Abstract
Objective:
This study aimed to evaluate the effect of e-cigarette use on nasal mucociliary clearance (NMC) by comparing e-cigarette users, traditional cigarette smokers, and nonsmokers. Although numerous studies have investigated the impact of cigarettes and other tobacco products on mucociliary clearance, research specifically focusing on e-cigarettes is limited.
Methods:
The study included 120 healthy participants, comprising 40 electronic cigarette users, 40 cigarette users, and 40 nonsmokers. NMC was evaluated using the saccharin test. Participants were asked to retain a saccharin particle on the inferior turbinate, and the time taken for the participant to perceive the sweet taste was recorded as the NMC time. The results were compared and analyzed statistically using the Statistical Package for the Social Sciences (SPSS).
Results:
The mean NMC time for e-cigarette users was 14.08 ± 5.99, which was significantly longer than that of nonsmokers, who had a mean NMC time of 10.80 ± 5.28 (P = .027), indicating impaired mucociliary clearance. The NMC time for cigarette smokers was 13.13 ± 6.97, which was also significantly prolonged compared to nonsmokers. No statistically significant difference was found between the groups regarding mean age.
Conclusion:
This study demonstrates that electronic cigarette use is associated with impairment of NMC, similar to conventional cigarette smoking. The findings suggest that e-cigarette use may have detrimental effects on the respiratory defense mechanism, potentially increasing the risk of respiratory infections and other pulmonary complications.
Introduction
Nasal mucociliary clearance (NMC) is a vital defense mechanism for both the upper and lower airways. This clearance system is responsible for trapping and removing inhaled particles, microorganisms, and other irritants from the respiratory tract.1-3 The mucociliary system relies on 3 key components: a sufficient amount of mucus, the right rhinological properties, and well-functioning cilia. 4 Disruption of this clearance system can lead to increased susceptibility to respiratory infections and other pulmonary complications. It is important to recognize that various conditions can impact NMC, including allergic rhinitis, nasal septal deviation, turbinate malformations, nasal polyps, sinonasal tumors, cigarette smoking, and genetic defects of the cilia. 4 Smoking is a leading cause of death and disease in humans. 5 Various substances found in cigarettes have been proven to be harmful to the respiratory cilia. Numerous studies have demonstrated that cigarette smoke, along with its various gaseous and particulate components, is linked to a range of structural and functional changes in the respiratory system. These changes include heightened oxidative stress, inflammation of the airways and lungs, disrupted anion transport, and abnormalities in both the ultrastructure and formation of cilia. 4
E-cigarettes have been popularized as being less harmful than traditional cigarettes. They are often considered less dangerous than traditional smoking because they avoid the inhalation of tobacco’s combustible by-products, which are more carcinogenic and harmful than nicotine alone. 6 However, e-cigarettes have been linked to various health issues, including cardiovascular effects such as a higher risk of heart attacks; thermal injuries from explosions leading to severe soft tissue and facial damage; and psychosocial effects related to addictive behavior.7-9 Pulmonary complications have been reported infrequently and sporadically. 7 The rapid rise in the popularity of electronic cigarettes (e-cigarettes) has prompted concerns about their potential health impacts, particularly on the respiratory system. E-cigarettes generate an aerosol through the heating of a liquid, which typically contains nicotine, propylene glycol, vegetable glycerin, and various flavorings. 10 Although e-cigarettes may expose users to fewer toxicants than traditional cigarette smoke, the effects of e-cigarette aerosols on the respiratory defense mechanisms, such as NMC, are not well characterized.
The saccharin test, as described by Anderson et al is an affordable, safe, straightforward, and effective method for evaluating NMC. 11 It has been suggested as a reliable screening tool for identifying abnormal NMC. Although the saccharin test is a widely accepted and noninvasive method for assessing NMC, it remains a subjective test reliant on patient response, as it measures the time taken for the perception of a sweet taste. Therefore, it is not a fully objective method. However, it is considered a reproducible and reliable test, as similar results have been obtained when repeated in healthy individuals under similar conditions. This reproducibility has been validated in the literature, including a recent systematic review by Caponnetto et al. 12
The aim of this study is to evaluate and compare the NMC function in electronic cigarette users, traditional cigarette smokers, and nonsmokers using the saccharin test.
