Abstract
Background:
Interventions such as steroids, endoscopic sinus surgery (ESS), biologics, or a combination of these are available for the treatment of chronic rhinosinusitis with nasal polyposis (CRSwNP). However, despite evidence supporting the efficacy of each approach, factors such as timing, cost, side effects, and patient preference need to be considered. The objective of this work was to provide a comparison between current biologics and considerations for each intervention when developing a treatment plan.
Methods:
Recent studies are referenced in a detailed description of current and developing biologics to compare U.S. Food and Drug Administration (FDA) indications, mechanism of actions, notable side effects, dosage, and cost. Considerations for timing of treatment and duration of treatment when analyzing the utility of ESS, biologics, or a combination of both are highlighted.
Results:
Currently, 3 biologics are FDA-approved for the treatment of inadequately-controlled CRSwNP and 2 biologics demonstrate promise as potential agents. The overall trend in literature suggests that the combination of ESS to relieve polyps and improve medication delivery and biologics to facilitate anti-inflammatory effects is clinically impactful.
Conclusion:
Evidence to date aligns with current recommendations that biologics are a promising maintenance option for CRSwNP patients who have failed both medical management and complete ESS.
Chronic Rhinosinusitis With Nasal Polyposis Background
Chronic rhinosinusitis (CRS) is defined as sinonasal inflammation persisting for more than 12 weeks with at least 2 cardinal symptoms confirmed by endoscopic or radiographic findings. 1 CRS with nasal polyposis (CRSwNP) is a phenotypic subgroup defined as meeting the criteria for CRS, with the additional presence of polyps. 1 Histologically, CRSwNP is characterized by excessive immune response, with disruption of the nasal epithelial barrier by basal cell proliferation, goblet cell metaplasia, and reduced ciliated cell differentiation. 2
CRSwNP may be further characterized by endotype. The most common endotypes associated with CRSwNP in North America are type 2 (T2), type 3 (T3), and type 1 (T1).1,3 With the T2 endotype, environmental insults induce a response characterized by increased tissue eosinophils, basophils, and mast cells, along with elevated type 2 T-helper cell (Th2) cytokines.4,5 These Th2 cytokines include interleukin-4 (IL-4), IL-5, IL-13, and IL-31.2,5,6 IL-4 has been found to increase epithelial permeability, decrease wound repair, and play a significant role in B-cell activation and immunoglobulin E (IgE) production. 2 IL-5 is not only essential for the maturation of eosinophils but also serves as a growth factor for B lymphocytes.4,7 IL-13 has been observed to play a role in goblet cell metaplasia. 2 Both IL-4 and IL-13 have been shown to drive Th2 cell differentiation, increase local production of Ig, and facilitate airway remodeling through influencing mucus secretion and matrix deposition.2,4
Studies have investigated how these Th2 cytokines are affected by upstream pathways, centered in the respiratory epithelium.4,8 Since the epithelium serves as a mechanical barrier to the environment, cytokines released by epithelial cells in response to injury play a critical signaling role in the immune response. Specifically, these epithelial cytokines include thymic stromal lymphopoietin (TSLP), IL-25, and IL-33. 4 TSLP contributes to mediating the differentiation of naïve T cells (Th0) into Th2 cells and augments the activity of a subtype of innate immune cells that release significant amounts of Th2 cytokines like IL-5 and IL-13. 4 IL-33 recruits and enhances the activity of Th2 cells as well as mast cells, basophils, and the production of IL-13.4,8 IL-25 upregulates the IL-25 receptor on Th2 cells, thus increasing the recruitment of eosinophils and production of the Th2 cytokines IL-4 and IL-5.4,8 Recent studies have also investigated the involvement of IL-9 and IL-17 in this epithelial signaling pathway. 9
As T2 is the most prevalent endotype, most patients with CRSwNP respond to Th2-targeted biologics. If a patient does not respond to these biologics and has no objective evidence of atopy (ie, positive allergy testing, eosinophilia), clinicians should consider whether the patient’s nasal polyposis (NP) may be of a different endotype. Patients with a T3 endotype would benefit from biologics targeting IL-17 or IL-22, which are not currently U.S. Food and Drug Administration (FDA)-approved for CRSwNP. 3 Similarly, while there are many biologics specific to the primary cytokines involved in the T1 endotype, such as TNF-alpha, IFN-gamma, and IL-12, they also lack FDA-approval for polyps. 3 From a practical standpoint, the lower prevalence of T1 in North America means that there is less economic impetus for study of these biologics for use with CRSwNP.
