Abstract
Objectives:
The study aims to investigate the possible side effects of isotretinoin use on the nasal mucosa with objective methods in the treatment of acne vulgaris.
Methods:
Before the treatment, nasal mucociliary clearance time (MCT) was measured in all patients. Also all patients were asked to complete the questionnaires about the nasal dryness (visual analog scale [VAS]), nasal obstruction (Nasal Obstruction Symptom Evaluation [NOSE]), and presence of epistaxis (Epistaxis Severity Score [ESS]). Both MCT and questionnaires were reevaluated in patients who completed the treatment.
Results:
The results of 101 patients were evaluated. Before treatment, mean duration of nasal mucociliary clearance (NMC) was 9.55 ± 1.30 minutes, nasal dryness (VAS) value was 2.7 ± 0.7, NOSE score was 2.1 ± 1.1, and ESS score was 1.2 ± 0.7; after treatment, the duration of NMC was 13.80 ± 2.29 minutes, VAS value was 3.3 ± 1.1, NOSE score was 3.2 ± 1.3, and ESS score was 2.1 ± 1.2 (P = .018, .150, .027, .011, respectively).
Conclusion:
The nasal mucosa is adversely affected in patients due to regular use of isotretinoin in the acne treatment, anamnesis should be checked in all nasal surgeries, and routine ear nose throat control should be recommended for these patients.
Introduction
Oral isotretinoin is the most effective acne treatment agent used in the treatment of severe acne for more than 25 years, and it affects all etiological factors involved in acne pathogenesis. 1 It significantly reduces sebum production from sebaceous glands and downscales sebaceous glands’ size, normalizes follicular keratinization, and prevents the development of microcomedones and comedones. 2 Mucociliary clearance is a defense mechanism that enables mucosa, which is in the nasal cavity with the particles, to move nasopharynx from the front part of the nose via wave actions of cilias. 3 Disruption of mucociliary clearance causes many pathologies such as rhinitis, sinusitis, otitis media, and odor disorders. 4 The results of the adverse effects of isotretinoin use on the nasal mucosa and mucociliary clearance time (MCT) have been reported in the literature. The aim of this study was to investigate the effects of isotretinoin use on nasal mucosa by mucociliary clearance measurement and other patient’s self-reported universal scales.
Materials and Methods
Clinical records of the patients were reviewed for study retrospectively. All patients who were diagnosed with acne vulgaris and treated with isotretinoin were followed up with routine blood tests before the beginning of treatment, with monthly periods during the treatment and at the end of the treatment. Patients who accepted isotretinoin treatment were consulted to the ear nose throat (ENT) clinic before and after treatment. After anamnesis and physical examination, information about the study was given and patients, who agreed to participate voluntarily and signed the consent form, were included in the study.
Patients with nasal surgery, allergic or nonallergic rhinitis and chronic sinusitis, upper respiratory tract infection in the last 4 weeks, any history of topical or systemic drug use, smoking and septum deviation, concha hypertrophy, nasal polyp, chronic otitis media, and systemic disease were excluded. Patients with chronic sinusitis, with nasal topical drug use, with systemic or topical steroid use, and disrupting the isotretinoin treatment were also excluded from the study at the final examination.
Systemic isotretinoin therapy was started in patients diagnosed with acne vulgaris as 0.5 to 0.75 mg/kg/d orally, and for the 6 months, the total dose was aimed to be 120 to 150 mg/kg. Nasal dryness (visual analog scale [VAS]; 0 none, 10 worst), nasal obstruction (Nasal Obstruction Symptom Evaluation [NOSE]), 5 and presence of epistaxis (Epistaxis Severity Score [ESS]) were examined just before and at the end of the treatment.
