Abstract
Objective:
To objectively assess nasal flow obstruction improvement through anterior active rhinomanometry (AAR) in patients with severe chronic rhinosinusitis with nasal polyps treated with dupilumab.
Methods:
Dupilumab-treated patients were evaluated at baseline (T0), 3rd month (T1) and 6th month (T2) after therapy initiation by AAR, and nasal airway resistances were measured using the Rinopocket ED200 system™. The Lund-Mackay Score (LMS), the Nasal Polyp Score (NPS) and the Lund-Kennedy Score (LKS) were assessed. Also, patients were required to fill in the Nasal Obstruction Symptom Evaluation scale, the 22-Item Sinonasal Outcome Test and the Visual Analogue Scale for sinonasal symptoms.
Results:
Twenty-one patients (n = 21) were included. By T1, both inspiratory and expiratory resistances, as well as total nasal resistance, showed a significant reduction. At T2, these improvements were sustained but did not differ significantly from T1. Patient-reported outcomes followed a similar trend, showing marked improvement at T1 but no further significant changes at T2. Conversely, endoscopic and radiologic outcomes (NPS, LKS, LMS) demonstrated significant improvements across all timepoints.
Conclusions:
This preliminary study shows the rapid effectiveness of dupilumab in relieving nasal obstruction. Interestingly, while endoscopic and radiologic assessments showed ongoing improvement at each time point, nasal airway resistance measured with AAR did not significantly decrease beyond the initial 3 months of treatment.
Keywords
Introduction
Chronic rhinosinusitis with nasal polyps (CRSwNP) is a common inflammatory condition significantly impacting patients’ quality of life (QoL). 1 The clinical presentation of CRSwNP includes a broad spectrum of symptoms, such as anterior and posterior rhinorrhea, headache, facial and periorbital pressure, as well as disturbances in smell, taste, sleep with possible implications even on the psychiatric sphere. 2 However, nasal congestion and obstruction are among the most frequently reported and debilitating symptoms. 3
Notably, monoclonal antibodies (mAbs), specifically targeting immune pathways involved in type 2 inflammation, have emerged as an effective therapeutic alternative for patients with recalcitrant disease who do not respond to standard medical or surgical treatments.4,5 Dupilumab, a mAb targeting interleukin-4 and interleukin-13 signaling pathways, was the first approved for CRSwNP, proving rapid and sustained efficacy in disease control across both clinical trials and real-world settings.6,7
The therapeutic efficacy of biologics in clinical trials is routinely assessed using multiple standardized parameters. 8 Endoscopic measures, such as the Nasal Polyp Score (NPS) and the Lund-Kennedy Score (LKS), allow for the evaluation of polyp size and sinonasal inflammation burden but fail to reflect patient’s subjective experience, immunological state or functional improvements resulting from treatment. Radiological parameters, including the Lund-Mackay-Score (LMS), can provide useful insights through the assessment of sinus opacification reduction but offer only a partial representation of treatment effectiveness. On the other hand, patient-reported outcomes (PROMs) and validated QoL questionnaires (eg, 22-Item Sinonasal Outcome Test (SNOT-22), Nasal Obstruction Symptom Evaluation (NOSE) scale, Visual Analogue Scale (VAS) for sinonasal symptoms severity) effectively assess symptom burden but lack the ability to objectively quantify functional improvement.
Based on these assumptions, we decided to introduce quantitative assessments to objectively assess functional recovery in patients treated with dupilumab for CRSwNP at our academic tertiary center. In particular, since nasal obstruction is recognized as one of the most debilitating symptoms and typically improves early after the initiation of dupilumab therapy, we have incorporated active anterior rhinomanometry (AAR) as a reliable tool for assessing changes in nasal airflow and resistance. Aware of the limitations of the procedure used, we believe that a comprehensive evaluation of therapy effectiveness in such a debilitating condition requires the integration of subjective symptom relief, objective anatomical changes and a quantitative assessment of functional recovery.