Ultimately, CRSwNP may present with a heterogenous histology. 3 Cases may be associated with both eosinophilic and neutrophilic infiltrates, highlighting the potential complexity of multiple overlapping processes. 3 Accordingly, future treatments for CRSwNP must look at targeting both the traditional T2 profile of cytokines, and those involved in T1 and T3 inflammation.
Immune Modulators for CRS
Biological immune modulators, or biologics, are a class of medications derived from biological processes. They include monoclonal antibodies that target specific components of the immune system. Instead of global immune suppression as seen with corticosteroids, biologic immune modulators precisely target specific steps in the inflammatory cascade.1,4 Given the significant overlap in mechanisms behind the pathogenesis of asthma and atopic dermatitis with those causing CRSwNP, certain biologics initially developed for the treatment of the former have been adapted for use in refractory CRSwNP.
Dupilumab (DUPIXENT) was the first biologic approved for inadequately-controlled CRSwNP by the FDA in 2019. 10 This was followed in 2020 by omalizumab (XOLAIR), which was FDA-approved as an add on maintenance therapy for CRSwNP in adult patients with inadequate response to nasal corticosteroids and evidence of IgE-mediated atopy in the form of positive in vitro or skin allergy testing. 6 Shortly after, the FDA-approved mepolizumab (NUCALA) as an add on maintenance treatment in adult patients with CRSwNP. 11 CRSwNP is not currently listed as an indication for benralizumab (FASENRA) as of the time of this publication. The FDA, however, has issued a complete response letter requesting additional clinical data, suggesting that this may be on the horizon. 12
Current Biologics
Dupilumab
Dupilumab (DUPIXENT) is an anti-IL-4Rα monoclonal antibody. As IL-4Rα is the receptor subunit for IL-4 and IL-13, dupilumab inhibits the downstream effects of both of these cytokines.1,2 It has also been shown to suppress leukotriene E4 levels, which accordingly decreases the activation of mast cells. 13 Consequently, dupilumab has been found to decrease endoscopic nasal polyp score (NPS), decrease Lund-Mackay Computed Tomography (CT) scores, improve nasal congestion, and improve sense of smell in patients undergoing treatment.1,14 It is FDA-approved for patients 12 years and older with severe CRSwNP who have not improved with other medical and surgical management interventions. 1 Dupilumab is self-administered subcutaneously every 2 weeks after an initial training session and observation period in the clinic setting.15,16 It is available in a prefilled syringe or a prefilled pen, both containing 300 mg of the medication. 15 Based on the current listed price and recommended dosage, the annual price is $49 400. 17
While rare, notable side effects include conjunctivitis and hypereosinophilia. 1 It should be noted that dupilumab should be used in caution in patients with high eosinophil levels due to potential unmasking of eosinophilic granulomatosis with polyangiitis (eGPA).
