Abstract
Objective:
Different criteria have been proposed for the response to biologic treatment to evaluate the efficacy of treatment among patients with chronic rhinosinusitis with nasal polyps (CRSwNP). This review aimed to summarize current response criteria and evaluate response rates of patients with CRSwNP after biologic therapies.
Methods:
We systematically searched PubMed, Embase, Web of Science, and the Cochrane Library from the inception to July 2024 to identify clinical studies exploring the response to biologic treatment in patients with CRSwNP.
Results:
Twenty-nine studies were included, comprising 3735 patients in 25 studies suitable for meta-analysis. These were various comprehensive and single criteria for response to biologic therapy in patients with CRSwNP. The comprehensive evaluation measures of patient responsiveness included 5 key outcome measures: nasal polyp size, systemic corticosteroid need, disease-specific quality of life, sense of smell, and impact of comorbidities. The single evaluation measure of treatment response included nasal polyp size, disease-specific quality of life, and sense of smell. The minimum clinically important difference and the thresholds of these parameters are used to determine the clinical improvement or the severity of the disease after treatment. These existing treatment response instruments categorized patients as follows: 4 (excellent, moderate, poor, and no response), 3 (good-to-excellent, moderate-poor, and no response), and 2 (response and no response). The pooled moderate-to-excellent response rate after biologic therapy assessed by comprehensive criteria was from 39.3% (95% confidence interval [CI], 0.231-0.670) to 87.7% (95% CI, 0.774-0.994). The pooled response rates evaluated by the single measures ranged from 62.9% (95% CI, 0.518-0.746) to 82.8% (95% CI, 0.801-0.857).
Conclusion:
The critical components and parameters of the response criteria for CRSwNP patients receiving biologic treatment have yet to be reached, leading to significantly heterogeneous response rates to biologics. The development of consistent criteria for the response to biologic treatment is critical to help define treatment efficacy.
Introduction
Chronic rhinosinusitis (CRS) is a chronic inflammatory sinonasal disease that affects 5% to 12% of the general population 1 and causes direct costs of $8.6 billion per year.2,3 CRS is initially classified into 2 phenotypes: CRS with and without nasal polyps (CRSwNP and CRSsNP), with the type of CRSwNP accounting for 18% of the total CRS. 4 Although maximum medical therapy was implemented, a study found that 62% of CRS patients still suffered from persistent symptoms. 5 Approximately 40% of patients with CRSwNP relapsed into nasal polyposis within 18 months after endoscopic sinus surgery. 6 Therefore, therapy must be revolutionized urgently for patients with CRSwNP to achieve long-term remission.
Recently, the endotype of CRSwNP, which explains the underlying pathophysiological mechanisms of the disease, has offered additional treatment options. 7 It has been estimated that about 80% of patients in Western countries with CRSwNP undergo a type 2 inflammatory profile, especially among those with comorbid asthma and aspirin-exacerbated respiratory disease (AERD).8-10 Type 2 CRS is typically characterized by elevated levels of type 2 cytokines (principally interleukin-4 [IL-4], IL-5, and IL-13), IgE, and eosinophils in both serum and tissues,11,12 which is significantly associated with the presence of nasal polyps, asthma, anosmia, and allergic mucin, 13 and is prone to relapsing. 14 In recent years, evidence has confirmed that monoclonal antibodies targeting the cytokine-mediated inflammatory cascade of type 2 have promising efficacy in sinonasal symptoms, quality of life, and reduced use of corticosteroids and sinus surgery.15-18 Current emerging biologics for CRSwNP include Dupilumab (anti-IL-4 receptor alpha, blocking IL-4/IL-13 signaling), Omalizumab (anti-IgE, reducing IgE levels in serum and tissues), Mepolizumab (anti-IL-5, inhibiting eosinophilic inflammation), Reslizumab (anti-IL-5, preventing IL-5 from binding to eosinophils), and Benralizumab (anti-IL-5 receptor alpha, inducing eosinophils apoptosis),11,12,19,20 which are administrated as add-on therapies by subcutaneous injection.
After the initial administration of biologics, accurate identification of responders is essential to help clinicians determine whether to discontinue or switch drugs, formulate subsequent treatment strategies, and reduce unnecessary medical expenses. A clear and applicable definition of the response to biological therapy in patients with CRSwNP is in urgent need for unity in expert consensus. 21 However, different response criteria were proposed to evaluate the success of biological therapies with multiple parameters, including the 2020 European Position Paper on Rhinosinusitis and Nasal Polyps (EPOS2020) response criteria, 22 2021 European Forum for Research and Education in Allergy and Airway Diseases (EUFOREA2021) response criteria, 23 the 22-item Sinonasal Outcome Test (SNOT-22), and the nasal polyps score (NPS). It should be noted that a fraction of patients still suffer from persistent symptoms after biological treatments despite the high cost associated with these biologics. Under different evaluation measures, the percentage of patients who would respond, especially with different biological treatment regimens, has not yet been confirmed.
To date, no studies have summarized common response evaluation measures applied in real-life studies or randomized controlled trials (RCTs) and investigated response rates of biologics utilizing different response criteria in patients with CRSwNP. Therefore, the main objectives of this systematic review and meta-analysis were (1) to summarize the various methods of evaluation of response to biological therapy and (2) to analyze the overall biological therapeutic response rates in different response criteria.
Methods
Search Strategy
The Statement of Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) was followed to perform the systematic literature search in PubMed, Embase, Web of Science, and the Cochrane Library, with search strategies as follows: (chronic rhinosinusitis OR nasal polyp) AND (biologics OR Dupilumab OR Mepolizumab OR Reslizumab OR Benralizumab OR Omalizumab) AND (response OR responder OR responders). The most recent search was conducted on July 23, 2024. In addition, a manual search of the reference lists in the included literature was performed to ensure the inclusion of all possible studies. The study protocol was registered in the PROSPERO database on September 23, 2024 (registration ID: CRD42024589900).
Eligibility Criteria and Study Selection
Articles meeting the following criteria were included: (1) Population: CRSwNP patients with or without comorbid asthma or AERD, regardless of prior steroid treatment, nasal rinse, or surgery. (2) Intervention: all types of biologics to control CRSwNP, commonly as an additional therapy to intranasal corticosteroids. (3) Comparison: Placebo, intranasal corticosteroids, or no treatment. (4) Outcomes: response rates to the treatment. We included all types of studies administering biologics for CRSwNP patients, including RCTs, cohort studies, and case series. Studies eligible for inclusion defined biologics treatment responses clearly in the original articles. The excluded criteria included non-English publications and the inclusion of pediatric patients. Studies with asthma and partially comorbid CRSwNP populations were also excluded if there were no independent data on CRSwNP populations.