The primary outcome of this study is to evaluate the efficacy and speed of dupilumab in improving nasal obstruction by assessing airflow variations at different timepoints using AAR. This approach aims not only to demonstrate the rapid effectiveness of dupilumab in relieving obstruction but also to underscore the importance of evaluating functional recovery for a comprehensive assessment of its overall therapeutic impact.
Materials and Methods
This single-center prospective observational study included consecutive patients with refractory CRSwNP who presented to our department between January 2023 and November 2023. All patients were prescribed dupilumab following evaluation by a multidisciplinary board consisting of allergologists, pulmonologists and rhinologists. Eligibility for dupilumab was determined based on the criteria established by the Italian Medicines Agency (Agenzia Italiana del Farmaco, Roma, Italia), which require CRSwNP patients to have persistent disease despite chronic intranasal corticosteroid therapy and either a history of systemic corticosteroid treatment (≥2 courses in the previous year) and/or endoscopic sinus surgery (with relapse or complications). Additionally, patients had to meet at least one of the following criteria: a NPS ≥5 and/or a SNOT-22 score ≥50.
As recommended by international guidelines, dupilumab was administered subcutaneously at a dose of 300 mg every 2 weeks, in combination with topical treatments such as nasal rinses and corticosteroids. 9 General characteristics and biometric data were collected from medical records. Atopy was defined as sensitization to at least 1 inhalant allergen.
Patients were evaluated at baseline (T0), 3 months (T1), and 6 months (T2) after therapy initiation. At each time point, patients underwent nasal endoscopy and CT scan with the assessment of NPS, LKS, and LMS. Also, patients had to fill out the SNOT-22 and VAS for sinonasal symptoms. The total SNOT-22 score was calculated by summing individual item scores. The global-VAS assessed the question, “How bad is my nasal condition overall?” Patient-reported nasal obstruction was assessed through specific validated tools: (i) the VAS for nasal obstruction (obstruction-VAS) and (ii) the NOSE scale. The first was shown to reliably reflect nasal obstruction compared to objective rhinomanometric findings. 10 The latter is a well-established and reproducible tool for assessing nasal obstruction symptoms. 11
At each examination, patients underwent nasal airflow assessment through AAR, and nasal airway resistances were measured using the Rinopocket ED200 system™ (Medi-care Solutions, Bologna, Italia; Figure 1). All measurements were conducted in an examination room at a temperature of 22°C to 24°C and 40% to 70% humidity. Before the examination, participants were asked to sit upright quietly for 20 minutes and to also maintain this posture through the measurements. In accordance with standard clinical practice, nasal airflow resistance was assessed separately for each nostril during both inspiratory and expiratory phases. Resistance values were determined based on the slope of pressure-volume curves analyzed at 150 Pa and expressed in Pa/cm3/second, following current guidelines. 12 Total inspiratory and expiratory resistances were ultimately recorded.

Anterior rhinomanometry interface. Each patient is required to breath nasally for 30 seconds with the opposite nostril blocked with an appropriate nasal plug. The mask is connected to a sensor (Rinopocket ED200™) for resistance analysis.
Outcomes were collected and compared across different time points using statistical analysis. Data analysis was performed using IBM® SPSS Software for Macintosh, Version 26.0 (IBM Corp, Armonk, NY, USA) and GraphPad, Version 8.0.2 (Boston, MA, USA). Statistical significance was set at an alpha level of .05. The minimum sample size was calculated based on an α-error of .05 and a β-error of .20 (statistical power = 80%). Improvements in continuous variables across time points were analyzed using 1-way repeated-measures analysis of variance (ANOVA). Mauchly’s Test for Sphericity was used to assess homogeneity of variances (ie, sphericity). In cases where the sphericity assumption was violated, the Greenhouse-Geisser correction was applied. Normality was confirmed using the Shapiro-Wilk test (P > .05). Pairwise analysis was conducted using the Bonferroni correction for multiple comparisons.
This study was conducted in accordance with the ethical principles of the Declaration of Helsinki and its subsequent amendments. Ethics approval was granted by the institutional review board (IRCCS-ICH-IEC/3402).