Omalizumab
Omalizumab (XOLAIR) is an anti-IgE monoclonal antibody that inhibits binding of IgE to its receptors on mast cells and basophils, thus mediating the allergic response. 1 Studies, while limited by sample size, suggest that omalizumab decreases polyp scores.1,4 Omalizumab is currently FDA-approved for moderate-to-severe allergic asthma, chronic idiopathic urticaria, IgE-mediated food allergy, and CRSwNP. 18 It is particularly useful for patients with severe CRSwNP and concomitant poorly-controlled allergic asthma. 1 Omalizumab is administered by subcutaneous injection in an office setting by a health care professional, given the elevated risk of anaphylaxis compared with other biologics. 18 Dosing is adjusted based on patients’ weight and pretreatment serum IgE. 19 Doses range from 75 to 600 mg, administered every 2 or 4 weeks. 19 Based on the current listed price, assuming a 65 kg body weight and 250 IU/mL of pretreatment serum IgE, the annual price is ~$45 000. 20
Primary areas of concern for adverse effects include malignancy, cardiovascular disease, and anaphylaxis. 4 This drug has the highest risk for anaphylaxis among all biologics used for CRSwNP, with studies estimating a rate of 0.09% to 0.2%.21-23
Mepolizumab
Mepolizumab (NUCALA) is an anti-IL-5 monoclonal antibody that directly neutralizes free IL-5 levels in serum, thus playing an important role in eosinophil maturation.1,4 Two trial studies suggest that mepolizumab decreases polyp size and the need for revision sinus surgery. 1 It is FDA-approved for CRSwNP and is especially suitable for patients with comorbid eosinophilic asthma. 1 In addition, mepolizumab is now FDA-approved for the treatment of eGPA. 24 This drug is self-administered by subcutaneous injection every 4 weeks, following an initial injection and observation period in the office setting. 25 The current estimated annual price is $44 268. 26
Its most common adverse effects are injection site reaction and oropharyngeal pain. 25 Notably, this drug is considered safer than dupilumab for use in patients with elevated eosinophil counts. 1
Benralizumab
Benralizumab (FASENRA) is a monoclonal antibody that binds to the IL-5Ra subunit on eosinophils, basophils, mast cells, and some B cells.1,4 This mechanism results in competitive inhibition of IL-5 and induced apoptosis of eosinophils and basophils, which is unique compared with mepolizumab which only targets eosinophils. 4 Historically, benralizumab has proven effective in reducing eosinophil counts in airway mucosa and sputum for patients with eosinophilic asthma. 4 Benralizumab is currently FDA-approved in the United States for severe asthma. 27 Like mepolizumab, benralizumab is now FDA-approved for the treatment of eGPA. 28 At time of publication, this drug is not yet FDA-approved for CRSwNP, though approval is currently being sought. Phase III clinical trials have been performed for CRSwNP and show promise in nasal polyp (NP) size and nasal obstruction. 29 Benralizumab may be administered via subcutaneous injection at home using a prefilled pen or in the office setting by a prefilled syringe. Dosing for eGPA is 30 mg every 4 weeks. 27 The current listed price is ~$66 000/year. 30 The most common adverse effects of benralizumab are headache and pharyngitis. 27
Tezepelumab
Tezepelumab (Tezspire) is a monoclonal antibody targeting TSLP, a cytokine produced by epithelial cells that has been shown to play an upstream role in the pathogenesis of asthma. 31 This cytokine is released on disruption of the respiratory epithelium and binds to receptors on immune cells, producing the downstream inflammatory response. 32 Tezepelumab was created to target this mechanism and is currently FDA-approved for severe asthma. 33 Given the upstream effects of TSLP, it has also been speculated that this drug could be effective in the treatment of CRSwNP. Preliminary results of a Phase III clinical trial (WAYPOINT) have demonstrated that tezepelumab provides statistically significant reduction in NPs in patients with CRSwNP. 31 The results of this trial are awaiting peer review and publication. 34 Tezepelumab may be administered as a subcutaneous injection via either prefilled pen at home or prefilled syringe in an office setting by a health care professional. 33 This 210 mg dose is administered every 4 weeks. 33 The current list price is ~$52 000/year. 35 The most-frequently-reported side effects of tezepelumab include pharyngitis, arthralgia, and back pain. 33
New Horizons
Additional biologics are currently in development for atopic dermatitis and asthma. Because of pathophysiological similarities with these conditions, many of these biologics show promise for CRSwNP.
Tralokinumab is an FDA-approved treatment option for moderate-to-severe atopic dermatitis. 36 Two identically-designed clinical trials demonstrated that tralokinumab was effective for improving the severity and symptoms of atopic dermatitis including pruritus, sleep disruption, and quality of life. 37 Tralokinumab was also investigated in phase 3 trials as a treatment for asthma; however, results were inconclusive.38,39 The mechanism of action of tralokinumab involves preventing IL-13 from binding to both IL-13Rα1 and IL-13Rα2. 40 This mechanism suggests that it could have efficacy in patients with CRSwNP, though this has not yet been assessed through clinical trials.