Screening and Data Extraction
The titles and abstracts of the retrieved literature were independently reviewed by 2 investigators (D.W. and S.X.), and any disagreement was referred to a third investigator for decision. Subsequently, 2 researchers (D.W. and S.X.) further screened the full text of the articles to decide the inclusion or exclusion of the selected studies, and discrepancies were discussed with the third researcher. Two researchers (D.W. and S.X.) independently used pre-designed tables for information extraction of each study, including first author, year of publication, country of origin, study design, patients, comorbidity, biologic therapy, follow-up duration, response criteria used, and response rate. Regarding the follow-up duration, the EPOS2020 criteria recommended that patients be scrutinized at 16 weeks and 12 months, respectively, to determine the response to biologic therapy, 22 whereas in the updated guidelines, the assessment should be at 6 and 12 months. 24 The follow-up periods of the studies we included were typically 6 and 12 months. To enable the biologics to take full advantage of their efficacy, we normally extracted data at the endpoint time if studies provided data at multiple follow-up time points and calculated the corresponding response rates. Again, the conflicts were resolved by discussion with the third investigator.
Quality Assessment
The quality of cohort studies was evaluated using the Newcastle-Ottawa Scale (NOS), which judges the quality of a study on 3 broad perspectives: the selection of study groups, the comparability of the groups, and the determination of exposure or outcome of interest. 25 A star system was used to quantitatively assess the quality of the study, with a rating ranging from 1 to 9 stars (highest quality). The risk of bias of case series was accessed by the Joanna Briggs Institute (JBI) Critical Appraisal Checklist for case series, which consists of 10 items that evaluate case series from inclusion criteria, disease problem assessment to clinical information report. 26 The risk of bias for each randomized controlled trial was assessed using the Cochrane Collaboration tool, which makes evidence-based judgments about the following elements: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome evaluation, incomplete outcome data, selective reporting, and other bias. 27 Each risk of bias element was evaluated as “low,” “high,” or “unclear.”
Statistical Analysis
Characteristics of different evaluation criteria were summarized in tables and narratively described. Depending on whether the evaluation criteria adopted in the studies encompassed multidimensional assessments, we categorized them into comprehensive evaluation criteria (incorporating 2 or more distinct items) and individual evaluation criteria (containing a single element). The studies providing data on response rates of CRSwNP patients to biological treatments, which were included in the single-rate meta-analysis, used these comprehensive criteria (EPOS or EUFOREA criteria) or individual criteria (NPS or SNOT-22 scores) for assessment. The heterogeneity between the studies was assessed using χ2 and I2 testing, with a fixed-effects model if I2 <50% and a random-effects model if I2 ≥50%. Subgroup analyses stratified by different biological agents were also performed to evaluate the results and explore the sources of heterogeneity. All statistical analyses were performed using R 4.3.3.
Results
Study Identification and Characteristics of the Study
The literature search initially yielded 2596 documents, and a total of 107 documents were entered into the full-text review after eliminating duplicates and screening titles and abstracts. Eventually, 29 studies met the eligibility criteria for inclusion in this review18,28-55 (Figure 1), including 20 cohort studies, 8 RCTs/post hoc studies of RCTs, and 1 case series. Of these, 25 studies reported response to treatment in CRSwNP patients who had been adequately treated with biologics according to comprehensive criteria (EPOS or EUFOREA criteria) and individual criteria (NPS or SNOT-22 scores). The remaining 4 studies used different response evaluation measures after treatment and were not meta-analyzed. Two post hoc analyses, both originating from the SINUS-24 (NCT02912468) and SINUS-52 (NCT02898454), 15 and another 2, both from the SYNAPSE study, 17 were jointly included in the meta-analysis due to different response criteria involved.
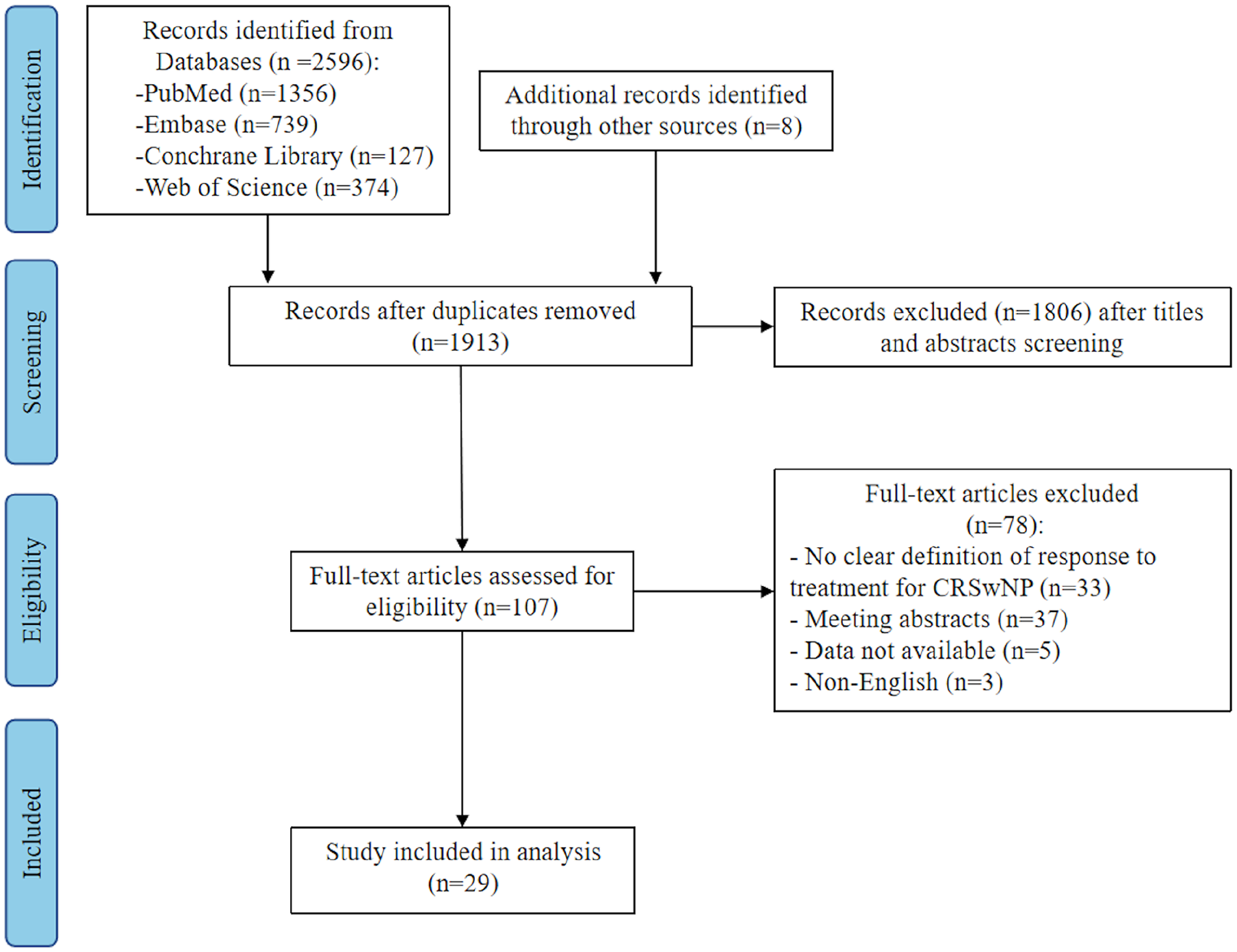
PRISMA flow diagram of the literature review.