Results
General Characteristics
Overall, 21 patients (males: 57.1%, n = 12/21) were included in statistical analysis. The mean age was 53.88 ± 8.31 years (range: 39-71, median: 55). All patients had undergone at least 1 prior functional endoscopic sinus surgery (mean: 2.35 ± 1.45, median: 2, range: 1-6). General demographics and characteristics are presented in Table 1.
Patients’ General Characteristics.
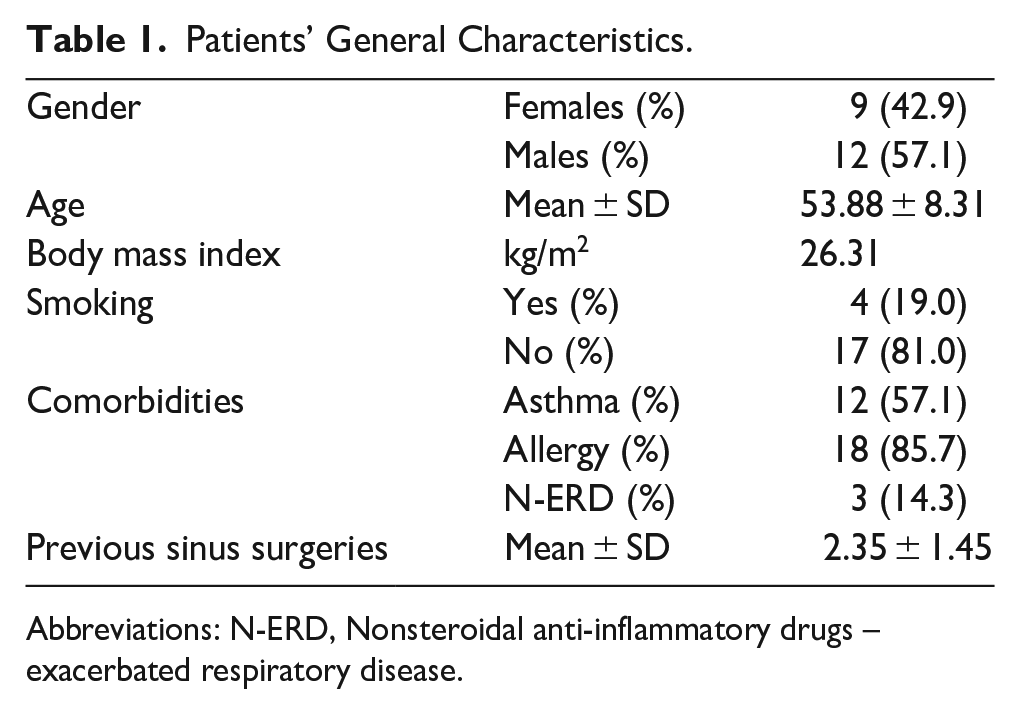
Abbreviations: N-ERD, Nonsteroidal anti-inflammatory drugs – exacerbated respiratory disease.
Nasal Flow Resistances
Mean nasal airflow resistance parameters at each timepoint are shown in Table 2.
Single-Side Inspiratory and Expiratory Nasal Airflow Resistances. Results Are Expressed in Pa/cm3/second.
