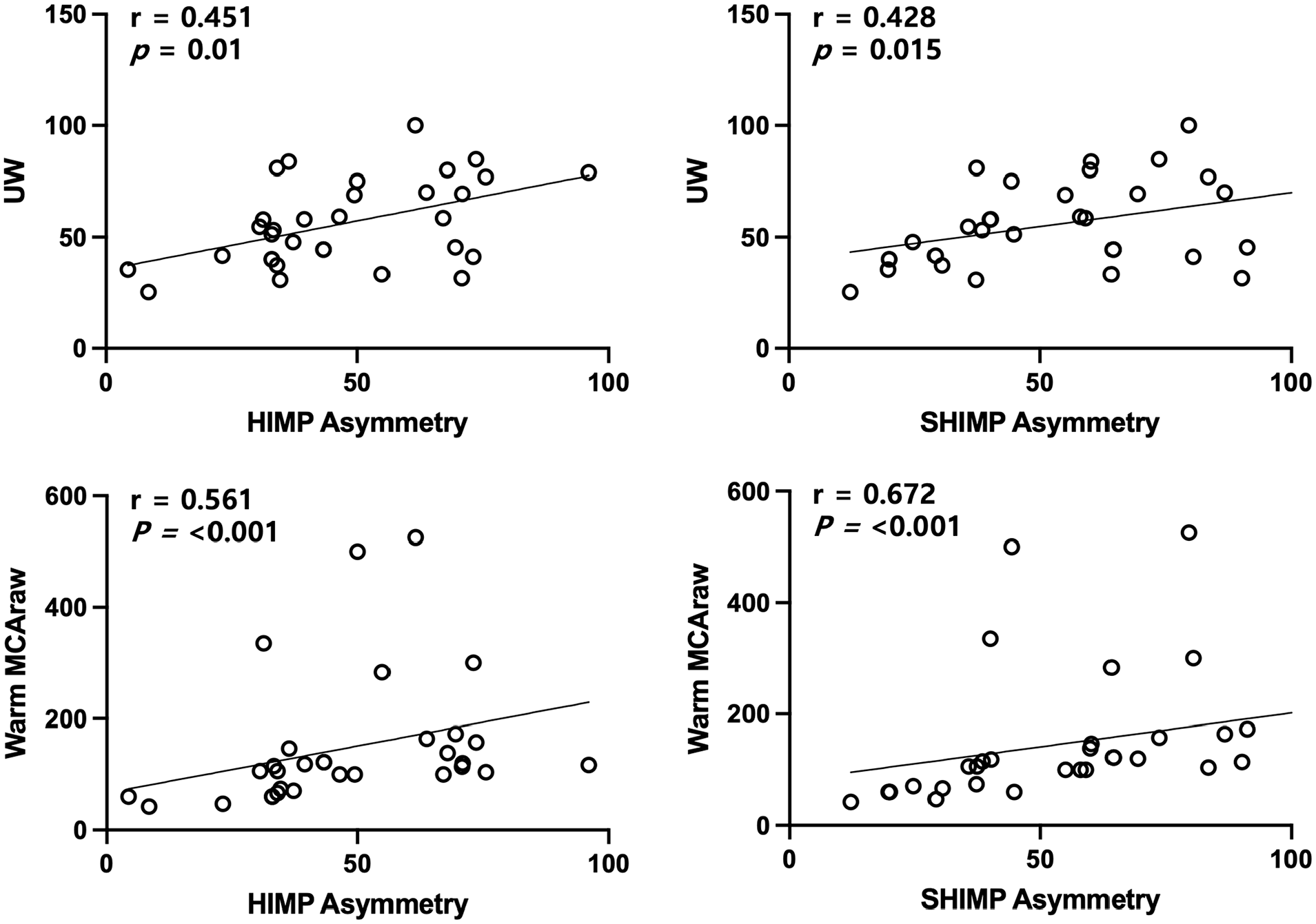
Correlation between video head impulse test asymmetry parameters and caloric asymmetry parameters. Significant positive correlations were observed in all panels, with the strongest correlation found between SHIMP asymmetry and warm MCA (r = 0.672, P < .001). The X- and Y-axes represent percentage values (%). UW, unilateral weakness; HIMP, head impulse paradigm; SHIMP, suppression head impulse paradigm; MCAraw, monothermal caloric asymmetry without spontaneous nystamgus adjustment.
We additionally analyzed the correlation between SN SPV and vHIT parameters. SN showed a moderate negative correlation with ipsilesional SHIMP gain (r = −0.461, P = .008) and SHIMP gain asymmetry (r = −0.486, P = .005). No significant correlations were observed between SN and ipsilesional or contralesional gains in HIMP or SHIMP, HIMP asymmetry, or UW.