Saccharin test was applied to the patients at the beginning and at the end of the treatment. All patients were rested half an hour before the test. The test was carried out at room temperature and in a sitting position with the patients’ heads tipped slightly forward. A 1-mm-diameter saccharin tablet was placed on the medial surface of the left inferior turbinate. 6 The time interval between the insertion of saccharin and the moment the patient felt the taste was accepted as MCT.
The data were analyzed by SPSS 22.0 (Armonk, New York) packet data program. The MCT results, VAS, NOSE, and ESS scores were evaluated by paired t test before and after treatment. The ratio of symptoms investigated was compared with χ2 test before and after treatment. The results which had significance values less than .05 were considered as statistically significant. This study was conducted in accordance with the Declaration of Helsinki. Ethics committee approval was received from the affiliated institution (2019/1415).
Results
A total of 108 volunteers were included in the study. The mean age was 21.4 ± 3.52 years. At the first examination, 63 (58.3%) of the patients were female and 45 (41.7%) were male. Seven patients were out of follow-up. A total of 101 patients completed the study. The mean age of the patients was 20.9 ± 3.95 years. Sixty (59.4%) participants were female and 41 participants (40.6%) were male. The mean MCT before the treatment was 9.55 ± 1.30 minutes and 13.80 ± 2.29 minutes at the end of the treatment. There was a statistically significant prolongation of mucociliary clearance after treatment (P = .018, paired t test; Figure 1). The obtained values of nasal dryness (VAS) were approximately 2.7 ± 0.7 before the treatment and 3.3 ± 1.1 at the end of treatment; the increase between values is significant (P = .015, paired t test). The mean NOSE scores of the patients before the treatment were 2.1 ± 1.1, and when the treatment was completed, it was 3.2 ± 1.3 (P = .027, paired t test). The ESS questionnaire was applied to patients with epistaxis, and the mean score was 1.2 ± 0.7 at the beginning of treatment and it was 2.1 ± 1.2 at the end of treatment (P = .011, paired t test). Five (4.9%) patients had a history of epistaxis in the last 6 months before the treatment, and this number increased to 60 (59.4%) when the treatment was completed (P = .021, χ2 test). Therewithal, the duration of NMC, nasal dryness (VAS), NOSE, and ESS scores obtained before and after treatment were not affected by age and gender (P > .050, paired t test). According to post hoc power analysis, the actual power (β) for the prolongation of MCT was calculated as 99%.
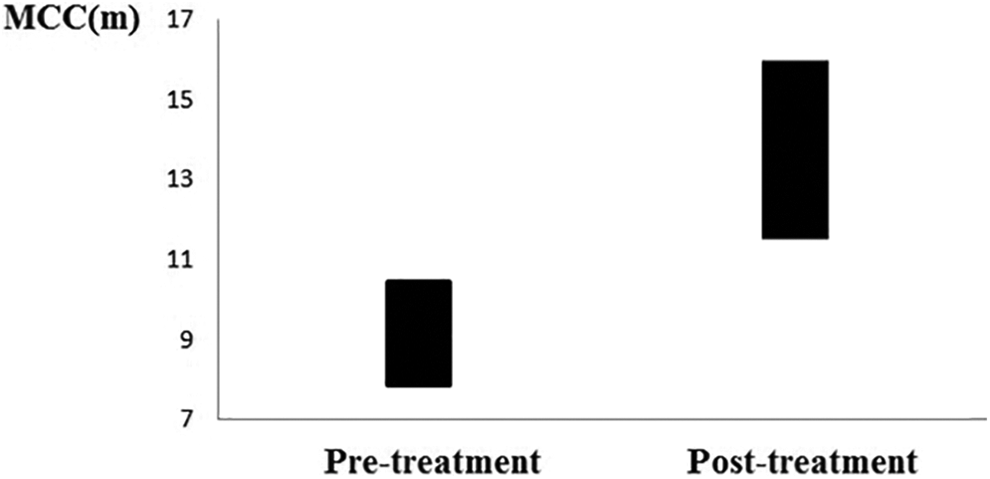
Mean of mucociliary clearance time before and after treatment. m indicates minutes; MCC, mucociliary clearance.