Detailed characteristics of the included studies for meta-analysis were exhibited according to different response criteria (Supplemental Tables 1–5). A total of 3745 patients treated with biologics were included in the meta-analysis of 25 studies, with asthma comorbidity between 56.5% and 100% and AERD comorbidity between 3% and 65.2% in all studies. The length of follow-up in all studies ranged from 4 to 24 months. The types of biologics selected for treatment included: Dupilumab (n = 12),27-38 Benralizumab (n = 6),40-45 Mepolizumab (n = 3),46-48 Omalizumab (n = 3),18,49,50 and 1 article 51 used Dupilumab, Omalizumab, or Mepolizumab without distinguishing between biologics.
Quality Assessment
Twenty cohort studies used NOS for quality assessment, and the results are shown in Supplemental Table 6. All studies scored between 6 and 9 (maximum 9) and were of medium to high methodological quality. One case series was evaluated using the JBI critical appraisal checklist for case series, and the results showed that 8 out of 10 questions were answered yes. Eight RCTs/post hoc analyses used the RoB2 tool for the assessment of bias risk, which indicated that the included studies had low risk of bias (Supplemental Figure 1), with the main source of bias being random sequence generation.
Different Evaluation Criteria of Treatment Response in Patients With CRSwNP
We divided the evaluation criteria into comprehensive and single criteria (Tables 1 and 2). Criteria are further classified as clinical improvement of parameters during treatment and evaluation of the disease severity after treatment based on implications of their specific content. Various objective measures and self-reported results were applied to assess CRS symptoms, quality of life, size of nasal polyps, and comorbidities.
Different Comprehensive Evaluation Measures of Response to Biologic Treatment.
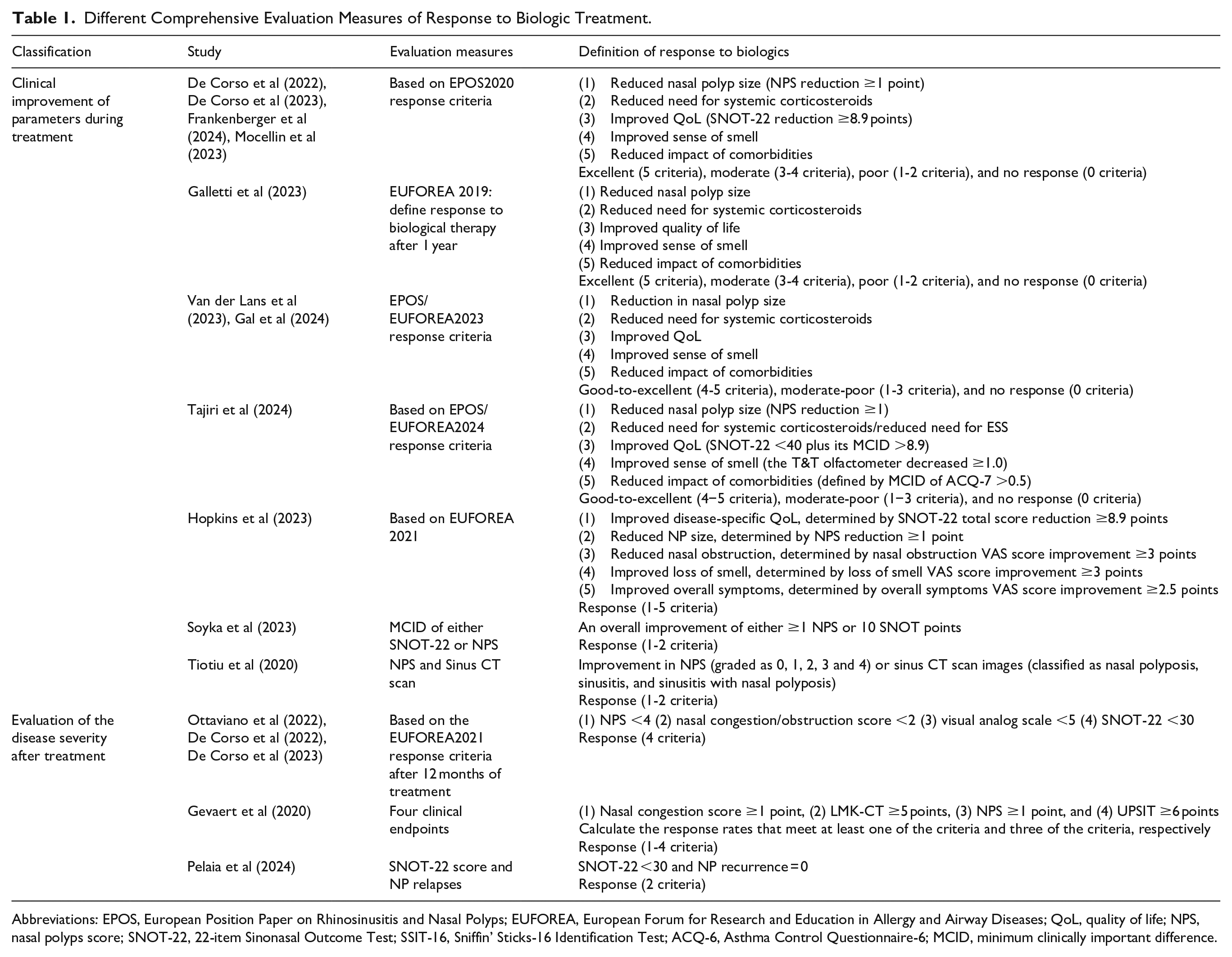
Abbreviations: EPOS, European Position Paper on Rhinosinusitis and Nasal Polyps; EUFOREA, European Forum for Research and Education in Allergy and Airway Diseases; QoL, quality of life; NPS, nasal polyps score; SNOT-22, 22-item Sinonasal Outcome Test; SSIT-16, Sniffin’ Sticks-16 Identification Test; ACQ-6, Asthma Control Questionnaire-6; MCID, minimum clinically important difference.
Different Single Evaluation Measures of Response to Biologic Treatment.