Materials and Methods
Our study was approved by the Ethical Committee of Trakya universtiy, desicion date/number: 23.10.2023/ 16/31. This cross-sectional study was conducted between May 2024 and August 2024, and participants were divided into 3 groups: nonsmokers, smokers, and e-cigarette users. The study protocol and participant flow are illustrated in Figure 1. The sample size in the study was determined based on a power analysis, assuming 80% power (1 − β = .80) and a 5% significance level (α = .05), with a medium effect size (Cohen’s d = 0.5).
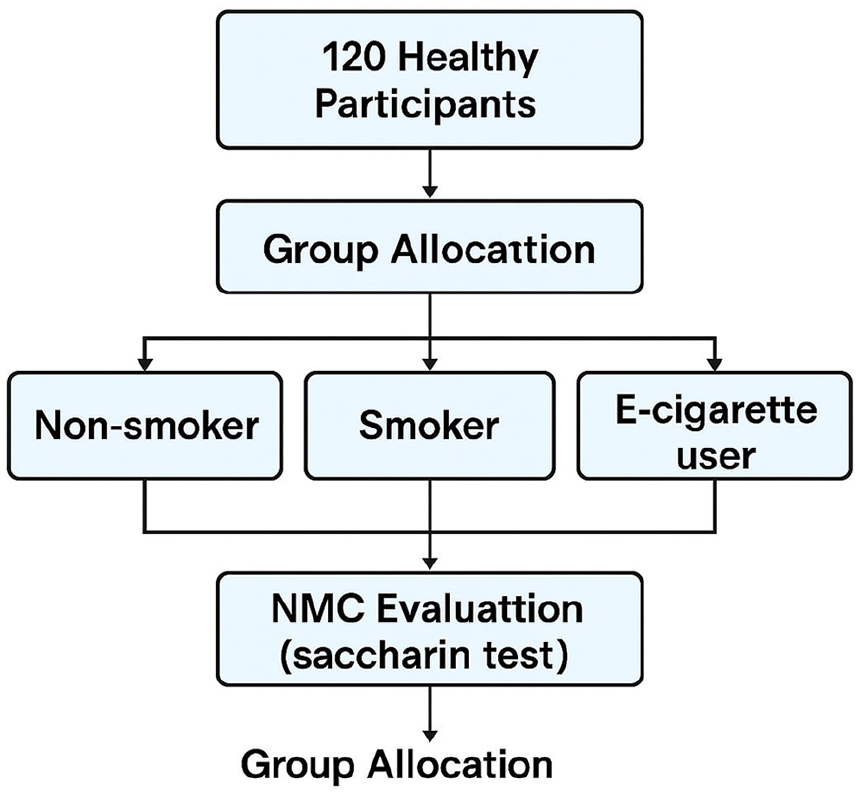
Flow diagram of the study design.
Participants included 120 healthy individuals between the ages of 16 and 61 years, with 40 participants in each group. Individuals were considered e-cigarette users if they had used e-cigarettes regularly for at least 6 months prior to the study, and cigarette smokers if they had smoked at least 1 pack per day for at least 6 months. Patients with upper respiratory tract infections, chronic obstructive pulmonary disease (COPD), bronchial asthma, severe systemic illnesses, pre-existing ciliary motility disorders, or any nasal conditions such as deviated nasal septum, allergic rhinitis, polyps, or masses were excluded from the study. Patients with a history of nasal surgery or trauma, those using antihistamines or any intranasal medication, and participants who did not provide consent were also excluded from the study. A comprehensive Ear, Nose, Throat examination was conducted for each patient to rule out any incidental nasal pathology, and all participants underwent a saccharin test.