Lebrikizumab is another FDA-approved treatment option for moderate-to-severe atopic dermatitis for patients who weigh at least 88 pounds. 41 Lebrikizumab is a monoclonal antibody that selectively targets IL-13 to inhibit binding to IL-13Rα1 and the formation of the IL-13Rα1/IL-4Rα receptor signaling complex. 42 As with tralokinumab, this drug’s mechanism targeting IL-13 binding suggests that it could have efficacy in patients with CRSwNP. This would need to be assessed further through clinical trials.
Nemolizumab is an FDA-approved treatment for prurigo nodularis. 43 Nemolizumab is an IL-31Rα antagonist. 44 Its mechanism of action involves blocking IL-31 signaling, thus suppressing the downstream immunologic effects of Th2 cells, eosinophils, basophils, and macrophages. 44 By targeting the downstream effects of these various cell types, it is conceivable that nemolizumab could eventually be used to treat CRSwNP.
Other therapeutic targets in development involve the OX40-OX40L pathway. OX40 is a co stimulatory T-cell receptor expressed on effector and regulatory T cells. 45 Its ligand, OX40L, is expressed on macrophages and mediated by TSLP.45,46 OX40-OX40L ligation induces Th2 differentiation.45,46 Consequently, OX40-OX40L poses as an attractive upstream target. 46
Treatment Strategy
Current Guidelines
The 2019 National Institute of Health proposed treatment strategy for CRSwNP begins with medical management, shifting to functional endoscopic sinus surgery (ESS) if initial medical management fails. 47 If medical management and ESS fail, biologics are indicated as an adjunct treatment to ongoing topical corticosteroids. 47 The 2019 European Forum for Research and Education in Allergy and Airway diseases (EUFOREA) takes a more aggressive stance in regard to the use of biologics, setting forth 5 criteria that should be assessed in treating patients with CRSwNP: (1) evidence of Th2 inflammation, characterized by the presence of eosinophilic airway inflammation and related cytokines IL-4, IL-5, IL-13, and/or detection of IgE, (2) systemic corticosteroid usage in the past 2 years, (3) significantly impaired quality of life, (4) significant smell loss, and (5) diagnosis of comorbid asthma. 48 If a patient is surgically naïve and meets 4 of the above criteria, they may be considered for biologic therapy according to EUFOREA standards. If the patient has already undergone ESS, the presence of 3 of the criteria are sufficient to consider biologic therapy. 48
ESS and Biologic Immune Modulators
Effective use of biologics in the treatment of CRSwNP requires consideration of the timing of their use relative to sinus surgery. The purpose of ESS is to restore dysfunctional sinuses by surgically removing diseased mucosa and bony partitions in order to open the natural drainage pathway.1,49 Studies suggest that a complete ESS is more beneficial than a limited ESS like a simple polypectomy.1,47 A complete ESS involves the removal of all bony septations between sinuses and wide opening of the natural ostia. Cohort participants with a more complete ESS were associated with fewer revision surgeries and a greater degree of improved postoperative 22-item Sinonasal Outcome Test (SNOT-22) scores.1,47 Removal of polyps is also believed to facilitate more effective topical corticosteroid delivery.1,50 Continued postoperative medical therapy, such as steroid irrigations, helps to control symptoms and mitigate polyp regrowth. 47
Early ESS Versus Early Biologic
For CRSwNP patients, initial medical management with topical steroids and irrigations is universally recommended by medical allergists and otolaryngologists alike. When this initial medical management fails, however, treatment strategies have begun to vary between otolaryngologists and medical allergists. Otolaryngologists typically recommend surgery after medical failure while some medical allergists may gravitate to earlier use of biologics. 51 Thus, it is important to compare outcomes between ESS and biologics.
Miglani et al performed a prospective multicenter cohort study comparing patients undergoing ESS with those using dupilumab, omalizumab, and mepolizumab at the 24th and 52nd week. 52 ESS and dupilumab had comparable SNOT-22 scores at both the 24th and 52nd week and similar smell identification at the 24th week. 52 Compared with omalizumab and mepolizumab, ESS was consistently more effective in terms of improvement in SNOT-22 scores and NPS. 52
Likewise, clinical trial data revealed that some patients who are started on early biologics ultimately require surgery. 5 Of these patients that demonstrated persistent symptoms and polyps, 1.1% were on dupilumab, 8.7% were on mepolizumab, and 18% were on omalizumab. 5
Comparing ESS With Biologic Therapy to Biologic Monotherapy
Studies have sought to compare the use of biologics alone (monotherapy) to combination therapy with both ESS and biologics. Garvey et al performed a retrospective matched cohort study comparing CRSwNP patients who received biologics only with patients on dupilumab or mepolizumab at the time of ESS. 50 The ESS-biologic cohort had significantly improved polyp burden and SNOT-22, which suggests that the combination of ESS to relieve polyps and improve medication delivery and biologics to facilitate anti-inflammatory effects has a significant clinical impact. 50 This fits with the authors’ clinical experience.