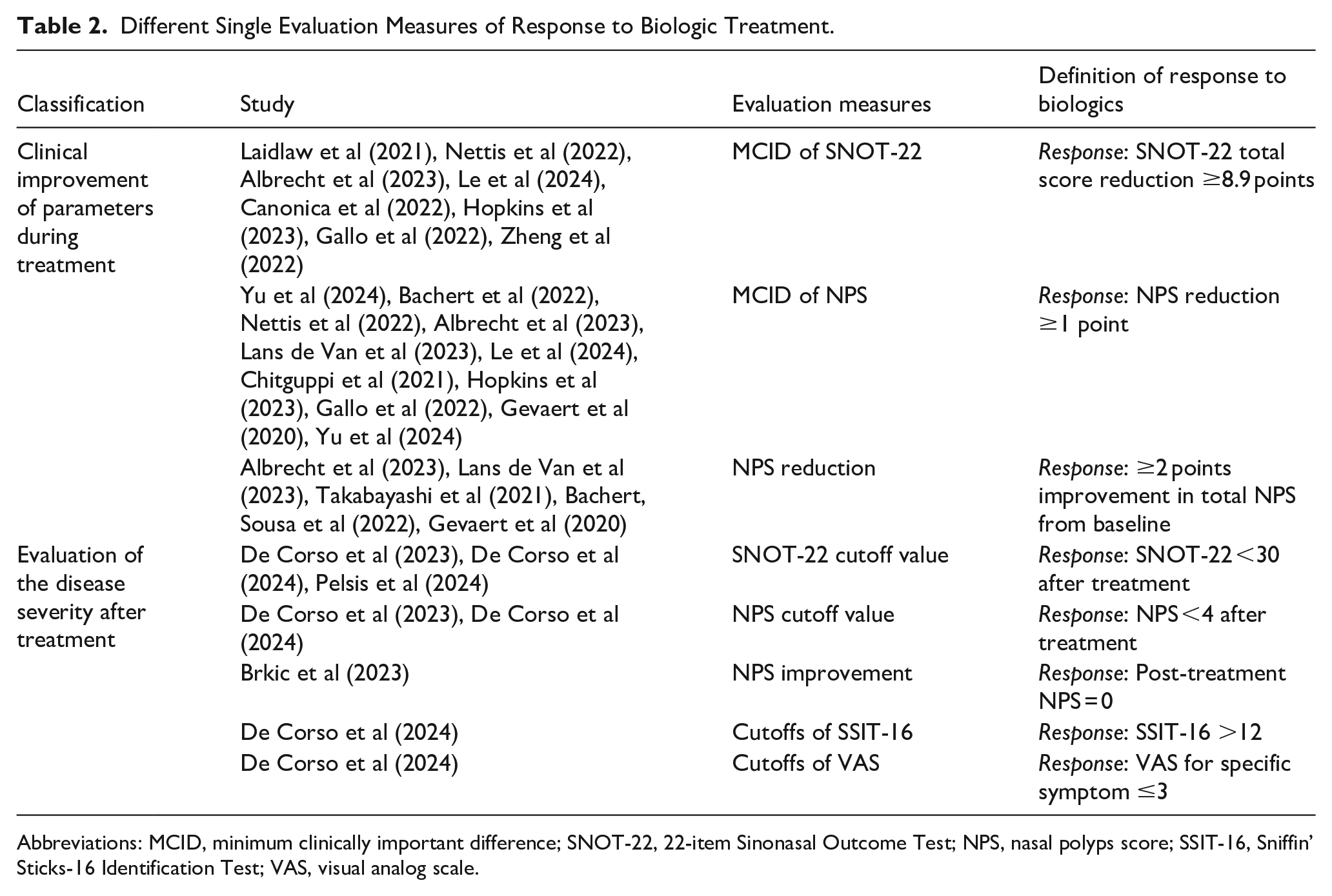
Abbreviations: MCID, minimum clinically important difference; SNOT-22, 22-item Sinonasal Outcome Test; NPS, nasal polyps score; SSIT-16, Sniffin’ Sticks-16 Identification Test; VAS, visual analog scale.
The comprehensive assessment criteria consist of 2 or more items that define responders to biological therapy in multiple dimensions (Table 1). To evaluate clinical improvement, experts in EPOS and EUFOREA proposed 5 items that are widely recognized and implemented to assess treatment response, including (1) reduced nasal polyp size, (2) reduced need for systemic corticosteroids, (3) improved quality of life, (4) improved sense of smell, and (5) reduced impact of comorbidities. 22 CRSwNP patients were classified into 3 response groups (no/no poor response, moderate/poor response, and good/excellent response) to 4 (no response, poor response, moderate response, and excellent response) according to the number of items satisfied after biologics treatment. As stated in 5 studies, the EUFOREA2019 and EPOS2020 criteria initially categorized patients who met all 5 criteria after biological treatment as excellent responders, 3 to 4 criteria as moderate responders, and 0 to 2 criteria as non-poor responders.28,29,31,51,52 In the latest EPOS/EUFOREA2023 and EPOS/EUFOREA2024, the definition of excellent response is updated to meet the above 4 to 5 criteria, which promote the portion of patients who can achieve an excellent response.24,56 In EPOS/EUFOREA2023, fulfilling 1 to 3 criteria after treatment is considered a moderate-poor response, and not fulfilling any criteria is considered no response. In addition, in EPOS/EUFOREA2024, patients who meet 2 to 3 criteria are considered moderate responders to biologics, and those who meet 0 to 1 criteria are considered non-poor responders. At 12 months of treatment, the EUFOREA2021 response criteria use the following 4 thresholds, which assess the severity of the disease after treatment to define a responder: NPS <4, nasal congestion/obstruction score <2, visual analog scale (VAS) <5, and SNOT <30. 23 Furthermore, 4 studies combined NPS with SNOT-22,45,53 sinus CT scan images, 54 and subjective complaints, 55 respectively, to define responders. This emphasizes the role of biologics in reducing nasal polyps, while highlighting the reliability of objective assessment measures.
In particular, in each aspect of the assessment of comprehensive criteria, clinical improvement or thresholds for the parameters were simultaneously addressed during the follow-up periods. Almost all studies used a decrease in NPS of more than 1 point or NPS <4 to determine whether nasal polyps shrink compared to pretreatment conditions. However, Pelaia et al considered patients reaching no NP relapse as eventual responses to biologics. 45 All studies used a decrease in the SNOT-22 score that exceeded 8.9 points to assess whether patients’ quality of life improved. Based on this, Tajiri et al added SNOT-22 <40 plus its minimum clinically important difference (MCID) >8.9 to define improved quality of life. 34 Comparatively, the effect of biological treatments on olfactory function has different criteria in all studies. The criteria for improved or normal olfactory function included an improvement of 4 points in the Sniffin’ Sticks-16 Identification Test (SSIT-16), 11 to 12 correct identifications of the Sniffin’ Sticks-12 Identification Test (SSIT-12), University of Pennsylvania Smell Identification Test 6 points, a decrease of 1.0 in the T&T olfactometer, which is a standardized olfactory function testing in Japan, 34 and a ≥3 points improvement in VAS score. Post-treatment Asthma Control Questionnaire-7 (ACQ-7) >19 or ACQ-7 >0.5 MCID in patients after treatment generally indicated well-controlled asthma and thus represented a clinical improvement in comorbidities. In addition, Van de Lans et al asked CRSwNP patients only one ACQ-7 question to measure asthma control: “How would you rate your asthma control in the last 4 weeks?” 32 which improved the research efficiency and somewhat simplified the evaluation process.