Saccharine clearance test: The Saccharin test, originally defined by Anderson et al in 1974 and later modified by Rutland and Cole in 1980, was utilized in both experimental groups. 11 The saccharin test was conducted by placing a 1 mm saccharin particle 1 cm from the anterior end of the on the inferior turbinate under direct visualization using a nasal endoscope or nasal speculum. Participants were instructed to sit in an upright position and not to sneeze, swallow, or move their tongue. The time taken for the participant to perceive the sweet taste of saccharin was recorded in minutes and seconds. All saccharin tests were conducted under standard clinical conditions. Room temperature (22-24°C) and relative humidity (40%-60%) were maintained and monitored during the study period to minimize environmental influence on NMC measurements. All participants in the smoker and e-cigarette user groups were instructed to abstain from smoking or vaping for at least 12 hours prior to the saccharin test, in accordance with the study protocol designed to minimize acute effects on mucociliary function.
Statistical Analysis
Data was analyzed using SPSS 20.0 software (IBM Corp. Released 2011. IBM SPSS Statistics for Windows, Version 20.0: IBM Corp., Armonk, NY, USA). Normality distribution of the numeric variables was tested by using the Shapiro-Wilk test. The One-way ANOVA test was used in the comparison of age among the 3 groups (nonsmokers, smokers, and e-cigarette users) due to normal distribution. However, the Kruskal-Wallis test was used in the comparison of NMC among the groups due to nonnormal distribution. The Mann-Whitney U test was used to compare the normal cigarette pack-year values between the smoker and e-cigarette user groups due to nonnormal distribution. The Chi-square test was used for comparing categorical data among the 3 groups. A P-value of less than .05 was set as the threshold for statistical significance.
Results
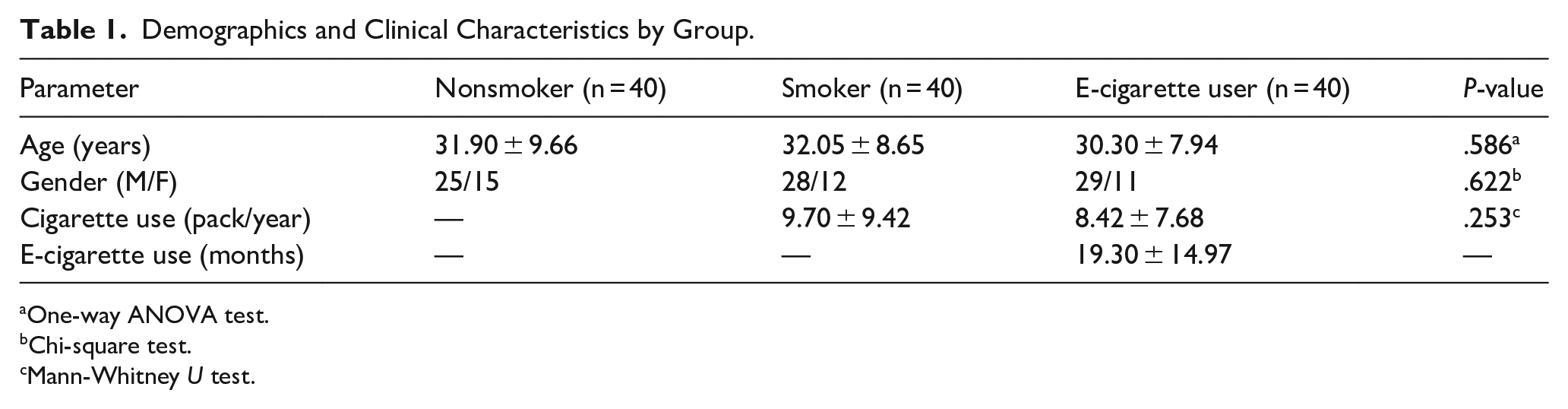