Cost
Duration of treatment is a key factor in considering cost between treatment plans. ESS may have an initial one time higher cost, but with sustained benefits, whereas biologics may have an initial lower cost, but require continued treatment and thus a greater net final cost. 51
The reported costs of ESS vary with sensitivity to extensivity of procedures, complications, and location. 53 A study sourced from charges from the health care system estimates ESS from the payer perspective to be ~$12K (adjusted to 2024 USD).53,54 In comparison, the cost of biologics range from $10K to $40K annually depending on frequency of use. 55 In an economic model evaluating the management of a patient for 36 years, the ESS strategy cost $50 436.99 with 9.8 quality-adjusted life year (QALY) in comparison with a dupilumab treatment strategy that cost $536 420.22 with a 8.95 QALY. 56 This 36 year model suggested that dupilumab was more costly and less effective. In another model, the ESS strategy costs $20 626 with a 1.526 QALY compared with a dupilumab treatment strategy that costs $194 764 with a 1.779 QALY. 57 This 10 year model suggested that dupilumab was 9 times more costly and only slightly more effective.
Choice of Biologic
Once the decision is made to employ a biologic, it is important to choose the right medication for a specific patient. There are no randomized controlled trials that directly compare the effectiveness of individual biologics to one another, when it comes to treating CRSwNP.47,52 However, individual studies and indirect comparisons highlight important differences in mechanism and recommendations.
These considerations have been included in the International Consensus Statement of Allergy and Rhinology (ICAR) 2021 guidelines. 1 Among the 3 FDA-approved biologics, dupilumab is the only drug in this category with level A evidence in the most recent ICAR document. 1 Omalizumab has been assigned level B evidence, with mepolizumab receiving level C evidence. Based on a self-reported survey study among American Rhinologic Society members, dupilumab was found to be the most common biologic used in surgery-naïve patients, ~25× that of omalizumab and ~16× that of mepolizumab. 58
Duration
Current evidence supports the assumption that biologics most likely require lifelong treatment to maintain efficacy.52,55,59 Symptom reoccurrence was observed to begin within 1 to 2 months of biologic cessation. 52 In various studies of biologics, findings suggest that following cessation, patients on dupilumab had worse symptoms and higher NPS. 59 Patients on omalizumab had higher NPS but with eosinophil counts below baseline levels, and patients on dupilumab had higher NPS and eosinophil counts returning to baseline. 59 For mepolizumab, it was observed in a pharmaceutical-funded phase III clinical trial study that patients experienced reduced NP size and reported improved symptoms over 24 weeks following the cessation of treatment after 52 weeks. 59 These results indicate an opportunity for future research into the duration of clinical impact after biologic cessation. Likewise, the prolonged duration of therapy for biologics should be considered in comparison with complete ESS.
Final Considerations
The estimated prevalence of CRSwNP is 4.2% in the United States and 4.3% in Europe. 57 Symptoms of CRSwNP pose a significant detriment to quality of life. Thus, it is valuable to understand the treatment options available. Factors to consider in developing a treatment strategy for CRSwNP include severity, comorbidities, quality of life, cost, potential side effects, and patient preference. Biologics provide an opportunity for continued research into targeting different aspects of the immunologic pathways involved in CRSwNP. Ultimately, given the balance between cost and benefit, evidence to date aligns with current recommendations, with biologics acting as a maintenance option for CRSwNP patients who have failed both medical management and complete ESS (Figure 1 and Table 1).