In terms of a single criterion, we also divided it into 2 categories to assess clinical improvement and disease severity (Table 2). Clinical parameters to evaluate improvement in patients treated with biologics included SNOT-22 MCID (SNOT-22 score reduction <8.9 points), NPS MCID (NPS reduction <1 point), and NPS reduction <2 points from baseline. After treatment, SNOT-22 <30, NPS <4, post-treatment NPS = 0, SSIT-16 >12, and VAS for specific symptoms 3 were used to assess responsiveness in several of the included studies. Among them, SNOT-22 and NPS remained the most widely used assessment methods.
Meta-Analysis
Response Rates to Biologics Based on Comprehensive Criteria
The response rates measured by comprehensive criteria are shown in Figures 2, 3A, and 3B, where the majority of the patients were treated with Dupilumab for 6 or 12 months. In the included studies, 4 articles classified meeting all 5 criteria as an excellent response,28,29,31,51 while 3 articles categorized meeting the 4 to 5 criteria as an excellent response.32-34 When defining the response to biologics according to the EPOS2020 response criteria, our analysis showed that 39.3% (95% CI [Confidence Interval], 0.231-0.670; I2 = 95%) and 49.7% (95% CI, 0.378-0.653; I2 = 95%) of CRSwNP patients treated with biologics (after 6 months of treatment, specifically) achieved excellent and moderated response, respectively, while the non-poor response rate was only 6% (95% CI, 0.024-0.147; I2 = 91%) (Figure 2). The good-excellent response rate notably reached 87.7% (95% CI, 0.774-0.994; I2 = 67%) when meeting 4 to 5 criteria was considered a satisfactory response (Figure 3A). Furthermore, the percentage of response to biologics (after 12 months of Dupiumab treatment, specifically) in patients with CRSwNP was 85.9% (95% CI, 0.660-1.000; I2 = 98%) according to EUFOREA2021 response criteria (Figure 3B).
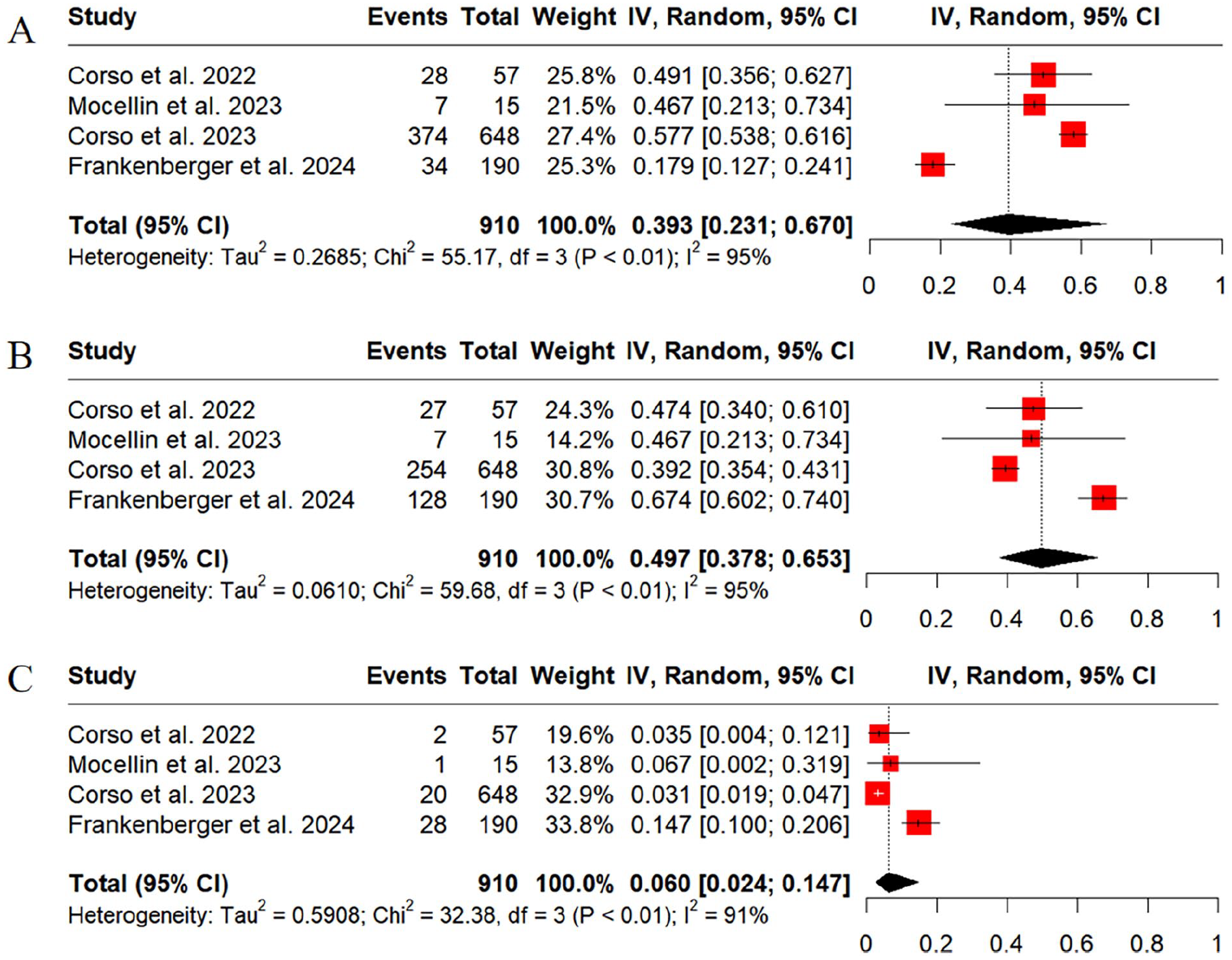
Meta-analysis of the response rates to biological agents defined by the EPOS2020 response criteria. Results for the excellent response (A), moderate response (B), and non-poor response (C).