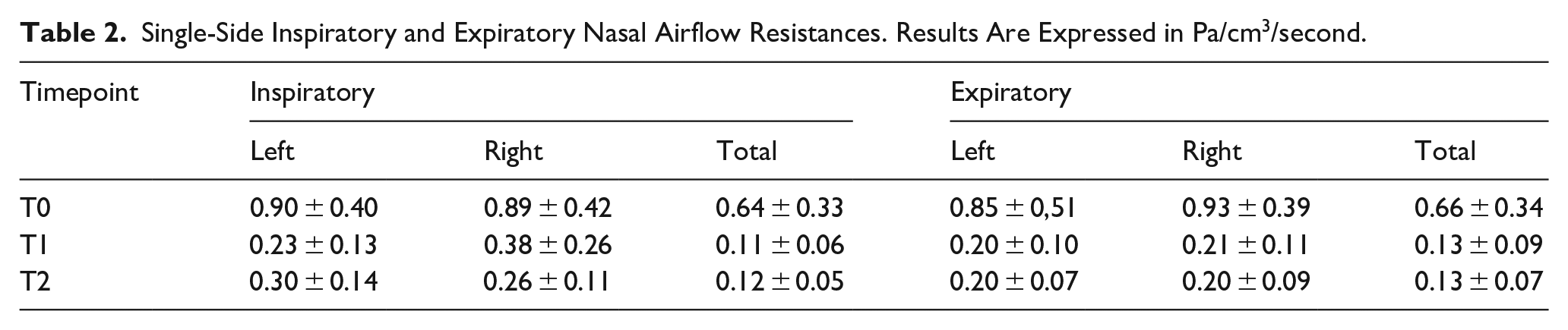
The minimum sample size to assess significant difference from the baseline was satisfied by the dimension of our sample for inspiratory, expiratory, and total resistances (n < 21).
Within-group ANOVA revealed a significant reduction in nasal airflow resistance across timepoints for all parameters (inspiratory resistance: left side, P = .022; right side, P = .019; expiratory resistance: left side, P = .028; right side, P < .001; total resistance: left side, P < .001; right side, P < .001).
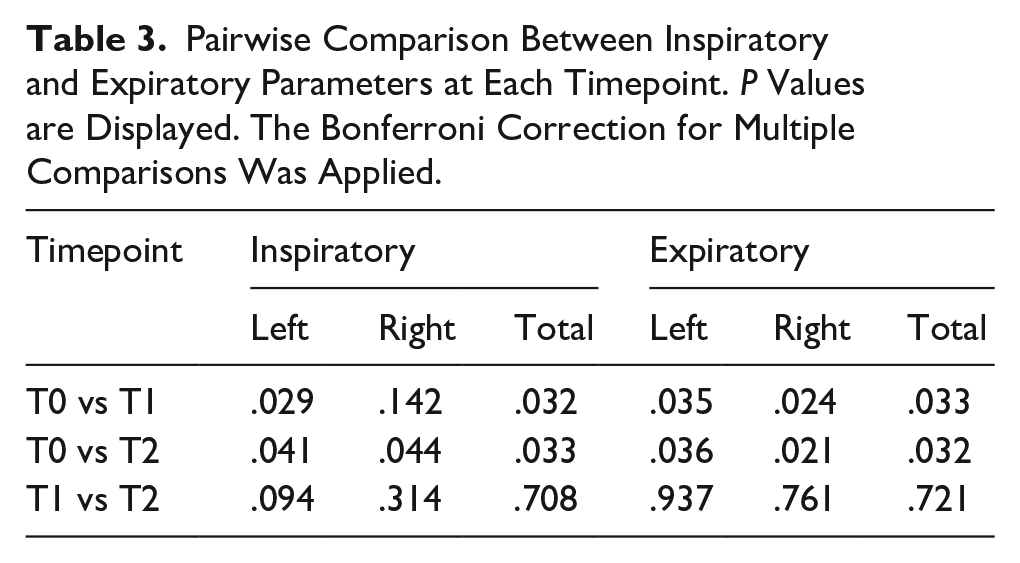
Pairwise comparisons (Table 3) showed a significant improvement in airflow resistance between baseline and subsequent timepoints, except for inspiratory right-side resistance between T0 and T1 (P = .142). Notably, no significant differences were observed between T1 and T2 for any parameter (P > .05, Table 3). Trends in single-side and total inspiratory and expiratory resistances are shown in Figure 2.
Pairwise Comparison Between Inspiratory and Expiratory Parameters at Each Timepoint. P Values are Displayed. The Bonferroni Correction for Multiple Comparisons Was Applied.