Discussion
Since the mucociliary system is one of the most important defense mechanisms of the airway and the force that provides the mucus transport is the continuous pulsing cilia, regulation of the ciliary pulse frequency has an important role in measuring the mucociliary system. 4 An abnormal ciliary function is known to predispose to infection in the upper and lower respiratory tract and middle ear. 7 Saccharin test is an inexpensive and easily applicable method to measure MCT, and with this test, the mean MCT in healthy individuals was reported as 17.17 ± 8.43 minutes. 8 Isotretinoin is the most effective treatment agent used in the treatment of acne. 1 Tasli et al 9 have previously demonstrated that in the first month of isotretinoin treatment, MCT prolonged significantly (9.9 ± 2.06 minutes) according to the saccharin test. In this study, the MCT was 13.80 ± 2.29 minutes at the end of 6 months and was significantly longer (P = .018). Tasli et al 9 did not detect a significant difference in the MCT in the first and third months of treatment but did not measure the time of NMC at the end of treatment. Gorpelioglu et al 10 found in their study including 40 patients with acne vulgaris had significantly longer MCT (15.9 ± 5.7 minutes) in the third month of treatment than the beginning of treatment (P < .001). In the same study, a positive and significant correlation was found between isotretinoin dose and MCT. 10 In rabbit model with isotretinoin, cilia loss in wound tissue and goblet cell loss even in normal mucosa have been shown histologically. 11 The weakness of this study is that it cannot support clinical outcomes with histological evidence.
The most common side effect of isotretinoin treatment is mucosal dryness, and concomitant cheilitis and dryness on eyes are reported to be 30%. 12 Although it is seen as a side effect that is not emphasized in the dermatology literature, it has been shown that the complaints related to nasal dryness increase twice in patients using isotretinoin compared to normal patients. 2 In this study, the mean VAS score of the patients was 2.7 ± 0.7 at the beginning of treatment and 3.3 ± 1.1 at the end of the treatment (P = .015). Similarly, Tasli 9 found nasal dryness in 9.5% of patients before treatment and in 43% of patients in the third month of treatment. In the same study, the mean VAS score (min: 0, max: 100) for nasal dryness was 0.47 ± 1.48 at the beginning of treatment, and in the third month of treatment, it was 17.17 ± 4.28 ± 6.0 (P < .05). 9 In a retrospective study with results of 3525 patients, Brzezinski et al 13 found that dry mouth complaints were 1.4% and epistaxis complaints were 47.2%, and nasal dryness was not mentioned. Similarly, nasal findings and symptoms were not been mentioned even though epistaxis rates calculated in the study, which includes analysis of 106 articles examining isotretinoin use by Bauer et al. 12 Only Vallerand et al 2 listed the side effects of ENT system as mouth and nasal dryness, epistaxis, rhinitis, and sore throat and reported that the prevalence of these symptoms was twice as high in patients receiving isotretinoin compared to the control group. Specific rates of ENT symptoms listed in this study were not mentioned. 2 The reason of this is that in literature, the nasal dryness is considered to be the underlying mechanism causing the decreased MCT (which is the objective finding of isotretinoin use) and epistaxis (which is the subjective finding of isotretinoin use). 2,9,10,13,14