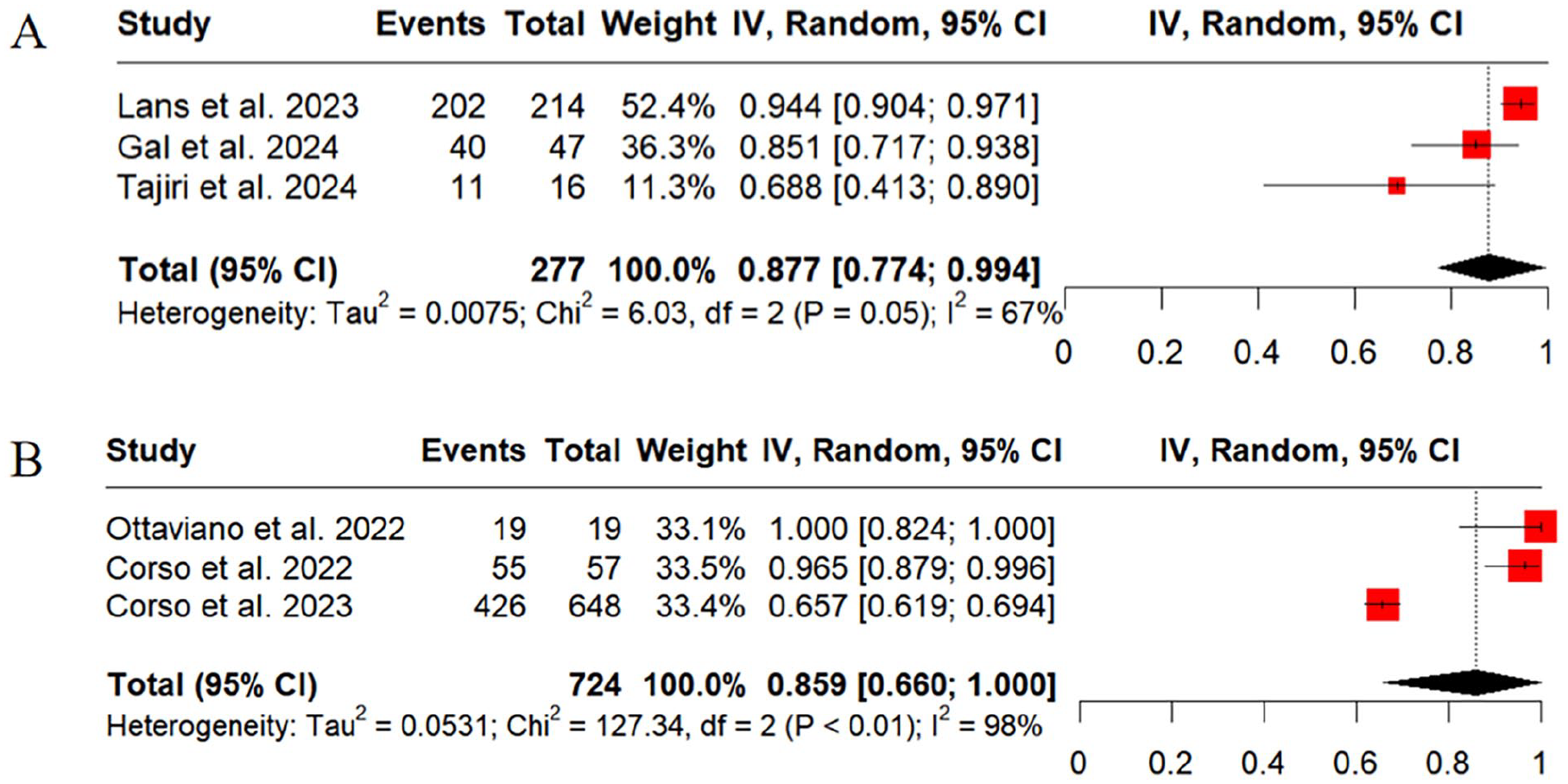
Meta-analyses of biological response rates of agents defined by different response criteria. Good-excellent response rate defined by the EPOS/EUFOREA2023/2024 response criteria (A) and response rates defined by the EUFOREA2021 response criteria (B).
Response Rates to Biologics Based on Single Criteria
The response rates to biologics based on individual response criteria were also analyzed. A total of 3,28,29,358,37-40,44,46,47,49 11,18,32,36-38,40-42,46,47,50 3,29,30,45 and 229,30 articles used EUFOREA2021 response criteria, MCID of SNOT-22, MCID of NPS, SNOT-22 <30, and NPS <4 as assessment criteria at the end of follow-up, respectively. When the types of biologics are not distinguished, the results showed that 77.6% (95% CI, 0.708-0.849; I2 = 85%) and 62.9% (95% CI, 0.518-0.746; I2 = 97%) patients achieved SNOT-22 8.9 points (Figure 4A) and improvement in NPS 1 point (Figure 5A) from baseline, respectively. Further subgroup analyses revealed that Dupilumab achieved the highest response rates, 85.0% (95% CI, 0.748-0.966; I2 = 94%) according to MCID of SNOT-22 (Figure 4A) and 84.7% (95% CI, 0.696-1.000; I2 = 98%) according to MCID of NPS (Figure 5A), respectively. Furthermore, 78.4% (95% CI, 0.562-1.000; I2 = 96%) of patients were successfully treated with SNOT-22 <30 at the end of follow-up (Figure 4B) and 82.8% (95% CI, 0.801-0.857; I2 = 0%) with NPS <4 (Figure 5C).
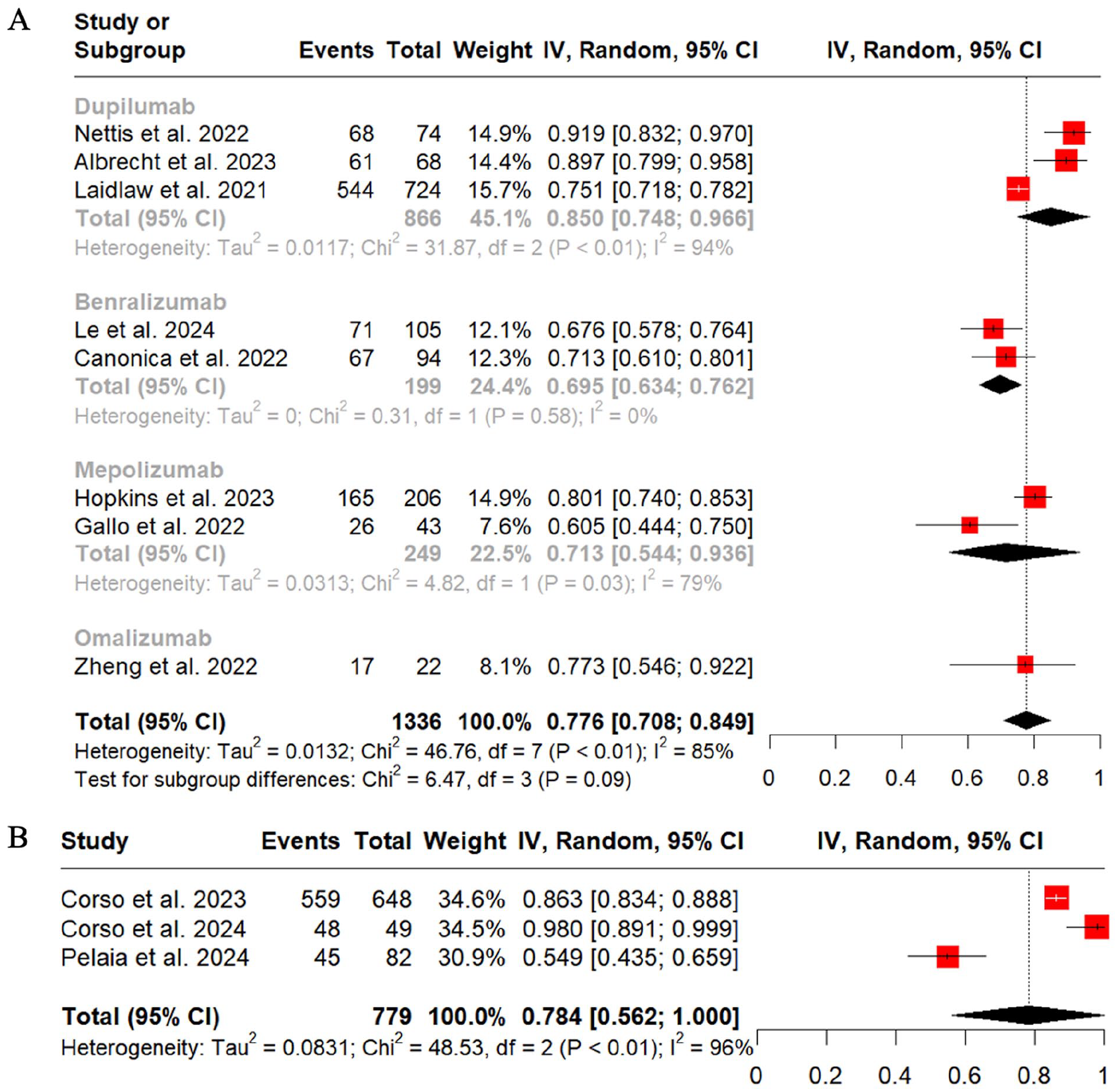
Meta-analyses of response rates to biological agents defined by single criteria, including SNOT-22 MCID (A) and SNOT-22 <30 (B). MCID, minimum clinically important difference; SNOT-22, 22-item sinonasal outcome test.