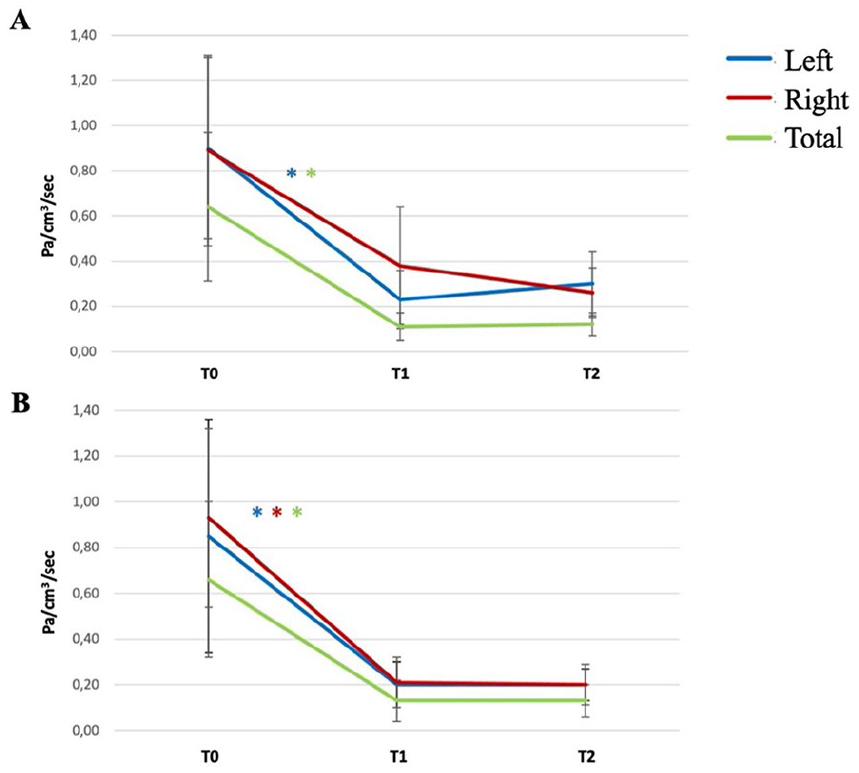
Mean nasal airflow resistance at each timepoint. Single-side and total values are displayed. Blue line: left-side resistance; red line: right-side resistance; green line: total resistance. Significant improvements at pairwise comparison are displayed by asterisks. (A) Inspiratory resistances; (B) expiratory resistances.
Endoscopic Outcomes
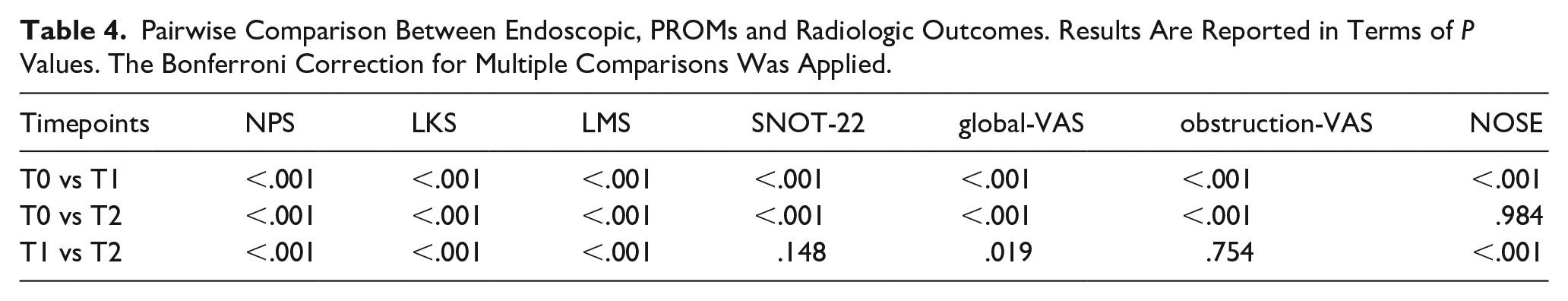
At the baseline, mean NPS and LKS were 6.10 ± 1.64 (range: 2-8) and 10.45 ± 1.85 (range: 6-14), respectively. In course of dupilumab treatment, mean NPS decreased to 3.48 ± 1.97 at T1 and 2.29 ± 2.19 at T2, whereas mean LKS decreased to 5.84 ± 2.27 at T1 and 4.16 ± 2.06 at T2. A significant improvement in endoscopic parameters across timepoints was observed at within-groups ANOVA (NPS: P < .001; LKS: P < .001). Similarly, pairwise analysis confirmed sustained and significant improvements in both outcomes at each time point (Table 4).