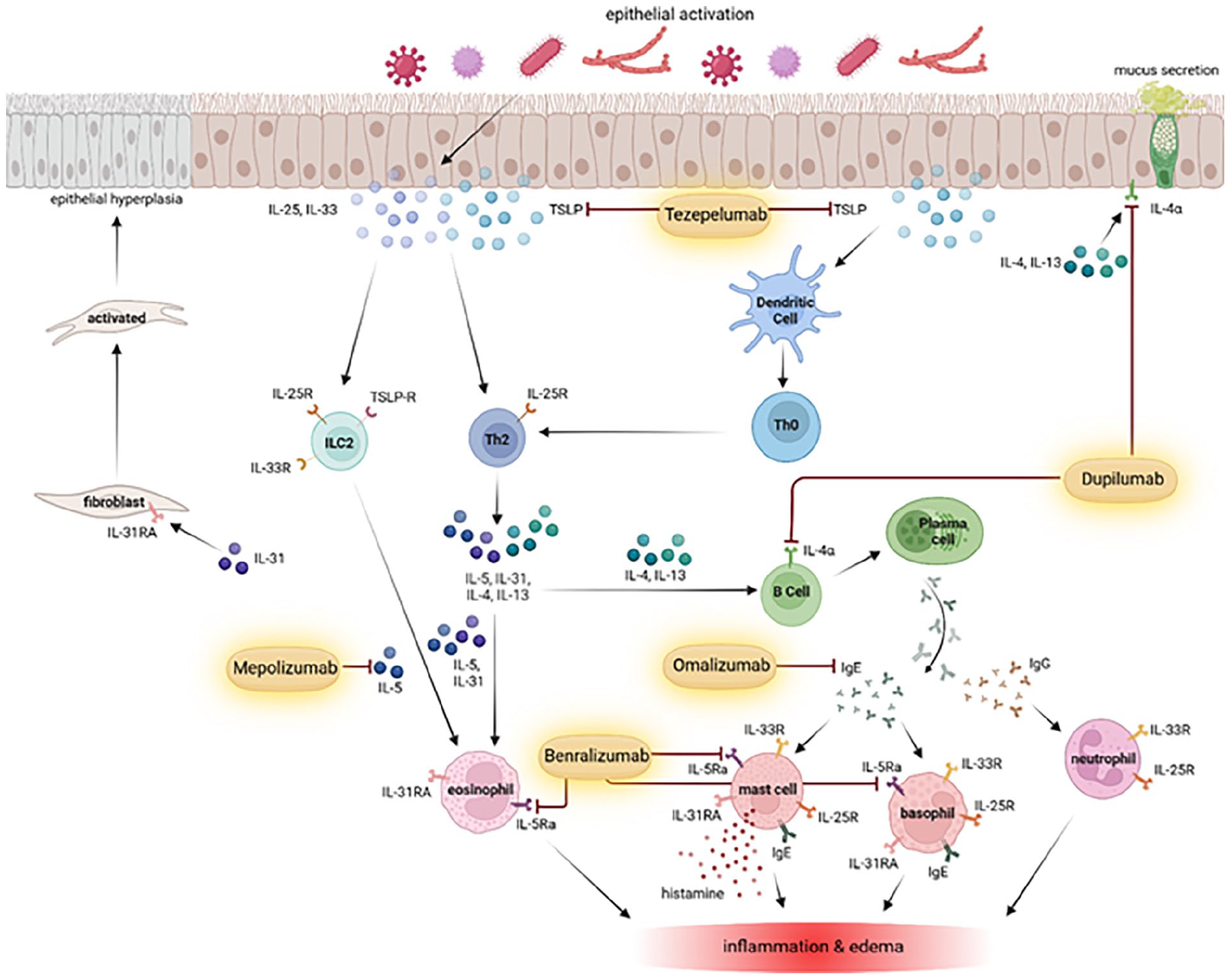
Pathophysiology of CRSwNP and therapeutic targets of current and promising biologics for the treatment of CRSwNP. Created in BioRender.com.
Comparison of Biologics for FDA Indications, Target, Mechanism, Notable Side Effects, Dose, and Cost.