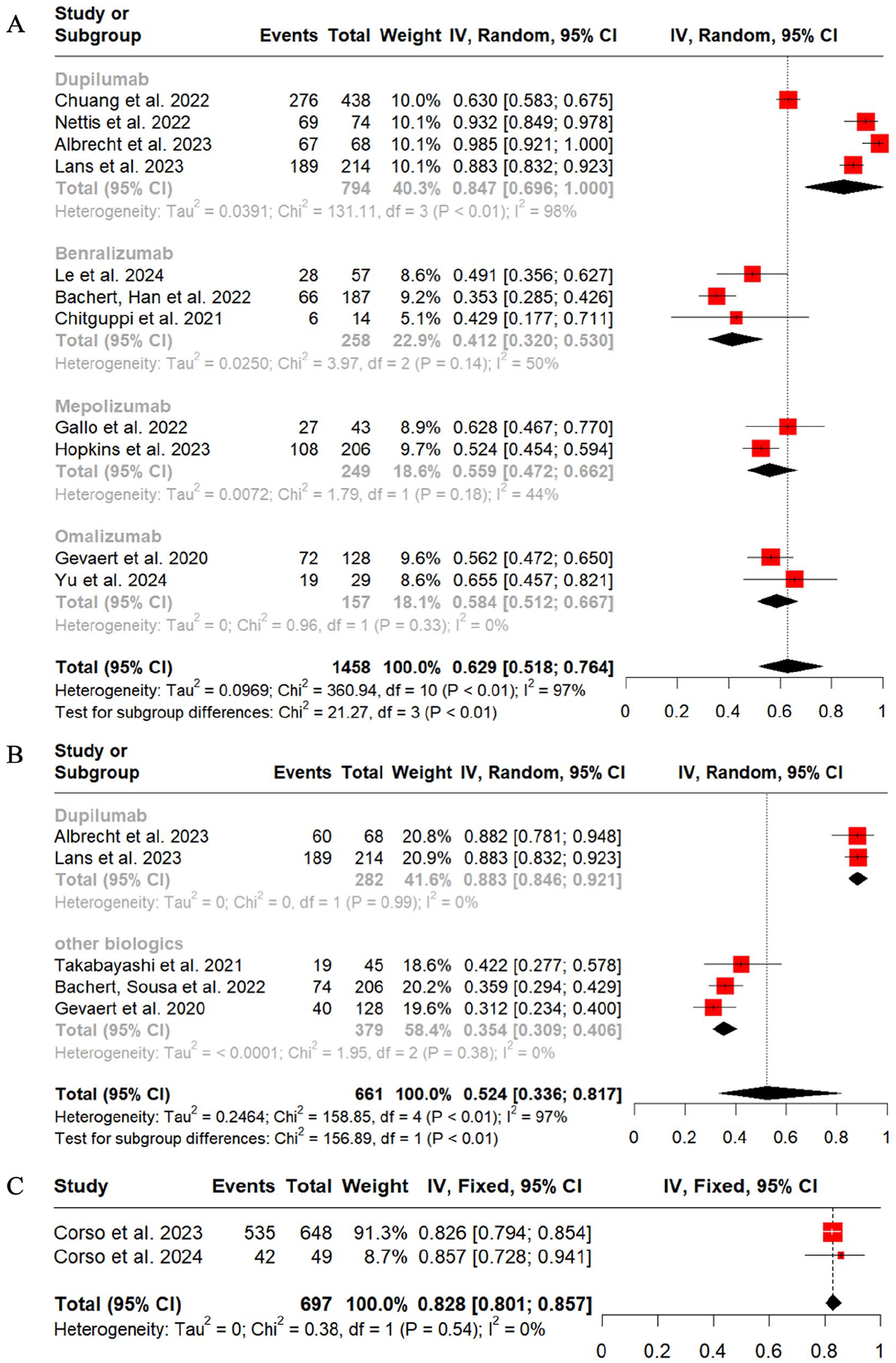
Meta-analyses of response rates to biological agents are defined by a single criterion, including MCID of NPS (A), NPS reduction 2 (B), and NPS <4 after treatment (C). MCID, minimum clinically important difference; NPS, nasal polyps score.
Discussion
Defining the response criteria with key outcome measures has remained a challenge due to inconsistency in endpoint reporting. This review found that there were both comprehensive and single criteria for response to biologic therapy. The criteria were further classified as clinical improvement of parameters during treatment and evaluation of the severity of the disease after treatment based on implications of their specific content. Although many studies have demonstrated the efficacy of biologics, it remains unclear what percentage of patients will ultimately become responders, especially with multiple evaluation methods. We summarize the current response criteria after biologic therapies based on different response criteria. This systematic review and meta-analysis of 29 studies is the first to summarize various applicable response criteria and to analyze the overall response rates to biologics among patients with CRSwNP.
We first summarize the characteristics of different response criteria for patients with CRSwNP treated with biologics. According to EPOS2020, biologics are recommended for uncontrolled CRSwNP patients who experience ineffective medical treatment and surgery, in addition to meeting 3 of the following 5 criteria: presence of type 2 inflammation, regular need for systemic corticosteroids, significant impact on quality of life, loss of smell, and comorbid asthma. 22 It also described the severity of the disease in a round way. Consequently, items in the response criteria assess disease remission and define biologic response components from similar aspects. We divided the evaluation criteria into comprehensive and single criteria, in which the comprehensive criteria are usually derived from EPOS or EUFOREA, and the single criteria used in the studies are often one of the comprehensive criteria. Obviously, a single valid criterion facilitates a quick and accurate determination of the efficacy of biologics and has better applicability in the real world; however, comprehensive criteria that assess the improvement of the CRS disease in multiple dimensions and thus yield more reliable results may be a preferred option when obtaining research data.