Pairwise Comparison Between Endoscopic, PROMs and Radiologic Outcomes. Results Are Reported in Terms of P Values. The Bonferroni Correction for Multiple Comparisons Was Applied.
PROMs
Prior to treatment initiation, mean scores for PROMs are as follows: SNOT-22: 55.84 ± 21.67 (range: 16-91), NOSE: 13.76 ± 5.12 (range: 7-20), global-VAS: 7.64 ± 1.86 (range: 4-10) and obstruction-VAS: 7.18 ± 2.04 (range: 3.20-10). At T1, all PROMs showed marked significant improvement (SNOT-22: 21.42 ± 14.90; NOSE: 5.52 ± 4.50; global-VAS: 3.36 ± 2.06; obstruction-VAS: 2.28 ± 2.16). At T2, further reductions were observed (SNOT-22: 16.16 ± 11.47; NOSE: 5.38 ± 4.26; global-VAS: 2.35 ± 1.93; obstruction-VAS: 1.94 ± 2.09), although pairwise comparisons did not show significance for SNOT-22, NOSE and obstruction-VAS compared with T1 (Table 4). Nevertheless, within-group ANOVA confirmed significant improvements in each PROM across the whole observation period (P < .001).
Sinus Opacification
At the baseline, mean LMS was 17.08 ± 3.37 (range: 9-22). During follow-up, mean scores were 9.50 ± 2.88 and 5.92 ± 1.67 at T1 and T2, respectively. A significant improvement in sinus opacification was observed across timepoints through within-groups ANOVA (P < .001). Pairwise comparison also confirmed sustained significant improvements in radiologic outcomes at each timepoint (Table 4).
Discussion
Nasal obstruction is a common and debilitating symptom in patients affected by CRSwNP. 3 This condition has proved to impact QoL by disrupting sleep, impairing olfaction, and reducing overall daily functioning. 13 Stull et al demonstrated that among various PROMs, nasal congestion alone is a significant predictor of patient-relevant outcomes, underscoring its clinical relevance. 14 In CRSwNP, persistent inflammation leads to mucosal edema and polyp formation, reducing nasal airflow and increasing airway resistance. 1 In a study by Mattos et al, 93% of CRS patients rated improvement in nasal obstruction as “extremely” or “very” important following endoscopic sinus surgery. 15 Similarly, a very rapid improvement in clinical and patient-reported parameters has been observed in CRSwNP patients treated with dupiliumab. 16 This improvement is associated with a reduction in polyp size, typically observed within the first few weeks of therapy, which relieves obstruction in key nasal passages, specifically the inferior and middle meatus, responsible for the majority of nasal airflow. 17 While endoscopic evaluations, radiological assessments, and PROMs are important indicators of therapy efficacy, incorporating quantitative assessments of functional recovery is essential for accurately characterizing dupilumab’s overall therapeutic impact on nasal obstruction.