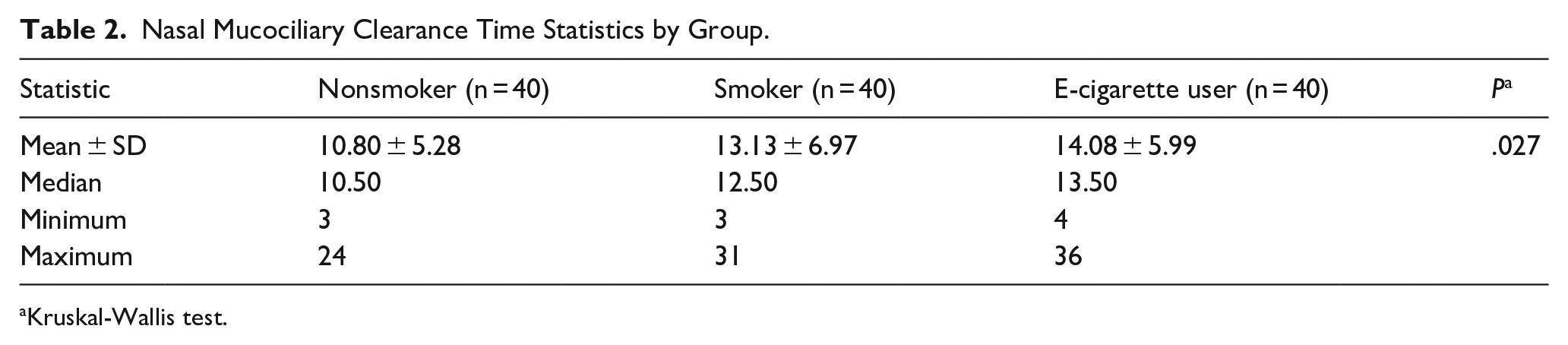
A total of 3 different groups were included in our study: 40 were nonsmokers, 40 were smokers, and 40 were in the electronic cigarette user group. Of the volunteers participating in the study, 38 were female (31.7%), and 82 (68.3%) were male. The mean age of the participants in the nonsmoker, cigarette smoker, and e-cigarette user groups were 31.90 ± 9.66, 32.05 ± 8.65, and 30.30 ± 7.94 years. There was no statistically significant difference between the groups in terms of mean age (F(2.117) = 0.488, P = .586). The usage duration for electronic cigarettes was determined in months, while for regular cigarettes it was determined in packs per year. In the group of regular cigarette smokers, the average usage amount was calculated as 9.70 packs/year ± 9.42; in the group of electronic cigarette users, the average usage duration was calculated as 19.30 months ± 14.97 (Table 1). Ninety-five percent of electronic cigarette users had a history of traditional tobacco smoking, and among these individuals, the average duration since cessation of traditional tobacco use was 11.2 ± 6.7 months. When comparing the regular cigarette pack years between the smoking and electronic cigarette groups using the Mann-Whitney U test, no significant difference was found (P = .253). The Kruskal-Wallis test was used to compare the NMC values among the 3 groups. The average NMC values were calculated as follows: 10.80 ± 5.28 in the nonsmoking group, 13.13 ± 6.97 in the smoking group, and 14.08 ± 5.99 in the electronic cigarette group (Figure 2). When comparing the groups in terms of NMC values, a significant difference was found between the electronic cigarette users and nonsmokers (P = .027) (Table 2). There was no statistically significant difference between the smoking and electronic cigarette groups (P = .765).
Demographics and Clinical Characteristics by Group.
One-way ANOVA test.
Chi-square test.
Mann-Whitney U test.