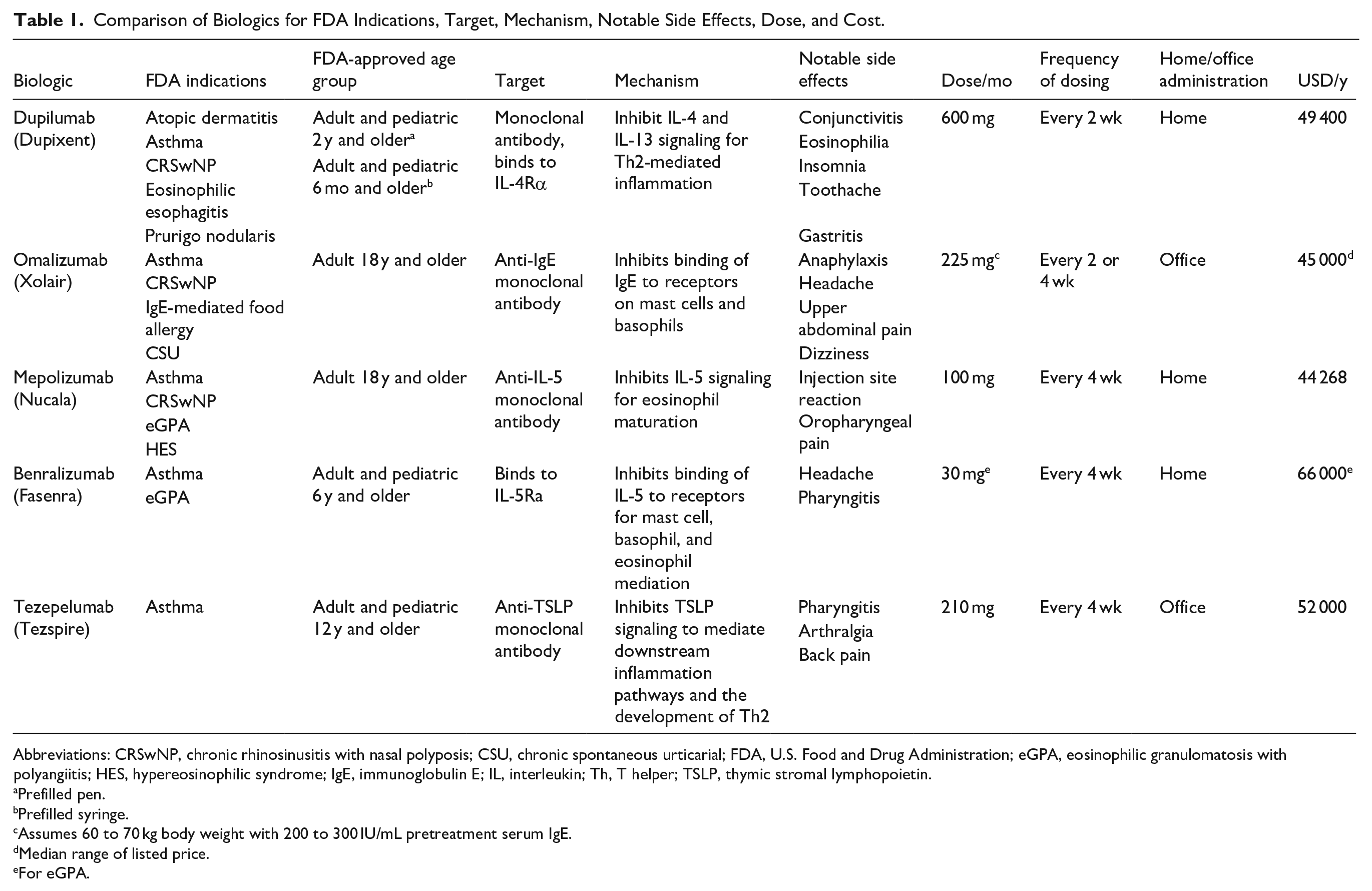
Abbreviations: CRSwNP, chronic rhinosinusitis with nasal polyposis; CSU, chronic spontaneous urticarial; FDA, U.S. Food and Drug Administration; eGPA, eosinophilic granulomatosis with polyangiitis; HES, hypereosinophilic syndrome; IgE, immunoglobulin E; IL, interleukin; Th, T helper; TSLP, thymic stromal lymphopoietin.
Prefilled pen.
Prefilled syringe.
Assumes 60 to 70 kg body weight with 200 to 300 IU/mL pretreatment serum IgE.
Median range of listed price.
For eGPA.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