In our study, we used AAR as an objective tool to assess nasal airflow resistance and track improvements over time. According to the 2012 European Task Force summary on diagnostic tools in rhinology, 18 nasal patency can be evaluated using different methods. AAR is considered the standard technique, as it provides a physiological measure of nasal airflow and resistance by recording pressure and flow during normal breathing. 19 Other techniques include the assessment of the peak nasal inspiratory and expiratory flow (PNIF and PNEF), a validated method that consists in measuring the force of inhalation and exhalation through the nose. 20 Acoustic rhinometry, on the other hand, evaluates nasal patency by analyzing the echoes of sound waves transmitted into the nasal cavity. 21
Despite different limitations, such as operator variability and patient positioning, AAR remains a sensitive and reliable tool for detecting changes in nasal patency. Unlike acoustic rhinomanometry or PNIF, AAR provides reproducible measurements that capture dynamic changes in the anterior nasal cavity, the most critical region for airflow regulation. 22 The perception of nasal patency is influenced not only by the amount of air passing through the nasal cavity but also by various factors related to the complex anatomy of the sinonasal region. One important aspect is the nature of airflow, as smooth, uninterrupted flow is perceived differently from turbulent or irregular flow. 23 In CRSwNP patients, nasal polyps not only physically obstruct airflow but also disrupt its dynamics, resulting in altered turbulence that contributes to the sensation of nasal blockage. 24 In addition, this compromised nasal patency can impair convective air currents, limiting the effective distribution of topical nasal therapies throughout the nasal and paranasal mucosa. As a result, a vicious cycle is established in which chronic inflammation continues to prevent medications from reaching the very anatomical sites where the disease originates. 25 The reduction in polyp volume, which typically occurs within the first few weeks of dupilumab therapy, 16 helps clearing the lower and anterior regions of the nasal cavity, leading to a rapid normalization of nasal air currents. This results in improved airflow resistance and enhances the distribution and effectiveness of concurrent intranasal medications.
Previous estimates in the healthy population reported mean unilateral nasal resistance values of 0.51 Pa/cm3/second at 150 Pa, with no significant difference between genders. 26 On the other side, a previous meta-analysis found the average total nasal airway resistance to be 0.25 Pa/cm3/second. 12 Based on these findings, we conventionally considered pathological values to be higher than 0.50 Pa/cm3/second for unilateral nasal airway resistance and 0.25 Pa/cm3/second for total nasal airway resistance. Our study included 21 CRSwNP patients, all of whom had previously undergone sinonasal surgery. Despite prior interventions, they continued to experience significant nasal obstruction, as reflected by high NPS, SNOT-22, and VAS scores.
Baseline airflow measurements confirmed markedly-elevated nasal airway resistance beyond normal thresholds. Remarkably, 3 months after initiating dupilumab therapy, both inspiratory and expiratory resistances showed a significant reduction, approaching normal levels. This rapid improvement was sustained at the 6th month without further significant change, highlighting the swift and lasting effect of dupilumab in alleviating nasal obstruction.
Our preliminary study aimed to highlight the importance of incorporating quantitative assessments of functional recovery in patients with CRSwNP undergoing treatment with dupilumab. While improvements in PROMs, endoscopic findings, and radiologic scores following dupilumab therapy are already well established in the literature, 27 objective functional measures remain underutilized. Given the strong association between nasal obstruction and impaired QoL in this population, 28 the ability to objectively demonstrate improved nasal airflow represents a critical advancement in treatment monitoring.
In the same way that many centers are now adopting olfactometry to assess recovery from hyposmia, 29 we propose the use of AAR to complement current evaluation methods. This approach enables a more comprehensive assessment of therapeutic effectiveness by capturing measurable improvements in nasal function.
This study is subject to limitations. The main limitations of this study are the limited number of included patients and its retrospective nature; however, minimum sample size to reach statistical significance was satisfied. Moreover, the inherent variability of AAR, due to factors such as operator technique and patient positioning, may have influenced the accuracy of resistance measurements and highlights the need for cautious interpretation of the results. The goal of this work was to emphasize the importance of quantitatively assessing functional responses to biologic therapies, rather than relying solely on PROMs or endoscopic and radiologic findings.
Conclusion
This preliminary study shows the rapid effectiveness of dupilumab in relieving nasal obstruction. Interestingly, while endoscopic and radiologic assessments showed ongoing improvement at each time point, nasal airway resistance did not significantly decrease beyond the initial 3 months of treatment. AAR may represent a suitable tool for evaluating treatment response in terms of nasal obstruction. Future research is warranted to validate the findings of this preliminary study and to identify the most appropriate method for assessing the early-functional response in CRSwNP patients treated with dupilumab.
Footnotes
Ethical Considerations
This study was conducted in accordance with the ethical principles of the Declaration of Helsinki and its subsequent amendments. Ethics approval was granted by the institutional review board (IRCCS-ICH-IEC/3402).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