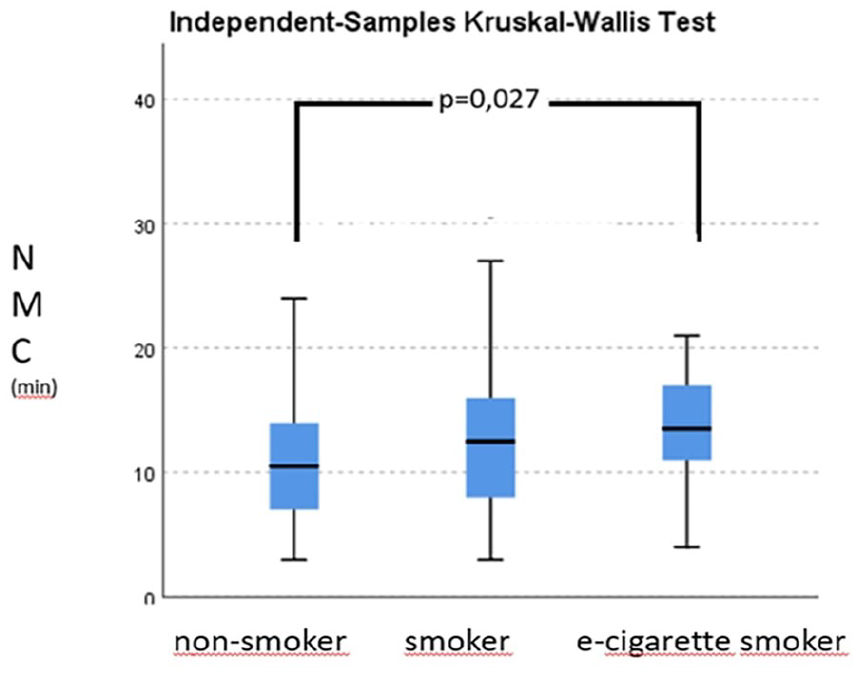
Kruskal-Wallis test results for smokers, nonsmokers, and e-cigarette smokers, showing the distribution in minutes (min).
Nasal Mucociliary Clearance Time Statistics by Group.
Kruskal-Wallis test.
Discussion
This study aimed to evaluate and compare the NMC function in electronic cigarette users, traditional cigarette smokers, and nonsmokers. The results demonstrated that the NMC times for both the e-cigarette group and the traditional cigarette group were significantly impaired compared to the nonsmoker group (P < .005). However, the degree of impairment in NMC was similar between the e-cigarette users and traditional cigarette smokers, indicating that e-cigarette use may have comparable adverse effects on NMC function as traditional smoking.
Worldwide, smoking significantly contributes to morbidity and mortality, particularly in developing countries.13,14 The World Health Organization estimates that tobacco use and exposure currently cause the deaths of approximately 6 million people worldwide each year. 15 Cigarette smoking is a major risk factor for several respiratory diseases, including COPD, lung cancer, and idiopathic pulmonary fibrosis. 16
The tobacco smoking industry has evolved significantly over the past decades, with the introduction of e-cigarettes as a potential smoking cessation aid. E-cigarettes have been marketed as a safer alternative to traditional cigarettes. Despite the growing popularity of e-cigarettes as a smoking cessation tool, there are still concerns about their long-term safety. These battery-operated devices were initially created to heat chemical substances containing nicotine and flavorings, producing a smokeless vapor known as aerosol for the user to inhale. 17 The solution in the e-cigarette cartridge, known as e-liquid or e-juice, typically consists of nicotine, flavoring ingredients in propylene glycol, and vegetable glycerin. In addition, some vaping devices include cannabis, which expands the composition and appeal of the aerosols. 18 The cartridge might also include heavy metals such as nickel, tin, lead, or chromium. 19 Although e-cigarettes have fewer components than traditional cigarettes, they still contain toxic and carcinogenic residues, such as formaldehyde, acetaldehyde, and acrolein. 20 While e-cigarettes may deliver nicotine in a less harmful manner than traditional cigarettes, they still expose users to potentially toxic chemicals that can impair NMC and contribute to respiratory problems. Various studies have shown that e-cigarettes are not harmless and may also adversely affect respiratory health.21,22 Specifically, e-cigarette use has been associated with an increased risk of lung injury, asthma, and other respiratory conditions. 21 This study provides additional evidence that e-cigarette use, similar to traditional cigarette smoking, can have detrimental effects on NMC. The impairment of NMC observed in e-cigarette users may potentially lead to increased susceptibility to respiratory infections and chronic lung diseases over time.