Discussion
This study demonstrated that in patients with VN, warm MCAraw exhibited a stronger correlation with vHIT gain asymmetry, using both the HIMP and SHIMP protocols, than with conventional UW or SN-adjusted warm MCA values (MCAadj). These findings suggest that warm MCAraw may reflect acute VOR dysfunction more accurately and serve as a practical and sensitive indicator of lateralized vestibular impairment in the early uncompensated phase of VN. In clinical practice, vHIT is not always feasible due to limitations such as cervical spine disorders, severe vertigo, or poor cooperation. In such cases, a simplified and less provocative caloric protocol—such as warm MCT—may offer a more tolerable and efficient alternative, especially in the acute phase. Our results reinforce the complementary nature of caloric testing and vHIT. While caloric testing assesses low-frequency VOR function, vHIT evaluates high-frequency responses. In clinical practice, combining these 2 modalities can provide a broader spectrum of vestibular function, particularly in acute settings where rapid and accurate assessment is crucial. Despite the growing use of vHIT, caloric testing remains an essential diagnostic tool in evaluating VN, particularly for identifying low-frequency VOR dysfunction not captured by vHIT. 5
The MCT, especially with warm air stimulation, has emerged as a simplified alternative to bithermal protocols. Several studies, including those by Bush et al, have demonstrated the diagnostic utility of warm air MCT, with some reporting sensitivity and specificity levels exceeding 90%.11,13,16 Lowering the diagnostic threshold to 10% has been shown to further enhance test accuracy. However, these studies often included diverse vestibular disorders and were not limited to acute VN, thereby limiting their generalizability. Similarly, Adams et al emphasized that MCA values show stronger correlations with bithermal caloric test-derived UW in patients with definite vestibular dysfunction, while correlations were reduced in borderline cases, suggesting that uncorrected values may better reflect clinical asymmetry in acute stages. 15 These prior results are consistent with our observation that unadjusted warm MCA showed stronger correlations with vHIT asymmetry than SN-corrected values.
Our findings further support the diagnostic relevance of warm MCT, particularly when SN is not mathematically corrected. The excitatory nature of warm stimulation may produce a broader dynamic range and greater sensitivity in revealing VOR asymmetry, 13 which explains the stronger correlation with vHIT gain asymmetry observed in this study. Moreover, the acute SN observed in VN may serve as a clinical marker of horizontal canal dysfunction, and its removal through correction procedures may inadvertently reduce diagnostic sensitivity. Our data suggest that SN, rather than acting as a confounding variable, may offer clinically-relevant insights into the extent of vestibular dysfunction. In support of this, we found that SN SPV was moderately correlated with both ipsilesional SHIMP gain (r = −0.461, P = .008) and SHIMP gain asymmetry (r = −0.486, P = .005). These findings suggest that SN intensity may reflect the degree of acute vestibular imbalance, particularly as captured by SHIMP-based parameters. Conversely, no significant correlation was observed between SN and HIMP gains, HIMP asymmetry, or caloric UW, highlighting the unique sensitivity of SHIMP to dynamic asymmetry in the acute phase of VN.
The reduced correlation observed after SN correction may be attributed to several factors. First, SN is affected by fixation suppression and testing conditions, which can result in imprecise baseline estimates. Second, traditional correction formulas assume SN to be a constant offset, failing to account for its dynamic properties, such as direction changes or gradual decay. Third, SN is likely a true reflection of acute vestibular imbalance, and its removal may obscure important physiological information. Therefore, in the context of acute VN, uncorrected SN-inclusive warm MCA values may better capture the underlying pathology than corrected values.
This study also highlights the complementary roles of the MCT and vHIT. Although caloric testing offers high sensitivity for detecting canal paresis, the vHIT provides specificity and practical utility, particularly in time-sensitive or resource-limited settings.8,9 Their combined use can enhance diagnostic accuracy, especially in cases where bithermal caloric testing is poorly tolerated or not feasible. Our results reinforce this complementary nature and suggest that even simplified MCT protocols, when interpreted alongside the vHIT, retain strong clinical relevance.
This study had several limitations. First, the study was not originally designed to evaluate the MCT in isolation; warm and cold responses were extracted from a bithermal protocol, raising the possibility of inter-stimulus effects despite a 5 minute interval between irrigations. Second, air caloric stimulation was used, which may have yielded different response profiles compared with water stimulation. Third, the study included only patients with acute VN; future research should assess the applicability of the MCT to other vestibular disorders. Fourth, we did not directly calculate the sensitivity and specificity of MCT within this cohort, limiting comparison with previous reports. Fifth, only horizontal HIMP were used in the vHIT, restricting assessment to the horizontal semicircular canals. As such, dysfunction of the anterior and posterior canals was not evaluated, which limits the generalizability of our findings to global vestibular function.
In conclusion, our findings suggest that warm MCT, particularly without SN adjustment, may more effectively reflect acute VOR dysfunction than previously recognized. By preserving clinically-relevant asymmetries during the early phase of VN, uncorrected warm MCA appears to align more closely with vHIT parameters. These results support the broader clinical utility of the MCT as a practical, efficient, and diagnostically-informative tool when used in conjunction with the vHIT, particularly in acute vestibular assessment.
Footnotes
Ethical Considerations
This retrospective study was approved by the Institutional Review Board (IRB) of Chosun University Hospital (approval number: 2025-05-032).
Consent to Participate
The requirement for informed consent for this chart review study was waived by the Chosun University Hospital IRB.
Consent for Publication
All authors have read and approved the final manuscript and consent to its publication.
Author Contributions
Gi-Sung Nam: concept, design, data acquisition and analysis, data interpretation, draft, editing, approval. Sung Il Cho: concept, design, data interpretation, draft, editing, approval.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a research fund from Chosun University Hospital, 2024.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data are available from the corresponding author upon reasonable request.