In comprehensive criteria, EPOS and EUFOREA focused on 5 aspects of nasal polyp size, systemic corticosteroid needs, quality of life, smell, and comorbidities to develop evaluation criteria, classifying CRS patients into 3 or 4 response groups according to the number of items met after treatment. The response of the included subjects was classified by EPOS2020 and EUFOREA2019 as no (0 criteria met), poor (1-2 criteria met), moderate (3-4 criteria met), or excellent (5 criteria met) in 5 studies.28,29,31,51,52 EPOS/EUFOREA2023 classifies responders into 3 groups, with those who meet 0 criteria as no responders, 1 to 3 criteria as poor-moderate responders, and 4 to 5 criteria as good-excellent responders.32,33 EPOS/EUFOREA2023 combines the 2 intermediate grades of EPOS2020, as clinical recommendations for poor to moderate responders are generally consistent, namely, continuing the biologics until re-evaluation of treatment response at 12 months or choosing a different treatment strategy based on the patient’s preference. 24 The updated criteria are considered more concise and practical. As suggested by Fokkens et al, 24 concerning the absence of comorbidities in some patients, which prevented the fulfilment of all 5 criteria, changing the definition of good-excellent response to meet 4 to 5 criteria resulted in an increase in the response rate from 39.3% to 87.7% in the pooled results. Our results also indicated that the EPOS 2020 response criteria appeared to be more stringent than the other criteria, with only 39.3% of the patients achieving an excellent response, while 62.9% to 87.7% of the patients under the other criteria ended up as responders, which is close to the 50% to 75% estimated in the EUFOREA expert board meeting in 2021. 23 This encourages more CRSwNP patients to continue biological therapies to avoid subsequent salvage treatments, such as surgery or additional courses of systemic corticosteroids. Furthermore, the updated expert opinion extends the evaluation time from 16 weeks to 6 months to allow sufficient time for biologics to reach their full potential.23,24 Comprehensive criteria also combine NPS with SNOT-22, sinus CT scan, or patients’ subjective complaints to jointly evaluate efficacy. Although appearing in a small minority of studies, the criteria included are still of some reference significance.
SNOT-22 and NPS were ranked as the 2 most commonly utilized scoring systems among single criteria. SNOT-22 is considered the most robust patient-reported questionnaire for assessing the condition of sinusitis symptoms and quality of life. 57 At the same time, NPS is an objective score indicator for scoring each side of the sinuses by nasal endoscopy and calculating total scores. 58 SNOT-22 and NPS have been used increasingly to assess disease severity and detect the efficacy of various treatments for CRS in recent years, including biologics. In general, the pooled response rates to biologics assessed by the thresholds of the 2 metrics were approximate, where 78.4% versus 82.8% of patients fell below the NPS and SNOT-22 scores cutoffs, respectively. However, more CRSwNP patients evaluated by SNOT-22 MCID (77.6%) were biologic responders than NPS MCID (62.9%), suggesting that patient-reported results may overstate data compared to objective measures. Similarly, a meta-analysis evaluating the impact of placebo on clinical trials of CRS medical treatments found that SNOT-22 demonstrated a significant improvement of 20% relative to baseline following the placebo administration, reaching 9.49 points on average. However, NPS did not show a significant difference before and after placebo. 59 Although objective measures often involve more costly and operator-intensive procedures such as nasal endoscopy, olfactory tests, and sinus CT scans, it is still clinically recommended for precise assessment of patients and to guide subsequent treatment.
Further stratified analyses demonstrated that Dupilumab outperformed all other biologics, resulting in the most significant proportion of patients with CRSwNP who successfully achieved SNOT-22 MCID and NPS MCID. Numerous network meta-analyses and indirect comparisons indicated that Dupilumab ranked the most beneficial for primary CRSwNP outcomes,60-63 which is similar to our finding. This may be due to the fact that inhibition of IL-4Rα would achieve dual blockade of IL-4 and IL-13 signaling, which were the key pathways driving type 2 inflammation. A study that included data from RCT of type 2 diseases such as CRSwNP, asthma, and atopic dermatitis found that Dupilumab significantly suppressed almost all biomarkers of type 2 inflammation, including serum thymus, activation-regulated chemokine, plasma eotaxin-3, serum IgE, and serum periostin, while serum eosinophil counts also decreased below baseline levels after a transient increase. 64 However, due to the lack of head-to-head comparison between biologics and convincing biomarkers, it is still beyond possible to select optimal biologics for CRSwNP patients. 65
The heterogeneity of the studies included in the meta-analysis of this study was significant, which was controlled by the random-effects model and explored by subgroup analyses for different biologic types. In Figures 4 and 5 of the results, subgroup analyses resulted in a significant reduction in heterogeneity by reducing I2 to below 50%, exhibiting that distinct biological treatments may be the main source of heterogeneity. It would be a limitation that we were unable to divide all studies into smaller units by follow-up study design, clinical characteristics of enrolled patients, biologic dose and regimen, and length of follow-up. This suggests that future research should develop a unified and standardized paradigm on the above aspects of the research process to obtain more rigorous and referable findings.
Conclusion
The present study provides a detailed summary of the current understanding and application of response criteria. There were 2 main frameworks to assess treatment response: comprehensive and single criteria, which lacked uniformity with respect to key outcome measures. Significantly heterogeneous response rates to biologics urge the development of a consistent criterion for treatment response to biologics to define treatment efficacy.
Supplemental Material
sj-docx-1-ear-10.1177_01455613251362034 – Supplemental material for Different Response Criteria and Response Rates for Biologic Therapy in Patients With Chronic Rhinosinusitis With Nasal Polyps: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-ear-10.1177_01455613251362034 for Different Response Criteria and Response Rates for Biologic Therapy in Patients With Chronic Rhinosinusitis With Nasal Polyps: A Systematic Review and Meta-Analysis by Shirong Xu, Yankun Li, Xingchen Pan, Yi Zhao and Dawei Wu in Ear, Nose & Throat Journal
Footnotes
Acknowledgements
None.
Ethical Considerations
This article does not contain any studies with human or animal participants.
Author Contributions
S.X.: methodology, literature search, data extraction analysis, formal analysis, investigation, writing, and draft preparation. X.J.: methodology, literature search, and data extraction. Y.L.: literature search, data extraction analysis, formal analysis, and investigation. Y.Z.: methodology, literature search, and data extraction. D.W.: conceptualization, Supervision, and critical revision.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Natural Science Foundation of China (82000954), Beijing Science and Technology Nova Program (Z201100006820086), Beijing Hospitals Authority Youth Program (QML20190617), Beijing Hospitals Authority Clinical Medicine Development of Special Funding (XMLX202136), and the Key clinical projects of Peking University Third Hospital (BYSYZD2023029).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author, DW, upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