To keep the paranasal sinuses healthy, the mucociliary clearance pathway serves as an innate defense system against organic and inorganic particles that enter the airway. It physically clears inhaled air through mucociliary flow, which consists of cilia covering the respiratory epithelium and a mucus layer. This system transports the mucus layer toward the nasopharynx. Then eliminated by swallowing or coughing. Disruption of this system can lead to the accumulation of mucus and inhaled particles, increasing the risk of respiratory infections and other airway diseases. A defect in this defense mechanism can cause inflammation and obstruction in the small airways, increase susceptibility to respiratory infections, contribute to lung damage, impair wound healing, and accelerate the progression of chronic respiratory diseases. 4 This study investigated the impact of electronic cigarette use on mucociliary clearance.
Although the saccharin test is a widely accepted and noninvasive method for assessing NMC, it remains a subjective test reliant on patient response. Rhinoscintigraphy, in contrast, provides an objective and quantitative assessment of NMC by utilizing radiolabeled particles and gamma camera imaging. This technique allows for more precise and reproducible measurement of ciliary transport time and mucociliary efficiency. However, its higher cost, limited accessibility, and radiation exposure make it less practical for routine clinical use or large-scale studies. The choice of the saccharin test in this study was based on its safety, ease of administration, and cost-effectiveness for screening purposes.
The current study showed that both traditional cigarette smoking and e-cigarette use resulted in significant impairment of NMC compared to nonsmokers (P < .05). In the studies conducted by Dulger et al and Chethana et al also found that the NMC was higher in smokers.16,23 In the systematic review conducted by Prasetyo and colleagues, published in 2020, it was reported that smoking impairs NMC. 24 In this review, it was reported that not only traditional cigarette smoking but also bidi, hookah, electronic cigarettes, and passive smoking harm the respiratory system and disrupt NMC. 24 In our study, it was found that NMC is also impaired in e-cigarette users. In the review published by Cherian and colleagues, it was reported that lung injury is statistically more common in e-cigarette users. 10 Exposure to e-cigarettes triggers various stress and inflammatory responses in the body areas that first come into contact with it: the mouth, nasal passages, trachea, bronchial system, and lungs. Exposure to propylene glycol, the primary component of e-cigarette liquid, has been linked to airway irritation, airway obstruction, and increased severity of dyspnea in individuals who did not previously experience these conditions.25,26 This study suggests that e-cigarette use may not be a harmless alternative to traditional smoking and may also have detrimental effects on respiratory health. Given these findings, healthcare providers should have comprehensive discussions with patients about the potential risks and benefits of e-cigarette use and consider alternative smoking cessation strategies that are evidence-based and approved by regulatory bodies, such as nicotine replacement therapy.27,28
Limitations
Limitations of this study include the relatively small sample size, as well as the lack of a longitudinal design to assess changes in NMC over time with continued e-cigarette or traditional cigarette use. In addition, the study did not assess the specific e-cigarette device and e-liquid characteristics that may contribute to the observed effects on NMC. Ninety-five percent of the e-cigarette users had a prior history of traditional cigarette smoking, which may introduce confounding effects and make it difficult to isolate the specific impact of e-cigarette use on NMC. This overlap should be considered a limitation when interpreting the results. Moreover, the substantial difference in average usage duration between traditional cigarette users (mean 9.7 years) and e-cigarette users (mean 19.3 months) may affect the comparability of NMC impairment between these groups. Further research with larger sample sizes and a longitudinal approach is needed to better understand the long-term impact of e-cigarette use on respiratory health.
Conclusions
This study found that both traditional cigarette smoking and e-cigarette use were associated with significant impairment of NMC compared to nonsmokers. The disruption of this crucial respiratory defense mechanism may increase the risk of respiratory infections and other chronic lung diseases in e-cigarette users, similar to traditional cigarette smokers. Healthcare providers should carefully consider the potential risks and benefits when discussing e-cigarette use with patients and promote evidence-based smoking cessation strategies.
Footnotes
Acknowledgements
None.
Ethical Considerations
This study was approved by the local ethics committee of the Trakya University ethics commitee desicion number 16/31. Written informed consents were obtained from all the participants for their anonymized information to be published in this article.
Consent to Participate
Written informed consent was obtained from the patients who agreed to take part in the study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Meeting Information
This study was presented as an oral presentation at the 42nd Turkish National ENT Congress (October 23-27, 2024) and received the second-place award.