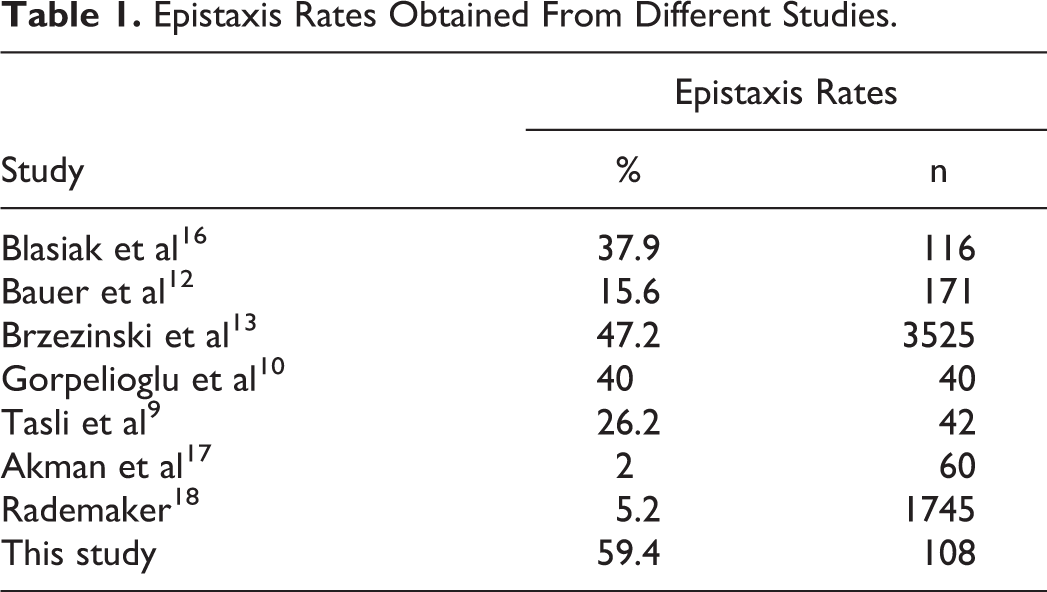
In our study, epistaxis was evaluated with ESS questionnaire. 15 Five (4.9%) patients had a history of epistaxis in the last 6 months before the treatment, and this number increased to 60 (59.4%) when the treatment was completed (P = .021). The mean ESS score was 1.2 ± 0.7 at the beginning and 2.1 ± 1.2 at the end of the treatment, and the increase was statistically significant (P = .011). Similarly, Tasli et al 9 found epistaxis complaint in 7% of the patients before treatment and 31% and 26% of the patients, respectively, in the first and third months of treatment. In the same study, the epistaxis score (min: 0, max: 100) was 0.35 ± 1.30 at the beginning, 2.61 ± 4.45 in the first month of treatment, and 2.26 ± 4.71 in the third month of treatment, and the mean scores of the first and third months were significantly higher than the beginning score (P < .050). In their review of 3525 patients, Brzezinski et al 13 reported that the complaint of epistaxis was found to be 47.2% and that they may require a short-term (1-5 days) interruption of treatment. The frequency of epistaxis varies between 11.6% 2 and 47.2% 13 in different studies (Table 1). Epistaxis in the treatment of isotretinoin is a side effect that should be emphasized as much as mucosal dryness and cellulitis.
Epistaxis Rates Obtained From Different Studies.
Conclusion
The common side effects of isotretinoin use are epistaxis and nasal dryness of the ENT system, but they are overlooked. In this study, it has been shown that regular use of isotretinoin causes a statistically significant prolongation of MCT. Epistaxis and nasal crusting were also detected. Therefore, isotretinoin treatment should be questioned in all nasal surgeries and patients receiving isotretinoin should be informed in terms of epistaxis and other nasal symptoms before treatment and followed up routinely. Nasal moisturizers can be routinely recommended for these patients before starting isotretinoin treatment. In our clinical practice, we recommend nasal moisturizers to patients at the beginning of isotretinoin therapy. It can also be recommended to use during the treatment and according to the severity of the complaints (epistaxis and crusting) within 6 months after the end of the treatment. In the first step, saline can be recommended to these patients. The pattern of administration may vary depending on the severity of the symptoms (eg, sun exposure), 4 to 6 times a day. It can also be preferred in ready-to-use products containing D-panthenol, Vaseline, glycerin, hypertonic solutions, and hyaluronic acid.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
